Abstract
Peanut (Arachis hypogaea L.) crown rot and root rot are common diseases caused by Aspergillus niger Van Tieghem. Early and accurate detection of A. niger is key to disease management. In this study, the design of two to five sets of loop-mediated isothermal amplification (LAMP) primers was based on the EglA, GOD, Tub, NRPS, Tan, CbhA, and CbhB genes of A. niger. Of these, primer set GOD-91 was selected for optimization of the three-factor LAMP system: the Bst DNA polymerase concentration, the concentration ratio of the inner and outer primers, and the concentration of Mg2+. In addition, the optimized LAMP reaction system for A. niger detection was validated for specificity, sensitivity, and on-site feasibility. The specificity test showed that A. niger could be specifically detected with the proposed method without cross-amplification of other pathogenic fungi DNA. Moreover, based on the sensitivity test, the lowest detection limit of this reaction system was 5.1 × 10−7 ng/µL pAN01 plasmid DNA, after which a standard curve was generated for the quantitative detection of A. niger. The LAMP method was further applied for field sample assessment before and after A. niger infection, successfully detecting A. niger presence in the samples collected in the field. This study yielded a sensitive, specific, and reproducible LAMP system that can be used to assess on-site samples within 45 min. It is an effective approach for the rapid and quantitative detection of A. niger.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Peanut (Arachis hypogaea L.) is among China’s most valuable oil crops (FAOSTAT, http://faostat.fao.org). Its yield and quality are significantly impacted by pests and diseases (Zhao et al. 2024). Peanut crown rot and root rot are fungal diseases caused by Aspergillus spp., occurring more frequently at the sowing and seedling stages. The cotyledons and hypocotyls are usually infected, leading to root crown or root neck rot (Kokalis-Burelle 1997; Xu et al. 2015). Peanut infection by the host pathogen Aspergillus niger Van Tieghem often leads to seedling shortage and peanut plant death (Matloob and Juber 2014). In regions where A. niger is epidemic, the only pragmatic approach to efficiently managing peanut crown rot and root rot is via the implementation of host resistance (Ruark and Shew 2010; Tsai and Brenneman 2018). There are presently no cultivated peanut kinds or lines that exhibit significant levels of resistance against A. niger-infected plants, as far as our knowledge extends. Crown rot and root rot are still major threats to peanut production. Therefore, a strategy for the timely, effective, and precise identification of A. niger might contribute to the enhancement of resistance breeding and management strategies for this ailment (Kokalis-Burelle 1997; Xu et al. 2015; Dufault and Brenneman 2023).
The common methods for the detection of A. niger and other Aspergillus spp. fungi include morphological identification, polymerase chain reaction (PCR), enzyme-linked immunosorbent assay (ELISA), and others (Silva et al. 2011; Richardson et al. 1982; Kim et al. 2020). High sensitivity, strong specificity, relatively rapid detection, simple and inexpensive equipment, and convenient end-product detection and quantification are among the characteristics of these methods. Moreover, they can have high specificity for detecting A. niger compared to other pathogenic fungi that share a life cycle or induce symptoms similar to root rot and crown rot disease. Nonetheless, they do have the potential to provide a rapid and quantitative source that can directly detect pathogens in soil and crop samples. Therefore, for applied uses, an approach that is quick, specific, easy, and inexpensive would be more desirable. For the rapid, sensitive, and specific detection of A. fumigatus, a significant number of researchers have effectively established LAMP (loop-mediated isothermal amplification)-based assays. These assays use different target genes as targets for the creation of LAMP primers (Tang et al. 2016; Trabasso et al. 2021; Jiang et al. 2021).
The biological activity of A. niger is controlled by various genes encoding proteins, such as cellobiohydrolases, glucose oxidases, endoglucanases, nonribosomal peptide synthetases, and tanninases, among others. Cellulases are widely found in various microorganisms and include three enzyme groups: endoglucanases, cellobiose hydrolases, and glucosidases. Cellobiohydrolase (CBH) is a crucial enzyme in the cellulase system, which may directly act on the crystalline region of cellulose microfibrils, hydrolyze β-1,4-glycoside bonds, and cleave two sugar molecules from cellulose. Its activity is associated with the pathogenicity of A. niger (Qin et al. 2017). Glucose oxidase (GOD) catalyzes glucose oxidation, resulting in gluconic lactone formation and hydrogen peroxide release (Zhou et al. 2001). Endosaccharase (EglA) is a hydrolase acting mainly on the amorphous region of the cellulose molecule. It can hydrolyze the β-1,4- glycosidic bonds in a random manner and can rapidly reduce the degree of cellulose aggregation by providing a large number of reaction terminals for cellulose exonucleases, leading to the production of a large number of oligosaccharides (Annamalai et al. 2016). Tannase (Tan) not only exists in plants rich in tannin but is also widely present in A niger, A. oryzae, and A. flavus. It can hydrolyze ester bonds and depsiphenol carboxyl bonds in gallic tannins, generating gallic acid and glucose (Shao et al. 2020; Kenji et al. 2014; Yu et al. 2007).
The LAMP assay is an innovative technique for amplification of nucleic acids that is conducted under isothermal conditions utilizing strand displacement activity Bst DNA polymerase. Amplicons containing single-stranded loops are produced using a set of four to six custom-designed primers that detect a minimum of six different sequences on the target DNA. This configuration enables the primers to bind to the sequences without requiring multiple rounds of thermal denaturation (Tsugunori et al. 2000; Nagamine et al. 2001). A portable fluorescent reader, such as the ESE Quant Tube Scanner (fitted with a battery pact), or a real-time PCR thermal cycler, may be used to quantify LAMP products following staining with fluorescent dye. The fluorescent reader has the advantage of providing a convenient method for rapid on-site detection (Nagamine et al. 2002; Lucchi et al. 2010; Njiru et al. 2012; Peng et al. 2013).
