Abstract
Purpose of Review
To summarize the current knowledge of diagnosis and management of patients with perioperative immediate hypersensitivity reactions (POH)
Recent Findings
The diagnosis and management of POH is a challenge for both allergologists and anesthesiologists. They are rare entities; its difficulty lies in its poor clinical specificity, at the time of the acute event, and also in the lack of standardization of its etiological study, which favors that multiple reactions are not adequately studied, with the subsequent risk of re-exposure to the causative agent.
Summary
POH investigation aims are to identify a culprit drug and safe alternatives, and to ensure safe future anesthesia, even if no culprit is identified. The diagnosis goes through the recognition and treatment in the acute phase with adrenaline, obtaining tryptase samples as a marker of suspected IgE mechanism, and the collection of all the substances involved in the reaction. Clinical history and skin tests are the fundamental diagnostic tools; also specific IgE, basophil activation test, and drug provocation tests with anesthetics in highly specialized centers can be used. We recommended that all patients with suspected POH undergo a thorough investigation, increasing the collaboration between allergologists and anesthesiologists and also creating specialized units integrated by both.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Suspected perioperative immediate hypersensitivity reactions (POH) are rare but can be life-threatening. The diagnosis is difficult to make and is not standardized.
POH investigation aims are to identify the culprit drug and safe alternatives, and also to ensure safe future anesthesia, even if no culprit is identified.
The purpose of this document is to update the information, and highlight the importance of the problem and the need for coordination between allergy and anesthesia services to address the problem.
Incidence and Causal Agents
The incidence has been estimated from 1:353 to 1:18.600 anesthetics procedures with high variability influenced by the heterogeneity of studies and by differences in terminology, local practice, and drug use [1,2,3, 4••]. Recent data from France [5] and the United Kingdom, Sixth National Audit Project (NAP6) [6••], suggest an estimated incidence of 1:10,000. The NAP6 only considered severe reactions, so it is estimated that considering all the incidence would be higher 1:7000 [6••].
Recent epidemiological studies highlight emerging agents such as antibiotics, chlorhexidine, dyes, macrogols, or sugammadex compared with classics such as muscle relaxants and latex [3, 6••, 7, 8].
Antibiotics and neuromuscular blocking agents (NMBAs) are the leading causes of allergy during the perioperative process [3, 6••, 7]. While reactions to antibiotics are increasing in many countries, NMBAs still represent a major cause in France, Belgium, Australia, and in the UK. The emergence of teicoplanin in the UK has supplanted amoxicillin or amoxicillin-clavulanic acid as a major cause and reflects its increasing use in patients with a label of penicillin allergy [6••].
Reactions to latex are decreasing with the implementation of primary and secondary preventive measures [9••]. The increased use of blue dyes in cancer surgery and of chlorhexidine as a disinfectant may justify the increase in reactions to these agents [3].
Recent studies suggest that local anesthetics are an anecdotal cause of POH [10].
Sugammadex which is used to reverse the effect of neuromuscular relaxation, especially of rocuronium, has spread universally, and hypersensitivity reactions and IgE-mediated anaphylaxis have been reported [11].
Ethylene oxide is another emerging agent, especially in patients undergoing surgery for neural tube closure defects or carriers of peritoneal dialysis catheters [12].
In the pediatric population, there are few epidemiological studies. The estimated incidence ranges from 1:1774 anesthesias under the study by GERAP [5, 7], with latex being the most prevalent agent in children from 2 to 14 years and muscle relaxants and antibiotics in adolescents 14 to 18 years, at 2.73:100,000 anesthesia in the NAP6 [6••], being the most prevalent agent atracurium and without any case caused by latex.
Mechanisms
The clinical presentation of the reactions is not specific, and it is impossible to distinguish between different mechanisms.
Only subsequent allergological evaluation can identify the cause and rule out the rest of the agents as involved. It is also necessary for the participation of anesthetists to rule out non-allergological symptoms related to the effects of anesthetic drugs and surgical management [9••, 13••, 14, 15, 16••] (Table 1).
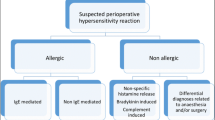
Hypersensitivity reactions may depend on 2 mechanisms: immunological mechanisms (allergic reactions) and non-immunological mechanisms [18]. From allergic reactions, IgE-mediated reactions represent about 60% of all reactions, and their severity can increase in subsequent surgeries [5, 16••, 19, 20]. Rarely, immunological reactions are mediated by IgG like protamin and dextrans [13••].
