Abstract
Nanotechnology is one of the new approaches introduced to improve agricultural production as the present ecosystem balance starts to decline significantly due to the extensive use of classical agriculture. Nanoparticles are suitable alternative materials in place of the excessive use of chemicals, pesticides, and fungicides in agricultural crops to resist various plant diseases and pests. Moreover, it has been demonstrated that the application of nanoparticles improves plant tolerance to various biotic stresses such as drought, heat, and salt. As nanoparticles have many positive effects in improving crop production and productivity, the adverse effects of some types of nanoparticles have been noted and observed. Therefore, understanding the pros and cons is very important for the efficient use of nanoparticles. In this review, the effect of nanoparticles on plants under various biotic and abiotic stresses is discussed. The effect of nanoparticle characteristics such as shape, size, and diameter on plant performance is also discussed. Also, the expression pattern of plant genes in response to the exposure to different nanoparticles is addressed in this review.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Agricultural crops are an essential source of raw materials needed for the feed and food industries. They provide human populations with food, fuel, textiles, feedstock, furniture, etc. [1]. Currently, the agricultural sector faces two main problems: a significant-rapid increase in the global population and the consequences of climate change. Both problems threaten the availability and quality of the agricultural crops. The increase in world population size resulted in a deterioration of nutrient lands and poor nutritional quality of important agricultural crops. Climate change is considered a severe problem for agricultural corps as it increases the harmful effect of biotic and abiotic stresses [2].
Breeding research could improve the production and productivity of agricultural crops. Moreover, the farmer can use a wide range of chemical fertilizers, pesticides, fungicides, etc., to improve nutritional quality and crop productivity [3]. However, breeding programs may take time to achieve their goals, and the extensive use of chemical fertilizers and pesticides raised severe concerns associated with human health. Therefore, looking for alternative methods and technologies is an imminent need to meet future demands from agricultural products.
Nanotechnology is one of the new-smart techniques that could be used to sustain agriculture [4]. Nanomaterials (NMs) refer to all materials with at least one dimension is less than 100 nm [5]. This extreme-small size, along with its unique properties, opened new possibilities to improve crop productivity under various environmental stresses. It was reported that more than 1300 different commercial nanomaterials could be used for many potential applications, including agriculture [6]. The composition and size are fundamental properties for synthesizing the nanomaterials (NMs) used in the agriculture sector. Nanomaterials can be either of natural or synthetic origin. Synthesized nanomaterials and modified naturally occurring nanomaterials are called engineered nanomaterials (ENs) and are classified into organic, inorganic, and hybrid materials, including surface-modified clay [6].
Understanding the effect of nanoparticles (NPs) on the agricultural crop is very important. Actually, the interaction between plant cells and the ENs can result in changing the modulation of plant gene expression and biological pathways that affect plant development and growth [7]. Moreover, the response to ENs differs by plant species. The effect of ENs on plants can vary with the stages of plant growth, method, and duration of exposure [7]. The shape, size, chemical composition, concentration, surface structure, aggregation, and solubility of ENs are essential characteristics that should be considered in understanding the effect of ENs on plants [8].
Some engineered NMs (ENs) have been reported to increase earlier plant germination and plant production [9]. Nanoparticles (NPs) can be presented to plants as nanofertilizers (e.g., nanozinc, titanium dioxide (TiO2), silica (Si), and iron) and nanopesticides, which can be safely used in improving crop productivity instead of the excessive use of chemicals [10]. Metal and metalloid NPs could be fruitfully used to alleviate the negative effect of abiotic stresses on crops [11]. On the other hand, it has been found that some nanoparticles have negative effects on crop production at a specific concentration. The negative effects could cause toxic effects and abnormal cell division. Silver nanoparticles (Ag NPs) and silver ions (Ag+) were found to decrease the mitotic index and caused a lot of chromosomal aberrations in the root tips of onion (Allium cepa) [12].
Studying and understanding nanoparticles’ positive and negative effects are significant to achieve high productivity in the crops. This review presents and discusses the negative and positive effects of different nanoparticles on some important agricultural crops.
General overview: nanomaterials
What would happen when the size of the material is reduced to be close to the Bohr radius (Nanoscale)? In 1857, Michael Faraday said, “a mere variation in the size of its particles gave rise to a variety of resultant colors” when he was discussing the optical properties of gold particles [13]. A hundred years later, in 1959, Richard P. Feynman, a physicist at California Institute of Technology, addressed the topic of nanomaterials for the first time in one of his classes when he said, “There is plenty of room at the bottom,” and he predicted that new technology will emerge based on the nanoscale materials [14]. Nowadays, the term nanomaterial is well established and refers to particles or assemblies of at least one dimension in the length scales of 1–100 nm range [15]. When the material size is reduced to the nanoscale, various novel promising chemical and physical properties are possible due to the electron-confinement effect and the fact that classical and quantum mechanics laws are no longer valid at the nanoscale dimensions [16].
The type of motion of the electrons allowed in a material determines its physical and chemical properties. In contrast, the electrons’ spatial confinement (space in which the electrons are confined) determines the type of motion of the electrons. Confined electrons in an atom or a molecule and unconfined (free) electrons are suffering two different types of electron motions [16]. The former is characterized by a quantized type of motion, while the latter has a non-quantized one. Critical size, usually in the nanoscale, arises due to the electrons’ spatial confinement; this size determines the material properties. A change in the material properties is often observed when its size is reduced below the critical size. At this regime, the material properties are size and shape-dependent. Consequently, modifying the shape or size of the nanoscale material may have an equivalent impact on changing the chemical composition or structure of the material on controlling (altering) a given physical property [17]. The significant difference in the nanomaterials’ physical and chemical properties for their bulk counterparts can be attributed to the spatial confinement of the electrons and the high surface-to-volume ratio observed in the nanoscale materials [16, 17].