This study focuses on developing and evaluating a specific real-time LAMP (RealAmp) assay for the direct and rapid quantitative detection of A. niger in soil samples or peanut plants that are naturally infected before planting or the development of root rot or crown rot symptoms. In this study, EglA, GOD, Tub, NRPS, Tan, CbhA, CbhB, and other A. niger target genes were used for primer screening and selection. The proportion of Bst DNA polymerase, the primer ratio, and the Mg2+ content were optimized as important reaction components of the LAMP reaction system by the implementation of a single-factor experiment coupled with an orthogonal experimental method. The method’s specificity and sensitivity were assessed, and the LAMP protocol’s applicability to field sample testing was validated by the use of samples that were infected both naturally and artificially. Further investigations into resistance breeding, root rot, crown rot disease management, and A. niger-peanut plant molecular interactions may be facilitated by the optimized RealAmp method developed in this work.
Materials and methods
Pathogen and plant material preparation
A. niger was isolated from peanut plants with typical crown rot symptoms, and the fungal strain was purified and morphologically and molecularly characterized. Strains of Aspergillus species (A. oryzae, A. terreus) were kindly provided by Professor Xianli Gao (Jiangsu University, Wuxi, China). Other pathogenic fungi, including Fusarium avenaceum, Pythium aristosporum were kept in our lab and propagated on freshly prepared potato dextrose agar (PDA) medium. The above pathogenic fungal strains were preserved in 30% glycerol and kept at −80 °C for long-term preservation.
A. niger artificial inoculation on peanut (including c.v. Zhenghua25, Luohua16, Zhuhua7, Yuhua143) seedlings executed in the manner stated previously (Xu et al. 2015; Matloob and Juber 2014). Harvesting of the infected peanut samples took place before the appearance of symptoms for further DNA isolation.
Soil sample inoculation
Conidiospores were prepared as previously described (Matloob and Juber 2014). Hemocytometer counting was utilized to adjust the obtained conidia to the intended concentration of 109 spores/mL for soil inoculation. Subsequently, we inoculated 10 g of twice-autoclaved soil substrates into 15 mL falcon tubes utilizing 1 mL titers of the A. niger conidiospore suspensions on the artificially infected soil samples. This was followed by a 10-day incubation of the inoculated soil samples at 25 °C. After three days of air-drying at room temperature, the specimens were crushed to a fine powder in liquid nitrogen. The obtained powder was subsequently preserved at −80 °C before DNA isolation.
DNA extraction
Conidial suspensions were filtered on sterile filters and collected for use in DNA extraction with the Mag-Bind® universal 96 Kit (Omega Bio-Tek, Norcross, GA, USA) as directed by the manufacturer or by employing a modified version of the CTAB method as outlined by Xu et al. (1999).
The E.Z.N.A.® SP Fungal DNA Kit (Omega Bio-Tek) was used as guided by the manufacturer to extract mycelial DNA from the fungal strains. Then, the E.Z.N.A.® SP Plant DNA Midi Kit (Omega Bio-Tek) was applied for the extraction of the genomic DNA from peanut plants. As reported earlier (Volossiouk et al. 1995; Wang and Chang 2003), the total genomic DNA was isolated from the soil samples (0.5 g, including around 108 teliospores).
Following the preparation of the soil and plant samples obtained from the field, DNA isolation was performed utilizing the mobile DH-WSW™ mini-DNA workstation (Double Helix Tech. Inc.).
LAMP primer design and screening
The Egla, GOD, Tub, NRPS, Tan, CbhA, and CbhB genes of A. niger were screened to select the optimum target gene for primer design for the A. niger LAMP assay. Based on the gene’s cDNA sequence, the PrimerExplorer V5 package (http://primerexplorer.jp/lampv5e/index.html; Eiken Chemical Co., Ltd., Tokyo, Japan) was utilized to construct six LAMP primers (outer primers F3 and B3; forward and backward inner primers (FIP and BIP, respectively); and loop primers, (LoopF and LoopB) for each gene. Every gene under evaluation was assigned between two and five sets of LAMP primers. Table S1 displays a list of these primers. The synthesis and HPLC purification of every primer were performed by TsingKe BioTech (Beijing, China). The basic local alignment search tool (BLAST) was employed to examine each primer’s specificity against non-redundant GenBank database sequences of fungal and human DNA.
Real-time LAMP reaction system optimization
Previously documented procedures (Cao et al. 2015, 2017) were used to perform the real-time LAMP assay for detecting A. niger LAMP primers. Then, 3 variables and 4 levels were used in a single-factor experiment to optimize the LAMP reaction. A 0.2 µM concentration of the outer primer was utilized in the field test and statistical analysis, with 1:4, 1:6, 1:8, and 1:10 set as the respective concentration ratios of the two primer pairs (outer primer F3/B3 and inner primer FIB/BIP). A total of 2.0 U, 4.0 U, 6.0 U, and 8.0 U of Bst DNA polymerase were added to the reaction mixture. The respective concentrations of Mg2+ were established as follows: 6.0, 7.0, 8.0, and 9.0 mM. Each experiment was performed in triplicate. In the LAMP optimization experiment, a cumulative sum of 16 combinations was sequentially assessed utilizing an L16 (45) orthogonal experimental design to prevent possible interference arising from interactions among distinct variables of the LAMP reaction (Table 1) (Ming 2005). There were two repetitions of each combination.
The CFX96™ real-time PCR detection system (Bio-Rad, Hercules, CA, USA) was utilized to execute the RealAmp reaction. Sixty cycles of the reaction below were performed: 63 °C for 30 s (stage 1), 63 °C for 15 s (stage 2), and 63 °C for 45 s (stage 3) followed by a melt curve (95 °C for 15 s, 65 °C to 95 °C increment 0.5 °C for 0.5 s). Following each cycle, this reaction was accompanied by the analysis of fluorescence signals utilizing a Bio-Rad CFX Maestro. An “S” amplification curve was used to classify a result as positive across all three response monitoring systems, while a linear or slightly oblique amplification curve indicated a negative result. Alternatively, the pathogen detection device ZYD-S1™ (Double Helix Tech. Inc., Guangzhou, China) was utilized to undertake the RealAmp assay at 63 °C for an hour as previously described (Cao et al. 2017). After the LAMP reaction, the products might also be recognized directly by visually examining the color of the solution, mixing, gently centrifuging it, and pre-adding 1 µL of SYBR Green I into the reaction solution. Positive reactions exhibited green fluorescence that was perceptible to the human eye, whereas negative reactions maintained an orange color.
