Abstract
Background
An adverse intrauterine environment reflected by low birth weight (LBW) and prematurity may induce fetal programming that favors kidney dysfunction in adulthood. We examined the association of LBW and prematurity with blood pressure (BP) and kidney function markers in non-diabetic, middle-aged adults without kidney disease from the Brazilian Longitudinal Study of Adult Health (ELSA-Brasil).
Methods
A cross-sectional analysis of 768 subjects aged 35–54 years was conducted. Comparisons were performed according to self-reported birth weight: LBW (< 2.5 kg) or normal birth weight (2.5–4.0 kg). Associations of LBW and prematurity with BP levels and kidney function markers "(estimated glomerular filtration rate [eGFR], albumin-creatinine ratio [ACR] and serum cystatin-C) were tested by multiple linear regression using adjustments based on Directed Acyclic Graphs. Propensity score matching was applied to control imbalances.
Results
Mean age of participants was 45.5 ± 4.6 years and 56.8% were female; 64 (8.3%) participants reported LBW and 39 (5.0%) prematurity. The LBW group had higher systolic (p = 0.015) and diastolic BP (p = 0.014) and ACR values (p = 0.031) and lower eGFR (p = 0.015) than the normal birth weight group, but no group difference for cystatin-C was found. The preterm group had higher mean levels of systolic and diastolic BP, but no difference in kidney function markers was evident. In a regression model adjusted for sex, skin color and family history of hypertension, both systolic and diastolic BP levels were associated with LBW, but this association disappeared after adding for prematurity, which remained associated with BP (p = 0.017). Having applied a propensity score matching, LBW was associated with ACR values (p = 0.003), but not with eGFR or BP levels.
Conclusion
The study findings of independent associations of prematurity with higher BP levels, and of LBW with markers of kidney function in adulthood, support that early life events may predict risk for hypertension and kidney dysfunction in adulthood. The study design precluded the inferring of causality, and prospective studies are needed to further investigate this hypothesis.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Based on the Developmental Origins of Health and Disease (DOHaD) theory, early life events can increase the risk of noncommunicable diseases in adulthood [1]. Nutrition-related and other stressors during pregnancy may result in low birth weight (LBW), considered a proxy of an adverse intrauterine environment [2] and commonly the result of intrauterine growth restriction or prematurity [3]. Given the short- and long-term adverse outcomes in LBW neonates, this condition remains a major public health concern. The World Health Organization (WHO) estimates that LBW occurs in 15–20% of all births, with rates varying widely according to country income level [4]. In Brazil, according to the Information System on Live Births–SINASC [5], LBW rates range from 7.4% in the Midwest to 8.4% in the Southeast region of the country. Compelling evidence has indicated that LBW may predict cardiometabolic and renal disorders [6, 7].
Perinatal distress has been associated with blood pressure elevation in adulthood [8]. Underlying mechanisms for the association of early life events with blood pressure elevation have been proposed in animal and human models [9, 10]. Changes in renin-angiotensin system gene expression, and activity of the angiotensin-converting enzyme and sympathetic nervous system, may contribute to the development of hypertension in adult life [11,12,13]. Sustained increased blood pressure levels with gradual glomerular damage may precipitate chronic renal failure.
Nephrogenesis begins at early stages of embryonic life with an increase in glomeruli number in the last weeks of gestation [14]. Insults during intrauterine life (undernutrition, toxic exposures and/or placental abnormalities, as well as maternal health-related factors) [15] can negatively impact renal functional reserve. These adverse events can induce fetal programming with resultant reduction in nephron formation and hypertrophy of the remaining nephrons, leading to adaptive hyperfiltration and increased risk of kidney dysfunction later in life [16, 17]. An early sign of hypertension-dependent glomerular damage is urinary protein loss [18]. Issues regarding whether LBW adults born at full-term or prematurely are prone to exhibit different long-term consequences, and whether these changes can be detected early in the natural history of hypertension and chronic kidney disease, remain understudied. Even with the available data pointing to kidney damage, such as a decrease in the number of nephrons or compensatory glomerular hypertrophy observed in the offspring of mothers who had insults during pregnancy, more evidence on their impact in functional reserve and structural renal changes are needed. The Brazilian Longitudinal Study of Adult Health (ELSA-Brasil) is a large cohort that provides an opportunity to investigate the relationship of exposures with several outcomes in adulthood [19, 20]. In particular, this allows the testing of associations of early life events with blood pressure levels and renal function markers in adults without overt kidney diseases. We examined the association of LBW and prematurity with blood pressure levels and kidney function markers in middle-aged, non-diabetic participants of ELSA-Brasil without kidney disease.
Methods
Study design and population
A cross-sectional analysis was carried out of the baseline cohort from the ELSA-Brasil, a multicenter cohort study aimed at investigating biological, behavioral, environmental, occupational, psychological, and social factors related to incidence and progression of diabetes and cardiovascular disease. Methodological details have been reported elsewhere [19, 20]. Briefly, from August 2008 to December 2010, ELSA-Brasil recruited employees aged 35–74 years working in universities and research institutions located in the Northeast, Southeast and Southern regions of Brazil, with varied socioeconomic conditions and skin color distribution. Sample size of ELSA-Brasil was calculated based on estimations of incidence of type 2 diabetes mellitus and myocardial infarction for the Brazilian population. The current study involved a random sample of 998 individuals without diabetes or cardiovascular disease aged 35–54 years, drawn from 5,061 participants at the São Paulo center of the ELSA-Brasil for whom information regarding serum cystatin C (sCys C) was available [21]. The sample showed the same demographic and socioeconomic distributions. The study was approved by the local Ethics Committee and informed consent was obtained from all participants.
