Abstract
The embryonic and larval development of volitionally spawned eggs of the serranid fish Marcia’s anthias, Pseudanthias marcia (Randall and Hoover 1993) is described and illustrated based on observations during spawning, hatching and larval rearing trials. Eggs were obtained from volitional spawning of P. marcia under captive conditions in Re-circulating Aquaculture System (RAS) in which the sea water is re - circulated at the rate of 1500 L per hour. Fertilized eggs of P. marcia were pelagic, non adhesive, transparent and measured 617.891 ± 14.9 μ (mean ± SD). Each egg had a single oil globule which measured in 125.866 ± 14.06 μ (mean ± SD).Cell division or two cell stage started forming after 14 min of collection of the fertilized eggs from the brood stock tank. Larval motility began after 14:30 h and hatching occurred after 14:50 h at 29 °C. Average total length of the newly hatched larvae was 1206.55 ± 100.02 μ and the yolk sac length was 826.00 ± 46.00 μ. On 3rd dph (days post hatch), the total length of the larvae was 1906.26 ± 32.3 μ. Pigmentations patterns were observed in freshly hatched larvae after 4 h of hatching, with the presence of eight pigmented areas on the dorsal surface of the larva and one spot on the posterior tip of the yolk sac. Notochord flexion stage was complete on 25 dph (TL- 4.85 mm). Between 32 and 34 dph larvae metamorphosed to adult shape. Adult coloration and forked caudal fin shape was observed after 50 days of post hatch and the total body length of the larvae was 42–43 mm.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Anthias (family: Serranidae) forms an important group of fishes in the tropical marine aquarium fish trade because of their vibrant colours. The Indo-Pacific genus Pseudanthias, to which Marcia’s Anthias, Pseudanthias marcia (Randall and Hoover 1993) belongs are commonly called as “jewels of the reef” or false anthias. They swim in groups and are compatible with other species, easily adapt to pellet feed and bivalve meat. Apart from this, these fishes fetch high prices from $ 6.36 to $ 42.38 /piece. These fishes are protogynous hermaphrodites and found as haremic aggregations with their distribution being confined to the Indo-Pacific region (Randall and Pyle 2001).
Several studies exists on the egg and larval development of serranid fishes, especially groupers (sub family: Epinephelinae) (Glamuzina et al. 2000; Sawada et al. 1999; Powell and Tucker Jr. 1992). Anil et al. 2018, reported the first brood stock maturation, volitional spawning, breeding and successful larval rearing of P. marcia in RAS under captive condition. However, there is hardly any study on the embryonic and larval development of the fishes of sub family: Anthiinae. Available reports are mostly on taxonomic studies (Randall and Hoover 1993; Randall and Pyle 2001; Allen 2005; Heemstra and Heemstra 2004; Heemstra et al. 2004; Randall and Heemstra 2006; Heemstra and Akhilesh 2012). Information on egg, embryonic development, duration of each embryonic stage etc. are essential in studies pertaining to larval abundance for population estimates, extracting embryonic stem (ES) cells for the cryo-preservation of fish embryos and in cell-mediated gene transfer and targeted gene mutations (Wolf and Ahne 1982; Glamuzina et al. 1998; Chen and Ye 2003). Information on gamete physiology, embryology, and larval ontogeny are essential to develop a captive breeding technique for targeted species and which in turn helps in brood stock management and development and standardisation of larval rearing techniques (Coward et al. 2002; Moorhead and Zeng 2010). Brooks et al. 1997 opined that information regarding larval size, duration of embryonic developmental stages, mouth gape, first feeding and swimming pattern of the larva are essential parameters to be known to assess the larval quality and for successful larval rearing.
To develop culture techniques of the species, studies on the characteristics of embryonic and larval development is important (Rahman et al. 2009; Holt 2003; Leu et al. 2015; Olivotto et al. 2017).
Most of the embryonic and larval developmental studies pertain to the families Pomacentridae and Gobiidae and the present study is the first attempt on Indo-Pacific species P. marcia. The present study describes the egg development, hatching, larval development and changes associated with growth and development of larvae, P. marcia reared in captivity.