Over the last two decades, nanomaterials have been extensively studied and classified based on their shape into (a) zero-dimensional nanomaterials such as spherical metal nanoparticles and quantum dots, (b) one-dimensional nanomaterials, for example, metal carbide and oxides nanorods, (c) two-dimensional nanomaterials, e.g., graphene and mxenes, and (d) three-dimensional nanomaterials such as nanoporous metals and inorganic nanocrystals [18,19,20,21,22] as shown in Fig. 1. Organic (e.g., liposomes & conducting polymers nanostructures), inorganic (e.g., metal, metal oxides & silica nanocomposites) and carbon-based nanomaterials (e.g., carbon nanodots) are another classification of the nanomaterials based on their chemical composition.
Nanomaterials can also be classified according to the synthesis methods into nanomaterials prepared by bottom-up or top-down strategies [18,19,20,21,22]. Among the different type of nanomaterials, metallic nanoparticles have aroused the attention of researchers due to their unique properties, which opened the gates for a wide range of potential applications in energy, agriculture, catalysis, optics, magnetic devices, magnetic separation, sensing, coatings, electronics, biotechnology, optoelectronics, national defense, etc. [15, 23,24,25,26,27,28]. As pointed earlier, when the size of material decreases, the chemical and physical properties of the material may change and new properties may arise [16, 17]. This is abundantly clear in metallic nanoparticles. For instance, gold is a noble metal with brilliant yellow color, has a very high melting point (1064 °C), and non-magnetic. However, gold nanoparticles display a wide range of colors extended from red to violet depending on the particle size.
Furthermore, the interaction between the gold nanoparticles and the electromagnetic field of light depends on the particle shape and size, where the gold nanorods give two plasmon peaks while the spherical gold nanoparticles display only one plasmon peak. Gold nanoparticles (2–3 nm) are no longer noble or non-magnetic, where they display considerable magnetism and prominent catalytic properties. Gold nanoparticles have a significantly lower melting point concerning the bulk gold where the melting point is a size-dependent property, and it dramatically decreases as the particle size reduces [29, 30]. The change in the physicochemical properties of a material when its size is reduced to the nanoscale (due to the dramatic increase in the fraction of surface atoms and so the high surface energy of the nanoparticles for the bulk counterparts) significantly affects the nature of the interactions between the nanoparticles and plant which affects the plant drought tolerance as discussed in the following sections.
Use of nanoparticles as inducers for crop improvement
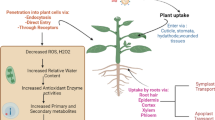
The application of nanofertilizers could be a potential approach to address issues of atmospheric changes, soil toxicity, and other environmental stresses that face the plant. Earlier studies reported that nanoparticles had positive and negative effects on the production and productivity of the crops (Table 1). The positive effects of nanoparticles have been reported for improving germination course [31], vegetative, and yield components [32] in different crop plants. Nanoparticles have displayed prominent outputs as inducers of tolerance to abiotic and biotic stresses (Fig. 2). In this regard, Hernández-Hernández et al. [33] illustrated that chitosan-polyvinyl alcohol hydrogels (Cs-PVA) + copper (Cu) NPs improved the salinity tolerance of tomato (Solanum lycopersicum) plants via increasing the content of β-carotene, phenols, vitamin C, and lycopene in stressed plants. In addition, they increased the antioxidant enzyme activities such as superoxide dismutase, catalase, ascorbate peroxidase, glutathione peroxidase, and phenylalanine ammonia-lyase. In another study, TiO2 NPs fertigation increased absolute and relative growth rates, unit leaf rate, and net assimilation rate of drought-imposed bread and durum wheat (Triticum aestivum) cultivars [34]. Moreover, the applied nanoparticle was found to increase leaf longevity by enhancing biomass duration, leaf area duration via improvement of chlorophyll content, and net photosynthetic rate.
The role of nanoparticles in enhancing plant growth and tolerance against metal stress has been studied extensively [35, 36]. Si NPs may enhance metal-stressed plants’ growth by improving the nutritional status, photosynthesis, morphology, and physiology of crops [36]. Zinc oxide nanoparticles (ZnO NPs), enhanced the zinc (Zn2+) concentration, photosynthetic pigments, antioxidant enzymes and reduced the cadmium (Cd2+) concentration in wheat grains. In another study, ZnO NPs combined with biochar decreased Cd2+ concentration in rice (Oryza sativa) [37] and maize (Zea mays) [38] in a short growth period. A study by Gil-Díaz et al. [39] stated that the application of zero-valent iron (Fe0) decreased the availability of heavy metals in calcareous or acidic soils. In another study about the effect of nanoparticles on mineral status in the growing media, Wang et al. [40] explored the potential of ceria nanoparticles (CeO2 NPs) to alleviate low (N deficiency) and high nitrogen (excess nitrogen) stresses compared to medium nitrogen application in rice. They illustrated that CeO2 NPs increased the N levels under N deficiency and reduced it under high N stress in roots and shoots. Moreover, CeO2 NP treatment enhanced N assimilation enzymes as glutamine synthetase (GS), glutamine oxoglutarate aminotransferase, and glutamate dehydrogenase (GDH), accounting for the high N content in plants under low nitrogen. Conversely, CeO2 NPs down-regulated the GS and GDH activity to reduce N accumulation under high nitrogen stress. The same study reported that CeO2 NPs reduced oxidative membrane and DNA damage by regulating the antioxidant enzyme system, proline and phytohormones’ levels under N-stress. Thus, the study of Wang et al. [40] was the first to report that CeO2 NPs could alleviate N stress in rice, while it might be a risk when the N supply is normal. Considering the effect of nanoparticles on UV stress, Lei et al. [41] found that nano-anatase (TiO2 NPs) promotes the antioxidant response of spinach plant (Spinacia oleracea) chloroplasts under UV-B irradiation. The nano-anatase treatment significantly decreased the oxidative stress caused by the accumulation of superoxide radicals, hydrogen peroxide, and malondialdehyde (MDA) contents, which mediated by the increased activity of catalase, superoxide dismutase (SOD), ascorbate peroxidase, guaiacol peroxidase, and elevating the oxygen evolution rate in spinach chloroplasts under UV-B radiation. Tripathi et al. [42] observed that pre-addition of Si-NPs protected wheat seedlings against UV-B stress through the nitric oxide-mediated triggering of antioxidant systems counteracted ROS-induced damages of photosynthesis. Another abiotic stress that adversely affects plant productivity is flooding stress. Various studies reported the importance of nanoparticles as protective agents against the negative disorders of flooding stress. In this regard, the effect of alumina (Al2O3) NPs of 30–60 nm was studied on soybean (Glycine max) plants under flooding conditions with the result that the root length increased while mitochondrial proteins related to glycolysis were suppressed [43]. Also, Al2O3 NPs of varying size and shape modulated cells’ scavenging activity by regulating the ascorbate/glutathione pathway [44].