The rest of the reactions are mediated by a non-immune inflammatory mechanism, including nonspecific activation of complement and nonspecific mast cell and basophilic mediator release [21], such as via the recently discovered MRGPRX2 receptor (Mas-related G protein-coupled receptor member X2) [22]. The latter would be involved in reactions to vancomycin, morphine, atracurium, and cisatracurium. Also, another group of non-immunologically mediated reactions would be related to the inhibition of cycloxygenase-1, as in the case of nonsteroidal anti-inflammatory drugs (NSAIDs) or the kinin-kallikrein system in bradykinin-induced angioedema [13••].
The different mechanisms and mediators are likely to contribute to the variation in the clinical presentation and severity of POH and may be correlated with an increase in serum tryptase [23•]. Grades I and II reactions are not life-threatening and more likely to be non-allergic (i.e., nonspecific activation of mast cells and basophils). Grades III and IV reactions are life-threatening, fulfill the criteria for anaphylaxis, and are more likely to be IgE-mediated [7, 24].
Diagnosis
The management of POH reactions is based on 2 well-differentiated steps in the initial diagnosis in the acute phase of the reaction and later the allergological investigation to confirm the causal agent (if it is possible).
Initial Diagnosis
In the acute phase, the anesthesiologist is responsible for the management of PHO. It consists of the diagnosis and obtaining biological samples for the determination of tryptase.
Recognizing the Reaction: Clinical Presentation
In addition to the complexity in the etiological diagnosis, it is also difficult the recognition of POH in the surgical area for their unspecific clinic and sudden appearance. The clinical symptoms must be addressed in a wide differential diagnosis with numerous events derived from the anesthetic technique, the pharmacological agents used, the surgical procedure itself, and the patient’s pathology [9••, 13••, 14, 15, 16••] (Table 1).
Most perioperative immediate allergic reactions occur around the time of induction of anesthesia [25,26,27,28].
In the clinical setting, it is important to consider anaphylaxis as a differential diagnosis when perioperative hypotension or bronchospasm does not respond to usual therapy or when cardiac arrest occurs unexpectedly during anesthesia [6••, 26, 29, 31].
Cardiovascular symptoms are the most frequent, especially in adults. There are less frequent entities such as paradoxical bradycardia or Kounis syndrome or acute coronary syndrome associated with anaphylaxis that may occur during POH. Skin symptoms may be absent in up to 20% of reactions, especially the most severe, appearing at the end of the resuscitation phase. Therefore, the absence of these should not be a reason to rule out the diagnosis of suspicion. Isolated angioedema is not common in POH, and it should make us suspect other entities, especially if it coincides with airway manipulation. Bronchospasm and increased airway pressure in the patient in mechanical ventilation, as a single symptom, are also not frequent, except in the asthmatic patient or with chronic obstructive pulmonary disease.
Most clinical guidelines use the modified Ringer and Mesmer classification for anesthesia [25, 32] to classify the clinical phenotypes of patients, the severity of reaction, and location of the patient after initial treatment of the reaction.
The severity and mortality of perioperative anaphylaxis may be influenced by a higher ASA physical status (Physical Status Classification System of the American Society of Anesthesiology), age, associated comorbidities (obesity and coronary artery disease), ongoing beta-blockers, and angiotensin-converting-enzyme inhibitor (ACE-I) treatment [6••, 25] and also the elevation of basal tryptase [33]. Subjects with a clonal mast cell disorder might be at higher risk of severe reactions with either specific or nonspecific triggers [17].
Serum Tryptase
This determination is essential for further diagnosis; it is currently recommended in all consensus documents on the management of POH [9••, 13••, 16••, 34••]. High tryptase is indicative of mast cell degranulation and is useful for differential diagnosis of the reaction. A tryptase sample should be taken between 1 and 3 h after a suspected POH and a baseline sample for comparison, obtained at least 24 h post reaction [9••, 25, 26]. The elevation of the tryptase is considered relevant when tryptase at the time of reaction is > 2 + 1.2 × baseline tryptase [9••, 14, 16••]. This formula has recently been validated for POH with sensitivity, specificity, positive predictive value, and negative predictive value of 78%, 91%, 98%, and 44% respectively [35•]. In patients who present suspected anaphylaxis, a normal tryptase level does not rule out an IgE-mediated anaphylactic event as false negatives occur [36]. Elevated baseline levels might be indicative of underlying clonal mast cell disorders, especially in severe POH [17, 37]. The high stability allows measuring a postmortem tryptase sampling if necessary [38].
