Abstract
Among the advantages of the index of average taxonomic distinction is that of generating a statistical reference framework to contrast the values observed from simple lists of species. This property is important for environmental monitoring studies, since it does not require control sites or few disturbed sites to know the environmental quality of a locality, which implies a decrease in operating costs and time savings. However, little research has been done on the effect of changes in the length (number of species) of species lists on the result and interpretation of this index. In this study, the effect of different lengths of species lists on this index was analyzed. Three species lists of benthic bivalve mollusks from the Cuban marine shelf were compiled: two local and one national. Our results indicated that changes in the length of the species lists affected the results and the interpretation of the average taxonomic distinctness index. In this sense, for the use of a local or national species list it is important to specify the objective of the research to be carried out. We also found that the combined use of species lists of different lengths can indicate the degree of deterioration of the sites analyzed, which is of vital importance for environmental monitoring studies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Changes and degradation in estuarine and coastal marine ecosystems is a widespread phenomenon worldwide as a result of human activities and climate change (Zhou et al. 2012). Various approaches have been taken to assess environmental quality, ranging from measurements that use the richness and relative abundance of species (e.g. Shannon index) to a variety of ecological índices (e.g. BOPA, BITS, BENTIX) that are structured with species tolerant or sensitive to particular environmental perturbations (Mistri and Munari 2008; Birk et al. 2012). However, both approaches present limitations for environmental monitoring purposes.
Measures that use the richness and relative abundance of species are strongly affected by sample size, sampling effort and natural environmental variability and do not show a monotonic response to anthropogenic disturbances (Leonard et al. 2006). Furthermore, the values of these measures must be compared with reference conditions (control or undisturbed sites) to univocally identify the state of an ecosystem (Bevilacqua et al. 2011). In the case of ecological indices, their limitation lies in their sensitivity to natural variability, their restricted use to certain types of habitat and sources of disturbance, which prevents their application to a wider array of environmental conditions (Salas et al. 2006; Tataranni and Lardicci 2010). To make up for all these limitations new diversity indices based on the taxonomic relationships of species have been created, within which average taxonomic distinctiveness may be a promising approach.
Clarke and Warwick (1998) created the average taxonomic distinctiveness index (Delta+, ∆+), which incorporates the identity of the species within a sample, calculating the average taxonomic distance at which two individuals are related to each other within a Linnean hierarchical classification taxonomic tree. This index requires a species list (or inventory of species) for its calculations, which makes it less dependent on the sampling effort (Clarke and Warwick 1999, 2001). In addition, it has low sensitivity to natural variability, which makes it easier to separate the effects caused by human activity. (Warwick and Clarke 1998; Leonard et al. 2006). The deviations from the observed values can be evaluated by comparing them with a range of expected values whose distribution is built with 95% confidence limits. This expected distribution (also called statistical reference framework) is obtained through a random sampling without replacement, obtaining a set of subsamples of m species from the lists of reference species. This ∆+ property does not require obtaining data on the abundance of species in sites less disturbed to identify the state of an ecosystem, saving time and resources. These properties allowed the application of ∆+ in studies assessing human and natural impacts on different environments and organisms (e.g. Arvanitidis et al. 2005; Ronowicz et al. 2018; Floerl et al. 2009; Capetillo-Piñar et al. 2016). To monitor the success of conservation and/or restoration measures (Reyes-Bonilla and Alavarez-Filip 2008; Stobart et al. 2009; Espinosa 2018; Achitte-Schmutzler et al. 2022), and in the analysis of diversity patterns in species assemblages, to prioritize conservation areas from an integrative perspective (Esqueda-González et al. 2014, Canales-Gómez et al. 2021).
An interesting but under researched aspect is the procedure to select the list of species that, after the randomization test, will serve as a reference to contrast with the observed values. Clarke and Warwick (1999) suggested that several lists of species could be used sensibly: local, global, geographic provinces, biogeographic, or the combined list of species of all studies carried out in a locality. In this context, they suggest that the choice of the species list is of secondary importance because it is not incorporated in the calculation of the indices, it simply provides a context for comparison. However, the species list (inventory) of a locality is the result of the combination of natural and human factors occurring in time and space. In this sense, extirpation and invasion of species by human disturbances, loss or deterioration of habitats, habitat types and extreme natural events (e.g. hurricanes), are known to affect the phylogenetic (taxonomic) structure of species associations (Ives and Helmus 2010; Bevilacqua et al 2011; Capetillo-Piñar et al. 2015, 2016; Morelli et al. 2016). Therefore, it is to be expected that such factors influence ∆+ values and consequently their interpretation.
After using lists of species with different lengths (local, regional and / or national), Bates et al. (2005) and Bevilacqua et al. (2012) show a certain effect of these in the results and interpretation of the average taxonomic distinctiveness, which refuted the suggestions of Clarke and Warwick (1999). These authors obtained different results that allowed them to corroborate the assumption that the average taxonomic distinctiveness is affected by natural variability and that it has little sensitivity to human impacts, regardless of the length of the lists of species used to calculate the reference condition. These results demonstrated the need to know the performance of ∆+ with respect to the statistical reference framework obtained from the list of species, given its importance in the significance of the values of this index in the context of environmental impact assessment and its cost-effectiveness and saving time in carrying out these studies that are so necessary for decision-making by the authorities.
Bates et al. (2005) have raised questions as to whether and how much the selection of the lists of reference species with different lengths (number of species) can affect the results and interpretation of the average taxonomic distinctiveness. In this sense, to know whether a species list is representative of a locality or biogeographic region is of great interest because of the implications on the results of environmental monitoring and coastal zone management.
