Abstract
Plants are crucial to human existence. They provide a source of sustenance, nutrient recycling, atmospheric replenishment, water cycling, and physiological health for life on Earth as well as in space. The human spaceflight realm poses unique challenges for engineers who develop facilities to conduct plant experiments, grow crops, and design biology-based life support systems for off-Earth habitation. Fractional or microgravity strongly influences fluid and thermal management directly and indirectly in both the organisms themselves and their engineered life support facilities. Scarce resources such as mass, volume, power, crew involvement, and data must be minimized through all mission phases. The current spaceflight facilities vary in complexity from simple Petri dishes to closed-loop feedback-controlled chambers that regulate biologically relevant parameters such as photosynthetic illumination intensity and quality, diurnal cycle, temperature, relative humidity, moisture, atmospheric constituency, and even fractional gravity. Learning how to grow plants efficiently and effectively will become increasingly relevant as humans journey farther and farther out into the solar system.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
The history of human and autonomous space flight is short and is continuously evolving. In 1961, Yuri Gagarin was the first human to travel into outer space. Eight years later astronauts were taking their first steps on the Moon. In 2022, fifty years after the last person walked on the Moon, multiple space agencies and international collaborators have committed to return to its surface. The intent is to build an orbital “Lunar Gateway” and eventually establish human settlements on its surface. After the Moon, Mars is the next logical step.
As humanity aims to extend its reach into the solar system, crewed missions of increasing length and complexity will be required. The engineered life support systems within the spacecrafts will need to be more efficient and self-sufficient to support long-term crew habitation. Food production, nutrient recycling, atmospheric replenishment, water cycling, and the physiological and psychological health of the crew members will all need to be considered and maintained. Growing plants in situ in bioregenerative life support systems (BLSS) can sustain these mission-critical and life sustaining elements in conjunction with abiotic life support systems. On Earth, explorers brought their plants and agricultural practices with them on their oversea or overland journeys. Modern solar sailors, astronauts, will do the same when they journey through the solar system.
2 A Brief History of Astrobotany
Plant and animal responses to space environments were studied long before humans overcame the Kármán line. Corn seeds and moss were launched into the upper atmosphere on a modified V2 rocket to test the effects of radiation exposure on biological systems9,28. Jack Myers studied green algae (Chlorella sp.) within BLSS in the 1950s, these systems provided the core elements to support plant life: light, water, carbon dioxide, and temperature controls27. The first space stations, Saylut and Skylab, were deployed in the 1970s and enabled fundamental astrobotany research, like the influence of microgravity on cytoplasmic streaming42. The SVET growth chamber, abord the Mir space station, produced the first crop of space-grown produce from seed10. The International Space Station (ISS), with its increased size and orbital lifespan, allows for more crew, crew time, and payloads devoted to science. The capacity to conduct complex and lengthy plant growth experiments in microgravity has grown over time. As space vehicular capabilities have improved, so have the systems for astrobotany research. Since 1970 more than 21 growth chambers have been developed and flown50. Through extensive engineering and design efforts, almost all growth environments contained more complex environmental control systems than their predecessors.
3 Bioregenerative Life Support Systems (BLSS)
Long duration missions will require in situ food production to provide nutrients and food diversity for crews, as the nutritional value of packaged food degrades over time4. On Earth, the study of plant cultivation within BLSS habitats for space missions has been a focus of research groups for years, some notable BLSSs are the EDEN ISS project21, the Hawai’i Space Exploration Analog and Simulation (HI-SEAS) project7, the Mars Desert Research Station35, the European Space Agency’s MELiSSA project44, and NASA’s Biomass Production Chamber46. Extensive research has gone into selecting suitable crops for space agriculture based on their crop readiness, edible biomass production, energy consumption per crop, and crew time required for plant husbandry21,36,48.
Excluding higher plants, algae grown in fluid bioreactors could support BLSS habitats27. Algae are relatively easy to rear, grow rapidly, have a high nutrient density and harvest index, recycle nutrients, can scrub injected carbon dioxide, and can clean wastewater6,18,44. Outside of BLSS, plants can provide psychological and therapeutic benefits to crew members30. Through the combined efforts of testing food crops and algae in BLSS, we have the technical merit to create BLSS in situ when there is space and resources to accommodate them.
