Abstract
Coronary computed tomographic angiography (CCTA) is an effective examination with high sensitivity to rule out obstructive coronary artery disease (CAD). It can provide anatomical information of coronary artery disease, such as the degree of stenosis, plaque characteristics, but has poor discriminatory power for hemodynamically significant lesions, which may lead to unnecessary referrals for invasive coronary angiography (ICA). Invasive fractional flow reserve (FFR) is generally considered as the gold standard for the functional evaluation of CAD. However, high cost and invasive characteristics of FFR limit its wide clinical application. Recently, non-invasive CT-derived fractional flow reserve (CT-FFR), a novel image post-processing technique combining the advantages of CCTA and FFR, allows us to obtain both anatomic and functional information of a coronary lesion. CT-FFR has been used in diagnosing and guiding clinical management of CAD patients. This review introduces the basic principles, key points of CT-FFR analysis, and current research progress of CT-FFR.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Coronary artery disease (CAD) is a serious cardiovascular disease, which is associated with poor clinical outcomes. As a non-invasive test, coronary computed tomography angiography (CCTA) has been considered as the preferred method of assessing CAD. CCTA has high sensitivity, negative predictive value (NPV), and high diagnostic value in low–mid-risk population. However, conventional CCTA cannot evaluate the effect of lumen stenosis on myocardial blood supply from a functional perspective, thus making it difficult to distinguish specific ischemic lesions. Invasive fractional flow reserve (FFR) has been established as the gold standard in evaluating functional significance of coronary stenosis, defined as the ratio between the maximum myocardial blood flow in the presence of an epicardial coronary stenosis and the maximum myocardial blood flow in the theoretical absence of the stenosis [1, 2]. Several clinical trials have validated that FFR can better predict clinical outcomes and guide clinical decision-making when compared with invasive coronary angiography (ICA) [3,4,5]. However, only 10–20% of revascularization procedures are performed according to FFR, because of its invasive and expensive nature [6]. Therefore, the development of non-invasive CCTA-derived fractional flow reserve (CT-FFR) has attracted widespread attention. Without additional scanning and drug administration, CT-FFR values of the entire coronary tree can be obtained through standardized routine CCTA images, providing both anatomical and functional information of coronary artery stenosis, enabling cardiac CT step into the new era of functional evaluation. This review introduces the basic principles, key points of CT-FFR analysis, and the current research progress of CT-FFR.
CT-FFR technology
The principle and workflow of CT-FFR
CT-FFR is an image post-processing technique for CCTA, which applies computational fluid dynamics (CFD) to conventional CCTA image data to simulate the physiological state of the coronary artery. In the mathematical model of blood, the blood is assumed as an incompressible Newtonian fluid, with a constant density and viscosity, and the blood flow can be calculated using a hydrodynamic basic control equation (Navier–Stokes equation). The following are the main principles of CT-FFR: (1) the coronary artery can provide enough blood to meet the consumption of the myocardium in resting state; (2) the resistance of microcirculation is inversely related to the coronary artery diameter, but not linearly proportional; and (3) the response of microcirculation to the maximum hyperemia of coronary artery is predictable [7,8,9,10]. Three elements are required: (1) describe the anatomic region of interest; (2) determine the “boundary condition” to define the physiological relationship between the variables at the boundary of the region of interest; and (3) understand the physical laws of fluid flow in the area. The blood flow of the coronary artery was obtained by the relationship between the volume of the myocardium and the blood flow. Then, the flow resistance of the coronary artery was obtained by the relationship model between the size of the blood vessel and the resistance, so as to calculate the microcirculation resistance of the coronary artery in the simulated maximal hyperemia state [7]. However, the large amount of computational requirements of the CFD model and the associated image segmentation process need supercomputers in core labs, preventing the technique from being applied to real-time on-site applications. Recently developed transluminal attenuation gradient (TAG)-based CT-FFR software used combined three-dimensional CFD principle and a machine learning (ML) algorithm to realize the on-site CFD-based CT-FFR application [11].
Researchers have also developed ML algorithms for the calculation of CT-FFR, which simplifies the calculation process of CT-FFR. ML-based CT-FFR technology determines the complex relationship between the anatomical structure of the coronary artery tree and its corresponding hemodynamics using a multi-layer neural network structure model and offline training. It uses a large database to simulate and train the model, and finally establishes a model that conforms to CFD rules and combines the known anatomical features of CCTA with FFR values [12].
The CT-FFR is usually measured at the 2–4 cm distal to the specific lesions. There are three essential steps for measuring CT-FFR values. In the case of CFD-based approaches, these comprise: (1) creation of a patient-special three-dimensional anatomic model of the coronary artery tree by conventional standardized CCTA image data. The underlying software can automatically extract blood vessel centerlines, and allow the operator to adjust center line and lumen contour; (2) determine the inlet and outlet boundary conditions to build a patient-specific hemodynamic model. The full automation software can be manually adjusted according to the needs, and then applies a CFD method to simulate the patient’s blood flow and vessel boundary state from the model. ML-based CT-FFR software does not need to determine boundary conditions. (3) combination of the physiological parameters and the CFD principle with anatomy model to calculate the coronary artery blood flow and blood pressure under maximum hyperemia [7, 12]. The CT-FFR value can be calculated at any position in the coronary artery tree in a single cardiac cycle; the results can be displayed by color-coded 3D images, where different colors code different ranges of the CT-FFR value.