Further studies revealed that nanoparticles exerted some biochemical upregulations as converting sugar metabolism to fermentation and deregulation of alcohol dehydrogenase enzyme [44]. In this regard, Mustafa et al. [45] stated that silver nanoparticles (2 ppm Ag NPs, 15 nm in size) treatment enhanced seedling growth under flooding stress. Ag NPs treatment had a significant role in reducing the abundances and transcript level of the glyoxalase II 3 and fermentation-related proteins revealing less cytotoxic by-products of glycolysis are produced in soybeans plants treated with Ag NPs as compared to flooded soybean. Moreover, the alcohol dehydrogenase 1 and pyruvate decarboxylase 2 genes were retarded in response to Ag NPs compared to flooding stress plants, revealing metabolic shift toward normal cellular processes.
As has been mentioned before, several reports documented that the application of nanomaterials prompted plant tolerance against abiotic stress. The advantages and challenges of NMs usage in agriculture are illustrated in Fig. 3. It has been demonstrated that several NMs act as anti-pathogenic agents and play vital roles in disease prevention [46, 47]. For instance, Au NMs coated with N-heterocyclic showed significant activity against bacteria, including gram-positive and multi-drug resistant strains [48]. Hao et al. [47] demonstrated that metal nanoparticles and carbon NMs reduced the turnip mosaic virus infection by increasing phytohormone levels. Cai et al. [49] reported the antiviral protective role of ZnO NPs against tobacco mosaic virus by regulating immunity and providing nutritional synergism in tobacco (Nicotiana benthamiana) plants. Abbai et al. [50] reported that the application of Si NPs on Ginseng (Panax ginseng) Meyer seedlings infected by Ilyonectria mors-panacis (the fungal causal agent of root rot disease) decreased disease-severity index and enhanced the tolerance against the pathogen, thereby improving root quality and yield. Younis et al. [32] developed silica (SiO2) NPs and chitosan-silica nanocomposites. These two NPs were found to control gray mold disease caused by Botrytis cinerea in grapes (Vitis vinifera). The nanoparticles application prevented the weight loss of bunches caused by pathogen infection. Hence, this could help in reducing the use of fungicides. A more recent study by Adeel et al. [51] investigated the role of nanoscale titanium dioxide (TiO2) and silver (Ag), C60 fullerenes, and carbon nanotubes (CNTs) as foliar exposure pre-inoculation with GFP-tagged tobacco mosaic virus (TMV). Plants treated with CNTs and C60 exhibited normal phenotype and lessened viral symptomology at 5 days post-infection compared to nanoscale TiO2 and Ag which failed to suppress the viral infection. This protective effect of CNTs and C60 treatments could be associated with reduction of viral coat protein transcript abundance and GFP mRNA expression. Moreover, the applied nanoparticles helped in keeping the chloroplast not effected by infection, maintained the photosynthesis process, upregulation of the defense-related phytohormones abscisic acid and salicylic acid.
Advanced research in the field of nanoparticles has led to production of genetically modified plants. In this regard, mesoporous Si NPs (MSNPs) can host several guest molecules like DNA, proteins, and agrochemicals because of their large surface area, adjustable pore sizes, three-dimensional open pore structure, and well-characterized surface properties [52]. The delivery of DNA and small-interfering RNA with surface-coated MSNPs, and re-recombinase through gold-plated MSNPs via particle bombardment in maize [53] have proved the potential of MSNPs to develop genetically modified plants. Nanobiolistics is emerging as a novel technique of plant genetic transformation, which requires optimization for broad-scale implementation [54]. Accordingly, such investigations are prefaced a new scenario in research that may prove futuristic aspects of food production under various abiotic/ biotic stresses.
Effect of nanoparticles on organs, cells, and subcellular organelles
It has been evidenced that some nanoparticle treatments exerted deleterious effects on the cellular compartments as well as the cell ultrastructure (Table 1). ZnO nanoparticles caused significant modifications in ryegrass (Lolium perenne), including altered root tip morphology, collapsed and vacuolated cortex, destroyed epidermis and root cap, and diminished vascular cylinder [55]. The treatment of maize and cabbage (Brassica oleracea) var. capitata with nanoparticles including Ag, ZnO, AgNO3, and ZnSO4 caused instrumental changes in the organs and organelles such as the increment of metaxylem counts and AgNO3 caused significant damage in maize root apical meristem [56]. Nickel oxide nanoparticles (NiO NPs) caused destructive changes in organelles of tomato with an agglomerated nucleus, high counts of peroxisomes, and destroyed mitochondrial cristae in root cells [57]. The Ag nanoparticles significantly altered the structure of chloroplast resulted in fewer interramal thylakoids [58]. In rapeseed (Brassica napus), the long-term exposure to ZnO NPs provoked instrumental anatomical and subcellular modifications such as decreasing the root tip diameter, size of epidermal, cortical cells, and increased counts of the stellar cells. It also decreased the size of pericycle cells, changed the shape and ultrastructure of chloroplasts of mesophyll cells, decreased size, and increased the number of plastoglobuli [28]. Copper oxide nanoparticles (CuO NPs) imposed similar effects on dotted duckmeat (Landoltia punctate), English oak (Quercus robur), and barley (Hordeum sativum distichum) via increasing plastoglobuli, decreasing mitochondria counts with abnormal shape, disorganized grana, and chloroplast membrane [59,60,61]. The exposure of eggplants (Solanum melongena) to cobalt oxide nanoparticles (Co3O4 NPs) showed its phytotoxicity by swelling mitochondria and causing cell death [62]. In wheat, across the whole life cycle, exposure to cerium oxide nanoparticles (CeO NPs) caused significant changes in leaf cells microstructure, swollen chloroplasts, abnormal nuclei, and disorganized thylakoids [63]. Cvjetko et al. [64], reported that in a concentration-dependent pattern, Ag NPs and AgNO3 NPs exerted destructive effects on different plant organs and organelles of tobacco, resulted in partially destroyed and vacuolated root cells, disintegrated nuclei, black points around and inside the cell wall of root cells, disorganized and smaller size plastids.