Bevilacqua et al. (2021) recorded a higher percentage of studies on taxonomic distinctiveness indices (predominatly ∆+) in the marine environment, which focused their analyses primarily on benthic marine mollusks (gastropods and bivalves).assemblages. Mollusks are a focal group for biodiversity studies in the marine environment, as them can represent an approximation to the total biodiversity of megazoobenthic organisms in a locality (Alcolado and Espinosa 1996). In addition, can be used to evaluate the status and management strategies in nature reserves and areas of conservation interest, as them are indicators of species richness in tropical and subtropical ecosystems (Espinosa and Ortea 2001). Bivalve mollusks are the second class with the highest number of species in this phylum. Since these organisms are numerically dominant in various habitat types and have a relative connection to environmental factors (e.g., sediment composition and particle size, water turbidity), can be used as bioindicators to characterize certain habitats and to assess and monitor the quality of the marine environment. (Espinosa 1992; Baqueiro-Cárdenas et al. 2007).
In the present study, the incidence of species list length change on ∆+ was tested through the representativeness of benthic marine bivalve mollusk species lists from two localities of the Cuban marine shelf with certain heterogeneity of soft-bottom habitats. It was also determined whether the average taxonomic distinctiveness is robust to natural variability, taking as a basis the different soft-bottom habitats present in each locality.
Materials and Methods
Study Localities
Cárdenas Bay (CB)
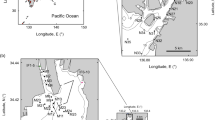
Cárdenas Bay is located between 23° 01´N and 81°18´W and 23°13´N and 80°42´W (Fig. 1). This bay is open and covers an area of 104 km2 with an average depth of 4 m (Álvarez and Quintana 1988). It has few freshwater tributaries, so coastal runoff is determined by the annual rainfall regime (Lluis-Riera 1981). The southern coastline of the city of Cárdenas is affected by 19 pollutant sources, whose main source of contamination is the organic matter produced by industrial and domestic wastewater from this city (González 1991). Its seabeds are muddy in its inner zone and sandier towards its outer zone, with the presence of the seagrass Thalassia testudinum (Banks and König 1805) (del Valle et al. 2008).
Batabanó Gulf (BG)
The BG is a semi-closed body of water located in the southwestern region of Cuba between 21°25' and 22°41' N and 80°52'and 84°00' W, (Fig. 1b). It has an approximate area of 21,305 km2, with an average depth of 6 m (Cerdeira et al. 2008). Most of the human settlements are confined on the North coast where pollutants from industrial, agricultural and domestic waste are produced and discharged into the interior of the gulf (Perigó et al. 2005). Its seabed is varied, with muddy and sandy bottoms without vegetation and/or a combination of these (sandy-muddy and muddy-sandy) with or without vegetation.In the seabed covered by vegetation, the species that dominant is the marine phanerogam T. testudinum with different densities and coverage (Cerdeira et al. 2008).
Obtaining the Databases for Each Study Localities
The details for each locality (e.g., number of sites and sampling gear, replicas, year or sampling period, number of species, nature of the data and origin of the data) are shown in Table 1. For the location of the sites see Fig. 1.
The systematic list of bivalve species for 2004 and 2007 was obtained through two research cruises. Several sampling sites from the years 2004 and 2007 were located in the same sites sampled in the period 1981–1985, while others were located in areas close to these in order to locate them in the same habitat to establish comparisons on a temporal scale.
The sampling of bivalves was carried out using epibenthic harrow and applied the methodology described by Alcolado (1990). The bivalves were identified to the species level, according to the criteria of Espinosa and Ortea (2001), Redfern (2013), Espinosa et al. (2005), and Mikkelsen and Bieler (2008). The systematic arrangement was based on Espinosa and Ortea (2012) and the WoRMS Editorial Board (2002).
Data Analysis
The Average Taxonomic Distinctness Index (Delta +, ∆ +) of the samples was estimated from the equation proposed by Clarke and Warwick (1998):
where: Wij is the taxonomic weight and is related to the taxonomic distance that exists through the Linnaean classification tree of any pair of individuals, the first being for species and the second for species j and S the total number of species in the sample. The taxonomic weight (Wij) increases as the taxonomic separation between the species is greater. In this work the values were given in such a way that there would be a constant increase from one level to another, as proposed by Clarke and Warwick (1999). The taxonomic categories used in this study were species, genus, family, order, subclass, and class.
Incidence of the Change in the Length of the Lists of Species on ∆+
The incidence of changes in the reference species lists on the average taxonomic distinctness was verified by estimating the index (∆ +) using three species lists: two lists formed from the species registered in each study location (local lists) and a list constructed with all the species of bivalves registered in other locations of the Cuban marine shelf (national list). This last list, which was obtained from Espinosa (2007), includes of all the bivalve species, described to date, that can be found on the Cuban continental shelf. The species lists for each locality presented a total of 41 (Supplementary material 1) species for CB, 86 for BG (Supplementary material 1) and 298 for the national list (Espinosa 2007).
Two analyses were performed to determine the effect of changes in the length of the reference species lists on ∆+. The first consisted in testing the null hypothesis that the bivalve associations present in each locality are taxonomically structured as if they were a random selection of the bivalve associations present in the national species list. In this case, both lists of species served as a reference to contrast the values of ∆+ observed in each locality. For this analysis, histograms (frequency of ∆+ values) obtained from 1000 random selections of m samples from the local and national species lists were constructed. These histograms reflected an expected distribution of ∆+ values, which was used to establish the contrasts with the observed values of this index in both locations.
The second analysis consisted of testing the null hypothesis that the bivalve species present in each habitat have a taxonomic structure representative of any of the samples obtained at random from the lists reference species of each locality and of the national. Ten types of soft-bottom habitats were identified between the two locations, which were characterized in three groups, according to Alcolado (1990) and Cerdeira et al. (2008). These were: a) Sandy habitat (S); b) muddy bottoms, with four types: 1- Muddy habitat (M), 2- Muddy habitat with organic contamination (MoM), 3- Disturbed muddy habitat by different anthropic activities (DM) and 4- Clay mudd habitat (CM) and c) bottoms covered with vegetation (Seagrass), with five types: 1- Sandy seagrass habitat (SS), 2- Muddy seagrass habitat (SM), 3- Muddy sand seagrass habitat (SSM), 4- Sandy muddy seagrass habitat (SMS) and 5—Seagrass in sediment deposited on rocky habitat (SR).