Materials and Methods
There were 12 fishes with 5 males and 7 females contributing to the egg production. Eggs of Pseudanthias marcia were collected from the RAS system setup in the hatchery (Anil et al. 2018) where fishes were allowed to spawn naturally. Once fishes started courtship behaviour, a 200 μ mesh egg collector was kept below the pipe attached to the overflow mechanism. All fish, used in the present study were handled in accordance with the guidelines for the care and use of fish in research by DeTolla et al. (1995) and the protocols were approved by ICAR-Central Marine Fisheries Research Institute, Kochi. The egg collector was observed regularly for floating eggs. For the embryonic developmental studies, fertilized eggs were collected from the spawning tank (RAS) soon after spawning. About 150–200 eggs were incubated in 500 mL glass container at 29 °C and the developmental stages were monitored until hatching. Salinity and pH of filtered and pre-treated sea water used for incubation was 35 ppt, and 8.1 respectively, and 50% of the sea water was changed twice daily. Eggs at the optic vesicle stage were collected from overflow conduit of RAS and used for rearing studies. The rearing trials were conducted in 500 L Fibreglass Reinforced Plastic (FRP) tanks, provided with green water medium and mild aeration in which these eggs were stocked at the rate of 10 no./L. From 1st dph onwards larvae were taken from the rearing tank.
Embryonic Development and Hatching of Pseudanthias marcia
Subsamples of 10–15 eggs were taken from the 500 mL glass container and observed under Leica DMCS microscope to examine the embryonic development. The characteristics of newly spawned and fertilized eggs were documented. Each time 10–15 fresh eggs were taken from 500 mL glass container for observation. The time taken for each stage of embryonic development from fertilized egg to 2, 4, 8, 16 and 32 cell stages, blastula, morula, gastrula, embryo formation and hatching were calculated and recorded. Diameter of the fertilized egg, yolk sac length and width, size of oil globule were measured in microns (μ).
Mouth Opening, Complete Yolk Sac and Oil Globule Absorption
Each time 10–15 larvae were taken from the 500 L rearing tank for observation and measurements. Yolk sac length, width and oil globule diameter were measured from 0 h of post hatch larvae to complete yolk sac absorption. The time taken for mouth opening was monitored at 4 h intervals.
Larval Morphology and Growth of Pseudanthias marcia
For the first three day, the same larvae were used for both the measurements of oil globule, yolk sac absorption and larval morphology. From 5 dph to 35 dph, 5–10 larvae were sacrificed from 500 L FRP tank for the study of larval morphology. Pigmentation pattern, morphological changes including fin spine and teeth formation, larval distribution in the tank and behavioural changes were observed and recorded. Water quality parameters such as temperature, salinity, pH, dissolved oxygen and ammonia were checked daily.
Results
Spawning and Embryonic Development
Fishes were allowed to spawn naturally in the tank and no hormonal induction was carried out. Spawning occurred in the early evening hours from 7.00–7.30 pm. Water quality parameters in the rearing tank were: salinity-33-35 ppt, temperature-28.0-29.0 °C, pH -8-8.2, dissolved oxygen- 4.5-5.01 mg/l and ammonia −0.001-0.004 ppm.
Egg Morphology and Hatching
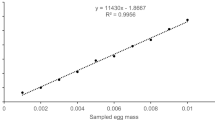
Details of embryonic development stages of Marcias anthias, P. marcia at 29 °C are given in Table 1 and Fig. 1 Fertilized eggs were spherical, transparent, pelagic and non-adhesive. Size of the individual eggs varied between 615 μ to 661 μ and the average size was 617.891 ± 14.9 μ (mean ± SD). Unfertilized eggs were opaque and settled at the bottom. The fertilized eggs had only one oil globule and the size ranged between 114 and 132 μ and had an average diameter of 125.866 ± 14.06 μ (mean ± SD) (Fig. 1A). Shortly after fertilization, the perivitelline space and blastodisc were formed. The eggs exhibited meroblastic discoidal cleavage. The blastodisc is like a disc shaped mass of dividing cells attached on the top of the yolk. First cell division was observed 14 min after fertilization resulting in the formation of 2 cell stage with 2 large equal sized blastomeres(Fig. 1B). After 39 min fertilization, the 2nd cleavage occurred which resulted in the formation of 4 equal sized blastomeres (Fig. 