LAMP specificity test
To confirm the LAMP assay specificity to A. niger, samples of DNA from other fungal species, including A. oryzae, A. terreus, F. avenaceum, F. graminearum, and P. aristosporum were evaluated in the assays. Moreover, negative and positive controls, respectively, were established in the LAMP reactions using the pathogenic microorganisms Staphylococcus aureus and Salmonella enterica.
LAMP sensitivity
To determine the sensitivity of the LAMP test for detecting A. niger, conventional PCR was employed to amplify a DNA fragment (202 bp) encompassing the LAMP target site of the gene in the A. niger genome. The primers utilized in this PCR were GOD-91 F (5′-TTGAGCGCCTGGACTGAG-3′) and GOD-91R (5′-TGGCATAGAACACCGTCATG-3′). The next step involved the purification of the acquired PCR product before cloning it into the pClone007 Versatile Simple Vector Kit (TsingKe BioTekas guided by the manufacturer. Sequencing was performed to confirm the resultant recombinant plasmid pAN1 before adjusting the concentration to 51 ng/µL and using a 10-fold serial dilution (1 × 10−1 to 1 × 10−8 copies) to dilute it, and thereafter combining it with isolated genomic DNA from the samples of soil or peanut plants. This recombinant plasmid was applied as a reference for evaluating the LAMP assay-related detection limit.
Quantitative real-time fluorescence PCR
The SYBR® Premix ExTaq™ kit (TaKaRa) was used to perform the real-time PCR (RT-PCR) assay in triplicate. Thereafter, the CFX96™ system (Bio-Rad) was utilized as directed by the manufacturer to implement each PCR amplification process with the total volume set to 25 µL. Previous descriptions (Cao et al. 2015) of the parameters for thermocycline were used.
Feasibility test of artificially inoculated samples
The A. niger conidiospore suspension was diluted 10-fold to 1.49 × 100 spores/g soil sample, which was then utilized as the LAMP amplification template. Regarding the peanut plant samples that were artificially inoculated with 1 mL (107 spore suspension), plant samples were collected at 1 DPI (days post inoculation) and 6 DPI before typical root rot or crown rot symptoms appeared. The genomic DNA was extracted, with the final concentration adjusted to 67.6 ng/µL, and served as the quantification reference.
Field LAMP assay evaluation
To ascertain the feasibility of the RealAmp assay diagnostic tool in samples collected under field conditions, a total of thirty-six peanut plants and seventy soil samples were acquired from either peanut-established fields or those that had never before undergone peanut planting. The RealAmp performed with the ZY1-S1™ pathogen detection device was employed to detect the presence of A. niger.
The A. niger DNA quantification results from the ZY1-S1™ in the field samples were statistically assessed according to a previous workflow (Cao et al. 2017). Briefly, the corresponding threshold time value was calculated relative to the standard curve formula, ascertainable using the aforementioned RealAmp detection sensitivity study with intentionally infected samples. Verification of the field samples RealAmp was performed by the amplification and sequencing of the LAMP. Furthermore, the conidiospores that were recovered from the aforementioned materials were propagated, followed by the amplification of the resulting DNA templates for sequencing validation.
Results
LAMP primer screening
To identify an optimal target gene to be used in the LAMP assay, a total of 39 sets of LAMP primers were designed that targeted seven A. niger genes, including CbhA, CbhB, Tub, GOD, NRPS, EglA, and Tan. No positive amplification was observed for the LAMP primer sets CbhA II-1, CbhB I-4, CbhB I-8, CbhB I-24, CbhB I-35, CbhB II-1, CbhB II-6, CbhB II-13, CbhB II-23, Tub-1, Nrps-1, Nrps-3, EglA-6, EglA-16, Tan-20, Tan-25, Tan-29, Tan-35, Tan-45, GOD-36, and GOD-86 (Table S1), indicating that they were not specific to the Aspergillus niger DNA. On the other hand, pseudo-positive amplification was observed for the primer sets CbhA I-5, CbhA I-11, CbhA I-16, CbhA I-25, CbhA II-10, CbhA II-14, CbhA II-28, CbhB I-13, CbhB II-16, Tub-3, EglA-10, EglA-13, GOD-50, and GOD-55 (Table S1). Thus, these primers could not be used to reproducibly or efficiently detect A. niger (Fig. S1). The primer sets CbhA I-37, CbhA II-7, GOD-91, and EglA-20 exhibited high amplification efficiency, good reproducibility, no false positives, and consistent melting curves. A. niger DNA was used to amplify the matching target segment, according to the LAMP amplification curve (Fig. 1).
Screening and selection of primers for the Aspergillus niger LAMP detection assay. The LAMP fluorescence signal (RFU) (a) vs. the Cq [threshold time (min)] amplification curve graph (b) was plotted automatically by the Bio-Rad CFX96 Real-Time PCR system. Four sets of primers targeting each of the three A. niger genes (CbhA, GOD, and EglA) were designed for the primer optimization experiment. NC indicates the reaction using negative control templates
Based on the above data, CbhA I-37, CbhA II-7, GOD-91, and EglA-20 were selected as suitable candidates for the LAMP test targeting A. niger. In addition, the GOD-91 primer set had the highest repeatability and the lowest Cq value (6.94 min reaction threshold time), making it the ideal choice for the LAMP assay, according to subsequent analysis (Fig. 1). To better optimize the A. niger LAMP test, GOD-91 was selected for this work. Table S2 lists the target sequences of the last set of primers utilized in the LAMP procedure.
LAMP system optimization
The preceding description of our screening of potential LAMP primers is drawn from our previous study. (Cao et al. 2015). We adjusted three LAMP reaction mixture parameters: the concentration ratio of the inner primer pair FIB/BIP to the outer primer pair F3/B3, the quantity of Bst DNA polymerase, and the content of Mg2+ to determine if we could enhance the LAMP reaction system for A. niger (Table 2). As shown in the univariate plot, the 8 U Bst DNA polymerase had the lowest reaction time and good amplification efficiency when the other parameters were kept constant [concentration of Mg2+ (8 mM), FIP/BIP (1.6 µM), and F3/b3 (0.2 µM)]. Regarding the Mg2+ concentration optimization, changing the Mg2+ content to 6 mM or 7 mM caused a remarkable rise in the amplification performance. On the other hand, amplification efficiency decreased when the MgSO4 concentration was 8 mM and 9 mM. Regarding the inner and outer primers’ concentration ratio, the amplification efficiency was similar at the ratio of 8:1 and 10:1, which was significantly higher than at 4:1 and 6:1 primer ratios (Fig. S2).