To determine the existence of significant differences in ∆+, the bivalve samples were grouped according to the type of habitat present in the sampling sites of each locality. The ∆+ values were calculated considering the species found in each habitat, contrasting with the expected values derived from the reference local and national lists of species.
To test the deviations of the ∆+ values observed in the different habitats of each locality, a probability funnel was built with 95% confidence contours (Clarke and Warwick 1998), from the reference lists local and the national species list. This probability funnel indicated the expected variance of the ∆+ values. If the observed values of ∆+ for each habitat are within the probability funnel, then the null hypothesis is accepted. The ∆+ values were calculated using the DIVERSE routine of the PRIMER v7 and PERMANOVA add on program (Clarke and Gorley 2015).
A permutational multivariate analysis of variance (PERMANOVA) (Anderson et al. 2008) was carried out on the Bray Curtis similarity matrix with the ∆+ values of the bivalve associations registered in the two locations separately. This was done in order to know the effects caused by habitat and period factors on ∆+. For CB, the habitat was used as a factor, which was divided into three groups: sandy habitat (one level), muddy habitat (three levels) and seagrass habitat (two levels). The probabilities (p) associated with the Pseudo-F value were obtained with 999 Monte Carlo permutations, under a model of unrestricted permutations. For BG, the factors periods and habitats were analyzed. The period factor (temporal) was fixed and with two levels, since the samples were taken in two different periods (decade of the 80' and 00'). The habitats factor (eight levels and nested in period) was considered as random and was used to know the incidence of spatial heterogeneity of each locality. The probabilities (p) associated with the Pseudo-F value were obtained under a reduced null model with 999 permutations (Anderson et al. 2008). All these analyzes were carried out using the PERMANOVA routine of the PRIMER v7 and PERMANOVA add on program (Clarke and Gorley 2015).
Due to the fact that biomass information was available for CB, a comparison analysis of biomass and abundance curves (ABC) proposed by Warwick (1986) in the habitat of Mud contaminated with organic matter (MoM) was carried out, to determine the level of impact (or stress) that the contamination had on the bivalve associations that inhabited it. This analysis was performed using the DOMINANCE Plot routine of the PRIMER v7 and PERMANOVA add on program (Clarke and Gorley 2015).
Results
Cárdenas Bay
The observed values of ∆+ of CB bivalve association when compared with the national species list fell outside their respective expected distribution (Fig. 2b). This was not the case for the local list (Fig. 2a). The result shows that the null hypothesis was rejected.
Histograms of the simulated ∆+ values (999 simulations) calculated from the list of reference species (a) local and (b) national. The length of the local species lists (n = 41) and the national (n = 298), correspond to the number of species observed in Cárdenas Bay and on the marine shelf respectively. The observed values of ∆+ for the entire Cárdenas Bay bivalve association are presented in the figures. The dashed line indicates where the observed value of ∆+ was located
The analysis with the list of reference species of this locality resulted that all the ∆+ values that characterized the habitats fell within the probabilistic limits at 95% of their respective tunnel (Fig. 3a). For the national list, habitat SS was the only one that fell outside and below the probability limit lower than 95% (Fig. 3b).
Probabilistic funnels of 95% of ∆+ from the list of reference species: local (a) and national (b) and the calculated values of the associations of bivalve mollusks recorded in the soft bottom habitats of Cárdenas Bay. S sandy habitat, M muddy habitat, CM clay muddy habitat, MoM muddy habitat with organic contamination, SS sandy seagrass habitat SM muddy seagrass habitat
The ∆+ values of the mud habitat with organic matter (MoM), were located within the expected limits of distribution for the two lists of reference species used in the analysis (Figs. 3a, b). This result was unexpected because these indices are supposed to be sensitive in detecting negative impacts on the environment.
To analyze the effect of organic pollution that this habitat presented, biomass abundance curves were constructed (Fig. 4). The result showed that the abundance curve did not remain above the biomass curve, and there was an interchange of both curves in a certain range of species, showing a moderate level of impact on the bivalve associations in this habitat.
The PERMANOVA test did not detect significant differences in the ∆+ values of the different habitats of this locality for any of the two lists of species used as a reference frame (Table 2), which shows that the habitats did not constitute a source of variation for the ∆+ of this locality.
Batabanó Gulf
The observed values of ∆+ of the BG bivalve association when compared with the local species list fell outside their respective expected distributions (Fig. 5a). This was not the case for the national list, whose value fell within their expected distributions (Fig. 5b). This result indicated that the null hypothesis that ∆+ of BG is equal to any of the samples (species associations) drawn at random from the local species list was rejected, except in comparisons to the national list.
Histograms of the simulated values of ∆+ (999 simulations) calculated from the local (a) and national (b) reference species list. The length of the local species lists (n = 86) and the national (n = 298), correspond to the number of species observed in the Batabanó Gulf locality and on the marine shelf respectively. The observed values of ∆+ for the entire Batabanó Gulf bivalve association are presented in the figures. The dashed line indicates the place where the observed value of ∆+ was located
The ∆+ values of the habitats present in BG, except for muddy sand seagrass (SSM) from the period 1981–1985, disturbed mud (DM) studied in 2004 and the sandy seagrass (SS) analyzed in 2007, fell within the probabilistic contour at 95% of their corresponding probabilistic funnels regardless of the reference species list (Fig. 6).