CT-FFR analysis software
There are three kinds of CT-FFR analysis software. (1) 3D-CFD-based CT-FFR analysis software: HeartFlow, Redwood City, CA, USA is a commercial software approved by The Food and Drug Administration (FDA) of the United States, which is computationally intensive and needs to be run on supercomputers in a core laboratory. It takes 4–6 h to calculate CT-FFR values and requires professional personnel for data procession, which limits its application in clinical practice around the world [7]. Newly developed uCT-FFR software (United Imaging Healthcare, Shanghai, China) has realized the on-site CT-FFR application (Fig. 1). (2) A reduced-order model-based CT-FFR analysis software. It has a small amount of computation and requires less anatomical models, so it can work in real time on local workstations. The software has high diagnostic efficiency with user friendliness and short operation time [13]. (3) ML-based CT-FFR analysis software adopts multi-layer neural network structure and studies the complex relationship between the anatomical structure of coronary artery tree and hemodynamics based on the geometric features of CCTA, such as vessel diameter, degree of stenosis and vessel length. Using ML software can save time and simplify operation, which is conducive to clinical application (Fig. 1) [13]. Due to the patent limitation of HeartFlow, the last two CT-FFR software have not been routinely applied in clinical practice.
ML-based CT-FFR and uCT-FFR in detecting lesion-specific ischemia. a Curved planar reformation image showing the plaque in the proximal segment of right coronary artery with moderate stenosis. b Invasive FFR showing the value of 0.77. c, d ML-based CT-FFR and uCT-FFR showing CT-FFR values of 0.79 and 0.77, respectively
Current evidences
Diagnostic performance of CT-FFR
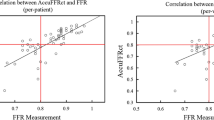
The first issue is to evaluate its diagnostic performance of CT-FFR. The DISCOVER-FLOW trial [14], DeFACTO study [15], and NXT trail [11] have successively proved that CT-FFR has a high diagnostic accuracy in identifying lesion-specific ischemia and good correlation with invasive FFR. Other scholars also investigated the diagnostic accuracy of ML-based CT-FFR as shown in Table 1 [16,17,18,19]. In a sub-study of the DISCOVER-FLOW trial, Min et al. [20] found that the diagnostic performance of CT-FFR was better than CCTA and remained stable among CCTA data with different image qualities. On a per-patient basis, the area under the receiver-operating characteristic curve (AUC) of CT-FFR was still higher than CCTA (p < 0.05) with an image quality score of Likert scale 0–3. In a large multicenter cohort, Coenen et al. compared the diagnostic performance of ML-based CT-FFR with CFD-based CT-FFR for the detection of hemodynamically significant stenosis [21]. They found that on a per-vessel basis, the diagnostic accuracy, sensitivity and specificity of ML-based CT-FFR were similar to CFD-based CT-FFR (78% vs. 78%, 81% vs. 82%, and 76% vs. 76%, respectively), without significant differences. The ML-based CT-FFR value shows an excellent correlation with CFD-based CT-FFR (r = 0.99). They also found that the specificity of ML-based CT-FFR was significantly improved from 38% of CCTA to 81% of ML-based CT-FFR. On a per-patient level, the addition of ML-based CT-FFR improved the specificity of CCTA from 11 to 76%. These results showed that the diagnostic performance of ML-based CT-FFR was equal to CFD-based CT-FFR. In addition, a Chinese multi-center CT-FFR study showed that uCT-FFR demonstrated a good correlation with invasive FFR (r = 0.69, p = 0.01). On a per-vessel level, the sensitivity and specificity of uCT-FFR were 89% and 91%, respectively. This was significantly better than CCTA (0.91 vs. 0.34, p < 0.001) and ICA (0.91 vs. 0.37, p < 0.001), especially in intermediate lesions (Fig. 1) [13]. Figures 1 and 2 provide some representative cases, showing the role of CT-FFR in detecting lesion-specific ischemia.