A subsequent assessment of the LAMP test was implemented by applying 16 parameter combinations in an orthogonal experimental method, building on the findings of the single-parameter trials (Table S3). The best settings for a LAMP system for detecting A. niger were assessed to be a combination of A4B4C1, taking into account the results of the single-parameter tests that were conducted before and the orthogonal experimental method (Table 1). Specifically, in this combination, the amount of Bst DNA polymerase was 8 U, the ratio of inner and outer primers was 10:1, and the Mg2+ concentration was 6 mM (Fig. 2). The optimized 25 µL mixed reaction system included 2.5 µL 10 × Isothermal Amplification buffer (NEB, Ipswich, MA, USA), 0.2 µM SYTO-9 fluorescent dye (Invitrogen, Carlsbad, CA, USA), 1.4 mM dNTP Mix, 2 µL template DNA, 6 mM MgSO4, 0.8 µM each of loop primers LF3 and LB3, 0.2 µM each of F3 and B3, 1.6 µM each of FIP and BIP, and 8 U of Bst 2.0 WarmStart DNA polymerase (NEB).
Reaction results based on different combinations of reaction components based on orthogonal testing. a Reactions of 16 combinations monitored by the amplification curves. b Corresponding melt curve peaks of the above reactions. 1–16 represent each combination assessed in the orthogonal test shown in Table S3. NC indicates reactions that share the same conditions with each combination except for the substitution with a negative control template, COMB, combination
LAMP reaction system specificity
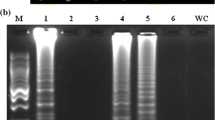
Five other plant fungal pathogens were tested using the same system to examine the A. niger-specificity of the GOD-91 LAMP primer set and the improved LAMP reaction. Reaction curve analysis revealed that GOD-91 primers selectively amplified A. niger target DNA sequences but not those of the other plant pathogens tested (Fig. 3a). A change in color from orange to green was visually observed in the LAMP product upon the identification of A. niger and positive control utilizing SYBR Green I. In contrast, it was observed that the coloration stayed orange for both the negative control as well as the other fungi examined (Fig. 3b). This was consistent with the agarose gel electrophoresis analysis of the LAMP assay amplicon, further validating the LAMP assay specificity in detecting A. niger (Fig. S3).
Specificity evaluation of the RealAmp assay for the detection of Aspergillus niger. a The RealAmp fluorescence (RFU) vs. the Cq [threshold time (min)] amplification curve graph was plotted automatically by the Bio-Rad CFX96 Real-Time PCR system. b Visual inspection of the RealAmp amplification products using SYBR Green I. The original orange color of SYBR green changed to green in the reaction mixture, indicating a positive result. The templates consisted of genomic DNA isolated from the following controls and pathogens: PC, positive control (Staphylococcus aureus); NC, negative control (Salmonella choleraesusi); A. niger, Aspergillus niger pAN01 plasmid DNA; 1, A. oryzae 2, A. terreus 3, Fusarium avenaceum 4, F. graminearum 5, Pythium aristosporum
LAMP assay sensitivity
For the sensitivity test, a 10-fold gradient dilution of pAN1 plasmid DNA was utilized, and it was combined with either DNA derived from soil or peanut genomic DNA. The preliminary experiment showed that amplification of LAMP products with exponential growth occurred within the range from 5.5 × 10−1 ng/µL to 5.5 × 10−6 ng/µL of plasmid DNA (Fig. 4a). The A. niger detection limit utilizing RealAmp was further determined in an experiment that used the pEasy-Pep plasmid serial dilutions between 0.55 ng/µL and 5.5 × 10−8 ng/µL. When combined with total DNA taken from soil or peanut plants, the RealAmp test could identify amounts of plasmid DNA as low as around 5.5 fg/µL, according to the amplification findings (Fig. S4), which agrees with the data from the preliminary experiment. Thereafter, a standard curve was constructed based on the association of the Cq value with the concentration of the initial plasmid DNA template, and an R2 > 0.99 indicated that the reaction met the linear regression requirements for quantification. (Fig. 4a).
Sensitivity evaluation of the Aspergillus niger RealAmp assay. a Sensitivity of the RealAmp assay conducted on the Bio-Rad CFX96 Real-Time PCR system. The RealAmp fluorescence (RFU) vs. the Cq [threshold time (min)] amplification curve graph and standard curve in which the threshold time was plotted against the amount of the initial template DNA on a logarithmic scale were plotted automatically by the CFX96 Real-Time PCR system. b Sensitivity test of real-time PCR for U. maydis detection. The real-time fluorescence units were plotted against the initial concentration of plasmid DNA ranging from 5.5 × 10−1 ng/µL to 5.5 × 10−7 ng/µL by the CFX96 Real-Time PCR system. The standard curve was generated using known Log10-transformed concentrations of a 10-fold serial dilution of the pEasy-Pep plasmid DNA and the corresponding threshold cycle (Ct) values
An RT-PCR experiment was conducted using serially diluted DNA template samples to contrast the sensitivity of the RealAmp detection technique with the traditional RT-PCR. The PCR product amplification was detected, and, a regression analysis was performed, with Cq plotted against initial template concentrations, demonstrating that the resulting standard curve was linear throughout a range of concentrations of eight magnitudes, from 10−1 to 10−8. Based on the amplification results, the RT-PCR detection limit was estimated at 5.5 × 10−6 ng/µL plasmid DNA (Fig. 4b).