Probabilistic funnels of 95% of ∆+ from the lists of reference species local and national and the calculated values of the associations of bivalve mollusks recorded in the soft bottom habitats of the Batabanó Gulf. S: sandy habitat, M: muddy habitat, DM: disturbed muddy habitat, SR: seagrass in sediment deposited on rocky habitat, SMS: sandy muddy seagrass habitat, SM: muddy seagrass habitat, SS: sandy seagrass habitat, SSM: muddy sand seagrass habitat. The numbers that appear above the habitat symbols refer to the period / year in which they were sampled. 1: 1981–1985 period, 2: 2004 year, 3: 2007 year
In the case of SSM habitat (from the period 1981–1985), ∆+ had higher values than expected according to the local list (Fig. 6a). The values of ∆+ in the DM habitat (studied in 2004) were lower than those expected in the two lists of reference species (Fig. 6a, b), while for the SS habitat (analyzed in 2007), ∆+ had a lower than expected value in the local reference list (Fig. 6a).
The PERMANOVA test indicated that the period had a significant influence on the variations of ∆+ of the bivalve associations recorded in the habitats of the BG, for the two lists of reference species (Table 3). In this case, the habitats had no effect on the variations of ∆+.
Discussion
The length of the species list depends on the size of the study region, so it is to be expected that the greater the spatial extent, the greater the length of the list. Furthermore, the composition of the species list is related to the environmental (or temporal) conditions. In this context, the differences observed in the contrasting ∆+ values of the bivalve associations located in the BG and CB with the two species lists due to the spatial extension and the ecological characteristics of each locality, which were reflected in the length and composition of their respective species lists.
The ∆+ values of each locality were expected to be representative of their local species list, a result that did not entirely coincide. This suggests that the ecological conditions (type and quantity of habitats and their intrinsic characteristics such as their extension, environmental conditions and level of anthropic impact) of each locality may have conditioned the responses of ∆+. In addition, there are underlying temporal effects (BG was sampled in two different decades (years 80' and 00') which may had influenced the length and specific composition of the species lists. Therefore, the compilation of a species list used as a reference condition to contrast the observed values of ∆+, implies the environmental conditions (natural and anthropic) of a locality, which shows the crucial importance that it has for the evaluation of impacted areas (Bevilacqua et al. 2012).
Aggregating sites with different numbers (composition) of species to obtain a more complete reference list may affect the significance of ∆+ in two directions. If the species added to the list are identified in the taxonomic tree, the mean values of ∆+ will not be affected (Ceshia et al. 2007). Consequently, taxonomic structures will co-exist and be identified with this list of references. However, if the opposite is true, slight increases in the variance of the mean ∆+ values will be observed, changing the width of the confines of the probabilistic tunnel and consequently the interpretation of the index (Bates et al. 2005). Both directions were observed in this study, which supports the criteria issued by these authors. For example, for the BG, the presence of a taxonomic structure that does not belong to its local list suggests the existence of other taxonomic structures (substructures) independent of this locality. The opposite was true for CB, where the taxonomic structure of bivalves was representative of their local list.
The taxonomic substructures in the BG could have originated from several causes such as: loss of quality and/or areas of benthic habitats (Cerdeira et al. 2008), increased salinity and organic pollution in areas close to its northern coast (Piñeiro 2006; Perigó et al. 2005) and the effects caused by hurricanes on its physical (e.g., increased water turbidity) and biological (e.g., decreased taxonomic diversity in benthic mollusks associations) environment (Capetillo-Piñar et al. 2016; Betanzos-Vega et al. 2019). In addition, several authors (Arias-Schreiber et al. 2008; Lopeztegui and Capetillo 2008; Capetillo-Piñar et al. 2015; Lopeztegui and Martínez 2020) have demonstrated the effects on benthic marine biota induced by the causes described above. In contrast to BG, in CB, low intensity affectations were detected on its southern coast, where a muddy bottom devoid of vegetation (seagrasses) was observed, due to the discharge of wastewater (Herrera and Espinosa 1988). Also, having a smaller spatial extent and habitat heterogeneity than the BG provides some confidence that the observed taxonomic structure is part of the reference list for this locality. This result was similar to that obtained by Ceshia et al. (2007), when assessing the biodiversity of the macroalgal flora in in the Gulf of Trieste (Northern Adriatic Sea).
By analyzing that for CB, at the local level, no significant (low or higth) ∆+ values were detected in the habitats studied; the robustness of ∆+ with respect to this type of natural variability is evident. The observed changes were due to length variation in the reference species lists and not to the effect of habitats. This result is consistent with the idea of Clarke and Warwick (1999) that some hábitats (naturally) may have low ∆+ values, but these are usually within the 95% confidence limits of the simulated distribution, contrary to what happens when are affected by human activities.
An unexpected result was the representativeness of ∆+ of the muddy habitat with organic contamination (MoM) in the two lists of reference species. For this habitat, a significant reduction in the value of ∆+ was expected, since it has been suggested that pollution significantly reduces its values (Clarke and Warwick 2001; Moulliot et al. 2005; Tan et al. 2010; Xu et al. 2011; Warwick and Clarke 1998; Warwick and Light 2002). However, although there is organic contamination, the ABC curves do not corroborate a relevant negative impact on the bivalve community.
The MoM habitat was located at a distance of 1 km from the coast, where it seems that organic contamination was more attenuated, since a survey carried out in an area closer to the coast, resulted in a virtual absence of macrobenthic life and the presence of thanatocenosis (Espinosa 1992). In this context, the ∆+ response demonstrated the existence of a low environmental impact, which induced sublethal changes in the taxonomic structure of the association of bivalves settled in the MoM habitat. This result supports the criteria of Bevilacqua et al. (2012) that low levels of environmental impact can generate changes in the taxonomic structure of the associations of species located in disturbed sites and these changes are not detected by ∆+.