1C) and the 8 celled stage was formed after 45 min of fertilization (Fig. 1D). The cleavages followed a meridional and equatorial pattern. After this, the cleavage became more or less irregular during which a drastic difference was observed in the size of the blastomeres and they segregated into less yolky upper micromeres and yolky lower macromeres. The micromeres divided at a faster rate than macromeres. This segregation process occurred within 59 min after fertilization (Fig. 1E). At the 32 cell stage (formed after 1 h and 13 min of fertilization), the cells in the centre appeared cut off from the yolk while the cells in the outer boundaries were merged with the underlying yolk (Fig. 1F). After morula stage (observed after 1 h 33 min of fertilization) (Fig. 1G), the spherical egg assumed a spherical shaped structure. Blastula stage appeared after 3 h 5 min. The first movement of cells were observed during the gastrula stage, due to the epiboly of the blastoderm cell over the yolk. The development of the egg from fertilization to gastrula stage took 7 h 2 min, resulting in the formation of a germ ring. Embryo formation began at 9 h: 55 min (Fig. 1H) and by this time the 3 germ layers were established with the blastopore closed. The region of the blastopore developed into anus while mouth developed from a new opening (Fig. 1I). At this stage, the larval body showed signs of segmentation with the myomeres making their first appearance. The appearance of optic vesicles was observed at 13 h:50 min (Fig. 1J). The eye development reached its peak which was demonstrated by the transformation of optic vesicles into optic cups and which in turn induced the formation of lens and its overlying epithelium develops into corneal layer (Fig. 1K). The larval body developed the functional circulatory system which was evident by the heart beat and at this stage it began to separate from the yolk sac (Fig. 1L). Larval motility began after 14 h: 30 min and hatching occurred at 14 h 50 min (Fig. 1M).The larva broke out of the capsule by extensive wriggling movements (Fig. 1N) and the hatched out larva (Fig. 1O).
(A-O) Egg development of Pseudanthias marcia. A, fertlilized egg; B, 2 celled stage; C, 4 celled stage; D, 8 celled stage; E, 16 celled stage; F, 32 celled stage; G, morula; H, blastula; I, gastrula; J, blastophore nearly closed; K, early embryo formation; L, optic vesicle formation; M, larval motility began; N, hatched larva; O, hatchling
Mouth Opening, Complete Yolk Sac and Oil Globule Absorption
On 1st dph, on hatching the average length and width of yolk sac were 826.00 ± 46.00 μ (mean ± SD) and 451.22 ± 20.54 μ (mean ± SD). The length of the yolk sac started decreasing from 826.00 μ (0 h) to 332.29 μ at 24 h, and measuring 88.4 μ at 48 h and by 72 h, the yolk sac was completely absorbed. Similarly, the average total length of oil globule at 0 h was120.39 μ by 48 h it was to an average size 35.93 μ and was utilized completely by 72 h of post hatch. Changes in yolk sac length are given(Fig. 2).
Larval Characteristics of P. marcia
Fertilized eggs of P. marcia hatched out within 14–16 h. The larvae came out of the egg membrane by twitching and wriggling movements.Yolk and the oil globules are the endogenous nutrient sources of the newly hatched larvae.Yolk-sac larva, characterized with huge yolk sac (1206.55 ± 100.02 μ) and single oil globule (measuring120.39 ± 17.90 μ) located at the anterior most tip of the yolk sac, were found floating in the surface layers and appeared transparent with unpigmented eyes, and with the mouth and anus not formed.Pigmentations began with hatching of egg. Hatchlings developed eight pigmented areas on the dorsal surface of the larva and one spot on the posterior tip of the yolk sac at 4 h of post hatching (Fig. 3a).
(a-j) Larval development of Pseudanthias marcia; a, hatchling with 8 pigment blotches on dorsal side; b, 1 dph larvae with triangular blotches; c, 2nd dph larvae with pectoral fin formation; d, 3 dph larvae after complete yolk absorption; e, 5 dph larvae (ventral view); f, 10 dph larvae; g, 15 dph larvae with elongated 2nd dorsal spine and opercular spines; h, 20 dph flexion larvae; i, 25 dph larvae; j, 34 dph larvae, settling stage
On 1st dph pigmentation pattern changed from 4 rows of pigments to 3 blotches of pigments in a triangular pattern (Fig. 3b). The anus was formed and appeared elongated. Eyes were non pigmented and pectoral fin bud formed by 24 h of post hatch.