LAMP assay assessment in artificially inoculated samples
To assess the efficacy of the LAMP test in detecting pathogens introduced artificially, soil and peanut plant samples were inoculated artificially. Genomic DNA was isolated from peanut seedlings pre-inoculated with A. niger at 1 DPI and 6 DPI before typical crown rot symptoms appeared for peanut samples infected artificially. The data obtained indicated that concentration levels as low as 6.76 × 10−2 ng/µL A. niger DNA could be detected by the RealAmp at 1 DPI (Fig. 5a). The RealAmp test did not identify any A. niger DNA in control plant samples without inoculation. In addition, the RealAmp assay was capable of detecting A. niger with high selectivity for levels as low as 1.49 × 102 spores/g of soil samples inoculated artificially (Fig. 5b).
Quantitative detection of artificially inoculated peanut plant and soil samples using the RealAmp assay. a Quantitative detection of A. niger in peanut seedlings inoculated with A. niger at 1 DPI (day post inoculation). Serially diluted template DNA (ranging from 100 to 10−3) obtained at1 DPI was used to test the sensitivity of RealAmp in detecting A. niger at the early stage of infection in the artificially inoculated peanut plants. b Detection of Aspergillus niger in the serial 10-fold dilutions ranging from 1.49 × 106 to 1.49 × 102 conidiospores per gram of artificially inoculated soil using the RealAmp assay. NC, negative control
RealAmp detection assessment in field samples
Since the RealAmp test is capable of detecting A. niger in samples that have been artificially infected, further assessment of the assay for the quantitative detection of A. niger in field samples was carried out. Firstly, samples were collected from diverse peanut varieties before the manifestation of symptoms. To ascertain whether or not peanuts are resistant to disease, the plants were planted in a field that was already infested with A. niger. The ZYD-S1™ detection system data showed that A. niger could be detected in samples from the peanut varieties Zhenghua 25, Zhuhua 7, and Yuhua 143, while no A. niger detection was observed in Luohua 16. These findings supported those of the CFX96™ RT-PCR assay (Fig. 6). In addition, 70 soil and 36 peanut plant samples were checked to determine whether A. niger was present by the RealAmp assay using the ZYD-S1™ system. Based on the results, 82 samples were tested positive, while 24 samples were tested negative (Tables S4 and S5).
Detection of Aspergillus niger in plants from various peanut varieties using the RealAmp assay for disease resistance testing. H1–H4, DNA isolated from seedlings of peanut varieties Zhenghua 25, Zhuhua 7, Yuhua 143, and Luohua 16 that were infected with A. niger; NC1–NC4, the genomic DNA isolated from healthy peanut seedlings of the four varieties
Discussion
Root rot and crown rot of peanuts caused by A. niger are considered to be economically important diseases throughout most areas where peanut is planted (Dufault and Brenneman 2023). To improve disease management as well as resistance breeding methods, it is extremely necessary to have a method that is both sensitive and effective for detecting A. niger either before or at a very early stage of pathogen infection (Buja et al. 2021). Microscopy has been used as a diagnostic tool to analyze the A. niger infection incidence in peanut plants (Matloob and Juber 2014; Suzui and Makino 1980; Nelson and Margaret 2018). Nonetheless, when analyzing asymptomatic plants or persons in the early stages of infection, this approach presents technological challenges and suffers from limited sensitivity (Patel et al. 2023). As previously described, PCR-based methods have emerged as a reliable and sensitive means of detecting A. niger (Sugita et al. 2004; Xu et al. 2015). Still, when applied in field scenarios, this approach can be tedious and cumbersome (von Hertwig et al. 2018). Our work here details the creation of a species-specific primer-based RealAmp approach for the detection of A. niger that is sensitive, rapid, and quantitative. A previously described LAMP assay for rapidly detecting Aspergillus species that produce fumonisin was highly valuable for performing direct monitoring “on-site” in the maize production and processing chain (Ferrara et al. 2020). To our knowledge, an effective tool for investigating in field conditions the latent infection and the initial phases of disease caused by A. niger is presently nonexistent, despite this pathogenic fungus being the causal agent of many plant disease epidemics. The LAMP system described here provides an alternative and effective solution for the early and accurate detection of A. niger. An “electricity-free” assay might be implemented by performing the LAMP reaction using an enhanced tube scanner. In addition, the test was designed using an appropriate methodology for crude DNA isolation “in-field” and a colorimetric approach for straightforward quantitative or naked-eye result evaluation, thus providing programs for the control of agricultural diseases, including those affecting peanuts, with a reliable and easy-to-use tool.
The A. niger functional genes (Egla, GOD, Tub, NRPS, Tan, CbhA, and CbhB) served as primer design targets when screening optimal LAMP primers. Research has linked these genes to specific phases in the A. niger infection life cycle. Therefore, finding the targeted gene could offer evidence of A. niger successfully infecting peanut plants. The LAMP technique was additionally developed using GOD-91, one of 39 sets of candidate primers since it showed the best amplification performance and stability (Figs. 1, S1).
Optimizing the reaction might be achieved by adjusting various parameters of the LAMP protocol. Two important factors for optimizing LAMP are the inner/outer primer ratio and the concentration of Bst DNA polymerase utilized in the reaction (Notomi et al. 2000; Kaymaz and Elitas 2021). In this work, the amplification improved dramatically upon increasing the amount of Bst DNA polymerase from 2 U to 4 U. Nevertheless, compared to the increase from 2 U to 4 U of Bst DNA polymerase, the increment from 4 U to 8 U resulted in a much smaller boost in amplification performance (Fig. S3). Optimum LAMP test findings were achieved using 8 U of Bst DNA polymerase in conjunction with the ideal inner/outer primer ratio and MgSO4 content (Fig. 2; Table 1). The efficiency improvement reached a plateau upon setting the Bst enzyme concentration to 8 U, which agrees with our previous findings (Cao et al. 2017). Based on the initial evaluation of LAMP primer ratios, the outer primer should be at 1/4–1/10 of the inner primer concentration. (Notomi et al. 2000; Nie 2005; Lamas et al. 2023). Herein, the ideal ratio (10:1) of the FIP/BIP to F3/B3 primers effectively resulted in a high-efficiency amplification without false positive results (Figs. 2, S3, and Table 1). It is expected that Mg2+ ions would affect the specificity of PCR and the activity of DNA polymerase (Saiki et al. 1988). Consequently, nonspecific amplification could cause false positives at very high Mg2+ concentrations. Previous reports indicated that a minimum of 4 mM Mg2+ is necessary for the occurrence of a noticeable reaction, with concentrations of 6 to 8 mM resulting in an optimum LAMP reaction (Nie 2005; Kaymaz and Elitas 2021). Single factor assessment indicated that the LAMP reaction with 6 mM or 7 mM Mg2+ exhibited higher amplification efficiency (Fig. S3). Further orthogonal experiments performed in this study indicated that combinations 1 and 3 (which employed 6 mM Mg2+) resulted in relatively high efficiency even without the optimization of primer ratios and Bst DNA polymerase (Fig. 2; Tables 1, S3). Similar findings have been reported in the LAMP test that used SYBR Green I to detect the sugarcane smut pathogen S. scitamineum, with 6.00 mM being the optimal Mg2+ ion concentration (Su et al. 2016). However, these findings differ from our previously described method (Cao et al. 2017) and other LAMP studies (Nie 2005; Tomita et al. 2008). The templates and primers utilized in these reactions, in addition to the varying concentrations of dNTPs, might account for this discrepancy.