The result obtained in the disturbed muddy habitat (DM) of the BG studied in 2004 was due to it being in an area affected by organic ammonia pollution (Montalvo et al. 2000; Perigó et al. 20002005) and by changes in sediment composition due to the decrease in terrigenous input (Guerra et al. 2005). The combination of these factors, magnified over time, could lead to this habitat being particularly present in BG, a fact that has been corroborated in this work. In this habitat, the species Chione cancellata (Linnaeus 1767) and Eurytellina nitens (Adams 1845) showed the highest abundances and distribution. These species can tolerate high salinities (e.g., C. cancellata) and high values of organic matter (e.g., E. nitens), according to Espinosa (1992). Other studies have reported similar results in this area (Capetillo-Piñar et al. 2015), as well as in this habitat type, which is found on the north coast of BG (Lopeztegui and Capetillo 2008; Lopeztegui and Martínez 2020).
Warwick and Clarke (1995, 1998) stated that ∆+ is not only affected by environmental pollution or other anthropogenic impacts, but also by the environmental characteristics of a given location or habitat. In this context, high and significant values of ∆+ were found in areas less influenced by anthropic effects, for example some seagrasses, which may be related to their intrinsic characteristics and not to human disturbances. This result supports the criterion of the sensitivity of ∆+ to natural variability, which would limit its potential to evaluate environmental quality (Bevilacqua et al. 2011, 2012). However, the fact that this same habitat, when analyzed in 2004 and 2007, fell within the confidence limits of the national list of reference species, casts doubt on the sensitivity of ∆+ to natural environmental variability. In this sense, it is necessary to consider the effect of variation in ∆+ values associated with natural and anthropogenic factors (or combination of these) over time, because of their implications for environmental monitoring studies and subsequent management measures in a locality.
The BG has been affected by the impact of hurricanes, which have increased in intensity and frequency in this locality from the 1980s to the 2000s (Puga et al. 2013). In this context, the low and significant values of ∆+ in the sand-muddy seagrass habitat of the year 2007 (high cyclonic activity) with respect to the period 1981- 1985 (low cyclonic activity), may be due to the effects of hurricanes and not to the characteristics of this habitat. Capetillo-Piñar et al. (2016) recorded a similar result. The above supports the criterion that ∆+ has low sensitivity to natural environmental variability (Hong et al. 2010; Leonard et al. 2006; Munari et al. 2009; Zhou et al. 2012).
The reference list may not adequately represent the taxonomic structure of the community of interest if it is too small (Bates et al. 2005). In this regard, it is suggested to use (broader) regional species lists that incorporate more species irrespectively of habitat preferences or to restrict reference lists to species inhabiting specific habitats so that deviations from ∆+ values correspond to human disturbances (Bevilacqua et al. 2021). However, in the present study it was shown that using local and regional species lists, ∆+ was able to discriminate anthropogenic effects. For example, the low and significant ∆+ values of the DM habitat studied in the period 2000 in the BG, obtained from its local (small) and national (larger) list, were due to human disturbance. In addition, the fact that this habitat type had the lowest values than expected in the national reference list confirmed that its level of disturbance was the most severe between the two locations studied. This demonstrated that the use of several species lists with different lengths can be used to determine the degree of severity of effects caused by human activities in a locality, which is of interest in environmental impact and monitoring studies.
Therefore, it is recommended to use the species list of a locality to know the environmental quality of a particular area, and even more so if species lists from several epochs are used. The results of this study provided evidence that each locality is a unique environmental unit, with its own natural and anthropogenic characteristics. These properties may or may not be present at a larger spatial scale, thus decreasing or increasing the discriminatory potential of ∆+. However, the use of a local reference species list and a larger spatial extent would help to determine the degree of severity of disturbed sites, giving a practical utility for decision making, corrective or mitigation measures for the protection or conservation of an area.
References
Achitte-Schmutzler HC, Avalos G, Oscherov EB (2022) Diversidad taxonómica (Araneae) en ambientes heterogéneos del sitio Ramsar Humedales Chaco, Argentina. Caldasia. https://doi.org/10.15446/caldasia.v44n1.83581
Adams CB (1845) Specierum novarum conchyliorum, en Jamaica repertorum, sinopsis. Proceedings of the Boston Natural History Society 2:1–17. Available online at https://www.biodiversitylibrary.org/page/9490685. Reviewed: 28 Mar 2021
Alcolado PM (1990) El bentos de la macrolaguna del Golfo de Batabanó, 161 pp. Editorial Academia, La Habana