4 Crop Production
Growth chambers aboard orbital space vehicles have enabled fundamental physiology and crop production research in space environments50. The first space-grown vegetables, radish and Chinese cabbage, were cultivated from seed in the SVET system within the Mir space station10. This demonstrated that crops could be grown from seed and that plant functions and morphology appeared to be normal. The SVET system used florescent lighting, ventilation fans, and sensors for temperature, humidity, and substrate moisture50.
As of 2023, there are two plant growth systems residing on the ISS, the Vegetable Production System (Veggie) and the Advanced Plant Habitat (APH)23 (Fig. 1). The Veggie and APH systems are on opposite ends of the technological spectrum. Veggie has minimal controls with only LEDs and a ventilation fan to autonomously control the growing environment and manual water addition via a syringe or passively coupled water reservoir. The APH on the other hand has extensive controls and sensors, including air speed, relative humidity, substrate moisture, CO2, ethylene scrubbing, and more. To date the edible crops grown within APH are dwarf wheat, Hatch chile peppers, and radish11,19, while the Veggie system has grown bok choy, mustard, kale, lettuce, and dwarf tomatoes12,19, 20. With the ISS circling in low earth orbit, efforts to supplement the crew diet with space-grown crops have not been prioritized; greater emphasis has been placed on fundamental plant biology studies.
5 Plant Physiology
Plant physiology research has provided us with a better understanding of plant functions and environmental responses, which can improve growth efforts on Earth and in space. Arabidopsis thaliana (A. thaliana) was the first plant to complete its lifecycle (from seed to seed) in space22. Following this inaugural generative study, A. thaliana, Brassica rapa, and Pisum sativum produced viable seed during space flight, with A. thaliana and Pisum sativum grown for multiple generations16,17,26,43. These experiments proved that multiple generations of plants can be grown in a low Earth orbit while experiencing microgravity.
A. thaliana, a model organism, is a favored plant for space research as its genome and physiological responses are well understood. Various experiments have recorded its unique responses in cell signaling, genomics, gravitropism, leaf chlorophyll content, phototropism, reproductive morphology, root composition, and root structure during space flight8,14,17, 25,31,32,34. A. thaliana presents engineering challenges. The seeds are tiny and typically sown or glued directly on a wicking substrate. Their small size makes them prone to drying out during the critical seedling establishment phase if the water delivery system cannot maintain adequate, uniform substrate moisture. Mature plants create considerable flower petal and leaf litter debris which remains airborne in the microgravity environment until it is trapped by airflow and mechanical filters. By understanding how plants respond to their environments aboard space vehicles, engineers can build more suitable plant growth systems.
6 Plant Growth Systems as Enablers of Research
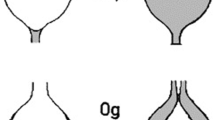
Generally, when plants are provided with the proper environmental conditions (atmosphere, light, media, nutrients, ventilation, water regime, etc.) they tend to adapt and grow readily in microgravity environments49. However, providing these goldilocks-like conditions is difficult. Plant growth facilities have continuously evolved to better control their environments50. Many modern growth facilities control and have sensors for lighting via LEDs, substrate moisture, forced air circulation, temperature, humidity, and atmospheric composition11,23,33. The improved environmental controls and monitoring in modern growth systems have generated high fidelity plant science studies. In addition to the aforementioned government-owned facilities, Veggie and APH, commercial hardware developers like Redwire Corporation provide researchers with other options such as the Passive Orbital Nutrient Delivery System (PONDS), fractional gravity Phytofuge modules, and the under development large-plant oriented scalable Greenhouse (Fig. 2). Fractional gravity platforms can be used to help understand the minimum threshold of gravity required for “normal” plant growth. Such information may help inform the next generation of spacecraft development to include the capacity to generate fractional gravity by rotation.
7 Plant Responses to Space Flight – Direct Effects
Microgravity itself has not shown to fundamentally alter plant growth or lifecycles, however, it holds great influence on the environment that envelope the plants. Gravity is fundamental to life on Earth. Plant roots are positively-gravitropic, they grow in the direction of the force of gravity, while stems are negatively gravitropic. Starch statolith deposition within root cells aid in sensing gravity and guide root growth14.Although plant growth in microgravity is often the variable of interest in astrobotany research, a new payload called the Phytopfuge allows researchers to study the effects of differential gravity on plant growth aboard the ISS. The Multi-use Variable g Platform (MVP) is a facility system with two centrifuges, each containing multiple cassettes that can be idle for microgravity conditions or be spun to produce fractional gravity ranging between 0.1 and 2.0 g. One such cassette type called Phytofuge provides growth conditions for seedlings in specially designed Petri dishes. The Phytofuge systems allows researchers to control the gravity variable and isolate effects of the space environment on plant growth.