CT-FFR in detecting lesion-specific ischemia. a–c A 64-year-old male with a noncalcified plaque in the distal segment of the right coronary artery (RCA) (a). Distal CT-FFR (b) and invasive FFR (c) of the lesion were 0.94 and 0.95, respectively. d–f A 51-year-old male with multiple calcified and mixed plaques in the proximal-middle segment of the left anterior descending artery (LAD) (d) Distal CT-FFR (e) and invasive FFR (f) of the lesion were 0.73 and 0.75, respectively
Comparison with other functional imaging modalities
Some recent clinical trials have compared the diagnostic accuracy of CT-FFR with other functional technologies, such as dynamic myocardial CT perfusion (CTP), cardiac magnetic resonance (CMR) stress perfusion imaging, and single-photon emission computed tomography (SPECT). In a prospective single-center study, Pontone et al. [22] compared the diagnostic accuracy of CTP plus CCTA with CT-FFR plus CCTA and found that the sensitivity, specificity, and AUC of CT-FFR plus CCTA were 86%, 75% and 0.878, respectively. The sensitivity, specificity, and AUC of dynamic CTP plus CCTA were 73%, 86%, and 0.876, respectively. There was no significant difference between CT-FFR plus CCTA and CTP plus CCTA (p = 0.4). However, the AUC of CCTA + CT-FFR + CTP was higher than that of CCTA + CTP (0.919 vs. 0.876; p = 0.016). This means that combining CCTA with CTP and CT-FFR can improve the diagnostic performance for the detection of lesion-specific ischemia. However, Li et al. [23] found that on a per-vessel analysis, ML-based CT-FFR was inferior to myocardial blood flow (MBF) derived from CTP for detecting lesion-specific ischemia with lower specificity and AUC (63% vs. 91%, p < 0.001, and 0.79 vs. 0.96, p < 0.001, respectively). However, CTP delivered higher radiation doses to the patients compared to CT-FFR. What must be further investigated is which modality is more appropriate. Many researchers have compared the role of CMR stress perfusion imaging with CT-FFR for detecting lesion-specific ischemia. In a recent study, Driessen et al. [24] compared the diagnostic performance of CT-FFR with CCTA, SPECT and positron emission computed tomography (PET). They found that CT-FFR had the highest sensitivity for vessel-specific ischemia among these methods (CT-FFR: 90% vs. CCTA: 68%, p < 0.001, CT-FFR: 90% vs. SPECT: 42%, p < 0.001, CT-FFR: 90% vs. PET: 81%, p = 0.03); while at a per-patient level, the specificity of CT-FFR was superior to SPECT and PET (CT-FFR: 65% vs. SPECT: 93%, p < 0.001, CT-FFR: 65 vs. PET: 87%, p = 0.001). Such comparative studies suggested that in future clinical work, we can learn from each other and take advantage of multiple tests to improve the diagnostic accuracy for the detection of lesion-specific ischemia. However, these results were taken from small sample studies; so to strengthen these findings, this must be further verified in large multicenter studies.
Factors affecting diagnostic accuracy of CT-FFR
The diagnostic performance of CT-FFR is affected not only by the algorithm itself, but also by the severity of calcification and CCTA image quality. Coronary calcified plaques can cause halo artifacts and interfere with the evaluation of coronary artery stenosis by CCTA (Fig. 2). Thus, many investigators have focused on the effect of coronary calcification on CT-FFR diagnostic performance. In the NXT subgroup study, Norgaard et al. [25] found that regardless of Agatston score (AS) > 400 or not, the accuracy and specificity of CT-FFR in diagnosing ischemia-specific lesions was higher than that of CCTA alone on a per-patient level (p < 0.001) and per-vessels level (p < 0.05) with similar sensitivity. Another study revealed that CT-FFR improved the diagnostic ability to identify specific ischemia in patients with mild and moderate calcium (calcium score ≤ 1000), and CT-FFR showed a good correlation with FFR (r = 0.81, p < 0.001) [26]. However, Tesche et al. [27] claimed the statistical difference in the diagnostic performance of ML-based CT-FFR between vessels with high AS > 400 and vessels with low-to-intermediate AS < 400. The AUC of vessels with high AS was inferior to that of vessels with low-to-intermediate (0.71 vs. 0.85, p = 0.004). In addition, the correlation between ML-based CT-FFR and invasive FFR was poor in the vessels with high AS > 400 (r = 0.35, p < 0.003). Whether coronary calcification will affect the diagnostic performance of CT-FFR is still controversial. At present, the main problem is that the sample size of patients with severe calcifications is small; so, the diagnostic performance of CT-FFR in a population with AS > 1000 remains to be further studied.
Image quality is another important factor affecting diagnostic performance of CT-FFR. Poor image quality will affect the accurate delineation of lumen and lesions by researchers, and a considerable number of patients had to be excluded from present studies because of poor image quality [15, 26, 28]. As early as 2013, Leipsic et al. pointed out that administration of β-blockers and nitroglycerin before CT image acquisition could improve the specificity (p = 0.03 and p = 0.05, respectively) of CT-FFR, but had no effect on sensitivity (p = 0.37 and p = 0.09, respectively) and overall accuracy (p = 0.58 and p = 0.97, respectively). Meanwhile, the presence of misalignment artifacts significantly reduced the sensitivity (p = 0.001) and accuracy (p = 0.03) of CT-FFR [29]. Recently, Xu et al. studied the effect of image quality on diagnostic performance at the vessel level and found that the group with high subjective image quality (Group score 4) had better specificity (0.98 vs. 0.86, p = 0.014) and higher AUC (0.93 vs. 0.80, p = 0.024) than the low image quality group (Group score 2) without sacrificing sensitivity. In addition, vessel enhancement could affect sensitivity (p = 0.003) of CT-FFR, while heart rate also had an impact on the specificity of CT-FFR (p = 0.015) and the positive predictive value (PPV) (p = 0.003) (Fig. 2) [30]. In summary, we need to strictly follow the scanning specifications of the cardiovascular CT guidelines, optimize CCTA image acquisition, control patients’ heart rate, reduce motion artifacts, and then improve the success rate and diagnostic accuracy of CT-FFR.