On most occasions, single-factor tests are performed to optimize the LAMP reaction, which, with all other variables held constant, involves adjusting several reaction conditions to find the best concentration ranges of a particular reaction component (Becherer et al. 2021). Unfortunately, it is difficult to obtain ideal experimental data since it is not possible to evaluate possible interactions among these components (Su et al. 2016). To address this issue, single-factor tests were implemented in conjunction with an orthogonal experimental method, allowing the proper identification and determination of the interactive effects between the various reaction protocols to establish the detection optimization of the LAMP test (Cao et al. 2017; Su et al. 2016; Shirshikov and Bespyatykh 2022). The findings of the single-factor trials served as the basis for the design of the orthogonal arrangement in this research, which was then further verified by the orthogonal experiment (Fig. 2; Tables 1, S3). In addition to the utilization of the amplification data of the orthogonal experiment (Fig. 2) following the description in previous reports (Cao et al. 2017; Su et al. 2016), the k-statistic was innovatively employed to determine the optimal conditions (Table 1). Hence, an optimized LAMP system suitable for detecting A. niger was successfully developed by performing a single-factor experiment followed by enhanced evaluation through an improved orthogonal experimental design.
The RealAmp assay that has been developed is capable of detecting A. niger at concentrations as low as 1.49 × 102 spores/g of soil sample (Fig. 5b), which shows a reduction in size by an order of magnitude when compared to a LAMP test previously documented for detecting Aspergillus species on maize kernels (Ferrara et al. 2020). Potentially inhibiting compounds in the soil that impede the efficacy of the reaction might account for the reduced detection sensitivity of RealAmp when combined with DNA derived from soil samples. A. niger infection does not result in the manifestation of outward or distinctive symptoms of peanut root rot or crown rot, unless under certain conditions. Thus, the precise identification of the RealAmp detection threshold based on samples inoculated artificially, such as the conidiospore-inoculated soil and conidia-inoculated peanut plants, may provide key guidance for future research on the interactions of plants and pathogens, management of diseases, and resistance breeding. The portable fluorescent reader ZYD-S1™, fitted with a battery pack is utilized for plant pathogen detection (Cao et al. 2017). The ZYD-S1™ represents a significant progression toward an “electricity-free” LAMP technique by utilizing a mobile DNA workstation to analyze field samples, including template preparation, amplification, and quantitative product detection. (Fig. 6, Table S4). The RealAmp assay that was developed in this study might serve as the cornerstone for integrated disease management practices and direct peanut cultivation to prevent the spread of A. niger. Incorporating these findings into studies of plant-pathogen interplay and disease-resistance breeding could potentially prove to be beneficial.
Conclusions
In this study, an effective technique was successfully developed for detecting and monitoring A. niger in the field. Seven effector genes of A. niger were evaluated for LAMP primer selection. An optimized RealAmp assay was developed by assessing different concentrations of Bst DNA polymerase and Mg2+ and the reaction system’s inner/outer primer ratio with an experiment using a single factor and an orthogonal design configuration. Additionally, the assay specificity was confirmed by the absence of amplification when other pathogenic fungi were tested. Notably, the RealAmp assay detection limit was approximately 5.5 × 10−6 ng/µL plasmid DNA (combined with environmental samples), or 1.49 × 102 conidiospores/g of artificially inoculated soil, and 6.76 × 10−2 ng/µL A. niger genomic DNA in the naturally infected peanut plants. Additionally, an enhanced tube scanner might be used to perform the assay, allowing for a “one-step” process including template creation and quantitative detection. The new assay has the potential to be applied effectively in the field as an efficient, rapid, and simple method for detecting A. niger.
Data availability
All data generated or analyzed during this study are included in this article.