Allergological Investigation
The etiological study is the responsibility of the allergologist. The aim is to confirm or disprove an allergic mechanism behind the reaction, and to identify culprit agent(s) and safe drugs, including suitable alternatives for the next anesthesia.
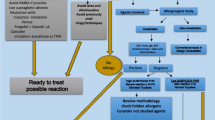
Figure 1 shows the management of patients with suspected POH who have not been studied before. We distinguish the management for urgent or elective procedures and also the diagnostic tests that have to be carried out in elective or urgent procedures when we have obtained or not the clinical information of the reaction. An important aspect is to determine which agents should be studied for whom clinical information of the previous reaction is not available. In these cases, skin testing should be performed with all the essential agents needed for anesthesia, including a NMBAs and an opioid [16••]. We must not forget that the ultimate goal of the study is to provide safe alternatives for further anesthesia.
As a general rule, we had to study all drugs involved regardless if we found a positive culprit drug, because there is a risk of more than one culprit involved [29]. When the culprit is positive, cross-sensitivity should be investigated, if relevant. For NMBAs, all available NMBAs should be tested, and for antibiotics, NSAIDs, and local anesthetics, at least one safe alternative should be identified.
The diagnosis of perioperative hypersensitivity reactions is based on the combination of the clinical history and tests performed once the reaction has resolved. These include skin tests, in vitro tests, and sometimes drug provocation testing. Also, in vitro determinations performed during the acute phase of the reaction are necessary.
POH can be produced for different mechanisms. The IgE-mediated reactions are the most severe, and in vitro tests and skin tests can identify them. Differential diagnosis with non-immunological reaction needs collaboration with anesthesiologists.
The diagnosis of nonspecific histamine release is mostly concluded after further investigations when all substances test negative, and this is corroborated by the history and a normal tryptase result [13••]. Positive identification of them would require specific drug provocation, but as in most of centers drug provocation testing (DPT) is not a standard, therefore, often diagnoses of exclusion.
Some guidelines recommend that drug allergy investigation is performed at least 4–6 weeks after the event [25, 26], but in some cases (clinical urgency), patients are tested (skin and specific IgE) within a few days of anaphylaxis [23•, 31, 39]. However, it would be prudent to consider repeating investigations a few weeks later if skin tests or in vitro test are negative after 1 to 2 months [23•, 40, 41].
Clinical History
It is essential to validate the rest of the diagnostic tests. Those patients without a medical history suggestive of POH should not be subject to study. Most reactions occur during anesthetic induction [25, 26, 28], 90% of cases, but this temporality should not be used to determine the probable causative agent.
Skin Testing
It is the most widely used method to identify the culprit agent in IgE-mediated reactions, comprising skin prick tests (SPTs) and intradermal tests (IDTs). Using optimal non-irritant concentrations for SPT and IDT, as is recently recommended in a position paper by the EAACI [9••], these tests should be performed by experienced personnel. When SPT is negative or inconclusive, IDT is performed. As patients with POH are considered high-risk, most guidelines recommend titrated skin testing for both SPT and IDT using 2–3 dilutions with 20-min intervals.
In Vitro Testing
Specific IgE Testing (sIgE)
It is not available to all agents that may be potentially involved; it can be used with latex, suxamethonium, morphine, pholcodine, bovine gelatine, protamine, chlorhexidine, ethylene oxide, and beta-lactam antibiotics [16••]. In the case of latex and chlorhexidine which has a high positive predictive value, its value is limited for the rest of the agents due to its low sensitivity and specificity [9••]. Results should ideally be interpreted in relation to the total IgE value. Its elevation indicates IgE sensitization, but it does not always rise to clinical allergy.