On 2nd dph, pectoral fin formed and anus became functional. The mouth opened with a mouth gap measuring76–80 μ.The average total length of larvae within 48 h post hatch was 1852.88 μ and eyes were pigmented (Fig. 3c).
On 3rd dph, eyes got densely pigmented and pectoral fins were well developed and the larvae measured 1906 μ. Yolk sac and oil globules were completely utilized by 3rd day (Fig. 3d).
An important event observed during the development of fishes is the flexion of notochord (Kendall et al. 1984). Based on this feature, the larval development could be divided into preflexion, flexion and post flexion stages. Pre-flexion stage was observed between 4 to 19 dph.
On 5dph, the larva had an average total length of 2.23 mm with well developed pectoral fin(Fig. 3e).Anal opening was elongated.
On 10 dph, the larval body still appeared transparent, with yellowish pectoral fin and golden yellow coloured gut. The formation of primordial fin fold was observed and eyes became fluorescent blue in colour. Larvae were mottled with golden yellow colour on the dorsal side along with intermittent black patches. The larvae swam vertically capturing the prey (artemia) occupying the entire water column. (Figure 3f).
On 14–15 dph, P .marcia larvae had well developed spiny dorsal with the 2nd dorsal spine and pelvic spine elongated. The tip of the spiny dorsal fin had melanophore formation. Larvae had developed canine teeth and pectoral fins with the round caudal fin started developing. Opercular or cheek spination started to appear and had 4 spines without any serrations. Angular part of opercle was orange-red in colour while the nape region had black pigments. Dorsal part of body became densely packed with melanophores.
On 17 dph, a notch developed between spiny and soft dorsal fin. The opercular spines persisted and the caudal fin became rounded in shape. Canine teeth became prominent. Larvae swam vertically and captured the prey organisms like Artemia nauplius and copepod adults (Fig. 3 g).
By 20 dph, 2nd dorsal spine and pelvic fin length decreased in relative length and the fins appeared normal. The larvae measured 4.85 mm average total length with the caudal fin acquires a round shape. Spiny dorsal fin had black bands. Eyes were fluorescent green in colour. Fin formula of the larvae was: D – X,16; A-II,10; P-I,5; Pectoral fin rays −17; Caudal fin rays-29. Pre-opercular cheek region had 9 spines where the first 3 spines were small in size, followed by two moderate sized spine (one above the angular spine which was elongated measuring 2.5 mm in length and one below) and remaining 3 were small sized spines (Fig. 3 h). Larval body became opaque with the opercular region having golden-yellow colour. In P. marcia the flexion stage formed when the larvae measured 4.85 mm (20 dph). On 21 dph, spiny dorsal fin started forming with 4 spines (2nd spine elongated) and resorption of median finfold continued. However, caudal fin was observed not detached fully from median finfold.
Between 23 and 25dph, the post flexion larval stage formed, resorption of fin folds were completed. Dorsal (spiny and soft), anal and caudal fins were fully developed and separated. Except for the spiny dorsal fin, which was pigmented with red colouration, all the other fin rays were transparent without any pigmentation. Preopercle region was heavily pigmented with red colour, and it extended to the base of the caudal peduncle region. Larval metamorphosis began by 25th day when the head and opercular region of the larvae developed intense pigment formation (Fig. 3i) and it continued till 34–35 dph during which the entire body became fully pigmented. Squamation took place between 25 and 30 dph.
Settlement of the larvae (body length − 22 mm) began by 34 dph, during which the nape region of the larvae were mottled with black pigments with a black line extending from nape region to caudal peduncle. Dorsal spines were pinkish in colour. Red colour appeared behind the eye region and a thin red coloured line was seen running parallel to the lateral line. The dorsal part of the larval body was greenish yellow in colour while the ventral part was translucent and mottled with red pigments. Appearance of fin spines and rays were similar to adults except body colour (Fig. 3j).
The adult colouration of vibrant pink started forming only after 45–50 days post hatch during which the larvae measured 42–43 mm in body length (Fig. 4).
Discussion
The present study aims to describe the sequential events of embryonic and larval development of Marcia’s anthias, P. marcia reared under captive conditions. There is very little published information on the early development of anthiid fishes except the first brood stock maturation volitional spawning, breeding and successful larval rearing of P. marcia in RAS under captive condition (Anil et al. 2018).