In a comprehensive study by Milewska et al. [65], positively and negatively charged gold (Au) NPs did not affect the cytoplasm density or the development of the dictyosomes of Golgi apparatus in Arabidopsis root cells; the neutral Au NPs reduced the density of cytoplasm and reduced the development of dictyosomes of Golgi apparatus. For mitochondria, the cells of the roots treated by the negatively charged Au NPs showed a denser matrix and rounded-shape mitochondria compared with the control treatment, while the remaining AuNPs did not display any effect. Compared with control, no changes had been observed in plastids when the roots were treated by neutral and positively charged Au NPs, but the plastids in cells of roots treated by negatively charged Au NPs had light stroma and no lamella or any other structures inside. No changes have been observed in the nuclei or the plasmodesmata of the cells treated by the different Au NPs compared to the control treatment. For cells treated by the different kinds of Au NPs, the vacuole size increased and was filled by various precipitations. The periplasmic space (i.e., the space between the cell wall and cell membrane) dramatically decreased when roots were treated by negatively charged Au NPs, while it became wider when roots were treated by positive and neutral Au NPs relative to control. Fedorenko et al. [66] reported that barley grown soils contaminated by CuO NPs showed shorter dense root hairs, larger cortical cell vacuoles, and destroyed mitochondria compared with control. Moreover, the author stated that significant modifications were observed in leaf-related parameters as thick leaf blades and destroyed chloroplasts.
The prominent effect of nanoparticles on the cell is the induction of the reactive oxygen species (ROS) production, causing significant cellular damages, including mitochondrial damage, plastid damage, and genotoxic effects due to DNA fragmentation, and chromosomal abnormalities, leading to cell death [67]. To sum up, nanoparticles effects on plant cells vary depending on the plant-specific factors, including (species, organ, and organelle), as well as particle-specific factors including (type, size, dose strength, and charge).
Nanoparticles and their effect on gene expression controlling plant growth and development
It was reported that multiwalled carbon nanotubes (MWCNTs) exaggerate the upregulation of stress-related gene expression in tomato seed germination and further affect the seedlings’ progress [68,69,70]. On the other hand, Lahiani et al. [71] demonstrated that MWCNTs activate seed germination and growth in soybean, maize, and barley seed coats by stimulating gene expression encoding several types of water channel proteins that belong to different gene families of aquaporins such as plasma membrane intrinsic proteins (PIPs), tonoplast intrinsic proteins (TIPs), and small and basic intrinsic proteins (SIPs). On another side, single-walled carbon nanotubes (SWCNHs) can influence stress signaling in tobacco plants and the expression of genes associated with cell growth [71]. Additionally, the treatment of MWCNTs enhances the expression of cell elongation, cell division, as well as stress-responsive genes. In the same context, Khodakovskaya et al. [68] revealed that MWCNTs nanotubes could enhance the growth of cultured tobacco cells by activating water channels and significant gene regulators of cell division and extension, such as NtPIP1, CycB, and NtLRX1 [68]. In a study by Frazier et al. [72], nano-TiO2 exposure to tobacco plants affected miRNAs’ expression profiles significantly. Low concentrations of TiO2 significantly induced miR395 and miR399 expression that might be responsible for reducing the growth of tobacco seedlings. MWCNTs improve the root and stem growth and peroxidase and dehydrogenase activity due to the accumulation of MWCNTs by roots followed by the translocation from roots to leaves [73]. The exposure of SWCNTs to maize seedlings promotes the growth of seminal roots [74]. These effects are related to the differential expression and upregulation of the associated genes encoding epigenetic modification enzymes, leading to global deacetylation of histone H3, similar to other abiotic stress response mechanisms. Syu et al. studied the effect of three different morphologies of Ag NPs on the molecular response of Arabidopsis. The three different sizes and shapes of Ag NPs induced gene expression involved in cellular events. For example, gene expression of indoleacetic acid protein 8 (IAA8), 9-cis-epoxycarotenoid dioxygenase (NCED3), and dehydration-responsive RD22. Also, Ag NPs activated the aminocyclopropane-1-carboxylic acid (ACC)-derived inhibition of root elongation in Arabidopsis seedlings. It was also observed that Ag NPs stimulate the buildup of reactive oxygen species and enhanced root development in the Arabidopsis plant by inducing the expression of some genes that regulated cellular processes like metabolism, hormone signaling pathways, and cell proliferation. In other studies, Koul et al. [70] reported Au NPs had a significant role in altering microRNAs expression levels, which regulated various morphological, physiological, and metabolic processes in plants. Moreover, it was discovered that proteins that were significantly altered during exposure to alumina (Al2O3) NPs are associated with energy metabolism, glycolysis, and lipid metabolism, and the cell wall was primarily enhanced when exposed to Al2O3 nanoparticles. The mRNA expression revealed that upregulation of NmrA likes negative transcriptional regulator when treated with Al2O3 nanoparticles [45]. Wang et al. [75] studied the gene profiles of Arabidopsis in response to ZnO NPs and ZnSO4. The expression of several genes encoding the synthesis of carotenoids augmented significantly, including geranyl pyrophosphate synthase (GGPS6), phytoene synthase (PSY), phytoene desaturase (PDS), and zeta-carotene desaturase (ZDS). Tumburu et al. [76] found that nano-titania and nano-ceria induced distinct transcriptomic responses in Arabidopsis rosette leaves and roots. Nano-titania upregulated transcripts related to photosynthesis, carbohydrate, lipid, and secondary metabolic pathways in leaves and roots. There was an overall down-regulation of these processes in both leaves and roots under the nano-ceria exposure. In canola roots and shoots exposed to ENMs, genes such as auxin-responsive protein and protein kinase decreased with increasing concentration of ZnO NPs, as revealed by the qPCR study. In A. thaliana exposed to ZnO NPs, zinc homeostasis genes like AtHMA3 and AtHMA4, macro-and microelement homeostasis were up hormone regulation genes like AtNAC1, AtASA1 were upregulated in roots but downregulated in the shoots. Yue et al. [77] found that lanthanum oxide (La2O3) NPs were affected by the expression of aquaporin genes like PIPs, TIPs, NIPs, and SIPs in the roots of maize seedling.