Eligibility criteria
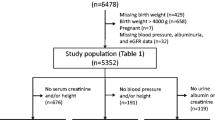
Of the initial 998 participants, 17 with newly diagnosed diabetes and 16 with low estimated glomerular filtration rate (eGFR < 60 mL/min/1.73 m2) were not included. Fifty individuals born with macrosomia, 18 with thyroid dysfunctions (TSH < 0.1 or ≥ 10.0 µUI/mL), eight underweight individuals due to consumptive syndrome (unwanted weight loss higher than 10% without apparent cause) and two with liver dysfunctions (ALT or AST levels three-fold the normal range) were excluded. Since macrosomia confers potential risk of metabolic disorders and our aim was to specifically analyze low birth weight and prematurity, macrosomia was an exclusion criterion. Participants with missing data regarding exposure (birth weight) or outcomes "(sCys C, estimated glomerular filtration rate [eGFR] and albumin-to-creatinine ration [ACR]) were also excluded (n = 119), as shown in Fig. 1. The excluded participants did not change the main composition of the final sample. A total of 768 participants were included in the present analysis.
Clinical and laboratory data
Interviews and anthropometric data were collected by trained personnel using standardized questionnaires [22]. Prematurity (yes or no) and birth weight were self-reported since birth records were not available for the entire sample. Participants were asked to report their weight at birth and the variable was classified into “ < 2.5 kg”, “2.5–4.0 kg”, “ > 4.0 kg” or “unknown”. All participants were asked to provide their specific birth weight and to recall their body weight at 20 years of age.
Sociodemographic and health factors of interest for this study were: age (years), sex (male or female), self-reported skin color (black, white, brown, yellow, and indigenous), parental history of diabetes and hypertension (yes or no) and educational levels of the participant and their mother. The latter was used as a socio-economic trajectory marker since it was available in the first wave of ELSA-Brasil. For the purpose of the present study, skin colors were grouped into white and non-white categories.
Blood pressure was measured using an Omron HEM 705CPINT device (Omron Co, Kyoto, Japan) after a 5-min rest in a sitting position. Three measurements were taken at 1-min intervals and mean values calculated. Body mass index (BMI) was calculated as weight in kilograms divided by height in meters squared. After overnight fasting, blood and urine samples were obtained, and participants then underwent a 2-h 75 g oral glucose tolerance test. Aliquots of biological samples were frozen at − 80 °C for further analyses [23, 24].
Fasting and 2-h post-load plasma glucose levels were measured using the hexokinase method (ADVIA Chemistry; Siemens, Deerfield, Illinois, USA) and glycated hemoglobin was evaluated by high-pressure liquid chromatography (HPLC) (Bio-Rad Laboratories, Hercules, California, USA), according to the National Glycohemoglobin Standardization Program (NGSP) certified method. Glomerular filtration rate was estimated using the equations proposed by the Chronic Kidney Disease Epidemiology Collaboration (eGFR CKD-EPI) [25]. Albuminuria was determined in a 12-h overnight urine sample by nephelometry and expressed as ACR. Urinary creatinine was measured using the kinetic Jaffe method (Advia 1200 Siemens, USA). Participants were instructed to avoid strenuous exercise and alcohol consumption for 24 h preceding the clinical visit. Serum cystatin C was measured using a human cystatin C ELISA kit (Elabscience Biotechnology, Houston, Texas, USA). Intra- and inter-assay coefficient of variability ranges were 5.05–5.38% and 3.29–6.48%, respectively.
Definitions
Prematurity was defined by an affirmative answer to the question: “Were you a premature baby, that is, were you born earlier than expected?”. Birth weight was classified into three categories: low birth weight (< 2.5 kg), normal birth weight (2.5–4.0 kg) and macrosomia (> 4.0 kg). Outcomes were blood pressure levels, eGFR, ACR and sCys C, and were analyzed as continuous variables.
Hypertension was diagnosed when systolic or diastolic blood pressure levels were ≥ 140 or 90 mmHg, respectively, or when the individual was on antihypertensive medication. Kidney function was considered altered when eGFR < 60 mL/min/1.73 m2 or urinary albumin excretion > 1000 mg/g creatinine, according to KDIGO 2012 [26]. Normal ranges of sCys C were 0.64–0.84 mg/L for men and 0.57–0.74 mg/L for women [27].
Diabetes and pre-diabetes were defined according to the ADA (American Diabetes Association) criteria [28]. Diabetes was diagnosed when fasting plasma glucose was ≥ 126 mg/dL or 2-h post 75-g glucose load > 200 mg/dL or glycated hemoglobin ≥ 6.5%. Prediabetes (yes or no) was defined as fasting plasma glucose between 100 and 125 mg/dL or 2-h post 75-g glucose load between 140 and 199 mg/dL or glycated hemoglobin 5.7–6.4%. Nutritional status was stratified by BMI into underweight (< 18.5 kg/m2), normal weight (18.5 and 24.9 kg/m2), overweight (25 and 29.9 kg/m2) and obese (> 30 kg/m2). Insulin sensitivity was assessed using the Homeostasis Model Assessment of Insulin Resistance (HOMA-IR) and considered a continuous variable [29].