CT-FFR for treatment decision-making in CAD and outcome
Current evidence showed that CT-FFR was safe and feasible for guiding treatment strategies of patients with suspected CAD, thus reducing unnecessary ICA. In the planned invasive test group of the PLATFORM trial (Prospective Longitudinal Trial of CT-FFR: Outcome and Resource Impacts), some patients without obstructive CAD were wrongly advised to plan invasive testing, and the proportion of these patients in the CT-FFR guided arm was much lower than in the usual care arm (12.4%, 24/193 vs. 73.3%, 137/187, r = 60.8%, p < 0.0001) [31]. In the CT-FFR guided arm, in 61% (117/193) of patients with planned ICA, invasive assessment was canceled and no events appeared during the 90-day and 1-year follow-up period [31, 32]. The RIPCORD study confirmed that compared with CCTA alone, CT-FFR changed the treatment decisions of 44% (88/200) patients with CAD including 72 patients who had a change in management category and 16 patients in whom the target vessel for PCI changed based on CT-FFR results [33]. The ADVANCE study, a multi-center, prospective study, evaluated the impact of CT-FFR on the choice of management strategies for CAD patients in in a real-world scenario. The study showed that in a core laboratory analysis, CT-FFR reclassified the treatment decisions of 66.9% of patients compared with CCTA alone. No major adverse coronary event (MACE) occurred within the 90-day follow-up period in any patients with negative CT-FFR [34, 35]. The results of the 1-year follow-up showed that 92.9% (2490/2679) of patients received only medical therapy based on the CT-FFR result, and none of them had any MACE in the aftermath [36]. If this non-invasive technique can be widely used for treatment decision-making in patients with CAD, many unnecessary invasive assessments and revascularization procedures can be reduced.
For patients with complex multi-vessel CAD, it is significant to distinguish the number of vessels that need intervention and to identify the location that causes lesion-specific ischemia so as to select an appropriate treatment management. Anatomical SYNTAX score (SYNERGY between PCI with TAXUS™ and Cardiac Surgery) was proposed to evaluate the complexity of lesions according to the pathological location, severity, bifurcation, calcification and other anatomical features [37,38,39]. In recent years, the non-invasive functional SYNTAX score guided by CT-FFR has been evaluated for multi-vessel disease [40,41,42,43]. The CT-FFR based functional SYNTAX score reclassified 30% of patients from the high/intermediate-risk group to the low-risk group [40]. The SYNTAX III REVOLUTION trial changed treatment decisions in 14 patients, including 13 patients who were planned for coronary artery bypass grafting (CABG) and were reclassified to percutaneous coronary intervention (PCI) [41, 42]. The secondary end point of the SYNTAX III REVOLUTION trial showed that the treatment selection of 6.6% patients was changed and treatment plan of 18.3% patients was modified after adding CT-FFR [43]. In the upcoming years, it is expected that CCTA combined with CT-FFR for anatomical and functional evaluation of multiple vessels will guide CABG. The value of CT-FFR in other complex lesions, such as bifurcation lesions, single-branch tandem lesions or diffuse lesions, should also be systematically studied in the future.
CT-FFR for prognosis evaluation in CAD
For patients with suspected CAD or acute chest pain, CT-FFR can predict MACE within 90 days, reduce the ICA rate in patients with non-obstructive CAD, and improves prognosis [14.44]. In the NXT study, 206 stable CAD patients were followed up with a median of 4.7 years. Ihdayhid et al. found that none of the patients with negative CT-FFR developed MACE, and the probability of MACE in the positive CT-FFR patients (CT-FFR ≤ 0.80) was higher than that in CCTA-positive patients, which showed that CT-FFR had an outstanding performance in predicting the prognosis of patients with CAD [45]. The ADVANCE study also showed that the lower the CT-FFR value, the higher the rate of revascularization (CT-FFR < 0.71: 57.5%, CT-FFR 0.71–0.75: 27.5%, CT-FFR 0.76–0.80: 15.9%, CT-FFR 0.81–0.85: 6.9%, CT-FFR 0.86–0.90: 3.9%, and CT-FFR > 0.90: 4.6%; p < 0.001). The rate of cardiovascular death or MI in patients with positive CT-FFR (≤ 0.80) was higher than in patients with negative CT-FFR (> 0.80) (0.80% vs. 0.20%, RR: 4.22, p = 0.01) [35]. Lu et al. retrospectively enrolled 181 patients from the PROMISE study (Prospective Multicenter Imaging Study for Evaluation of Chest Pain) and performance of CT-FFR according to MACE was observed within 90 days. The result indicated that on a per-patient level, CT-FFR ≤ 0.80 was superior to ICA (p < 0.001) and CCTA (p < 0.001) in predicting MACE [46].