Enhancement of secondary metabolites through nano-treatment
Secondary metabolites play a significant role in plants’ survival, protection against pests, insect attack, mechanical injury, and other biotic and abiotic stresses. The application of nanoparticles has been reported as potential elicitors for the production of secondary plant metabolites. For example, Jasmonate (JA) hormone enhances various plant defense responses, along with the biosynthesis of defensive secondary metabolites [78]. Nanoparticles could play a significant role in regulating the expression of genes for jasmonate production in treated cells. On the other hand, cobalt nanoparticles have been used in cell suspension cultures of Sweet wormwood (A. annua) to elicit artemisinin secondary metabolites. The expression levels of SQS and DBR2 genes were declined at different concentrations of cobalt nanoparticles. This decline in the expression of SQS and DBR2 genes might cause enhanced production of artemisinin content at high concentrations of the cobalt nanoparticles [79].
The intervention of nanoparticles in photosynthesis
Rubisco (a complex of Rubisco and Rubisco activase) is an important enzyme for photosynthesis process to catalyze the incorporation of carbon dioxide into biological compounds. Nano-anatase TiO2 enhances the photosynthetic carbon assimilation by activating Rubisco that could promote Rubisco carboxylation, thereby increasing plants’ growth [80]. Ze et al. [81] found a significant increase in expression of light-harvesting complex II (LHCII) b gene in Arabidopsis upon TiO2 NPs exposure. This high LHCII content in the thylakoid membrane increased the light absorption efficiency in the chloroplast. Linglan et al. [71] studied the impact of nano-anatase on the molecular mechanism of carbon reaction. They suggested that the nano-anatase-induces marker gene for Rubisco activase (RCA) mRNA and enhances protein levels and activities of Rubisco activase, which result in the improvement of the Rubisco carboxylation and the high rate of photosynthetic carbon reaction. Wang et al. [75] investigated the gene profiles of Arabidopsis in response to ZnO NPs and ZnSO4. RT-PCR indicated a 50% reduction in the expression of chlorophyll oxygenase (CAO), chlorophyll synthase (CHLG), and photosystem structure gene photosystem 1 subunit D2 (PSAD2), photosystem 1 subunit E-2 (PSAE2), photosystem 1 subunit K (PSAK), and photosystem 1 subunit N (PSAN) in 300 mg/L ZnO NP-treated plants.
Conclusion
Improving the productivity and production of important crops can be achieved using advances in nanotechnology. However, it is important to understand the action and effects of nanoparticles when they are applied to field crops. The effects of these nanoparticles depend on their characteristics such as shape, size, and concentration. It is expected that the use of nanoparticles will continue gaining more and more importance in agriculture as some of them improved tolerance to various biotic and abiotic stresses. There are very few studies on the effect of these nanoparticles on the genetic material which is very important to genetically improve crops. Expanding our knowledge of the genetic changes is very important and urgently needed to be addressed. Also, plant species expressed different interactions with the nanoparticles. All changes induced by the nanoparticles in plant morphological traits, physiological parameters, and gene expressions are very important to improve field crops to meet the future food demand. Last, researchers should pay attention to the toxicity of nanomaterials applied in the agricultural field.
Availability of data and materials
Not applicable.
References
Yunlong C, Smit B (1994) Sustainability in agriculture: a general review. Agric Ecosyst Environ. https://doi.org/10.1016/0167-8809(94)90059-0
Sallam A, Alqudah AM, Dawood MFA et al (2019) Drought stress tolerance in wheat and barley: advances in physiology, breeding and genetics research. Int J Mol Sci. https://doi.org/10.3390/ijms20133137
Francis DM, Sood N, Gokhale T (2020) Applications of metal nanoparticles in agriculture. In: Pawar S (ed) Progress and prospects in nanoscience today applications of metal nanoparticles in agriculture. Nova Science Publishers, Inc.