Statistical analysis
Continuous variables with normal distribution were expressed as mean ± standard deviation (SD) and compared using Student’s t-test. Non-normal distributed parametric variables were expressed as median and interquartile range and compared using the Wilcoxon rank test. Categorical variables were expressed as absolute and relative frequencies and compared using the chi-squared test.
Associations of LBW (exposure) with outcomes (blood pressure levels and kidney function parameters) were initially analyzed by simple linear regression. Directed Acyclic Graphs (DAGs) were used to build theoretical models to analyze independent associations of exposure with outcomes in multiple linear regression analyses. The DAG is a causal diagram which allows the input of scientific evidence regarding the relationships among variables in graphics software to reach the ideal set of covariables (minimum sufficient adjustment) for the model to prevent bias and overadjustments [30, 31]. For the construction of the DAG for blood pressure and kidney function, several confounder variables were considered, such as age, sex, skin color, weight at age 20, maternal education level, prematurity, income, current BMI, smoking and others as shown in Figure S1 of the Supplementary material (Panels A and B). The figures were created using DAGitty software, version 3.0 (www.dagitty.net).
Having applied the DAG, the covariables selected for adjustments in the linear regression models regarding the association of LBW with blood pressure levels were: race/skin color, sex, parental history of hypertension, and prematurity. Linear regression models for the associations of LBW with kidney function parameters (eGFR, ACR and sCys C) were adjusted for skin color and prematurity.
Considering the contrasting sample sizes of the groups with normal and LBW, as well as possible selection bias due to the observational nature of the study, propensity score matching was employed to create more comparable groups [32, 33]. The nearest neighbor-matching algorithm, within a caliper of 0.1 SD of logit function of propensity score was used. Firstly, to apply the propensity score matching, a multiple logistic regression model was used, adjusted for DAG-based covariates (blood pressure was adjusted for skin color, sex, parental history of hypertension and prematurity, while kidney function markers were adjusted for skin color and prematurity), and the probability of each participant being LBW versus normal birth weight was estimated. Balance between the groups was assessed by comparing each covariate. When standardized mean difference lay in the –0.1 to 0.1 range, groups were considered balanced. This matching was able to reduce all covariate imbalance in the sample. Using the matched sample, multiple linear regression analyses were then performed to test associations of LBW with blood pressure and kidney function markers (outcomes) adjusted for the same DAG-based covariates. Tests were performed using “MatchIt”, “rbounds”, “Matching”, “twang” and “survey” packages in the R statistical environment.
Additionally, sensitivity analyses were performed by selecting the participants with self-reported preterm birth (prematurity). Also, when blood pressure levels were outcomes, sensitivity analyses were carried out excluding participants who reported use of antihypertensive agents (Supplementary Table S1). Since similar results were obtained in both analyses, preterm and hypertensive participants were retained for subsequent analysis.
All analyses were performed using R Project for Statistical Computing (R version 3.5.2) and statistical significance was set at a p value of 0.05.
Results
In the sample of 768 participants, mean age was 45.5 ± 4.6 years, 56.8% were female, and 60% reported white skin color. A total of 64 (8.3%) participants reported being LBW and 39 (5.0%) born preterm. The LBW group comprised participants that were predominantly female (54.6%), white skinned (56.0%) and had mothers with low educational level (71.9%). These characteristics showed a similar pattern in the preterm subgroup (67.0% female and 61.0% white skin color).
A total of 133 participants fulfilled the diagnostic criteria for hypertension, which was more frequent in the LBW than in the normal birth weight group (16.2% versus 29.7%, p = 0.024). Among the hypertensive participants in the normal and LBW groups, 8.1% and 14.0% were receiving antihypertensive treatment, respectively.
Overall, cardiometabolic risk profile and kidney function of the total sample were within normal ranges, except for being generally overweight (mean 26.0 kg/m2 ± 4.15) and having borderline fasting plasma glucose (102.0 mg/dL ± 7.5). Mean values of systolic (116.7 ± 14.7 mmHg) and diastolic (74.9 ± 10.5 mmHg) blood pressure, eGFR (89.6 ± 13.8 mL/min/1.73 m2), ACR (11.0 ± 39.0 mg/g creatinine) and sCys C (0.74 ± 0.61 mg/L) were within normal ranges.
Low birth weight participants were, on average, older than normal birth weight subjects (47.0 ± 5.5 years versus 45.3 ± 4.8 years, p = 0.027) and had a higher prevalence of low educational level (6.3 versus 3.7%, p = 0.025). Mean BMI values were lower at age 20 (21.2 ± 2.81 versus 20.7 ± 3.0 kg/m2, p = 0.196) than at current age (26.2 ± 4.1 versus 26.0 ± 4.4 kg/m2, p = 0.688) for both normal and LBW participants, respectively, with no difference between subgroups.
Participants with LBW had higher mean blood pressure and ACR, and lower eGFR, than individuals with normal birth weight, but there were no group differences for sCys C (Table 1).