Some retrospective studies also combined CT-FFR with different measurements to confirm the predictive power of the combined effect on MACE. For example, during a median of 19.5-month follow-up in 48 patients with acute coronary syndrome (ACS), CT-FFR ≤ 0.80 combined with Framingham risk score and CAD-RADS ≥ 3 could predict MACE (AUC 0.78, p = 0.032) [47]. In another study, 82 patients with suspected CAD were followed for a median of 18.5 months and the results showed that CT-FFR ≤ 0.80, CCTA stenosis ≥ 50% and plaque characteristics could better predict MACE with a high AUC (0.94) [48]. In conclusion, these results suggested that CT-FFR can reduce ICA examinations and play an important role in the evaluation of short-term and middle-term prognosis of CAD.
Other applications of CT-FFR
CT-FFR has also been used in patients with myocardial bridging (MB) (Fig. 3) and anomalous origin of coronary arteries. Zhou et al. first studied the value of CT-FFR in patients with MB and they found that CT-FFR values of MB were lower than in controls (p < 0.05) and that positive CT-FFR values were associated with chest pain symptoms. The diastolic stenosis degree of MB could be used to predict abnormal CT-FFR values [49]. They also demonstrated that CT-FFR had a high diagnostic performance in MB patients with proximal atherosclerosis plaques with using invasive FFR as reference standard. They also investigated the role of CT-FFR in predicting proximal plaque of MB and found that the formation of proximal plaque of MB can be predicted based on ML-based CT-FFR, while the CT-FFR value and change in CT-FFR value (the difference between proximal CT-FFR value and distal CT-FFR value) were independent predictors, suggesting that hemodynamic factors play an important role in the formation of proximal plaque of MB [50]. CT-FFR has also been used to investigate the role of an anomalous aortic origin of the right coronary artery [51,52,53]. Tang et al. investigated the role of CT-FFR in patients with anomalous origin of the right coronary artery from the left coronary sinus, and found that patients with abnormal CT-FFR had a higher prevalence of typical angina (29.4% vs. 7.8%, p = 0.025) and atypical angina (29.4% vs. 6.5%, p = 0.016) [53]. These studies suggest that clinicians should adopt appropriate treatment strategies in those patients with positive CT-FFR value.
CT-FFR in myocardial bridging. a, b Superficial MB in the mid-segment of the LAD in a 67-year-old male. a Curved planar reformation image showing MB in the mid-segment of the LAD. b CT-FFR image, a negative CT-FFR value (0.96) distal to the MB was observed. c, d Deep MB in the mid-segment of the LAD in a 53-year-old male. c Curved planar reformation image showing MB in the mid-segment of LAD. d CT-FFR image, a positive CT-FFR value (0.68) distal to the MB is observed
Cost-effectiveness analysis
CT-FFR can guide clinical treatment decisions and improves the prognosis of patients with a reduction in overall medical costs. Hlatky et al. reported that CT-FFR guided treatment costs in patients with CAD were 30% lower than that of an invasive FFR strategy ($7674 vs. $10 702) [54]. A substudy of the PLATFORM trial assessed the effective use of medical resources and the impact on patients’ quality of life after CT-FFR-guided clinical decision-making, and conducted a cost analysis during the follow-up period of 90 days and 1 year, respectively [33, 55]. It was found that the quality of life of patients in the CT-FFR-guided group was improved compared with the conventional decision-making group, and the cost of follow-up diagnosis and treatment was reduced by 32% and 33%, respectively. The NXT trial also suggested that CT-FFR-guided strategy resulted in a 32% reduction in cost compared with an ICA-guided strategy [56]. In patients with acute chest pain, there was no significant difference in resource utilization and overall medical costs between CCTA group (n = 258) and CT-FFR group (n = 297) within 90 days, but the medical costs of patients with negative CT-FFR were lower [44]. Taken together, CT-FFR can reduce medical costs and shows an improvement in quality of life, although more clinical evidence is needed.
Future challenges and perspectives of CT-FFR
Numbers of research studies have shown that CT-FFR has excellent diagnostic performance to identify lesion-specific ischemia and can safely guide treatment decision-making in patients with CAD. Furthermore CT-FFR can predict prognosis and clinical outcome of patients, and results in lower medical costs. However, CT-FFR is still in the stage of clinical research and has not been widely used in clinic practice. The application of CT-FFR still faces many challenges.
First, there is only one US FDA-approved commercially available algorithm. However, very long post-processing time in the core laboratory in addition to high cost limits its wide clinical application, including the emergency environment. Thus, development of workstation-based CT-FFR algorithms to reduce measurement time is warranted. Several workstation-based algorithms have been introduced but are for research purposes only. Second, CT-FFR is an image post-processing technology, and image quality is directly related to diagnostic performance of CT-FFR. In the DeFACTO study, 31 patients were excluded for poor image quality (11%, 31/285, 252 enrolled) [15]. In the NXT trials, 47 patients were excluded because of non-valuable image (13%, 47/365) [11]. In the study of Kawaji et al. and Tesche et al., 6% (3/48, 43 enrolled) and 5% (8/166, 104 enrolled) of patients were excluded due to poor image quality, respectively [26, 29]. Many reasons can lead to impaired image quality, such as electrocardiogram registration errors, motion artifacts from high heart rate and respiratory movements, coronary artery enhancement, and image noise. Third, the current CT-FFR studies mostly focus on the diagnostic accuracy, treatment decision-making and predicting prognosis of CAD patients, but the functional evaluation of CT-FFR for target vessels in patients who have undergone revascularization is unknown. CT-FFR only evaluates the hemodynamic significance of epicardial vessel lesions, which is unable to diagnose microvascular dysfunction. In the future, researchers can expand the application and indication of CT-FFR and explore the value of CT-FFR in patients after revascularization. Finally, most of the current studies do not make full use of CCTA data, for example, the combination of high-risk plaque features should be able to provide more prognosis information.