Mishra S, Singh A, Keswani C, Singh HB (2014) Nanotechnology: exploring potential application in agriculture and its opportunities and constraints. Biotech Today 4:9–14. https://doi.org/10.5958/2322-0996.2014.00011.8
Thomas R, Jasim B, Mathew J, Radhakrishnan EK (2012) Extracellular synthesis of silver nanoparticles by endophytic Bordetella sp. isolated from Piper nigrum and its antibacterial activity analysis. Nano Biomed Eng. https://doi.org/10.5101/nbe.v4i14.p183-187
Saranya S, Aswani R, Remakanthan A, Radhakrishnan EK (2019) Nanotechnology in agriculture. In: Panpatte DG, Jhala YK (eds) Nanotechnology for agriculture: advances for sustainable agriculture. Springer, pp 1–17
Chandrika KP, Singh A, Tumma MK, Yadav P (2018) Nanotechnology prospects and constraints in agriculture. In: Dasgupta N, Ranjan S, Lichtfouse E (eds) Environmental nanotechnology. Environmental chemistry for a sustainable world, vol 14. Springer
Kole C, Kole P, Randunu KM et al (2013) Nanobiotechnology can boost crop production and quality: first evidence from increased plant biomass, fruit yield and phytomedicine content in bitter melon (Momordica charantia). BMC Biotechnol 13:37. https://doi.org/10.1186/1472-6750-13-37
Servin AD, White JC (2016) Nanotechnology in agriculture: next steps for understanding engineered nanoparticle exposure and risk. NanoImpact. https://doi.org/10.1016/j.impact.2015.12.002
Zhang B, Zheng LP, Yi Li W, Wen Wang J (2013) Stimulation of artemisinin production in Artemisia annua hairy roots by Ag-SiO2 core-shell nanoparticles. Curr Nanosci. https://doi.org/10.2174/1573413711309030012
Khan MN, Mobin M, Abbas ZK et al (2017) Role of nanomaterials in plants under challenging environments. Plant Physiol Biochem 110:194–209
Fouad AS, Hafez RM (2018) The effects of silver ions and silver nanoparticles on cell division and expression of cdc2 gene in Allium cepa root tips. Biol Plant. https://doi.org/10.1007/s10535-017-0751-6
Faraday M (1857) Experimental relations of gold (and other metals) to light. Phil Trans R Soc 147:145–181
Appenzeller T (1991) The man who dared to think small. Science (80-). https://doi.org/10.1126/science.254.5036.1300
Mody V, Siwale R, Singh A, Mody H (2010) Introduction to metallic nanoparticles. J Pharm Bioallied Sci. https://doi.org/10.4103/0975-7406.72127
El-Sayed MA (2001) Some interesting properties of metals confined in time and nanometer space of different shapes. Acc Chem Res. https://doi.org/10.1021/ar960016n
Heath JR (1995) The chemistry of size and order on the nanometer scale. Science (80-) 270:1315–1316
Farghaly AA, Huba ZJ, Carpenter EE (2012) Magnetic field assisted polyol synthesis of cobalt carbide and cobalt microwires. J Nanoparticle Res. https://doi.org/10.1007/s11051-012-1159-z
Farghaly AA, Khan RK, Collinson MM (2018) Biofouling-resistant platinum bimetallic alloys. ACS Appl Mater Interfaces. https://doi.org/10.1021/acsami.8b02900
Farghaly AA, Lam M, Freeman CJ et al (2016) Potentiometric measurements in biofouling solutions: comparison of nanoporous gold to planar gold. J Electrochem Soc. https://doi.org/10.1149/2.0101604jes
Khan RK, Farghaly AA, Silva TA et al (2019) Gold-nanoparticle-decorated titanium nitride electrodes prepared by glancing-angle deposition for sensing applications. ACS Appl Nano Mater. https://doi.org/10.1021/acsanm.8b02354
Xue B, Farghaly AA, Guo Z et al (2016) Monoclinic hydroxyapatite nanoplates hybrid composite with improved compressive strength, and porosity for bone defect repair: biomimetic synthesis and characterization. J Nanosci Nanotechnol. https://doi.org/10.1166/jnn.2016.10919
Torres Galvis HM, Bitter JH, Khare CB et al (2012) Supported iron nanoparticles as catalysts for sustainable production of lower olefins. Science (80-). https://doi.org/10.1126/science.1215614
Bahadur D, Giri J, Nayak BB et al (2005) Processing, properties and some novel applications of magnetic nanoparticles. Pramana - J Phys. https://doi.org/10.1007/bf03010455
Duguet E, Vasseur S, Mornet S, Devoisselle JM (2006) Magnetic nanoparticles and their applications in medicine. Nanomedicine 1:157–168
Majkova E, Jergel M, Yamamoto M et al (2007) Advanced nanometer-size structures. Acta Phys Slovaca. https://doi.org/10.2478/v10155-010-0087-7
Tartaj P, Del Puerto MM, Veintemillas-Verdaguer S et al (2003) The preparation of magnetic nanoparticles for applications in biomedicine. J. Phys. D. Appl, Phys
Vatta LL, Sanderson RD, Koch KR (2006) Magnetic nanoparticles: properties and potential applications. Pure Appl Chem 78:1793–1801
Huang X, Jain PK, El-Sayed IH, El-Sayed MA (2007) Gold nanoparticles: interesting optical properties and recent applications in cancer diagnostics and therapy. Nanomedicine 2:681–693
Eustis S, El-Sayed MA (2006) Why gold nanoparticles are more precious than pretty gold: Noble metal surface plasmon resonance and its enhancement of the radiative and nonradiative properties of nanocrystals of different shapes. Chem Soc Rev. https://doi.org/10.1039/b514191e
Younes NA, Hassan HS, Elkady MF et al (2020) Impact of synthesized metal oxide nanomaterials on seedlings production of three Solanaceae crops. Heliyon. https://doi.org/10.1016/j.heliyon.2020.e03188
Younes NA, Dawood MFA, Wardany AA (2019) Biosafety assessment of graphene nanosheets on leaf ultrastructure, physiological and yield traits of Capsicum annuum L. and Solanum melongena L. Chemosphere. https://doi.org/10.1016/j.chemosphere.2019.04.097