The preterm subgroup also had significantly higher mean systolic and diastolic blood pressure levels, but no difference in kidney function markers was detected (Supplementary Table S2).
In multiple linear regression analysis, LBW was associated with blood pressure levels, but this association did not persist after adjustment for prematurity, which remained associated with systolic and diastolic blood pressure (Table 2). Associations of LBW with eGFR and ACR in the fully adjusted models had borderline significance (p = 0.05) (Table 3). In a separate analysis of only preterm participants, prematurity was independently associated with both systolic and diastolic blood pressure (Supplementary Table S3).
Imbalance of variables
Initially, the sample had 64 LBW participants. Propensity score matching was applied, yielding sub-samples with matched participants (normal and LBW) as follows: (a) 48 matched participants for systolic and 61 matched participants for diastolic blood pressure; (b) 44 matched participants for eGFR; and (c) 49 matched participants for ACR with the same proportion of normal and LBW participants in each sub-sample. The matching approach rendered all covariates appropriately balanced (standardized mean difference of between –0.1 and 0.1) for further analyses. Imbalance for all variables was assessed before and after matching to compare improvement by matching (Supplementary Table S4).
Associations of LBW with blood pressure and kidney function markers after propensity score matching
The multiple linear regression model, adjusted for skin color and prematurity, showed that being born with low weight was directly associated with ACR values (β 1.34; 95%CI 0.47–2.20; p = 0.003). Even when reaching adequate balance, LBW was not associated with eGFR or with systolic and diastolic blood pressure levels (Table 4).
Discussion
We hypothesized that being born with LBW is associated with higher blood pressure levels and impaired kidney function markers in adulthood, indicating an insult during intrauterine life. Our findings corroborate this hypothesis since we found that all LBW participants showed higher blood pressure levels, and exhibited lower eGFR and higher ACR than those with normal birth weight. Enhancing our analysis by propensity score matching and DAG-based adjustments on multiple linear regression boosted confidence in the associations found between early-life events and outcomes in adulthood. We suggest that individuals with LBW may be at increased risk of renal abnormalities even when routine parameters are within normal values.
Hypertension is a major cause of chronic kidney disease (CKD) and both are prevalent disorders globally [34, 35]. Additionally, these conditions increase the risk for cardiovascular events and deaths [36]. Therefore, several studies have sought to identify predictors for the prevention of these diseases. There is evidence that the pathophysiological process starts in early life, congruent with the DOHaD theory [37, 38]. Our main results are in line with the DOHaD, which holds that insults in fetal life induce programming and that LBW is a surrogate of this condition. An interesting finding of the present study was that prematurity was associated with blood pressure levels, yet LBW was not. Previous studies have reported an association of prematurity with elevated blood pressure levels [39, 40]. Proposed mechanisms linking prematurity to higher blood pressure levels in adult life involve pre- and postnatal factors. Premature birth has been associated with increased vascular resistance [41, 42], endothelial dysfunction [43], immature autonomic blood pressure regulation [44] and high sympathetic nervous system activity [13]. The extent of these abnormalities is dependent on the fetal adaptations and cardiovascular system programming in response to an adverse intrauterine environment during a critical period of development [45]. It is known that the full number of nephrons is achieved at between 32 and 36 weeks of gestation and that glomerular filtration starts during intrauterine life [14, 46]. Therefore, prematurity may cause morphological and functional renal changes that lead to hemodynamic disturbances and hypertension [47]. Most preterm infants have accelerated postnatal growth during the first years of life [48], where this compensatory catch-up growth is commonly associated with rapid body weight gain and obesity in childhood [49]. We speculate that the overweight participants in the present study have an increased risk of hypertension later in life.
Several nutritional, toxic or emotional (maternal anxiety or depression) determinants of LBW – regardless of gestational age at delivery – can impact adult kidney function [50,51,52], assessed by a number of parameters such as GFR, serum creatinine, sCys C and albuminuria in clinical practice. Elevated ACR is a recognized biomarker of kidney dysfunction, even when eGFR is within normal ranges [53]. In the present study, mean values of eGFR and ACR were significantly worse in the LBW group compared to the other groups.
Furthermore, having employed propensity score matching, a strong association of LBW was found with albuminuria, but not with eGFR or sCys C. We emphasize that ACR might be a useful marker for early detection of kidney dysfunction in adults born with LBW. The higher albuminuria seen in LBW individuals might have been due, in part, to fetal glomerular morphological alterations and prenatal programming. This notion is supported by evidence demonstrating that LBW individuals have fewer nephrons [54], lower glomerular volume at birth [55], abnormal glomeruli structure with enlarged Bowman’s capsule and altered glomerular tufts [56, 57], factors that, in the long-term, could result in increased glomerular permeability and albuminuria. The present sample of LBW individuals without overt nephropathy may have had glomerular hypertrophy of the remaining nephrons and hyperfiltration. The current mean eGFR of this group was within the normal range, as was albuminuria, but remained significantly lower than the group with normal birth weight. Whether ACR, periodically measured, serves as a suitable marker of early kidney dysfunction in LBW individuals requires investigation by appropriately designed studies. This is relevant considering the synergistic impact of the nephron mass on the development of hypertension favoring CKD and cardiovascular disease later in life.