Conclusions
In conclusion, CT-FFR can realize one-stop shop evaluation of coronary anatomy and function of lesion-specific ischemia, guide subsequent revascularization as a “gatekeeper” for ICA, reduce medical costs and improve the prognosis of patients. However, more evidence is needed to further promote the wide clinical application of CT-FFR to benefit more CAD patients.
Abbreviations
- CAD:
-
Coronary artery disease
- CCTA:
-
Coronary CT angiography
- FFR:
-
Fractional flow reserve
- ICA:
-
Invasive coronary angiography
- CT-FFR:
-
Computed tomography-derived fractional flow reserve
- CFD:
-
Computational fluid dynamics
- TAG:
-
Transluminal attenuation gradient
- ML:
-
Machine learning
- CTP:
-
CT perfusion
- CMR:
-
Cardiac magnetic resonance
- SPECT:
-
Single-photon emission computed tomography
- PET:
-
Positron emission tomography
- MACE:
-
Major adverse coronary event
- CABG:
-
Coronary artery bypass grafting
- PCI:
-
Percutaneous coronary intervention
- ACS:
-
Acute coronary syndrome
- MB:
-
Myocardial bridging
References
Pijls NHJ, van Son JAM, Kirkeeide RL, et al. Experimental basis of determining maximum coronary, myocardial and collateral blood flow by pressure measurements for assessing functional stenosis severity before and after percutaneous transluminal coronary angioplasty. Circulation. 1993;86:1354–67.
De Bruyne B, Baudhuin T, Melin JA, et al. Coronary flow reserve calculated from pressure measurements in humans. Validation with positron emission tomography. Circulation. 1994;89:1013–22.
Pijls NH, De Bruyne B, Peels K, et al. Measurement of fractional flow reserve to assess the functional severity of coronary-artery stenoses. N Engl J Med. 1996;334:1703–8.
Bech GJ, De Bruyne B, Pijls NH, et al. Fractional flow reserve to determine the appropriateness of angioplasty in moderate coronary stenosis: a randomized trial. Circulation. 2001;103:2928–34.
Tonino PA, De Bruyne B, Pijls NH, et al. Fractional flow reserve versus angiography for guiding percutaneous coronary intervention. N Engl J Med. 2009;360:213–24.
De Bruyne B, Fearon WF, Pijls NH, et al. Fractional flow reserve guided PCI for stable coronary artery disease. N Engl J Med. 2014;371:1208–17.
Taylor CA, Fonte TA, Min JK. Computational fluid dynamics applied to cardiac computed tomography for noninvasive quantification of fractional flow reserve: scientific basis. J Am Coll Cardiol. 2013;61:2233–41.
Glagov S, Weisenberg E, Zarins CK, et al. Compensatory enlargement of human atherosclerotic coronary arteries. N Engl J Med. 1987;317:1604.
Zarins CK, Zatina MA, Giddens DP, et al. Shear stress regulation of artery lumen diameter in experimental atherogenesis. J Vasc Surg. 1987;5:413–20.
Kamiya A. Adaptive regulation of wall shear stress to flow change in the canine carotid artery. Am J Physiol. 1980;239:H14–21.
Norgaard BL, Leipsic J, Gaur S, et al. Diagnostic performance of noninvasive fractional flow reserve derived from coronary computed tomography angiography in suspected coronary artery disease: the NXT trial (Analysis of Coronary Blood Flow Using CT Angiography: next Steps). J Am Coll Cardiol. 2014;63:1145–55.
Tesche C, De Cecco CN, Albrecht MH, et al. Coronary CT angiography-derived fractional flow reserve. Radiology. 2017;285:17–33.
Tang CX, Liu CY, Lu MJ, et al. CT FFR for ischemia-specific cad with a new computational fluid dynamics algorithm: a Chinese multicenter study. JACC Cardiovasc Imaging. 2019. https://doi.org/10.1016/j.jcmg.2019.06.018.
Koo BK, Erglis A, Doh JH, et al. Diagnosis of ischemia-causing coronary stenoses by noninvasive fractional flow reserve computed from coronary computed tomographic angiograms. Results from the prospective multicenter DISCOVER-FLOW (Diagnosis of Ischemia-Causing Stenoses Obtained Via Noninvasive Fractional Flow Reserve) study. J Am Coll Cardiol. 2011;58:1989–97.