Alcolado PM, Espinosa J (1996) Empleo de las comunidades de moluscos marinos de fondos blandos como bioindicadores de la diversidad del megazoobentos y de la calidad ambiental. Iberus 14(2):79–84
Álvarez M, Quintana M (1988) Caracterización sedimentológica preliminar de la Bahía de Cárdenas. Reporte De Investigación Del Instituto De Oceanología 3:1–16
Anderson MJ, Gorley RN, Clarke KR (2008) PERMANOVA+ for PRIMER: Guide to Software and Statistical Methods, 214 pp. PRIMER-E, Plymouth
Arias-Schreiber M, Wolf M, Cano M, Martínez-Daranas B, Marcos Z, Hidalgo G, Castellano S, del Valle R, Abreu M, Martíne JC, Díaz J, Areces A (2008) Changes in benthic assemblages of the Gulf of Batabanó (Cuba)- results from cruises undertaken during 1981–85 and 2003–04. Pan America Journal Aquatic Sciences 3(1):49–60
Arvanitidis C, Chatzigeorgiou G, Koutsoubas D, Kevreskidis T, Dounas C, Eleftheriou A, Koulouri P, Mogias A (2005) Mediterranean lagoons revisisted: weakness and efficiency of the rapid biodioversity assessment techniques in a severely fluctuating environment. Helgol Mar Res 59:177–186. https://doi.org/10.1007/s10152-005-0216-8
Banks J, Konig KD (1805) Thalassia testudinum. In: Annals of Botany (Eds)König & Sims V(2): 96. Source: The World Checklist of Vascular Plants (WCVP)
Baqueiro-Cárdenas ER, Luz B, Goldaracena-Islas CG, Navarro JR (2007) Los moluscos y la contaminación. Una revisión. Rev Mex Biodivers 78(Suplemento, Octubre):1–7 Recuperado de. https://repositorio.unam.mx/contenidos/4109425. Reviewed: Feb 2021
Bates CR, Saunders GW, Chopin T (2005) An assessment of two taxonomic distinctness indices for detecting seaweed assemblage responses to environmental stress. Bot Mar 48:231–243. https://doi.org/10.1515/BOT.2005.034
Betanzos-Vega A, Capetillo-Piñar N, Lopeztegui-Castillo A, Garcés-Rodríguez Y, Tripp-Quezada A (2019) Parámetros meteorológicos, represamiento fluvial y huracanes. Variaciones en la hidrología del golfo de Batabanó, Cuba. Rev Biol Mar Oceanogr 54(3):308–318. https://doi.org/10.22370/rbmo.2019.54.3.2024
Bevilacqua S, Anderso MJ, Ugland KI, Somerfield PJ, Terlizzi A (2021) The use of taxonomic relationships among species in applied ecological research: Baseline, steps forward and future challenges. Austral Ecol 46:950–964. https://doi.org/10.1111/aec.13061
Bevilacqua S, Sandulli R, Plicanti A, Terlizzi A (2012) Taxonomic distinctness in Mediterranean marine nematodes and its relevance for environmental impact assessment. Mar Pollut Bull 64:1409–1416. https://doi.org/10.1016/j.marpolbul.2012.04.016
Bevilacqua S, Fraschetti S, Musco L, Guarnieri G, Terlizzi A (2011) Low ensitiveness of taxonomic distinctness indices to human impacts: Evidences across marine benthic organisms and habitat types. Ecol Ind 11:448–455. https://doi.org/10.1016/j.ecolind.2010.06.016
Birk S, Bonne W, Brucet S, Courrat A, Poikane S, Solimini A, van de Bund W, Zampoukas N, Hering D (2012) Three hundred ways to assess Europe´s Surface waters: An almost complete overview of biological methods to implement the water framework directive. Ecol Ind 18:31–41. https://hal.inrae.fr/hal-02596654. Reviewed: Jan 2021
Canales-Gómez E, Peña-Joya KE, Téllez-López J (2020) Diversidad taxonómica alfa y beta del ensamble de peces continentales de la Cuenca del río Ameca, México. Rev Mex Biodivers. https://doi.org/10.22201/ib.20078706e.2021.92.3408
Capetillo-Piñar N, Espinosa J, Tripp-Valdez A, Tripp-Quezada A (2016) The impact of cyclonic activity during 1981–1985 and 2004–2009 on taxonomic diversity of mollusks in the Gulf of Batabanó, Cuba. Hidrobiológica 26(1):121–131. https://doi.org/10.24275/uam/izt/dcbs/hidro/2016v26n1/Tripp
Capetillo-Piñar N, Villalejo-Fuerte MT, Tripp-Quezada A (2015) Distinción taxonómica de los moluscos de fondos blandos del Golfo de Batabanó. Cuba Latin American Journal of Aquatic Research 43(5):856–872. https://doi.org/10.3856/vol43-issue5-fulltext-6
Cerdeira S, Lorenzo-Sánchez S, Areces-Molleda A, Martínez-Bayón C (2008) Mapping of the spatial distribution of benthic habitats in the Gulf of Batabanó using Lansat-7 images. Cienc Mar 34(2):213–222
Ceshia C, Falace A, Warwick RM (2007) Biodiversity evaluation of the macroalgal flora of the Gulf of Trieste (Northern Adriatic Sea) using taxonomic distinctness indices. Hydrobiologia 580:43–56. https://doi.org/10.1007/s10750-006-0466-8
Clarke KR, Warwick RM (1998) A taxonomic distinctness index and its statistical properties. J Appl Ecol 35:523–531. https://doi.org/10.1046/j.1365-2664.1998.3540523.x
Clarke KR, Warwick RM (1999) The taxonomic distinctness measure of biodiversity: weighting of step lengths between hierarchical levels. Mar Ecol Prog Ser 184:21–29. https://doi.org/10.3354/meps184021
Clarke KR, Warwick RM (2001) A further biodiversity index applicable to species lists: variation in taxonomic distinctness. Mar Ecol Prog Ser 216:265–278. https://doi.org/10.3354/meps216265
Clarke KR, Gorley RN (2015) PRIMER v7: User manual/tutorial, 296 pp. PRIMER-E, Plymouth
del Valle RG, Abreu Pérez M, Rodríguez R, Solís-Marín FA, Laguarda-Figueras A, Durán González AL(2008) Equinodermos (Echinodermata) del occidente del Archipiélago Sabana-Camagüey, Cuba. Rev Biol Trop 56 (3): 19–35. https://doi.org/10.15517/RBT.V56I3.27022
Espinosa J (1992) Sistemática y Ecología de los Moluscos Bivalvos Marinos de Cuba. Tesis presentada en opción al grado científico de Doctor en Ciencias Biológicas. Academia de Ciencias de Cuba. Instituto de Oceanología. La Habana, Cuba. 125 p