References
Annamalai N, Rajeswari MV, Balasubramanian T (2016) Endo-1,4-β-glucanases: role, applications and recent developments. In: Gupta V (ed) Microbial enzymes in bioconversions of biomass. Biofuel and Biorefinery technologies, vol. 3. Springer, Cham. Switzerland, pp 37–45. https://doi.org/10.1007/978-3-319-43679-1_3
Becherer L, Borst N, Bakheit M, Frischmann S, Zengerle R, Stetten F (2021) Loop-mediated isothermal amplification (LAMP)–review and classification of methods for sequence-specific detection. Anal Methods 12:717–746. https://doi.org/10.1039/C9AY02246E
Buja I, Sabella E, Monteduro AG, Chiriacò MS, De Bellis L, Luvisi A, Maruccio G (2021) Advances in plant disease detection and monitoring: from traditional assays to in-field diagnostics. Sens (Basel) 21(6):2129. https://doi.org/10.3390/s21062129
Cao Y, Li Y, Li J, Wang L, Cheng Z, Wang H, Fan Z, Li H (2015) Rapid and quantitative detection of Pythium inflatum by real-time fluorescence loop-mediated isothermal amplification assay. Eur J Plant Pathol 144:83–95. https://doi.org/10.1007/s10658-015-0752-2
Cao Y, Wang L, Duan L, Li J, Ma J, Xie S, Shi L, Li H (2017) Development of a real-time fluorescence loop-mediated isothermal amplification assay for rapid and quantitative detection of Ustilago maydis. Sci Rep 7:13394. https://doi.org/10.1038/s41598-017-13881-4
Dufault NS, Brenneman T (2023) Disease of Peanut. In: Hollier CA, Padgett GB, Draper MA (eds) Disease of filed crops, second edn. APS Press, St. Paul, MN, USA, pp 161–174
Ferrara M, Logrieco AF, Moretti A, Susca A (2020) A loop-mediated isothermal amplification (LAMP) assay for rapid detection of fumonisin producing Aspergillus species. Int J Food Microbiol 90:103469. https://doi.org/10.1016/j.fm.2020.103469
Jiang L, Gu R, Li X, Mu D (2021) Simple and rapid detection Aspergillus fumigatus by loop-mediated isothermal amplification coupled with lateral flow biosensor assay. J Pure Appl Microbiol 131:2351–2360. https://doi.org/10.1111/jam.15092
Kaymaz SV, Elitas M (2021) Optimization of loop-mediated isothermal amplification (LAMP) reaction mixture for biosensor applications. MethodsX 8:101282. https://doi.org/10.1016/j.mex.2021.101282
Kenji A, Ryu S, Hiroshi N (2014) Purification and some properties of yeast tannase. Agr Biol Chem 40:79–85. https://doi.org/10.1080/00021369.1976.10861997
Kim WB, Park C, Cho SY, Chun HS, Lee DG (2020) Development of multiplex real-time PCR for rapid identification and quantitative analysis of Aspergillus species. PLoS ONE 15:e0229561. https://doi.org/10.1371/journal.pone.0229561
Kokalis-Burelle N (1997) Aspergillus crown rot. In: Kokalis-Burelle N, Porter DM, Rodríguez-Kábana R, Smith DH, Subrrhmanynm P (eds) Compendium of peanut disease: a guide to the identification, diagnosis, and control of peanut diseases and disorders, second edn. APS Press, St. Paul, MN, USA, pp 7–8
Lamas A, Azinheiro S, Roumani F, Prado M, Garrido-Maestu A (2023) Evaluation of the effect of outer primer structure, and inner primer linker sequences, in the performance of loop-mediated isothermal amplification. Talanta 260:124642. https://doi.org/10.1016/j.talanta.2023.124642
Lucchi NW, Demas A, Narayanan J, Sumari D, Kabanywanyi A, Kachur SP, Barnwell JW, Udhayakumar V (2010) Real-time fluorescence loop-mediated isothermal amplification for the diagnosis of malaria. PLoS ONE 5:13733. https://doi.org/10.1371/journal.pone.0013733
Matloob AAH, Juber KS (2014) First report of peanut crown rot disease caused by Aspergillus niger in Iraq and its biological control. J Exp Biol Agr Sci 2:171–177
Ming DX (2005) Orthogonal design. In: Ming DX (ed) Field test and statistical analysis. Science, Beijing, China, p 294
Nagamine K, Watanabe K, Ohtsuka K, Hase T, Notomi T (2001) Loop-mediated isothermal amplification reaction using a nondenatured template. Clin Chem 47:1742–1743. https://doi.org/10.1093/clinchem/47.9.1742
Nagamine K, Hase T, Notomi T (2002) Accelerated reaction by loop-mediated isothermal amplification using loop primers. Mol Cell Probe 16:223–229. https://doi.org/10.1006/mcpr.2002.0415
Nelson M, Margaret M (2018) Occurrence of aflatoxigenic Aspergillus species in peanut varieties in Busia and Kisii central districts, Kenya. Open J Med Microbiol 8:98–108. https://doi.org/10.4236/ojmm.2018.84009
Nie X (2005) Reverse transcription loop-mediated isothermal amplification of DNA for detection of Potato virus Y. Plant Dis 89:605–610. https://doi.org/10.1094/PD-89-0605
Njiru ZK, Yeboah-Manu D, Stinear TP, Fyfe JA (2012) Rapid and sensitive detection of Mycobacterium ulcerans by use of a loop-mediated isothermal amplification test. J Clin Microbiol 50:1737–1741. https://doi.org/10.1128/JCM.06460-11
Notomi T, Okayama H, Masubuchi H, Yonekawa T, Watanabe K, Amino N, Hase T (2000) Loop-mediated isothermal amplification of DNA. Nucleic Acids Res 28:e63. https://doi.org/10.1093/nar/28.12.e63
Patel R, Mitra B, Vinchurkar M, Adami A, Patkar R, Giacomozzi F, Lorenzelli L, Baghini MS (2023) Plant pathogenicity and associated/related detection systems. A review. Talanta 251:123808. https://doi.org/10.1016/j.talanta.2022.123808
Peng J, Zhan Y, Zeng F, Long H, Pei Y, Guo J (2013) Development of a real-time fluorescence loop-mediated isothermal amplification assay for rapid and quantitative detection of Fusarium oxysporum f. sp. niveum in soil. FEMS Microbiol Lett 349:127–134. https://doi.org/10.1111/1574-6968.12305
Qin HB, Li MF, Yang HJ (2017) Heteroexpression of cellobiohydrolase a gene of Aspergillus niger in Pichia pastoris. Biotechnol 27:330–336. https://doi.org/10.16519/j.cnki.1004-311x.2017.04.0054
Richardson MD, Stubbins JM, Warnock DW (1982) Rapid enzyme-linked immunosorbent assay (ELISA) for Aspergillus fumigatus antibodies. J Clin Pathol 35:1134–1137. https://doi.org/10.1136/jcp.35.10.1134