Min JK, Leipsic J, Pencina MJ, et al. Diagnostic accuracy of fractional flow reserve from anatomic CT angiography. JAMA. 2012;308:1237–45.
Renker M, Schoepf UJ, Wang R, et al. Comparison of diagnostic value of a novel noninvasive coronary computed tomography angiography method versus standard coronary angiography for assessing fractional flow reserve. Am J Cardiol. 2014;114:1303–8.
Coenen A, Lubbers MM, Kurata A, et al. Fractional flow reserve computed from noninvasive CT angiography data: diagnostic performance of an on-site clinician-operated computational fluid dynamics algorithm. Radiology. 2015;274:674–83.
Kruk M, Wardziak Ł, Demkow M, et al. Workstation-Based Calculation of CTA-Based FFR for Intermediate Stenosis. JACC Cardiovasc Imaging. 2016;9:690–9.
Yang DH, Kim YH, Roh JH, et al. Diagnostic performance of on-site CT-derived fractional flow reserve versus CT perfusion. Eur Heart J Cardiovasc Imaging. 2017;18:432–40.
Min JK, Koo BK, Erglis A, et al. Effect of image quality on diagnostic accuracy of noninvasive fractional flow reserve: results from the prospective multicenter international DISCOVER-FLOW study. J Cardiovasc Comput Tomogr. 2012;6:191–9.
Coenen A, Kim YH, Kruk M, et al. Diagnostic accuracy of a machine-learning approach to coronary computed tomographic angiography-based fractional flow reserve: result from the MACHINE consortium. Circ Cardiovasc Imaging. 2018;11(6):e007217.
Pontone G, Baggiano A, Andreini D, et al. Dynamic stress computed tomography perfusion with a whole-heart coverage scanner in addition to coronary computed tomography angiography and fractional flow reserve computed tomography derived. JACC Cardiovasc Imaging. 2019. https://doi.org/10.1016/j.jcmg.2019.02.015.
Li Y, Yu M, Dai X, et al. Detection of hemodynamically significant coronary stenosis: CT myocardial perfusion versus machine learning CT fractional flow reserve. Radiology. 2019;293:305–14.
Driessen RS, Danad I, Stuijfzand WJ, et al. Comparison of coronary computed tomography angiography, fractional Flow reserve, and perfusion imaging for ischemia diagnosis. J Am Coll Cardiol. 2019;73:161–73.
Norgaard BL, Gaur S, Leipsic J, et al. Influence of coronary calcification on the diagnostic performance of CT angiography derived FFR in coronary artery disease: a substudy of the NXT trial. JACC Cardiovasc Imaging. 2015;8:1045–55.
Kawaji T, Shiomi H, Morishita H, et al. Feasibility and diagnostic performance of fractional flow reserve measurement derived from coronary computed tomography angiography in real clinical practice. Int J Cardiovasc Imaging. 2017;33:271–81.
Tesche C, Otani K, De Cecco CN, et al. Influence of coronary calcium on diagnostic performance of machine learning CT-FFR: results from MACHINE registry. JACC Cardiovasc Imaging. 2020;13:760–70.
Tesche C, De Cecco CN, Baumann S, et al. Coronary CT angiography-derived fractional flow reserve: machine learning algorithm versus computational fluid dynamics modeling. Radiology. 2018;288:64–72.
Leipsic J, Yang TH, Thompson A, et al. CT angiography (CTA) and diagnostic performance of noninvasive fractional flow reserve: results from the determination of fractional flow reserve by anatomic CTA (DeFACTO) study. AJR Am J Roentgenol. 2014;202:989–94.
Xu PP, Li JH, Zhou F, et al. The influence of image quality on diagnostic performance of a machine learning-based fractional flow reserve derived from coronary CT angiography. Eur Radiol. 2020;30:2525–34.
Douglas PS, Pontone G, Hlatky MA, et al. Clinical outcomes of fractional flow reserve by computed tomographic angiography-guided diagnostic strategies vs. usual care in patients with suspected coronary artery disease: the prospective longitudinal trial of FFR(CT): outcome and resource impacts study. Eur Heart J. 2015;36:3359–67.
Douglas PS, De Bruyne B, Pontone G, et al. 1-year outcomes of FFRCT-guided care in patients with suspected coronary disease: the PLATFORM study. J Am Coll Cardiol. 2016;68:435–45.
Curzen NP, Nolan J, Zaman AG, et al. Does the routine availability of CT-derived FFR influence management of patients with stable chest pain compared to CT angiography alone?: the FFRCT RIPCORD study. JACC Cardiovasc Imaging. 2016;9(10):1188–94.
Chinnaiyan KM, Akasaka T, Amano T, et al. Rationale, design and goals of the HeartFlow assessing diagnostic value of non-invasive FFR in coronary care (ADVANCE) registry. J Cardiovasc Comput Tomogr. 2017;11:62–7.
Fairbairn TA, Nieman K, Akasaka T, et al. Real-world clinical utility and impact on clinical decision-making of coronary computed tomography angiography-derived fractional flow reserve: lessons from the ADVANCE Registry. Eur Heart J. 2018;39:3701–11.