In microgravity, phototropism becomes the primary sense for plant growth and orientation, generally roots are negatively phototropic, and shoots are positively phototropic5,13,14. Thus, proper lighting is imperative for astrobotany research. In the last 30 years plant lighting technology and capabilities have increased by leaps and bounds. Early growth chambers either used fluorescent lights or cabin light50. Today, all growth chambers on the ISS use tunable LEDs, in which the light intensity and color are both precisely controlled. This allows researchers to study the effects and influence of light intensity and color on plant growth in microgravity. LEDs are also more energy efficient than fluorescent lamps, which can reduce to total power draw of a payload or increase its light output.
Stresses linked to space flight have been observed in A. thaliana, Brassica rapa, Zinnia sp., and many other plants31, 38,39,41. Water related stresses can arise when soil moisture setpoints optimized in unit gravity are used in microgravity40. Root hypoxia occurs when there is limited available oxygen in the root zone often from overwatering and a lack of convective gas exchange; this is a common stress in space-grown plants31,34. To address these issues, engineers have devised solutions to better control the water content within the rooting substrates, including the use of agar-based media, floral foam, and embedded porous tubes. The APH contains porous tubes to deliver or remove water from the rooting substrate via regulating pressures placed on the tubes. The PONDS hardware is passive; water is separately stored in a reservoir, wicks embedded into the rooting substrate are hydraulically connected to the reservoir and water is drawn into the root zone through capillary action. Engineers can also manage root zone gas exchange operationally by iteratively adding water and then permitting the root zone to dry out. This technique is sometimes referred to as 'ebb and flow’.
Radiation is another stressful factor in spaceflight. The effects of cosmic radiation on plants are not well understood and are difficult to study. Spacecraft in low Earth orbit are largely protected from cosmic radiation held within the Van Allen Belts. During the Artemis 1 mission, the Orion capsule carried A. thaliana seeds on its journey through the high radiation within the Van Allen Belts, lunar orbit, and back to Earth29,51. Multiple studies have exposed seeds to the space environment by affixing containers to exterior platforms on the ISS. The Biorisk experiments exposed seeds for 13 months. The majority of seeds remained viable, though some species had reduced germination percentages1. A. thaliana and tobacco seeds were placed on the exterior of the ISS for 558 and 682 days, some with UV protection and others without. UV protected seeds germinated while unprotected seeds did not37. The Artemis and Biorisk studies will show how cosmic radiation affects seeds and will inform best practices for seed preservation and storage. As humans take prolonged voyages through outer space, radiation shielding for the astronauts and their biologically active components and payloads will need to be implemented.
8 Plant Responses to Space Flight – Indirect Effects
In microgravity there is no buoyancy-driven convection for liquids or gases. As a result, leaf boundary layers are larger and air molecules do not naturally mix which can limit photosynthesis and transpiration15,34. Forced air throughout plant canopies helps to lessen the leaf boundary layer and mix the air, therefore all modern growth chambers have at least one circulation or exhaust fan.
In sealed growth chambers the atmospheric composition of carbon dioxide, ethylene, and other volatile organic compounds (VOCs) influences plant growth and development. Elevated ethylene concentrations affected the morphology of A. thaliana roots in a Biorack experiment and prevented wheat from forming viable seed in SVET3, 8. Through these lessons learned, most modern systems scrub VOCs from the chamber atmosphere using a variety of engineering solutions including photocatalytic systems and filters packed with granular potassium permanganate. Carbon dioxide is both consumed by plants through photosynthesis during the day and produced by plants metabolically during the night. Thus, closed systems must include feedback control that both scrubs carbon dioxide, typically using a lithium hydroxide filter, and adds carbon dioxide from a compressed gas bottle. At a minimum, chambers have ventilation fans to exchange chamber air with cabin air to prevent the buildup of VOCs and maintain a relatively stable carbon dioxide environment.