Spawning of P. marcia occurs at evening hours as observed in many egg scatterers, the eggs were spawned individually into the water. The pelagic egg stage encompasses a critical stage in the life-history of many organisms and is prone to high mortality (Hirst and López-Urrutia 2006). Eggs attached to substrate are spawned mostly during morning hours and are less vulnerable to predation as they are deposited in protected areas and they often receive parental care and these strategies offer better protection to eggs (Brinley 1939; Myrberg Jr et al. 1967; Goulet 1995; Anil et al. 2012; Rohini Krishna et al. 2016). Hatching time for P. marcia is only 15 h and such fast hatching helps in reducing the time of the most vulnerable stage of life history of the fish, which in turn helps them to escape from predators. From an aquaculture point of view, short incubation periods leading to the formation of altricial fish larvae without mouth at hatching or larvae with small mouth opening and underdeveloped digestive system which make them difficult to rear under captivity and would require very small live feed like super small rotifer or copepod nauplii as done in the present study.
There is no information available regarding the duration of embryonic development and hatching, larval size and yolk sac absorption of the species P. marcia of sub family anthiine. However, the embryonic development and hatching of many grouper species of family Serranidae has been reported. The embryonic development of a new hybrid Grouper, tiger grouper, Epinephelus fuscoguttatus x giant Grouper, E. lanceolatus took 18–19 h after fertilization at 28 °C temperature and 30 ppt salinity (Chng and Senoo 2008) whereas in dusky grouper, Epinephelus marginatus it was reported to last for 30 h at 23 °C(Glamuzina et al. 1998) and 24 h at 25.5 °C in goldblotch grouper, Epinephelus costae(Glamuzina et al. 2000). Embryonic development of P. marcia lasts for about 14–16 h at 29 °C temperature and 35 ppt salinity. The average size egg of P. marcia was observed in the present study was 617.891 ± 14.9 μ (615 μ to 661 μ) and it could be the smallest egg reported among serranids. James et al. (1997) reported an average egg diameter of 757.3 μ for camouflage grouper E. polyphekadion whereas Tamaru et al. (1996) reported a range of 769–832 μ for E. microdon which are among the smaller sized eggs from serrranids when compared to many other members of the family (Tucker Jr 1991; Watanabe et al. 1995). Similarly the average size of P. marcia hatchling was also found to be smaller (1206.6 ± 100.02 μ) than those reported for groupers (1.4–1.98 mm in groupers). Newly hatched larvae of brown spotted grouper, E. tauvina (Nazar and Hussain 1980) and gold blotch grouper E. costae (Glamuzina et al. 2000), were 2.25 mm and 1.76 ± 0.048 mm, respectively, while those reported for dusky grouper, Epinephelus marginatus (Glamuzina et al. 1998) and Malabar grouper, E. malabaricus were 1.52 ± 0.07 mm 1.71 ± 0.16 to 1.84 ± 0.10 (Yoseda et al. 2006). But other characteristics of P. Marcia larvae like large yolk sac, head and body shapes, short intestine tract were similar to the groupers (Ukawa et al. 1966, Hussain and Higuchi 1980, Lim 1993, Doi et al. 1991, Duray et al. 1997, Glamuzina et al. 1998, Rasem et al. 1997, Glamuzina et al. 2000, Leu et al. 2005, Yoseda et al. 2006).
Elongated second dorsal and pelvic spine is characteristic features of the grouper larvae (E.septemfasciatus) (Kitajima et al. 1991). Similar findings of well-developed dorsal and ventral spine was also reported in giant grouper, E.lanceolatus (García-Ortega et al. 2013) on 14 dph and the fins acquired normal shape after 38 dph. Like groupers, P. marcia also had well developed 2nd dorsal spine and pelvic spine on 14–15 days of post hatch though the elongation was not as much as that reported in groupers and by 34 dph, the larvae acquired the final fin shape.
In P. marcia notochord flexion stage was completed on 25 dph. Larval metamorphosis began by 25 dph and was completed around 50 dph with adult coloration. In giant grouper, Epinephelus lanceolatus metamorphosis began by 25 dph with head coloration and completed by 38 dph with the formation of entire body pigmentation and two bands on the posterior part of the body (García-Ortega et al. 2013).