Hernández-Hernández H, González-Morales S, Benavides-Mendoza A et al (2018) Effects of chitosan–PVA and Cu nanoparticles on the growth and antioxidant capacity of tomato under saline stress. Molecules. https://doi.org/10.3390/molecules23010178
Dawood MFA, Abeed AHA, Aldaby EES (2019) Titanium dioxide nanoparticles model growth kinetic traits of some wheat cultivars under different water regimes. Indian J Plant Physiol. https://doi.org/10.1007/s40502-019-0437-5
Asgari F, Majd A, Jonoubi P, Najafi F (2018) Effects of silicon nanoparticles on molecular, chemical, structural and ultrastructural characteristics of oat (Avena sativa L.). Plant Physiol Biochem. https://doi.org/10.1016/j.plaphy.2018.03.021
Hussain HA, Men S, Hussain S et al (2019) Interactive effects of drought and heat stresses on morpho-physiological attributes, yield, nutrient uptake and oxidative status in maize hybrids. Sci Rep. https://doi.org/10.1038/s41598-019-40362-7
Ali S, Rizwan M, Noureen S et al (2019) Combined use of biochar and zinc oxide nanoparticle foliar spray improved the plant growth and decreased the cadmium accumulation in rice (Oryza sativa L.) plant. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-019-04554-y
Rizwan M, Ali S, Zia ur Rehman M et al (2019) Alleviation of cadmium accumulation in maize (Zea mays L.) by foliar spray of zinc oxide nanoparticles and biochar to contaminated soil. Environ Pollut. https://doi.org/10.1016/j.envpol.2019.02.031
Gil-Díaz M, Pinilla P, Alonso J, Lobo MC (2017) Viability of a nanoremediation process in single or multi-metal(loid) contaminated soils. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2016.09.071
Bowne JB, Erwin TA, Juttner J et al (2012) Drought responses of leaf tissues from wheat cultivars of differing drought tolerance at the metabolite level. Mol Plant 5:418–429. https://doi.org/10.1093/mp/ssr114
Lei Z, Mingyu S, Xiao W et al (2008) Antioxidant stress is promoted by nano-anatase in spinach chloroplasts under UV-B radiation. Biol Trace Elem Res. https://doi.org/10.1007/s12011-007-8028-0
Taran N, Storozhenko V, Svietlova N et al (2017) Effect of zinc and copper nanoparticles on drought resistance of wheat seedlings. Nanoscale Res Lett 12:60. https://doi.org/10.1186/s11671-017-1839-9
Mustafa G, Komatsu S (2016) Insights into the response of soybean mitochondrial proteins to various sizes of aluminum oxide nanoparticles under flooding stress. J Proteome Res. https://doi.org/10.1021/acs.jproteome.6b00572
Mustafa G, Sakata K, Hossain Z, Komatsu S (2015) Proteomic study on the effects of silver nanoparticles on soybean under flooding stress. J Proteomics. https://doi.org/10.1016/j.jprot.2015.03.030
Mustafa G, Sakata K, Komatsu S (2015) Proteomic analysis of flooded soybean root exposed to aluminum oxide nanoparticles. J Proteomics. https://doi.org/10.1016/j.jprot.2015.08.010
Cai Y, Xu W, Wang M et al (2019) Mechanisms and uncertainties of Zn supply on regulating rice Cd uptake. Environ Pollut. https://doi.org/10.1016/j.envpol.2019.07.077
Hao Y, Xu B, Ma C et al (2019) Synthesis of novel mesoporous carbon nanoparticles and their phytotoxicity to rice (Oryza sativa L.). J Saudi Chem Soc 23:75–82. https://doi.org/10.1016/J.JSCS.2018.05.003
Feng Y, Chen W, Jia Y et al (2016) N-Heterocyclic molecule-capped gold nanoparticles as effective antibiotics against multi-drug resistant bacteria. Nanoscale. https://doi.org/10.1039/c6nr03317b
Cai L, Liu C, Fan G et al (2019) Preventing viral disease by ZnONPs through directly deactivating TMV and activating plant immunity in: Nicotiana benthamiana. Environ Sci Nano. https://doi.org/10.1039/c9en00850k
Abbai R, Kim YJ, Mohanan P et al (2019) Silicon confers protective effect against ginseng root rot by regulating sugar efflux into apoplast. Sci Rep. https://doi.org/10.1038/s41598-019-54678-x
Adeel M, Farooq T, White JC et al (2021) Carbon-based nanomaterials suppress tobacco mosaic virus (TMV) infection and induce resistance in Nicotiana benthamiana. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2020.124167
Torney F, Trewyn BG, Lin VSY, Wang K (2007) Mesoporous silica nanoparticles deliver DNA and chemicals into plants. Nat Nanotechnol. https://doi.org/10.1038/nnano.2007.108
Martin-Ortigosa S, Peterson DJ, Valenstein JS et al (2014) Mesoporous silica nanoparticle-mediated intracellular cre protein delivery for maize genome editing via loxP site excision. Plant Physiol. https://doi.org/10.1104/pp.113.233650
Cunningham FJ, Demirer GS, Goh NS et al (2020) Nanobiolistics: an emerging genetic transformation approach. In: Rustgi S, Luo H (eds) Methods in molecular biology. Humana
Lin D, Xing B (2008) Root uptake and phytotoxicity of ZnO nanoparticles. Environ Sci Technol. https://doi.org/10.1021/es800422x
Pokhrel LR, Dubey B (2013) Evaluation of developmental responses of two crop plants exposed to silver and zinc oxide nanoparticles. Sci Total Environ. https://doi.org/10.1016/j.scitotenv.2013.02.059
Faisal M, Saquib Q, Alatar AA et al (2013) Phytotoxic hazards of NiO-nanoparticles in tomato: a study on mechanism of cell death. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2013.01.063
Jiang HS, Qiu XN, Li GB et al (2014) Silver nanoparticles induced accumulation of reactive oxygen species and alteration of antioxidant systems in the aquatic plant Spirodela polyrhiza. Environ Toxicol Chem. https://doi.org/10.1002/etc.2577
Lalau CM, Mohedano RA, Schmidt ÉC et al (2014) Toxicological effects of copper oxide nanoparticles on the growth rate, photosynthetic pigment content, and cell morphology of the duckweed Landoltia punctata. Protoplasma. https://doi.org/10.1007/s00709-014-0671-7