The effects of moderately elevated carbon dioxide (CO2) concentrations (600–1000 ppm) have been studied for horticultural purposes on Earth and often bolster plant growth24. The CO2 levels on the ISS range between 3000 and 5000 ppm. Previous spacecraft had even higher concentrations. Multiple studies have shown that hyper-saturated CO2 environments generally cause no harm to plants and can increase crop biomass production for many species, though Chinese cabbage growth was inhibited in ISS-like CO2 conditions2, 45,46,–47. Thus, plant growth environments need to control CO2, ethylene, and VOC’s or conduct frequent cabin air exchanges to create optimal atmospheric compositions40,49.
9 Engineering Challenges
Engineers developing a facility for plant growth are essentially designing a miniature life support system with similar complexities and attributes to those developed on a larger scale to keep humans alive and thriving in space. While mission success is clearly important to plant researchers, the first and paramount concern for the engineer is crew safety. Whereas an engineer of terrestrial systems might select off the shelf components based purely on their function, spaceflight engineers must think deeper. There are many design considerations and trade spaces facing an engineer of space-borne plant growth systems. A few of them will be explored here.
Resources are notoriously scarce in spaceflight. Both mass and volume cost a small fortune to loft into low orbit, and no amount of money changes the fact that only a handful of vehicles currently traffic to and from orbit in any single year. Once on orbit, mass is no longer a constraining factor, but volume continues to be problematic. Orbiting platforms like the ISS have only so much volume. Period. That volume must be partitioned between active scientific facilities, life support systems, and storage space for supplies and consumables. Other constraining resources include power, commanding and telemetry bandwidth, crew time, and the availability of supporting equipment like cold stowage, videography/photography, and analytical tools like gloveboxes and microscopes. Crew on the ISS spend the majority of their time maintaining critical space station systems, conducting tasks such as space walks, and exercising to counteract the long-term negative physiological effects of spaceflight. Some weeks there have been as few as 40 work hours available to perform science by the combined efforts of the entire crew. Confronted by all these simultaneous resource constraints, the payload engineer must be ruthlessly efficient in every aspect of design and operations concept.
Consider all the facets of just one challenge: nonmetallic materials of construction burn. A fire on a spacecraft is a catastrophic event. All but the remotest scenarios that result in a fire must be assessed, mitigated, and verified. What are the flammability characteristics of nonmetallic materials? Are there ignition sources and flame propagation paths either from the vehicle or other parts of the payload itself? What are the toxic deflagration products of such combustion? How do the characteristics change in a spacecraft that may have an elevated oxygen environment? Answers to these questions often drive the use of atypical materials such as Ultem®, Kynar®, and Nomex® and strategies that compartmentalize or segregate flammable materials to aid in fire suppression should the unthinkable occur.
Materials used in construction and operation consumables pose interesting challenges to the designer of plant growth payloads. Flammability has already been discussed. Blowing agents used to manufacture foams like insulation can outgas and build up in a closed-loop system to levels that are harmless to humans but profoundly influential to plants. Many plant growth systems employ chemical or photochemical atmospheric scrubbers to remove volatile organic compounds originating from foams, plastics, and even the plants themselves. Biocompatibility of all materials must be considered. Metallic construction of a plant growth chamber might seem logical to mitigate fire hazards, but since most orbital growth chambers are small, the plants will likely make contact with the walls as they mature, and many metals are phytotoxic. A better plan is to coat metallic walls with a more biologically inert material with good flammability properties such as Teflon®.
Substrates that plants root either in or on must be sufficiently porous to facilitate water flow, nutrient delivery, and atmospheric gas exchange. In the absence of gravity, the wetting characteristics of such materials are dominated by capillary flow, often leading to flooding and overwatering. Common substrates include semi-solid gels and crushed granular clay. Frangible materials must be avoided as they can liberate material that, depending on size, can be a nuisance as an irritant in the eye or as life-threatening as an asphyxiation hazard.
The spaceflight environment aboard a crewed vehicle can also pose unique threats to the hardware itself. Elevated radiation degrades electronics; commonly hard drives and charge coupled devices suffer the most though even integrated circuit chips have been known to fail. The lack of buoyancy-driven convection causes heat to build up in odd places or can cause condensation to occur, sometimes deep within electronics. Heat does not rise from a hot object and water does not run down to the lowest physical level in space. Instead, a localized “spherical gradient” of heat or blob of water can grow undetected until problems occur. Often mere fractions of a degree in temperature across a surface can yield such deleterious effects. Two-phase flows, such as water and entrained air in plumbing, do not easily segregate in the absence of gravity. The resulting fluid manifests as a foam of varying density, viscosity, and thermal properties to the detriment of a cooling system or condensate recovery in a closed-loop environment.