Information generated in the present study will help to assess the quality of embryos and standardise the larval rearing techniques(Brooks et al. 1997;Coward et al. 2002; Moorhead and Zeng 2010) and also to standardise the step-by-step procedures to manipulate embryos including dechorionation, and cell transplantation (Porazinski et al. 2010), extracting embryonic stem (ES) cells (Chen and Ye 2003), to assess the quality of embryos (Brooks et al. 1997;Coward et al. 2002; Moorhead and Zeng 2010) .
References
Allen GR (2005). Appendix 4, list of the reef fishes of Madagascar, in: McKenna, S. a., Allen, G. R. (Eds.), a rapid marine biodiversity assessment of Northwest Madagascar. Conservation International, Washington, D.C. Bulletin of the Rapid Assessment, 31, pp.102–124
Anil MK, Santhosh B, Prasad BO, George RM (2012) Broodstock development and breeding of black-finned anemone fish Amphiprion nigripes Regan, 1908 under captive conditions. Indian Journal of Fisheries 59(1):77–82
Anil MK, Gomathi P, Raheem PK, Raju B, Philipose KK, Gopalakrishnan A (2018) Captive brood stock development, breeding and seed production of Anthid fish (family: Serranidae) Marcia's anthias,Pseudanthias marcia in recirculation aquaculture system (RAS). Aquaculture 492:265–272. https://doi.org/10.1016/j.aquaculture.2018.03.043
Brinley FJ (1939). Spawning habits and development of Beaugregory (Pomacentrus leucostictus). Copeia, 185–188. https://doi.org/10. 2307/1436876
Brooks S, Tyler CR, Sumpter JP (1997) Egg quality in fish: what makes a good egg? Reviews in Fish Biology & Fisheries 7:387–416. https://doi.org/10.1023/A:1018400130692
Chen ZSL, Ye XSH (2003) Establishment of a pluripotent embryonic cell line from sea perch (Lateolabrax japonicus) embryos. Aquaculture 218(1–4):141–151. https://doi.org/10.1016/S0044-8486(02)00570-7
Chng CL, Senoo S (2008) Egg and larval development of a new hybrid grouper ,Tiger grouper Epinephelus fuscoguttatus x Giant grouper Epinephelus lanceolatus. Aquaculture science 56(4):505–512
Coward K, Bromage NR, Hibbitt O, Parrington J (2002) Gamete physiology, fertilization and egg activation in teleost fish. Rev Fish Biol Fish 12:33–58
DeTolla LJ, Srinivas S, Whitaker BR, Andrews C, Hecker B, Kane AS, Reimschuessel R (1995) 1995. Inst Lab Anim Res J 37(4):159–173. https://doi.org/10.1093/ilar.37.4.159
Doi M, Munir MN, Nik-Razali NL, Zulkifli T (1991) Artificial propagation of the grouper, Epinephelus suillus, at the Marine Finfish Hatchery in Tanjong Demong, Terengganu, Malaysia, Kertas Pengembangan Perikanan Bil,167 ,1–41
Duray MN, Estudillo CB, Alpasan LG (1997) Larval rearing of the grouper Epinephelus suillus under laboratory conditions. Aquaculture 150:63–76. https://doi.org/10.1016/S0044-8486(96)01467-6
García-Ortega A, Daw A, Hopkins K (2013). Feeding hatchery-produced larvae of the giant grouper, Epinephelus lanceolatus. In: Conference: Hatchery Technology for high Quality Juvenile Production: Proceedings of the 40th U.S.-Japan Aquaculture Panel Symposium, At: Honolulu, Hawaii, USA, volume: U.S. Dept. commerce, NOAA Tech. Memo. NMFS-F/SPO
Glamuzina B, Glavic N, Tutman P, Kozul V, Skaramuca B (2000) Egg and early larval development of laboratory reared gold blotch grouper, Epinephelus costae (Steindachner, 1878) (Pisces, Serranidae). Sci Mar 64:341–345
Glamuzina B, Skaramuca B, Glavic N, Kozul V (1998) Preliminary studies on reproduction and early life stages in rearing trials with dusky grouper, Epinephelus marginatus (Lowe, 1834). Aquac Res 29:769–771. https://doi.org/10.1046/j.1365-2109.1998.29100769.x
Goulet D (1995) Temporal patterns of reproduction in the Red Sea damselfish Amblyglyphidodon leucogaster. Bull Mar Sci 57:582–595
Heemstra E, Heemsptra P, Smale M, Hooper T, Pelicier D (2004) Preliminary checklist of coastal fishes from the Mauritian island of Rodriguez. J Nat Hist 38:3315–3344