Espinosa J, Ortea J, Caballero M, Moro L (2005) Moluscos marinos de la península de Guanahacabibes, Pinar del Río, Cuba, con la descripción de nuevos taxones. Avicennia 18:1–84
Espinosa J, Ortea J (2001) Moluscos del Mar Caribe de Costa Rica: desde Cahuita hasta Gandoca. Avicennia 4:77
Espinosa J, Ortea J (2012) Inventario de los moluscos marinos de la Península de Guanahacabibes. En: Memorias del Proyecto: Fortalecimiento de la gestión del desarrollo integral y sostenible de la Península de Guanahacabibes, Reserva de Biosfera, Pinar del Río, Cuba. (Camacho, A., J., Baena, G., G. and Leyva, P.G. Eds.). Colaboración Cuba-Canadá. Editorial Científico- Técnica, La Habana, Cuba, 107–333
Espinosa J (2007) Moluscos. En: R. Claro (Ed.): La Biodiversidad Marina de Cuba. III Diversidad de organismos. Editorial Academia, La Habana, 46–51
Espinosa NA (2018) Cambios en la comunidad de peces arrecifales del Caribe Mexicano como medida para evaluar la efectividad de Areas Marinas Protegidas. Tesis presentada en opción al grado de Maestra en Ciencias Marinas y Costera con Mención en Manejo Sustentable. https://rep.uabcs.mx:80/handle/23080/250. Reviewed: July 2021
Esqueda-González MC, Ríos-Jara E, Galván-Villa CM, Rodríguez-Zaragoza FA (2014) Species composition, richness and distribution of marine bivalve molluscs in Bahia de Mazatlán, México. Zookeys 399: 49–69. https://doi.org/10.3897/zookeys.399.6256
Floerl O, Inglis GJ, Gordon DP (2009) Patterns of taxonomic diversity and relatedness among native and non-indigenous bryozoans. Divers Distrib 15(3):438–449. https://www.jstor.org/stable/20532114. Reviewed: May 2021
González H (1991) Heavy metals surveys in sediments of five important Cuban Bays. Biogeochemistry 14:113–128. https://doi.org/10.1007/BF00002901
Guerra R, Chávez ME, Hernández K, Tristá E (2005) Cambios sedimentarios en la cuenca marina sur de la provincia Habana. Revista Ciencias De La Tierra y El Espacio 1(8):35–44
Herrera A, Espinosa J (1988) Características de la fauna de bivalvos de la Bahía de Cárdenas. Cuba Reporte De Investigaciones Del Instituto De Oceanología 12:1–21
Hong Z, Chaik H, Zhinan EZ (2010) Taxonomic distinctness of macrofauna as an ecological indicator in laizhou bay and adjacent waters. Journal of Ocean of University of China. Oceanic and Coastal Sea Research 9:350–358. https://doi.org/10.1007/s11802-010-1717-x
Ives AR, Helmus MR (2010) Phylogeneticmetrics of community similarity. Am Nat 176:E28–E42. https://doi.org/10.1086/656486
Leonard DR, Clarke KR, Somerfield PJ, Warwick RM (2006) The application of an indicator based on taxonomic distinctness for UK marine biodiversity assessments. J Environ Manage 78:52–62. https://doi.org/10.1016/j.jenvman.2005.04.008
Linnaeus CV (1767) Systema naturae per regna tria naturae: clases secundum, ordines, géneros, especies, cum characteribus, differentiis, synomiis, locis. ed. 12. 1., Regnum Animale. 1 y 2. Holmiae [Estocolmo], Laurentii Salvii. pp. 533-1327. Available online at http://www.biodiversitylibrary.org/item/83650#5. Reviewed: 28 Mar 2021
Lluis-Riera M (1981) Condiciones hidrológicas de la plataforma nororiental de Cuba durante febrero de 1976. Archivo Científico Técnico, Instituto De Oceanología 161:1–32
Lopeztegui A, Martínez D (2020) 35 años de cambio en densidad y biomasa del megazoobentos del golfo de Batabanó, Cuba, e implicaciones para la langosta, Panulirus argus (Decapoda: Palinuridae). Rev Biol Trop 68(4):1346–1356. https://doi.org/10.15517/RBT.V68I4.41209
Lopeztegui A, Capetillo N (2008) Macrozoobentos como estimador del potencial alimentario para la langosta espinosa (Panulirus argus) en tres zonas al sur de Pinar del Río. Cuba Boletín Del Centro De Investigaciones Biológicas 42(2):187–203
Martínez-Estalella N, Alcolado PM (1990) Características generales de las comunidades de moluscos de la macrolaguna del Golfo de Batabanó. In: Alcolado PM (ed) El bentos de la macrolaguna del Golfo de Batabanó. Editorial Academia, La Habana, Cuba, pp 53–74
Mikkelsen PM, Bieler R (2008) Seashells of Southern Florida. Living marine mollusks oh the Florida Keys and adjacent regions. Bivalves, 503 pp. Princeton, University Press, New Jersey, USA
Mistri M, Munari C (2008) BITS: a smart indicator for soft-bottom, non-tidal lagoons. Mar Pollut Bull 56:587–599. https://doi.org/10.1016/j.marpolbul.2007.12.002
Montalvo JF, Perigó E, Espinosa J, García IA (2000) Prospección de variables hidroquímicas de calidad ambiental en la zona litoral entre el río Hatiguanico y Majana, costa suroccidental de Cuba. Contribución a La Educación y La Protección Ambiental 1:186–190
Morelli ML, Ibáñez-Alamo Y, Jokimaki JD, Mand J, Tryjanowski R, Moller P (2016) Evidence of evolutionary homogenization of bird communities in urban environments across Europe. Glob Ecol Biogeogr 25:1284–1293. https://doi.org/10.1111/geb.12486
Mouillot D, Gaillard S, Aliaume C, Verlaque M, Belsher T, Troussellier M (2005) Ability of taxonomic diversity indices to discriminate coastal lagoon environments based on macrophyte communities. Ecol Ind 5:1–17. https://doi.org/10.1016/j.ecolind.2004.04.004
Munari C, Warwick RM, Mistri M (2009) Monitoring with benthic fauna in Italian coastal lagoons: new tools for new prospects. Aquat Conserv Mar Freshwat Ecosyst 19(5):575–587. https://doi.org/10.1002/aqc.1005