We elected to measure sCys C based on previous reports suggesting its greater accuracy than creatinine for detecting decreased renal function [58,59,60]. SCys C determination, combined with creatinine, can be used in GFR estimation (KDIGO 2012) [26]. However, in the present sample with normal eGFR, sCys C failed to detect kidney dysfunction in LBW individuals.
This study has limitations in relation to recall bias, given that data about early life events as well as body weight at 20 years were collected retrospectively. These self-reported data could not be validated due to the non-availability of birth records or other validation methods, such as medical records, for all participants. However, our sample cohort was middle-aged and previous studies have shown that perinatal-related events can be accurately reported during adult life [61,62,63]. We also used the exposure variable (LBW or prematurity) as a categorical variable, thus this may have reduced the statistical power of the analysis. This variable was chosen so as to minimize information bias from participants unable to accurately report their birth weight. The frequency of self-reported prematurity in our sample was comparable to the rate reported in the Brazilian population at large [5]. Despite being non-representative of the general Brazilian population, employees enrolled in ELSA-Brasil represent a heterogeneous sample regarding socioeconomic and ethnic-racial background [19]. As far as ACR is concerned, the lack of a direct measurement of muscle mass in which creatinine is generated could be a limitation. Taking into consideration that the distributions of BMI values in each subgroup were very similar, and the lack of difference between mean urinary creatinine between normal birth weight and LBW groups, it is unlikely that urinary concentrations of creatinine interfered in our ACR results. Therefore, we considered ACR measurement as a reliable method for the assessment of proteinuria in our sample [64].
A strength of the present study was its methodological approach. DAG was employed to avoid overadjustments [30, 31] when performing the regression analysis. However, although several covariates were incorporated into our theoretical model, including body weight at age 20, other exposures throughout the participants’ life course were unavailable. Size difference of the groups with normal birth weight and LBW was also a concern; therefore, propensity score matching was applied to reduce potential selection confounders from observational studies [32, 33] and sufficiently balanced groups were achieved.
In conclusion, prematurity was found to be associated with higher blood pressure levels and LBW with albuminuria in adulthood. Considering the magnitude of the ELSA-Brasil and potentialities of its cohort [20, 24], our findings are important to raise awareness of early life events and to be alert to a subset of healthy adults born with LBW who are at increased risk for hypertension and kidney dysfunction. Long birth cohort studies could help confirm the hypothesis raised in this study.
Data Availability
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
References
Barker DJ, Fall CH (1993) Fetal and infant origins of cardiovascular disease. Arch Dis Child 68(6):797–799. https://doi.org/10.1136/adc.68.6.797
Jaddoe WV, Witterman JCM (2006) Hypotheses on the fetal origins of adult diseases: Contributions of epidemiological studies. Eur J of Epidemiol 21:91–102. https://doi.org/10.1007/s10654-005-5924-5
Wilcox AJ (2001) On the importance and the unimportance of birthweight. Int J Epidemiol 30:1233–1241. https://doi.org/10.1093/ije/30.6.1233
WHO (2014) Low birth weight policy brief 2014. Global nutrition targets 2025. World Health Organization, Geneva
de Souza Buriol VC, Hirakata V, Goldani MZ, da Silva CH (2016) Temporal evolution of the risk factors associated with low birth weight rates in Brazilian capitals (1996–2011). Popul Health Metr 14:15. https://doi.org/10.1186/s12963-016-0086-0
Würtz P, Wang Q, Niironen M, Tynkkynen T, Tiainen M, Drenos F et al (2016) Metabolic signatures of birthweight in 18 288 adolescents and adults. A meta-analysis. Int J Epidemiol 45(5):1539–1550. https://doi.org/10.1093/ije/dyw255
Das SK, Mannan M, Faruque ASG, Ahmed T, Mcintyre HD, Mamun AA (2016) Effect of birth weight on adulthood renal fuction: a bias-adjusted meta-analytic approach. Nephrology 21(7):547–565. https://doi.org/10.1111/nep.12732
Zhao M, Shu XO, Jin F, Yang G, Li HL, Liu DK, Wen W, Gao YT, Zheng W (2002) Birthweight, childhood growtha and hypertension in adulthood. Int J Epidemiol 31(5):1043–1051. https://doi.org/10.1093/ije/31.5.1043
Alexander BT, Dasinger JH, Intapad S (2015) Fetal programming and cardiovascular pathology. Compr Physiol 5(2):997–1025. https://doi.org/10.1002/cphy.c140036