Heemstra PC, Heemstra E (2004). Coastal fishes of southern Africa.NISC and SAIAB.I-xxiv + 1-488
Heemstra PC, Akhilesh KV (2012) A review of the anthiine fish genus Pseudanthias (Perciformes: Serranidae) of the western Indian Ocean, with description of a new species and a key to the species. International Journal of Ichthyology 18(3):121–164
Hirst A, López-Urrutia A (2006) Effects of evolution on egg development time. Marine Ecological Progress Series 326:29–35
Holt GJ (2003) Research on culturing the early life history stages of marine ornamental species. In: Cato, J.C., Brown, C.L.(Eds.), marine ornamental species: collection, culture and conservation. Iowa state press. 251–254
Hussain NA, Higuchi M (1980) Larval rearing and development of the brown-spotted grouper, Epinephelus tauvina (Forskal). Aquaculture 19:339–350. https://doi.org/10.1016/0044-8486(80)90082-4
James CM, Al-Thobaiti SA, Raseem BM, Carlos MH (1997) Breeding and larval rearing of the camouflage grouper Epinephelus polyphekadion (Bleeker), in the hypersaline waters of the Red Sea coast of Saudi Arabia. Aquac Res 28:671–681. https://doi.org/10.1046/j.1365-2109.1997.00908.x
Kendall AW, Ahlstrom EH, and Moser HG (1984). Early life history stages of fishes and their characters. In: H.G. Moser, W.J. Richards, D.M. Cohen, M.P. Fahay, a.W. Kendall and S.L. Richardson (editors), ontogeny and systematics of fishes, international Symposium dedicated to the memory of Elbert Halvor Ahlstrom, La Jolla, California, 15-18 august 1983. Am. Sot. Ichth. Herbs, spec. Publ. No. 1, Allen Press Inc., Laurence, USA, 11-24
Kitajima C, Takaya M, Tsukashima Y, Arakawa T (1991) Development of eggs, larvae and juveniles of the grouper, Epinephelus septemfasciatus, reared in laboratory. Japanese Journal of Ichthyology 38:47–55
Leu MY, Liou CH, Fang LS (2005) Embryonic and larval development of the Malabar grouper, Epinephelus malabaricus (Pisces: Serranidae). Journal of Marine Biological Association of the United Kingdom 85:1249–1254
Leu MY, Sun YH, Meng PJ (2015) First results of larval rearing and development of the bluestriped angelfish Chaetodontoplus septentrionalis (Temminck & Schlegel) from hatching through juvenile stage with notes on its potential for aquaculture. Aquaculture Research 46:1087–1110. https://doi.org/10.1111/are.12265
Lim LC (1993) Larvi culture of the greasy grouper Epinephelus tauvina and brown-marbled grouper E. fuscoguttatus in Singapore. Journal of World Aquaculture Society 24:262–274
Moorhead JA, Zeng C (2010) Development of captive breeding techniques for marine ornamental fish: a review. Rev Fish Sci 18(4):315–343
Myrberg A Jr, Brahy D, Emery AR (1967) Field observations on the reproduction of the damselfish, Chromis multilineata (Pomacentridae) with additional notes on general behaviour. Copeia 4:819–827
Nazar A, Hussain H (1980) Larval rearing and development of the brown spotted grouper , Epinephelus tauvina (Forskal). Mariculture and Fisheries Department, Kuwait Institute for Scientific Research, Aquaculture, 19, 339–350
Olivotto I, Leu MY, Blázquez M (2017) Life cycles in marine ornamental species - fishes as a case. In: Calado R, Olivotto I, Planas M, Holt GJ (eds) Marine ornamental species aquaculture. John Wiley & Sons Ltd., West Sussex, pp 23–49. https://doi.org/10.1002/9781119169147.ch3
Porazinski SR, Wang H, Furutani-Seiki M (2010) Dechorionation of Medaka embryos and cell transplantation for the generation of chimeras. Journal of Visualized Experiment, 46, 1–4, http://www.jove.com/details.php?id=2055, doi: https://doi.org/10.3791/2055
Powell AB, Tucker JW Jr (1992) Egg and larval development of laboratory-reared Nassau grouper, Epinephelus striatus (Pisces, Serranidae). Bull Mar Sci 50:171–185