Perigó AE, Montalvo JF, Martínez–Canals M, Ramírez O, Suárez G, Simanca J, Perigó A, Martínez C, Pérez DM (2005) Presiones Antropogénicas y su relación con la Calidad Ambiental de la Ecorregión del golfo de Batabanó, Impactos y Respuestas. Revista CENIC. Ciencias Biológicas 36. Número Especial. https://www.redalyc.org/articulo.oa?id=181220525073. Reviewed: Apr 2021
Perigó E, Montalvo JF, Penié I, Rodas L, Martínez M, Pérez R, Simanca J (2000) Calidad ambiental en zonas litorales y arrecifes coralinos del Golfo de Batabanó y de los estuarios del río Las Casas y La Coloma. In: Memorias del V Congreso de Ciencias del Mar. MARCUBA 2000. Palacio de las Convenciones de la Habana, Cuba, del 4 al 8 de diciembre del 2000. CD ROM
Piñeiro R (2006) Influencia del aporte fluvial en la zona marino costera suroccidental del Golfo de Batabanó. Cuba Revista Cubana De Investigaciones Pesqueras 24(1):28–31
Puga R, Piñeiro R, Alzugaray R, Cobas LS, de León ME, Morales O (2013) Integrating anthropogenic and climatic factors in the assessment of the Caribbean spiny lobster (Panulirus argus) in Cuba: Implications for fishery management. International Journal of Marine Science 3(6):36–45. https://doi.org/10.5376/ijms.2013.03.0006
Redfern C (2013) Bahamian Seashells. 1161 species from Abaco, Bahamas, 501 pp. Bahamianseashells.com, Inc., Boca Raton, Florida, USA
Reyes-Bonilla H, Álvarez-Filip L (2008) Long-term changes in taxonomic distinctness and trophic structure of reef fishes at Cabo Pulmo reef, Gulf of California. Proceedings of the 11th International Coral Reef Symposium, Ft. Lauderdale, Florida, 8(18):790–794
Ronowicz M, Kuklinski P, Włodarska-Kowalczuk M (2018) Diversity of kelp holdfast-associated fauna in an Arctic fjord-inconsistent responses to glacial mienral sedimentation across different taxa. Estuar Coast Shelf Sci 205:100–109. https://doi.org/10.1016/j.ecss.2018.01.024
Salas F, Patrício J, Marcos C, Pardal MA, Pérez-Ruzafa A, Márquez JC (2006) Are taxonomic distinctness measures compliant to other ecological indicators in assessing ecological status? Mar Pollut Bull 52:817–829
Stobart B, Warwick RM, González C, Mallol S, Díaz D, Reñones O, Goñi R (2009) Long-term and spillover effects of a marine protected área on an exploted fish community. Mar Ecol Prog Ser 384:47–60. https://doi.org/10.3354/meps08007
Tan X, Shi X, Liu G, Xu H, Nie P (2010) An approach to analyzing taxonomic patterns of protozoan communities for monitoring water quality in Songhua River, northern China. Hydrobiologia 683:193–201. https://doi.org/10.1007/s10750-009-0040-2
Tataranni M, Lardicci C (2010) Performance of some biotic índices in the real variable world: a case study al different spatial scales in North-Western Mediterranean Sea. Environmental Polution 158(1):26–34. https://doi.org/10.1016/j.ecss.2018.01.024
Warwick RM (1986) A new method for detecting Pollution effects on marine macrobenthic communities. Mar Biol 92:557–562
Warwick RM, Clarke KR (1995) New ‘biodiversity’ measures reveal a decrease in taxonomic distinctness with increasing stress. Mar Ecol Prog Ser 129:301–305. https://doi.org/10.3354/meps129301
Warwick RM, Clarke KR (1998) Taxonomic distinctness and environmental assessment. J Appl Ecol 35:532–543. https://doi.org/10.1046/j.1365-2664.1998.3540532.x
Warwick RM, Light J (2002) Death assemblages of mollusks on St. Martin´s Flats, Isles of Scilly: a surrogate for regional biodiversity. Biodivers Conserv 11:99–112
WoRMS Editorial Board (2002) World Register of Marine Species. VLIZ, Oostende. https://www.marinespecies.org. Reviewed: June 2021
Xu H, Jiang Y, Khaled AS, Al-Rasheid S, Al-Farraj SA, Song W (2011) Application of an indicator based on taxonomic relatedness of ciliated protozoan assemblages for marine environmental assessment. Environ Sci Pollut Res 18:1213–1221. https://doi.org/10.1007/s11356-011-0476-6
Zhou H, Zhang Z, Liu X, Hua E (2012) Decadal change in sublittoral macrofaunal biodiversity in the Bohai Sea, China. Mar Pollut Bull 64:2364–2373. https://doi.org/10.1016/j.marpolbul.2012.08.014
Acknowledgements
The authors are very grateful to Camila Perera for her help in editing the manuscript. Furthermore, we want to be grateful to the reviewers, who contributed to improve the paper and especially to Erika Becker for her timely criticisms and suggestions.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
Norberto Capetillo-Piñar: Conceptualization, Methodology, Investigation, Writing- Original draft preparation. Manuel Zetina Rejón: Investigation, Writing- Reviewing and Editing. Alejandro Bosch Callar: Investigation, Writing- Reviewing and Editing. Arturo Tripp Quezada: Writing- Reviewing and Editing. José Espinosa Sáenz: Investigation, Writing- Reviewing and Editing. Yuliesky Garcés: Investigation, Writing- Reviewing and Editing. Susana Perera-Valderrama: Investigation, Writing- Reviewing and Editing.
Corresponding author
Ethics declarations
Conflict of Interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Capetillo-Piñar, N., Rejón, M.Z., Perera-Valderrama, S. et al. Do Changes in the Length of the List of Reference Species Influence the Results of the Average Taxonomic Distinctness?. Thalassas 38, 1013–1024 (2022). https://doi.org/10.1007/s41208-022-00445-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41208-022-00445-1