Broadly defined, operations considerations exert a huge influence upon the engineering behind growing plants in space. Much like growing plants on Earth, growing plants in space can only utilize so much automation. The eyes, hands, and minds of astronauts are necessary for plants to flourish in space. Perhaps the most obvious operational considerations that must be incorporated into designs are “human factors”. Switches must include guards or covers to prevent inadvertent activation by crew. Labels must be standardized, obvious, and intuitive. Component quantities and locations are tracked using bar code labels. Velcro, elastic bands, and mesh bags are critical to gathering and managing objects that would otherwise simply float away. Less obvious are considerations about how crew must exert forces to push, pull, crank, or relocate objects. Objects in space have no weight, but they still have mass which means they also still have inertia. Fundamental physics tells us that every force exerts an equal and opposite force. Crew must often maintain their position using one hand or feet inserted through fabric loops on the floor in order to perform an operation.
Logistical “operations” must also be addressed. Spaceflight hardware must be handed over to the launch vehicle for stowing as much as nine weeks prior to flight. Only biologically active components, like seeds, can be handed over a few days before launch. Once on orbit, weeks or even months can elapse before a facility is activated. Once active, plant investigations are among the longest research campaigns crew perform, often taking 30, 60, or even 120 days to complete. If a plant investigation yields samples that must be analyzed, this analysis is mainly performed postflight back on Earth. There are very few analytical tools on the current space station that can be used to analyze plant samples, and analysis takes even more precious crew time. Most samples that return are either chemically preserved or frozen. This too represents logistical challenges. The number of “cold stowage assets” available to return samples is limited. Priority is often given to frozen biomedical samples used to assess crew health and performance.
There are a myriad other engineering aspects of growing plants in space. We have only taken a shallow dive into the deep pool of knowledge that exists in the space industry. Most importantly, this review is meant to open the eyes and minds of the next generation of space agricultural engineers. Few academic institutions provide a full course of study necessary for young engineers eager to design the next generation of spaceflight plant growth facilities, but it is these brilliant young minds that will take gardening, crop production, and eventually a closed-loop biological life support system, to the moon, Mars, and the stars.
References
Baranov VM, Novikova ND, Polikarpov NA, Sychev VN, Levinskikh MA, Alekseev VR, Okuda T, Sugimoto M, Gusev OA, Grigor’ev AI (2009) The Biorisk experiment: 13-month exposure of resting forms of organism on the outer side of the Russian Segment of the International Space Station: preliminary results. Dokl Biol Sci 426:267–270
Burgner SE, Nemali K, Massa GD, Wheeler RM, Morrow RC, Mitchell CA (2020) Growth and photosynthetic responses of Chinese cabbage (Brassica rapa L. cv. Tokyo Bekana) to continuously elevated carbon dioxide in a simulated Space Station “Veggie” crop-production environment. Life Sci Space Res 27:83–88
Campbell WF, Salisbury FB, Bugbee B, Klassen S, Naegle E, Strickland DT, Bingham GE, Levinskikh M, Iljina GM, Veselova TD et al (2001) Comparative floral development of Mir-grown and ethylene-treated, earth-grown Super Dwarf wheat. J Plant Physiol 158:1051–1060
Cooper M, Perchonok M, Douglas GL (2017) Initial assessment of the nutritional quality of the space food system over three years of ambient storage. NPJ Microgravity 3:17
Correll MJ, Kiss JZ (2002) Interactions Between Gravitropism and Phototropism in Plants. J Plant Growth Regul 21:89–101