Rahman MM, Miah MI, Taher MA, Hasan MM (2009) Embryonic and larval development of guchibaim, Mastacembelus pancalus (Hamilton). Journal of Bangladesh Agricultural University 7(1):193–204
Randall JE, Hoover JP (1993) Pseudanthias marcia, a new serranid fish from Oman. Revue Françaisd’Aquariologie 1993(20):47–52
Randall JE, Pyle RL (2001) Four new serranid fishes of the anthiine genus Pseudanthias from the South Pacific. Raffles Bulletin of Zoology 49(1):19–34
Randall JE, Heemstra PC (2006) Review of the indo-Pacific fishes of the genus Odontanthias (Serranidae: Anthiinae), with descriptions of two new species and a related genus. Indo-Pacific Fishes 38:1–32
Rasem BM, James CM, Al-Thomaiti SA, Carlos MH (1997) Spawning of the camouflage grouper, Epinephelus polyphekadion (Bleeker) in the hyper saline waters of Saudi Arabia. Asian Fisheries Science 9:251–259
Rohini Krishna MV, Anil MK, Neethu Raj P, Santhosh B (2016) Seed production and growth of Neopomacentrus cyanomos (Bleeker, 1856) in captivity. Indian Journal of Fisheries 61(3):50–56
Sawada Y, Kato K, Okada T, Kurata M, Mukai Y, Miyashit S, Murata O, Kumai H (1999) Growth and morphological development of larval and jevenile Epinephelus bruneus (Perciformes: Serranidae). Ichthyol Res 46:245–257. https://doi.org/10.1007/BF02678510
Tamaru CS, Carlstromtrick C, Fitzgerald WJ and Ako H (1996) Induced final maturation and spawning of the marbled grouper, Epinephelus microdon captured from spawning aggregations in the Republic of Palau, Micronesia Journal of World Aquaculture Society, 27 (4), 363–372
Tucker, JW Jr (1991) Induced spawning and culture of Nassau grouper, Epinephelus striatus. (abstract). Journal of World Aquaculture Society, 22 (3), 60A
Ukawa M, Higuchi M, Mito S (1966) Spawning habits and early life history of a serranid fish, Epinephelusakaara (Temmincket Schlegel). Japanese Journal of Ichthyology 13:156–161
Wolf K, Ahne W (1982) Fish cell culture. In: advances in cell culture Marmorosh K. (ed.) academic press, New York. 2, 305-328
Watanabe WO, Ellis SC, Ellis EP, Head WD, Kelley CD, Moriwake A, Lee CS, Bianfang PK (1995) Progress in controlled breeding of Nassau grouper (Epinephelus striatus) broodstock by hormone induction. Aquaculture 138:205–219
Yoseda K, Dan S, Sugaya T, Yokogi K, Tanaka M, Tawada S (2006) Effects of temperature and delayed initial feeding on the growth of Malabar grouper (Epinephelus malabaricus) larvae. Aquaculture 256:192–200. https://doi.org/10.1016/j.aquaculture.2006.01.031
Acknowledgements
This research work was carried out with the funding from Indian council of Agricultural Research (ICAR) AINP1007134 under the project entitled “All India Net work Project on Mariculture (AINP-M)” in the hatchery of Vizhinjam Research center of ICAR-CMFRI Trivandrum, Kerala. The authors are thankful to all the staff members of Vizhinjam Research.
Centre for their cooperation and support.
Availability of Data and Materials
The datasets supporting the conclusions of this article are available.
Author information
Authors and Affiliations
Contributions
P. Gomathi conceived the study and wrote the manuscript, M. K. Anil has designed the rearing system(RAS), P.K Raheem collected the data and performed the experiments, P. Neethu Raj analyzed the data, M.V Rohini Krishna assisted in manuscript preparation, Ambarish P.Gop has maintained the broodstock of fishes and S.Surya has collected eggs for the study. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
P, G., K, A.M., K, R.P. et al. Egg and Larval Development of Serranid Fish Marcia’s Anthias, Pseudanthias marcia (Subfamily: Anthiinae) Spawned and Reared under Captive Condition. Thalassas 36, 23–31 (2020). https://doi.org/10.1007/s41208-020-00193-0
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41208-020-00193-0