Basophil Activation Test
This test is based on flow cytometry with different strategies to identify basophils (anti-IgE, CCR3, CRTH2, and CD203c) and to measure their activation (CD63 and CD203c) after the stimulation with the culprit drug or their metabolites [42]. This test can be used in cases of IgE-mediated and non-IgE-mediated hypersensitivity reactions (e.g., opiates) for identifying the culprit drug, cross-reactivity, or safe alternative drugs. It must be performed by experienced laboratories [9••]. Its realization does not imply a risk for the patient and allows us to assess a large number of agents. Their role in the study of POHs would reside in cases where other in vitro or cutaneous tests are negative or inconclusive, before carrying out [43] complex and risk provocation tests, as in the case of muscle relaxants. Its usefulness has also been assessed in the case of agents such as sugammadex or opiates where we do not have a specific IgE yet and where skin tests have limitations due to the nonspecific release of histamine produced by some of them.
The sensitivity of basophil activation test (BAT) for NMBAs varies between 36 and 92%, and the specificity between 81 and 100% [23•, 44]. BAT complements skin tests in the diagnostic workup and also enables assessment of cross-reactivity between NMBAs [23•, 45]. Because negative skin tests do not always guarantee the subsequent safe use of the NMBA [46], there are cases where BAT is positive in a patient with a skin test of negative [47, 48], and also, BAT can confirm the diagnosis of rocuronium-induced mast cell activation in patients with negative skin tests [49].
Drug Provocation Testing
It is considered the gold standard test in drug allergy and is used to establish a diagnosis when other tests are negative [43]. It is a common practice in allergy services for many drugs, but the anesthetic agents involved in these reactions added complexity due to the strong pharmacological effects, and DPT has not been recommended in the investigation of POH. Recently, the European [9••] and Spanish guidelines [16••], and ISPAR group [50••] advised the use of DPT in perioperative allergy investigation. They consider there is a place for DPT in perioperative allergy investigation due to the false negative result on conventional testing which can have fatal consequences.
The aim is to exclude sensitization to the culprit drug or to test a safe alternative [51, 52].
Its main indication is when clinical suspicion of allergy is high, but skin tests, in vitro tests, or both, are equivocal or negative; where false positive skin test results are suspected; and finally, when no other reliable diagnostic tests are available.
In the Allergy Anesthesia Unit of Hospital Central de la Cruz Roja, we usually perform DPT with all the drugs involved in the reaction, including anesthetics (propofol, opiates, and other induction agents). If the clinic is suggestive and the rest of the tests (SPT, IDT, and specific IgE) are negative, a four-step continuous iv protocol with increases in doses and infusion rate every 15 min is usually performed until reaching full therapeutic doses. In the case of NMBAs, we previously do BAT.
It is important to emphasize that DPT should be undertaken in close collaboration between anesthesiologists and allergologists in highly specialized centers. DPT should be performed by trained personnel, in an appropriate clinical setting, with access to resuscitation facilities, after a thorough evaluation of the clinical history, and after appropriate allergological workup. A careful individual risk-benefit analysis should be made, and the benefit should always clearly outweigh the risks.
Patient comorbidity, factors related to the reaction, and the future indication for individual drugs should be considered. Assessment of patient comorbidity includes age, medical history (especially cardiac or pulmonary), systemic mastocytosis or elevated baseline serum tryptase, current medications, and the physical status of the patient usually classified using the ASA system [50••, 53••].
Treatment and Management
Treatment of the Reaction
The mortality of POHs is close to 4% [25, 54], and one of the variables associated with the risk of this is the delay in diagnosis and the administration of adrenaline [13••, 55]. Its treatment is similar to other forms of anaphylaxis, and the only therapeutic measures that have proven effective in treatment are the initiation of cardiopulmonary resuscitation maneuvers, and the administration of fluid therapy, oxygen therapy, and adrenaline. The adrenaline administration is not contraindicated in any patient as long as its dose is titrated according to the severity of the reaction and the clinical response or associated adverse effects [13••]. The sugammadex has been suggested in rocuronium refractory anaphylaxis; however, actual studies show conflicting evidence, and we cannot recommend its use as clinical practice in this indication [11].
Table 2 summarizes the basic therapeutic measures in anaphylaxis according to the degree of severity and the location of the patient after it. In refractory anaphylaxis cases, after using repeated bolus adrenaline and/or fluids, we will consider using norepinephrine adrenaline, glucagon, and/or vasopressin or its analogs. Once the resuscitation with adrenaline is finished, we can use corticosteroids and antihistamines, although their efficacy is not demonstrated in the acute treatment of the reaction [34••].