Jansson T, Lambert GW (1999) Effect of intrauterine growth restriction on blood pressure, glucose tolerance and sympathetic nervous system activity in the rat at 3–4 months age. J Hypertens 17:1239–1248. https://doi.org/10.1097/00004872-199917090-00002
Franco MCP, Casarini DE, Carneiro-Ramos MS, Sawaya AL, Barreto-Chaves MLM, Sesso R (2008) Circulating renin-angiotensin system and catecholamines in childhood: is there a role for birthweight? Clin Sci (Lond) 114(5):375–380. https://doi.org/10.1042/CS20070284
Martinez-Aguayo A, Aglony M, Bancalari R, Avalos C, Bolte L, Garcia H, Loureiro C, Carvajal C, Campino C, Inostroza A, Fardella C (2012) Birth weight is inversely associated with blood pressure and serum aldosterone and cortisol levels in children. Clin Endocrinol (Oxf) 76(5):713–718. https://doi.org/10.1111/j.1365-2265.2011.04308.x
Boguszewski MCS, Johannsson G, Fortes LC, Sverrisdottir YB (2004) Low birth size and final height predict high sympathetic nerve activity in adulthood. J Hypertens 22(6):1157–1163. https://doi.org/10.1097/00004872-200406000-00017
Hinchliffe SA, Sargent PH, Howard CV, Chan YF, Velzen DV (1991) Human intrauterine renal growth expressed in absolute number of glomeruli assessed by the disector method and Cavalieri principle. Lab Invest 64(6):777–784
Black MJ, Sutherland MR, Gubhaju L, Kent AL, Dahlstrom JE, Moore L (2013) When birth comes early: effects on nephrogenesis. Nephrology (Carlton) 18:180–182. https://doi.org/10.1111/nep.12028
Lucas SR, Costa Silva VL, Miraglia SM, Zaladek Gil F (1997) Functional and morphometric evaluation of offpring kidney after intrauterine undernutrition. Pediatr Nephrol 11(6):719–723. https://doi.org/10.1007/s004670050374
Luyckx VA, Brenner BM (2005) Low birth weight, nephron number and kidney disease. Kidney Int Suppl 97:S68-77. https://doi.org/10.1111/j.1523-1755.2005.09712.x
Ritz E, Nowicki M, Fliser D, Hörner D, Klimm HP (1994) Proteinuria and hypertension. Kidney Int Suppl 47:S76-80
Schmidt MI, Duncan BB, Mill JG, Lotufo PA, Chor D, Barreto SM, Aquino EM, Passos VM, Matos SM, Molina Mdel C, Carvalho MS, Bensenor IM (2015) Cohort profile: Longitudinal Study of Adult Health (ELSA-Brasil). Int J Epidemiol 44(1):68–75. https://doi.org/10.1093/ije/dyu027
Aquino EM, Barreto SM, Bensenor IM, Carvalho MS, Chor D, Duncan BB, Lotufo PA, Mill JG, Molina Mdel C, Mota EL, Passos VM, Schmidt MI, Szklo M (2012) Brazilian Longitudinal Study of Adult Health (ELSA-Brasil): objectives and design. Am J Epidemiol 175(4):315–324. https://doi.org/10.1093/ije/dyu027
Almeida-Pititto B, Ribeiro-Filho FF, Lotufo PA, Bensenor IM, Ferreira SRG (2015) Novel biomarkers of cardiometabolic risk are associated with plasma glucose within non-diabetic range. The Brazilian Longitudinal Study of Adult Health – ELSA-Brasil. Diabetes Res Clin Pract 109(1):110–116. https://doi.org/10.1016/j.diabres.2015.04.021
Bensenor IM, Griep RH, Pinto KA, Faria CP, Felisbino-Mendes M, Caetano EI, Albuquerque Lda S, Schmidt MI (2013) Routines of organization of clinical test and interviews in the ELSA-Brasil investigation center. Rev Saude Publica 47(Suppl 2):37–47. https://doi.org/10.1590/s0034-8910.2013047003780
Fedeli LG, Vidigal PG, Leite CM, Castilhos CD, Pimentel RA, Maniero VC, Mill JG, Lotufo PA, Pereica AC, Bensenor IM (2013) Logistics of collection and transportation of biological samples and the organization of the central laboratory in the ELSA-Brasil. Rev Saude Publica 47(Suppl 2):63–71. https://doi.org/10.1590/s0034-8910.2013047003807
Pereira AC, Bensenor IM, Fedeli LM, Castilhos C, Vidigal PG, Maniero V, Leite CM, Pimentel RA, Duncan BB, Mill JG, Lotufo PA (2013) Design and implementation of the ELSA-Brasil biobank: a prospective study in a Brazilian population. Rev Saude Publica 47(Suppl 2):72–78. https://doi.org/10.1590/s0034-8910.2013047003822
Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF, Feldman HI, Kusek JW, Eggers P, Van Lente F, Greene T, Coresh J (2009) CKD-EPI (chronic kidney disease epidemiology collaboration). A new equation to estimate glomerular filtration rate. Ann Intern Med 150(9):604–612. https://doi.org/10.7326/0003-4819-150-9-200905050-00006. (Erratum in: Ann Intern Med 2011;155(6):408)
Kidney Disease Improving Global Outcomes - KDIGO 2012. Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease (2013) Official Journal of the International Society of Nephrology. Kidney Int Suppl 3(1):7–10
Galteau MM, Goyun M, Gueguen R, Siest G (2001) Determination of serum cystatin C: biological variation and reference values. Clin Chem Lab Med 39(9):850–857. https://doi.org/10.1515/CCLM.2001.141
American Diabetes Association (2015) Classification and diagnosis of diabetes. Diabetes Care 38(Suppl 1):S8–S16