Ruark SJ, Shew BB (2010) Evaluation of microbial, botanical, and organic treatments for control of peanut seedling diseases. Plant Dis 94(4):445–454. https://doi.org/10.1094/PDIS-94-4-0445
Saiki R, Gelfand D, Stoffel S, Scharf S, Higuchi R, Horn G, Mullis K, Erlich H (1988) Primer directed enzymatic amplification of DNA with a thermostable DNA polymerase. Science 239:487–491. https://doi.org/10.1126/science.2448875
Shao Y, Zhang YH, Zhang F, Yang QM, Weng HF, Xiao Q, Xiao AF (2020) Thermostable tannase from Aspergillus niger and its application in the enzymatic extraction of green tea. Molecules (Basel) 25:952. https://doi.org/10.3390/molecules25040952
Shirshikov FV, Bespyatykh JA (2022) Loop-mediated isothermal amplification: from theory to practice. Russ J Bioorg Chem 48(6):1159–1174. https://doi.org/10.1134/S106816202206022X
Silva DM, Batista LR, Rezende EF, Fungaro MH, Sartori D, Alves E (2011) Identification of fungi of the genus Aspergillus section nigri using polyphasic taxonomy. Braz J Microbiol 42:761–773. https://doi.org/10.1590/S1517-83822011000200044
Su YC, Yang YT, Peng Q, Zhou DG, Chen Y, Wang ZQ, Xu LP, Que YX (2016) Development and application of a rapid and visual loop-mediated isothermal amplification for the detection of Sporisorium scitamineum in sugarcane. Sci Rep 6:23994. https://doi.org/10.1038/srep23994
Sugita C, Makimura K, Uchida K, Yamaguchi H, Nagai A (2004) PCR identification system for the genus Aspergillus and three major pathogenic species: Aspergillus fumigatus, Aspergillus flavus and Aspergillus niger. Med Mycol 42(5):433–437. https://doi.org/10.1080/13693780310001656786
Suzui T, Makino T (1980) Occurrence of Aspergillus crown rot of peanut caused by Aspergillus niger Van Tieghem. Ann Phytopath Soc 46:46–48. https://doi.org/10.3186/JJPHYTOPATH.46.46
Tang Q, Tian S, Yu N, Zhang X, Jia X, Zhai H, Sun Q, Han L (2016) Development and evaluation of a loop-mediated isothermal amplification method for rapid detection of Aspergillus fumigatus. J Clin Microbiol 54:950–955. https://doi.org/10.1128/JCM.01751-15
Tomita N, Mori Y, Kanda H, Notomi T (2008) Loop-mediated isothermal amplification (LAMP) of gene sequences and simple visual detection of products. Nat Protoc 3:877–882. https://doi.org/10.1038/nprot.2008.57
Trabasso P, Matsuzawa T, Arai T, Hagiwara D, Mikami Y, Moretti ML, Watanabe A (2021) Development and validation of LAMP primer sets for rapid identification of Aspergillus fumigatus carrying the cyp51A TR46 azole resistance gene. Sci Rep 11:17087. https://doi.org/10.1038/s41598-021-96651-7
Tsai YC, Brenneman TB (2018) Early-season fungicide programs for peanut disease management. Plant Health Prog 19:103–106. https://doi.org/10.1094/PHP-05-17-0029-RS
Tsugunori N, Hiroto O, Harumi M, Toshihiro Y, Keiko W, Nobuyuki A, Tetsu H (2000) Loop-mediated isothermal amplification of DNA. Nucleic Acids Res e63. https://doi.org/10.1093/nar/28.12.e6
Volossiouk T, Robb EJ, Nazar RN (1995) Direct DNA extraction for PCR-mediated assays of soil organisms. Appl Environ Microb 61:3972–3976. https://doi.org/10.1128/aem.61.11.3972-3976.1995
von Hertwig AM, Sant’Ana AS, Sartori D, da Silva JJ, Nascimento MS, Iamanaka BT, Pelegrinelli Fungaro MH, Taniwaki MH (2018) Real-time PCR-based method for rapid detection of Aspergillus niger and Aspergillus welwitschiae isolated from coffee. J Microbiol Meth 148:87–92. https://doi.org/10.1016/j.mimet.2018.03.010
Wang PH, Chang CW (2003) Detection of the low-germination-rate resting oospores of Pythium miriotylum from soil by PCR. Lett Appl Microbiol 36:157–161. https://doi.org/10.1046/j.1472-765x.2003.01287.x
Xu ML, Melchinger AE, Lübberstedt T (1999) Species-specific detection of the maize pathogens Sporisorium reiliana and Ustilago maydis by dot blot hybridization and PCR-based assays. Plant Dis 83:390–395. https://doi.org/10.1094/PDIS.1999.83.4.390
Xu ML, Yang JG, Wu JX, Chi YC, Xie LH (2015) First report of Aspergillus niger causing root rot of peanut in China. Plant Dis 99:284. https://doi.org/10.1094/PDIS-05-14-0530-PDN
Yu XW, Li YQ, Zhou SM, Zheng YY (2007) Synthesis of propyl gallate by mycelium-bound tannase from Aspergillus niger in organic solvent. World J Microb Biot 23:1091–1098. https://doi.org/10.1007/s11274-006-9338-7
Zhao K, Li Z, Ke Y, Ren R, Cao Z, Li Z, Wang K, Wang X, Wang J, Ma Q, Cao D, Zhao K, Li Y, Hu S, Qiu D, Gong F, Ma X, Zhang X, Fan G, Liang Z, Yin D (2024) Dynamic N6 -methyladenosine RNA modification regulates peanut resistance to bacterial wilt. New Phytol 242(1):231–246. https://doi.org/10.1111/nph.19568
Zhou YF, Zhang XE, Liu H, Zhang CG, Cass AE (2001) Cloning and expression of Aspergillus niger glucose oxidase gene in methylotrophic yeast. Chin J Biotechnol 4:400–405. https://doi.org/10.13345/j.cjb.2001.04.011
Acknowledgements
This study was supported by grants from the National Key Research and Development Program of China (2022YFD1200400), the Joint Funds of Natural Science Foundation of Henan Province (222301420026), China Agriculture Research System of MOF and MARA (CARS-13), the Henan Province Agriculture Research System (S2012-5), Henan provincial scientific and technological research project (242102110255). We thank Professor Xianli Gao (Jiansu University, Wuxi, China) for providing strains of Aspergillus spp.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethical approval
The authors confirm that there are no ethical issues in publication of the manuscript.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Dai, X., Cao, Y., Yu, M. et al. Rapid and quantitative detection of Aspergillus niger Van Tieghem using loop-mediated isothermal amplification assay. J Plant Pathol (2024). https://doi.org/10.1007/s42161-024-01674-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42161-024-01674-4