The treatment guided by the clinical presentations is a general principle including special situations such as a pregnant woman (the parturient should be positioned with left uterine displacement so that aortocaval compression is avoided) or pediatrics patients where fluid therapy with crystalloids (20 ml kg−1, repeated as needed) is recommended [26, 56, 57••], and epinephrine remains the drug of choice, but there is no consensus on dosage [25, 26, 56, 57••].
Immediate Management After Reaction
Once the patient has been treated and stabilized after a suspected POH, communication with the surgical team during the reaction is essential to determine the suspension, continuation, or urgent completion of the surgery. This decision will depend on the type of surgery (e.g., elective vs emergency) and its indication [58]. Outcomes were similar in cases where surgery had been abandoned or proceeded with (once initial stabilization had been achieved) for all grade I to III cases of POH [59]. However, in cases that occur before the start of elective surgery, the most prudent could be the suspension.
Also, the anesthesiologist will be in charge according to the severity of the reaction, to locate the patient or proceed to his medical discharge (Table 2).
All patients should be observed in a monitored area for a minimum of 6 h from the onset of the reaction. Most patients with grades III and IV reactions will require admission to an intensive care department [34••].
Informing the Patient
Before the patient is discharged, the anesthesiologist should give him/her a clinical report containing all relevant information about the reaction, including its severity, any treatment administered, and all agents involved. The patient should be warned that if anesthesia is needed again before the allergology investigation, it should only be for emergency procedures.
Referral for Allergological Evaluation
All grades II–IV reactions and grade I reactions with generalized urticaria, erythema, or both should be referred for allergy investigation [9••]. Ideally, investigations should take place in specialized clinics with collaboration between anesthesiologists and allergologists with experience in perioperative allergy investigation [9••, 60].
It is recommended to send all the information through a standardized data referral document, filling in all the agents used and chronological narrative of events. Copies of anesthetic charts, drug charts, including premedication, surgical notes, and operating room documentation, should be collected [9••, 14, 16••]. Examples of referral documents can be found on www.nationalauditprojects.org.uk/NAP6-Resources (from NAP6) and in recently published Spanish or European Academy of Allergy and Clinical Immunology (EAACI) guidelines [9••, 16••].
Any compound that the patient was exposed to within a 1–2-h timeframe (depending on the route of administration) before symptom onset could be the potential culprit [9••]. Remind that only the compounds documented will be investigated [61], so those not necessarily recorded on the anesthetic chart such as gels, sprays, hemostatic agents, chlorhexidine, excipients, and blue dyes should be meticulously registered too.
Prevention
It would include a series of measures aimed to avoid POH or reduce if their severity occurs.
- 1.
Detection in pre-anesthesia consultations of risk patients. The main identified risk factor of perioperative drug anaphylaxis is a previously uninvestigated POH [62••]. The anesthesiologist should refer to the allergy consultation those patients with previous POH or suspicious reactions, not explained by another cause, who are scheduled for elective surgery. It should not be referred for study to patients with allergies to drugs that are not used during the anesthetic act or with a history of atopy or food allergies [62••]. Urgent or non-delayable surgery, for example, oncology, implies other recommendations, which as a final objective, shall be to avoid all agents that might be involved in the previous reaction (Fig. 1).
- 2.
Avoid the presence of latex in surgical areas and the use of low-latex proteins and powder-free gloves to reduce latex sensitization [62••]. The risk of sensitization is higher in patients younger than 1 year, especially in procedures for the correction of neural tube closure defects and genitourinary malformations. Therefore, it is advisable to develop strategies to eliminate latex in pediatric hospitals. Clinical features suggestive of previous latex-induced immediate hypersensitivity require subsequent investigation. Latex avoidance is required in proven or highly suspected latex allergy avoidance if the delay is not compatible with the surgery [62••].
- 3.
There is no evidence to avoid the use of propofol in egg, soy, or peanut allergy [9••, 62••, 63, 64]
- 4.
Several guidelines on POH recommend that iv antibiotics should be administered before anesthetic induction, in the hospitalization or pre-anesthesia room, provided that its administration window is respected [6••, 9••, 16••, 28, 60]. In the case of presenting a POH, this would simplify the etiological diagnosis since a few suspected drugs are involved. It has also been suggested to administer antibiotics as a slow infusion [24].