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC (1985) Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28(7):412–419
Hernán MA, Robins JM (2006) Instruments for causal inference: an epidemiologist’s dream? Epidemiology 17(4):360–372. https://doi.org/10.1097/01.ede.0000222409.00878.37
Greenland S, Pearl J, Robins JM (1999) Causal diagrams for epidemiologic research. Epidemiology 10(1):37–48
Austin PC (2011) An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivar Behav Res 46(3):399–424. https://doi.org/10.1080/00273171.2011.568786
Haukoos JS, Lewis RJ (2015) The propensity score. JAMA 314(15):1637–1638. https://doi.org/10.1001/jama.2015.13480
Romagnani P, Remuzzi G, Glassock R, Levin A, Jager KJ, Tonelli M, Massy Z, Wanner C, Anders HJ (2017) Chronic kidney disease. Nat Rev Dis Primers 3:17088. https://doi.org/10.1038/nrdp.2017.88
WHO (2017) Global Health Observatory (GHO) data. Raised blood pressure. World Health Organization, Geneva
Arnett DK, Blumenthal RS, Albert MA, Buroker AB, Goldberger ZD et al (2019) ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease: a report of the College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 140(11):e596–e646. https://doi.org/10.1161/CIR.0000000000000678
Barker DJ (1999) Fetal origins of cardiovascular disease. Ann Med 31(Suppl 1):3–6. https://doi.org/10.1080/07853890.1999.11904392
Sutherland MR, Gubhaju L, Moore L, Kent AL, Dahlstrom JE, Horne RSC, Hoy WE, Bertram JF, Black MJ (2011) Accelerated maturation and abnormal morphology in the preterm neonatal kidney. J Am Soc Nephrol 22(7):1365–1374. https://doi.org/10.1681/ASN.2010121266
Juonala M, Cheung MMH, Sabin MA, Burgner D, Skilton MR et al (2015) Effect of birth weight on life-course blood pressure levels among children born premature: the Cardiovascular Risk in Young Finns Study. Hypertension 33:1542–1548. https://doi.org/10.1097/HJH.0000000000000612
de Jong F, Monuteaux MC, Elburg RM, Gillman MW, Belfort MB (2012) Systematic review and meta-analysis of preterm birth and later systolic blood pressure. Hypertension 59(2):226–234. https://doi.org/10.1161/HYPERTENSIONAHA.111.181784
Lazdam M, de la Horra A, Pitcher A, Mannie Z, Diesch J, Trevitt C, Kylintireas I et al (2010) Elevated blood pressure in offspring born premature to hypertensive pregnancy: is endothelial dysfunction the underlying vascular mechanism? Hypertension 56:159–165. https://doi.org/10.1161/HYPERTENSIONAHA.110.150235
Leeson CP, Kattenhorn M, Morley R, Lucas A, Deanfield JE (2001) Impact of low birth weight and cardiovascular risk factors on endothelial function in early adult life. Circulation 103:1264–1268. https://doi.org/10.1161/01.cir.103.9.1264
Bourque SL, Gragasin FS, Quon AL, Mansour Y, Morton JS, Davidge ST (2013) Prenatal hypoxia causes long-term alterations in vascular endothelin-1 function in aged male, but not female, offspring. Hypertension 62(4):753–758. https://doi.org/10.1161/HYPERTENSIONAHA.113.01516
Skilton MR, Viikari JSA, Juonala M, Laitinen T, Lehtimäki T, Taittonen L, Kähönen M, Celermajer DS, Raitakari OT (2011) Fetal growth and preterm birth influence cardiovascular risk factors and arterial health in young adults: the Cardiovascular Risk in Young Finns Study. Arterioscler Thromb Vasc Biol 31(12):2975–2981. https://doi.org/10.1161/ATVBAHA.111.234757
Barker DJP (2007) The origins of the developmental origins theory. J Intern Med 261(5):412–417. https://doi.org/10.1111/j.1365-2796.2007.01809.x
Osathanondh V, Potter EL (1963) Development of human kidney as shown in by microdissection. III. Formation and interrelationship of collecting tubules and nephrons. Arch Pathol 76:290–302
Bertagnolli M, Luu TM, Lewandowski AJ, Leeson P, Nuyt AM (2016) Preterm birth and hypertension: is there a link? Curr Hypertens Rep 18(4):28. https://doi.org/10.1007/s11906-016-0637-6
Giannì ML, Roggero P, Liotto N, Amato O, Piemontese P, Morniroli D, Bracco B, Masca F (2012) Postnatal catch-up fat after late preterm brith. Pedriatr Res 72(6):637–640. https://doi.org/10.1038/pr.2012.128
Ou-Yang MC, Sun Y, Liebowitz M, Chen CC, Fan ML, Dai W, Chuang TW, Chen JL (2020) Accelerated weight gain, prematurity, and the risk of childhood obesity: a meta-analysis and systematic review. PLoS ONE 15(5):e0232238. https://doi.org/10.1371/journal.pone.0232238
Silverwood RJ, Pierce M, Hardy R, Sattar N, Whincup P, Ferro C, Savage C, Kuh D, Nitsch D (2013) Low birth weight, later renal function, and the roles of adulthood blood pressure, diabetes, and obesity in a British birth cohort. Kidney Int 84(6):1262–1270. https://doi.org/10.1038/ki.2013.223
White SL, Perkovic V, Cass A, Chang CL, Poulter NR, Spector T, Haysom L, Craig JC, Salmi IA, Chadban SJ, Huxley RR (2009) Is low birth weight an antecedent of CKD in later life? A systematic review of observational studies. Am J Kidney Dis 54(2):248–261. https://doi.org/10.1053/j.ajkd.2008.12.042