Eckart P (1996) Spaceflight life support and Biospherics. Kluwer Academic Publishers & Microcosm Press, Dordrecht
Ehrlich JW, Massa G, Wheeler R, Gill TR, Quincy C, Roberson L, Binsted K, Morrow R (2017) Plant growth optimization by vegetable production system in HI-SEAS analog habitat. AIAA SPACE Astronautics Forum Expos. https://doi.org/10.2514/6.2017-5143
Guisinger MM, Kiss JZ (1999) The influence of microgravity and spaceflight on columella cell ultrastructure in starch-deficient mutants of Arabidopsis. Am J Botany 86:1357–1366
Henry JP, Ballinger ER, Maher PJ, Simons DG (2009) Classics in Space Medicine: animal studies of the subgravity state during rocket flight. Aviat Space Environ Med 80:747–747
Ivanova TN, Bercovich YuA, Mashinskiy AL, Meleshko GI (1993) The first “space” vegetables have been grown in the “SVET” greenhouse using controlled environmental conditions. Acta Astronaut 29:639–644
John S, Abou-Issa F, Hasenstein KH (2021) Space flight cultivation for Radish (Raphanus sativus) in the advanced plant habitat. Gravit Space Res 9:121–132
Khodadad CLM, Hummerick ME, Spencer LE, Dixit AR, Richards JT, Romeyn MW, Smith TM, Wheeler RM, Massa GD (2020) Microbiological and nutritional analysis of lettuce crops grown on the international space station. Front Plant Sci 11:199
Kiss JZ, Mullen JL, Correll MJ, Hangarter RP (2003) Phytochromes A and B mediate red-light-induced positive phototropism in roots. Plant Physiol 131:1411–1417
Kiss JZ, Wright JB, Caspar T (1996) Gravitropism in roots of intermediate-starch mutants of Arabidopsis. Physiol Plant 97(2):237–244
Kitaya Y, Hirai H, Shibuya T (2010) Important role of air convection for plant production in space farming. Biol Sci Space 24:121–128
Kuang A, Popova A, Xiao Y, Musgrave ME (2000) Pollination and embryo development in Brassica rapa L. in microgravity. Int J Plant Sci 161:203–211
Link BM, Busse JS, Stankovic B (2014) Seed-to-seed-to-seed growth and development of Arabidopsis in microgravity. Astrobiology 14:866–875
Lizzul A, Lekuona-Amundarain A, Purton S, Campos L (2018) Characterization of Chlorella sorokiniana, UTEX 1230. Biology 7:25
Massa G, Romeyn M, Spencer L, Johnson C, Poulet L, Wheeler R (2021) NCERA-101 Station report from Kennedy Space Center, FL, USA
Massa G, Newsham G, Hummerick ME, Caro JL, Stutte GW, Morrow C, Wheeler RM (2015) Preliminary species and media selection for the veggie space hardware. Gravit Space Res 1:96–106
Meinen E, Dueck T, Kempkes F, Stanghellini C (2018) Growing fresh food on future space missions: environmental conditions and crop management. Sci Hortic 235:270–278
Merkys AI, Laurinavicius RS (1983) Complete cycle of individual development of Arabidopsis thaliana (L.) Heynh. plants on board the Salyut-7 orbital station. Dokl Akad Nauk SSSR 271:509–512
Monje O, Richards JT, Carver JA, Dimapilis DI, Levine HG, Dufour NF, Onate BG (2020) Hardware validation of the advanced plant habitat on ISS: canopy photosynthesis in reduced gravity. Front Plant Sci 11:673
Mortensen LM (1987) Review: CO2 enrichment in greenhouses. Crop Responses Scientia Horticulturae 33:1–25
Musgrave ME, Kuang A, Brown C, Mathews S (1998) Changes in Arabidopsis leaf ultrastructure, chlorophyll and carbohydrate content during spaceflight depend on ventilation. Ann Bot 81:503–512
Musgrave ME, Kuang A, Tuominen LK, Levine LH, Morrow RC (2005) Seed storage reserves and Glucosinolates in Brassica rapa L. grown on the international space station. JASHS 130:848–856
Myers J (1954) Basic remarks on the use of plants as biological gas exchangers in a closed system. J Aviation Med 25:407–411
NASA Technical Report (2010) Birthplace of America’s Missile and space activity. Report # 449089
NASA Press Release (2022) Life beyond earth: effect of space flight on seeds with improved nutritional value | science mission directorate. https://science.nasa.gov/biological-physical/investigations/life-beyond-earth
Odeh R, Guy CL (2017) Gardening for therapeutic people-plant interactions during long-duration space missions. Open Agric 2:1–13