Manuscript A (2012) NIH Public Access. 59:3485–3498. https://doi.org/10.1021/jf104517j.Interaction
Olchowik J, Bzdyk RM, Studnicki M et al (2017) The effect of silver and copper nanoparticles on the condition of English oak (Quercus robur L.) seedlings in a container nursery experiment. Forests. https://doi.org/10.3390/f8090310
Faisal M, Saquib Q, Alatar AA et al (2016) Cobalt oxide nanoparticles aggravate DNA damage and cell death in eggplant via mitochondrial swelling and NO signaling pathway. Biol Res. https://doi.org/10.1186/s40659-016-0080-9
Du W, Gardea-Torresdey JL, Ji R et al (2015) Physiological and biochemical changes imposed by CeO2 nanoparticles on wheat: a life cycle field study. Environ Sci Technol. https://doi.org/10.1021/acs.est.5b03055
Cvjetko P, Zovko M, Štefanić PP et al (2018) Phytotoxic effects of silver nanoparticles in tobacco plants. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-017-0928-8
Milewska-Hendel A, Zubko M, Stróż D, Kurczyńska EU (2019) Effect of nanoparticles surface charge on the Arabidopsis thaliana (L.) roots development and their movement into the root cells and protoplasts. Int J Mol Sci. https://doi.org/10.3390/ijms20071650
Fedorenko AG, Minkina TM, Chernikova NP et al (2020) The toxic effect of CuO of different dispersion degrees on the structure and ultrastructure of spring barley cells (Hordeum sativum distichum). Environ Geochem Health. https://doi.org/10.1007/s10653-020-00530-5
Rahman IA, Padavettan V (2012) Synthesis of silica nanoparticles by sol-gel : size-dependent properties, surface modification, and applications in silica-polymer nanocomposites—a review. J Nanomater. https://doi.org/10.1155/2012/132424
Khodakovskaya MV, De Silva K, Nedosekin DA et al (2011) Complex genetic, photothermal, and photoacoustic analysis of nanoparticle-plant interactions. Proc Natl Acad Sci USA. https://doi.org/10.1073/pnas.1008856108
Singh S, Tripathi DK, Dubey NK, Chauhan DK (2016) Effects of nano-materials on seed germination and seedling growth: striking the slight balance between the concepts and controversies. Mater Focus. https://doi.org/10.1166/mat.2016.1329
Koul A, Kumar A, Singh VK et al (2018) Exploring plant-mediated copper, iron, titanium, and cerium oxide nanoparticles and their impacts. In: Tripathi DK, Ahmad P, Sharma S, Chauhan DK, Dubey NK (eds) Nanomaterials in plants, algae, and microorganisms. Academic Press
Linglan M, Chao L, Chunxiang Q et al (2008) Rubisco activase mRNA expression in spinach: Modulation by nanoanatase treatment. Biol Trace Elem Res. https://doi.org/10.1007/s12011-007-8069-4
Frazier TP, Burklew CE, Zhang B (2014) Titanium dioxide nanoparticles affect the growth and microRNA expression of tobacco (Nicotiana tabacum). Funct Integr Genomics. https://doi.org/10.1007/s10142-013-0341-4
Smirnova E, Gusev A, Zaytseva O et al (2012) Uptake and accumulation of multiwalled carbon nanotubes change the morphometric and biochemical characteristics of Onobrychis arenaria seedlings. Front Chem Sci Eng. https://doi.org/10.1007/s11705-012-1290-5
Yan S, Zhao L, Li H et al (2013) Single-walled carbon nanotubes selectively influence maize root tissue development accompanied by the change in the related gene expression. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2012.12.013
Wang X, Yang X, Chen S et al (2016) Zinc oxide nanoparticles affect biomass accumulation and photosynthesis in Arabidopsis. Front Plant Sci. https://doi.org/10.3389/fpls.2015.01243
Tumburu L, Andersen CP, Rygiewicz PT, Reichman JR (2017) Molecular and physiological responses to titanium dioxide and cerium oxide nanoparticles in Arabidopsis. Environ Toxicol Chem. https://doi.org/10.1002/etc.3500
Yue L, Ma C, Zhan X et al (2017) Molecular mechanisms of maize seedling response to La2O3 NP exposure: water uptake, aquaporin gene expression and signal transduction. Environ Sci Nano. https://doi.org/10.1039/c6en00487c
Misra P, Shukla PK, Pramanik K et al (2016) Nanotechnology for crop improvement. In: Kole C, Kumar D, Khodakovskaya M (eds) Plant nanotechnology. Springer, pp 219–256
Ghasemi B, Hosseini R, Dehghan Nayeri F (2015) Effects of cobalt nanoparticles on artemisinin production and gene expression in Artemisia annua. Turk J Botany. https://doi.org/10.3906/bot-1410-9
Gao F, Hong F, Liu C et al (2006) Mechanism of nano-anatase TiO2 on promoting photosynthetic carbon reaction of spinach: Inducing complex of Rubisco-Rubisco activase. Biol Trace Elem Res. https://doi.org/10.1385/BTER:111:1:239
Ze Y, Liu C, Wang L et al (2011) The regulation of TiO2 nanoparticles on the expression of light-harvesting complex II and photosynthesis of chloroplasts of arabidopsis thaliana. Biol Trace Elem Res. https://doi.org/10.1007/s12011-010-8901-0
Funding
No funding was received for conducting this study.
Author information
Authors and Affiliations
Contributions
DA, MF, YM, AF, AS performed literature search, data collection, and wrote the review; MA designed all graphical presentation of this study, AS contributed to the study conception and design. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Research involving human participants and/or animals
Not applicable.
Informed consent
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
El-Moneim, D.A., Dawood, M.F.A., Moursi, Y.S. et al. Positive and negative effects of nanoparticles on agricultural crops. Nanotechnol. Environ. Eng. 6, 21 (2021). https://doi.org/10.1007/s41204-021-00117-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s41204-021-00117-0