Ritz E, Amann K, Koleganova N, Benz K (2011) Prenatal programming-effects on blood pressure and renal function. Nat Rev Nephrol 7(3):137–144. https://doi.org/10.1038/nrneph.2011.1
de Jong PE, Gansevoort RT (2009) Focus on microalbuminuria to improve cardiac and renal protection. Nephron Clin Pract 111:c204-211. https://doi.org/10.1159/000201568
Hughson M, Farris AB 3rd, Douglas-Denton R, Hoy WE, Bertram JF (2003) Glomerular number and size in autopsy kidneys: the relationship to birth weight. Kidney Int 63(6):2113–2122. https://doi.org/10.1046/j.1523-1755.2003.00018.x
Mañalich R, Reyes L, Herrera M, Melendi C, Fundora I (2000) Relationship between weight at birth and the number and size of renal glomeruli in humans: a histomorphometric study. Kidney Int 58(2):770–773. https://doi.org/10.1046/j.1523-1755.2000.00225.x
Zohdi V, Sutherland MR, Lim K, Gubhaju L, Zimanyi MA, Black MJ (2012) Low birth weight due to intrauterine growth restriction and/or preterm birth: effects on nephron number and long-term renal health. Int J Nephrol. https://doi.org/10.1155/2012/136942
Rodríguez MM, Gómez AH, Abitbol CL, Chandar JJ, Duara S, Zilleruelo GE (2004) Histomorphometric analysis of postnatal glomerulogenesis in extremely preterm infants. Pediatr Dev Pathol 7(1):17–25. https://doi.org/10.1007/s10024-003-3029-2
Laterza OF, Price CP, Scott MG (2002) Cystatin C: an improved estimator of glomerular filtration rate? Clin Chem 48(5):699–707
Fassett RG, Venuthurupalli SK, Gobe GC, Coombes JS, Cooper MA, Hoy WE (2011) Biomarkers in chronic kidney disease: a review. Kidney Int 80(8):806–821. https://doi.org/10.1038/ki.2011.198
Qiu X, Liu C, Ye Y, Li H, Chen Y, Fu Y, Liu Z, Huang X, Zhang Y, Liao X, Liu H, Zhao W, Liu X (2017) The diagnostic value of serum creatinine and cystatin C in evaluating glomerular filtration rate in patients with chronic kidney disease: a systematic literature review and meta-analysis. Oncotarget 8(42):72985–72999. https://doi.org/10.18632/oncotarget.20271
Chin HB, Baird DD, McConnaughey DR, Weinberg CR, Wilcox AJ, Jukic AM (2017) Long-term recall of pregnancy-related events. Epidemiology 28(4):575–579. https://doi.org/10.1097/EDE.0000000000000660
Promislow JHE, Gladen BC, Sandler DP (2005) Maternal recall of breastfeeding duration by elderly women. Am J Epidemiol 161(3):289–296. https://doi.org/10.1093/aje/kwi044
Tomeo CA, Rich-Edwards JW, Michels KB, Berkey CS, Hunter DJ, Frazier AL, Willett WC, Buka SL (1999) Reproducibility and validity of maternal recall of pregnancy-related events. Epidemiology 10(6):774–777
Cirillo M (2010) Evaluation of glomerular filtration rate and of albuminuria/proteinuria. J Nephrol 23(2):125–132
Acknowledgements
The authors would like to acknowledge the participation of the 5,061 individuals recruited for this study, without them this study and those based on the ELSA-Brasil cohort would not have been possible.
Funding
The current study was supported by a grant from the São Paulo Research Foundation (Fundação de Amparo à Pesquisa do Estado de São Paulo—FAPESP—Protocol 2009/15041-9). Also it was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nı́vel Superior—Brasil (CAPES)—(Finance Code 001). The baseline ELSA-Brasil study was supported by the Brazilian Ministry of Health (Science and Technology Department), the Brazilian Ministry of Science and Technology, and CNPq-National Research Council (# 01 06 0010.00 RS, 01 06 0212.00 BA, 01 06 0300.00 ES, 01 06 0278.00 MG, 01 06 0115.00 SP, 01 06 0071.00 RJ).
Financial interestThe authors have no financial interests to disclose.
Author information
Authors and Affiliations
Consortia
Contributions
Study design, analysis, interpretation and preparation of the manuscript: JB, BA-P and SF. Acquisition of data and critical revision for the manuscript content: PL and IB. All authors listed have made a substantial, direct, and intellectual contribution to the work and approved it for publication.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical disclosures
The study was approved by the National Commission on Ethics Research (CONEP) and the local ethics committee, the Research Ethics Committee (CEP) under registration number 76 of the University of São Paulo (HU-USP). The patients/participants provided written informed consent to participate in this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Branda, J.I.F., de Almeida-Pititto, B., Bensenor, I. et al. Associations of prematurity and low birth weight with blood pressure and kidney function in middle-aged participants of the Brazilian Longitudinal Study of Adult Health: ELSA-Brasil. J Nephrol 36, 1373–1382 (2023). https://doi.org/10.1007/s40620-022-01549-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40620-022-01549-w