Paul A-L, Daugherty CJ, Bihn EA, Chapman DK, Norwood KLL, Ferl RJ (2001) Transgene expression patterns indicate that spaceflight affects stress signal perception and transduction in Arabidopsis. Plant Physiol 126:613–621
Paul A-L, Levine HG, McLamb W, Norwood KL, Reed D, Stutte GW, William Wells H, Ferl RJ (2005) Plant molecular biology in the space station era: utilization of KSC fixation tubes with RNAlater. Acta Astronaut 56:623–628
Paul A, Wheeler RM, Levine HG, Ferl RJ (2013) Fundamental plant biology enabled by the space Shuttle. Am J Bot 100:226–234
Porterfield DM (2002) The biophysical limitations in physiological transport and exchange in plants grown in microgravity. J Plant Growth Regul 21:177–190
Poulet L, Doule O (2014) Greenhouse automation, illumination and expansion study for mars desert research station. 65th International Astronautical Conference, Toronto, Canada, p 11
Poulet L, Zeidler C, Bunchek J, Zabel P, Vrakking V, Schubert D, Massa G, Wheeler R (2021) Crew time in a space greenhouse using data from analog missions and Veggie. Life Sci Space Res 31:101–112
Tepfer D, Leach S (2017) Survival and DNA damage in plant seeds exposed for 558 and 682 days outside the International Space Station. Astrobiology 17:205–215
Schuerger AC, Amaradasa BS, Dufault NS, Hummerick ME, Richards JT, Khodadad CL, Smith TM, Massa GD (2021) Fusarium oxysporum as an Opportunistic Fungal Pathogen on Zinnia hybrida Plants Grown on board the International Space Station. Astrobiology 21:1029–1048
Stout SC, Porterfield DM, Briarty LG, Kuang A, Musgrave ME (2001) Evidence of Root Zone Hypoxia in Brassica rapa L. grown in microgravity. Int J Plant Sci 162:249–255
Stutte GW, Monje O, Wheeler RM (2012) Plant science in reduced gravity: lessons learned. ASGSR, New Orleans, p 13
Sugimoto M, Oono Y, Gusev O, Matsumoto T, Yazawa T, Levinskikh MA, Sychev VN, Bingham GE, Wheeler R, Hummerick M (2014) Genome-wide expression analysis of reactive oxygen species gene network in Mizuna plants grown in long-term spaceflight. BMC Plant Biol 14:4
Summerlin LB (1977) Skylab, classroom in space. NASA Special Public 19770022245:195
Sychev VN, Levinskikh MA, Gostimsky SA, Bingham GE, Podolsky IG (2007) Spaceflight effects on consecutive generations of peas grown onboard the Russian segment of the International Space Station. Acta Astronaut 60:426–432
Wheeler RM (2017) Agriculture for space: people and places paving the way. Open Agric 2:14–32
Wheeler RM, Fitzpatrick AH, Tibbitts TW (2019) Potatoes as a crop for space life support: effect of CO2, irradiance, and photoperiod on leaf photosynthesis and stomatal conductance. Front Plant Sci 10:1632
Wheeler RM, Mackowiak CL, Stutte GW, Sager JC, Yorio NC, Ruffe LM, Fortson RE, Dreschel TW, Knott WM, Corey KA (1996) NASA’s biomass production chamber: a testbed for bioregenerative life support studies. Adv Space Res 18:215–224
Wheeler RM, Mackowiak CL, Yorio NC, Sager JC (1999) Effects of CO2 on stomatal conductance: Do stomata open at very high CO2 concentrations? Ann Bot 83:243–251
Wheeler RM, Strayer RF (1997) Use of bioregenerative technologies for advanced life support: some considerations for BIO-Plex and related testbeds. Technical Memorandum 113229
Wolff SA, Coelho LH, Zabrodina M, Brinckmann E, Kittang A-I (2013) Plant mineral nutrition, gas exchange and photosynthesis in space: a review. Adv Space Res 51:465–475
Zabel P, Bamsey M, Schubert D, Tajmar M (2016) Review and analysis of over 40 years of space plant growth systems. Life Sci Space Res 10:1–16
Zea L, Piper SS, Gaikani H, Khoshnoodi M, Niederwieser T, Hoehn A, Grusin M, Wright J, Flores P, Wilson K et al (2022) Experiment verification test of the Artemis I ‘Deep Space Radiation Genomics’ experiment. Acta Astronaut 198:702–706
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Reed, D.W., Vanden Bosch, C.A. Engineering Perspectives of Growing Plants in Space. J Indian Inst Sci 103, 797–805 (2023). https://doi.org/10.1007/s41745-023-00369-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41745-023-00369-6