Patel MR, Nørgaard BL, Fairbairn TA, et al. 1-Year Impact on medical practice and clinical outcomes of FFR: the ADVANCE registry. JACC Cardiovasc Imaging. 2020;13:97–105.
Serruys PW, Onuma Y, Garg S, et al. Assessment of the SYNTAX score in the syntax study. EuroIntervention. 2009;5:50–6.
van Gaal WJ, Ponnuthurai FA, Selvanayagam J, et al. The syntax score predicts periprocedural myocardial necrosis during percutaneous coronary intervention. Int J Cardiol. 2009;135:60–5.
Serruys PW, Morice MC, Kappetein AP, et al. SYNTAX Investigators Percutaneous coronary intervention versus coronary-artery bypass grafting for severe coronary artery disease. N Engl J Med. 2009;360:961–72.
Collet C, Miyazaki Y, Ryan N, et al. Flow reserve derived from computed tomographic angiography in patients with multivessel CAD. J Am Coll Cardiol. 2018;71:2756–69.
Cavalcante R, Onuma Y, Sotomi Y, et al. Non-invasive Heart Team assessment of multivessel coronary disease with coronary computed tomography angiography based on SYNTAX score II treatment recommendations: design and rationale of the randomized SYNTAX III Revolution trial. Euro Interv. 2017;12:2001–8.
Collet C, Onuma Y, Andreini D, et al. Coronary computed tomography angiography for heart team decision-making in multivessel coronary artery disease. Eur Heart J. 2018;39:3689–98.
Andreini D, Modolo R, Katagiri Y, et al. Impact of fractional flow reserve derived from coronary computed tomography angiography on Heart Team treatment decision-making in patients with multivessel coronary artery disease: insights from the SYNTAX III REVOLUTION Trial. Circ Cardiovasc Interv. 2019;12:e007607.
Chinnaiyan KM, Safian RD, Gallagher ML, et al. Clinical use of CT-Derived fractional flow reserve in the emergency department. JACC Cardiovasc Imaging. 2020;13:452–61.
Ihdayhid AR, Norgaard BL, Gaur S, et al. Prognostic value and risk continuum of noninvasive fractional flow reserve derived from coronary CT angiography. Radiology. 2019;292:343–51.
Lu MT, Ferencik M, Roberts RS, et al. Noninvasive FFR derived from coronary CT angiography: management and outcomes in the PROMISE trial. JACC Cardiovasc Imaging. 2017;10:1350–8.
Duguay TM, Tesche C, Vliegenthart R, et al. Coronary computed tomographic angiography-derived fractional flow reserve based on machine learning for risk stratification of non-culprit coronary narrowings in patients with acute coronary syndrome. Am J Cardiol. 2017;120:1260–6.
von Knebel Doeberitz PL, De Cecco CN, Schoepf UJ, et al. Impact of coronary computerized tomography angiography-derived plaque quantification and machine-learning computerized tomography fractional flow reserve on adverse cardiac outcome. Am J Cardiol. 2019;124:1340–8.
Zhou F, Tang CX, Schoepf UJ, et al. Fractional flow reserve derived from CCTA may have a prognostic role in myocardial bridging. Eur Radiol. 2019;29:3017–26.
Zhou F, Tang CX, Schoepf UJ, et al. Machine learning using CT-FFR predicts proximal atherosclerotic plaque formation associated with LAD myocardial bridging. JACC Cardiovasc Imaging. 2019;12:1591–3.
Miki T, Miyoshi T, Watanabe A, et al. Anomalous aortic origin of the right coronary artery with functional ischemia determined with fractional flow reserve derived from computed tomography. Clin Case Rep. 2018;6:1371–2.
Tahir H, Ahmad S, Awan MU, et al. Anomalous origin of left anterior descending artery and left circumflex artery from right coronary sinus with malignant left anterior descending artery course: role of coronary CT angiography derived fractional flow reserve in decision making. Cureus. 2018;10:e3220.
Tang CX, Lu MJ, Schoepf JU, et al. Coronary computed tomography angiography-derived fractional flow reserve in patients with anomalous origin of the right coronary artery from the left coronary sinus. Korean J Radiol. 2020;21:192–202.
Hlatky MA, Saxena A, Koo BK, et al. Projected costs and consequences of computed tomography-determined fractional flow reserve. Clin Cardiol. 2013;36:743–8.
Hlatky MA, De Bruyne B, Pontone G, et al. Quality-of-life and economic outcomes of assessing fractional flow reserve with computed tomography angiography: platform. J Am Coll Cardiol. 2015;66:2315–23.
Kimura T, Shiomi H, Kuribayashi S, et al. Cost analysis of non-invasive fractional flow reserve derived from coronary computed tomographic angiography in Japan. Cardiovasc Interv Ther. 2015;30:38–44.
Funding
Supported by The National Key Research and Development Program of China (2017YFC0113400 for L.J.Z.).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, Y.T., Xu, P.P., Zhou, F. et al. Coronary computed tomography angiography derived flow fractional reserve: the state of the art. Chin J Acad Radiol 3, 84–93 (2020). https://doi.org/10.1007/s42058-020-00039-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42058-020-00039-x