- 5.
The assessment of patients with a history of allergy to beta-lactam antibiotics in the preoperative period has been the subject of discussion because of the risk of a higher rate of surgical infections and perioperative morbidity and mortality [65]. The fact that only between 10 and 30% of patients with a history of reaction to beta-lactam antibiotics are truly allergic, it has led certain authors to justify the use of cephalosporins in low-risk patients [66]. Although there is no unanimous agreement on this, these patients should be a subject of attention to assessing the most appropriate strategy for acting before them.
- 6.
The reported cases of anaphylaxis to chlorhexidine products are increasing [6••, 62••]; most of the reactions occurred after the use of urological lubricants containing chlorhexidine or central venous catheters impregnated with it. To minimize the risk of sensitization, central venous catheters, impregnated with chlorhexidine, is not recommended routinely in all settings, only after strict risk-benefit assessment [67]. The US Centers for Disease Control and Prevention considers the routine use of antiseptic urethral lubricants ‘unnecessary’ [68].
- 7.
Clinical guidelines suggest that there is no evidence [26, 56] or unlikely [25, 26] for antihistamines and steroids to prevent IgE-mediated reactions. Premedication with antihistamines 2–3 days prior to anesthesia and slow administration of the drugs may reduce or prevent mild reactions caused by nonspecific histamine release from mast cells and basophils and histamine-releasing effect of drugs (for example opioids, NMBAs, vancomycin, and thiopentone) [9••, 16••, 26, 69].
Conclusions
-
1.
The diagnosis of POH is complicated due to the nonspecific clinical symptoms, the number of drugs, and the different mechanisms involved.
-
2.
The prompt recognition of the reaction and correct treatment are necessary to ensure a proper outcome. Adrenaline is essential for the treatment of these reactions.
-
3.
All patients with suspected POH undergo a thorough investigation to avoid the morbidity and mortality associated with repeat episodes of avoidable anaphylaxis.
-
4.
It is mandatory to investigate all substances involved in the reaction even though a culprit drug was found because more than one can contribute to the reaction.
-
5.
Serum tryptase elevated suggests IgE-mediated POH reaction. When it is elevated and all results are negative, the patient should be reevaluated (reassessing methodology and hidden allergens).
-
6.
As sensitivity and specificity are not 100% for any test, the results of single tests should always be interpreted in the context of relevant clinical information and serum tryptase results. Authors recommend performing several tests both to avoid false positives and to increase the sensitivity of the tests [41, 70].
-
7.
Only some centers can do BAT, which has demonstrated high specificity and usefulness even in cases of negative skin tests [9••, 13••].
-
8.
Only highly specialized centers perform DPT with anesthetic drugs. All this leads us to conclude that the creation of these centers should be important [13••, 50••].
Unmet needs
The diagnosis and management of POH involve great complexity and sometimes can be inadequate or inconclusive. For this reason, it is necessary to implement a series of improvement measures:
- 1.
Collaboration between anesthesiologists and allergologists is very important in order to improve the diagnosis and treatment of POH and these will allow:
Improvement of communications of perioperative reactions.
Establishing local referral documentation so anesthesiologists can collect the data of reaction in a standardized manner, and it can reach the allergy departments properly.
Including perioperative anaphylaxis treatment packs, containing a laminated treatment algorithm with an emphasis on iv epinephrine and fluids, and instructions for tryptase sampling [9••, 16••, 26, 34••, 57••], also including adrenaline prefilled syringes when they are available.
Improving the challenge test with anesthetic agents.
Improving the dissemination of management guidelines developed in recent years.
Creating specific units integrated by allergologists and anesthesiologists, with appropriate knowledge and experience in the investigation and management of drug allergy [9••]. These units may constitute a true regional or state network, which will allow the development of epidemiological studies, protocols for action and diagnosis, and training programs.
Developing clinical scores to facilitate rapid recognition of reactions in a complex environment [71•].
Implementing clinical simulation programs for anesthesiologists to optimize the recognition and management of reactions [72]. Also, methods of safely diluting epinephrine should be part of perioperative anaphylaxis training.
- 2.
Future challenges in perioperative allergy investigation include the continued optimization of skin and in vitro tests, specific IgE, and BAT to minimize the need for DPT.
- 3.
Auditing re-expositions to anesthesia after drug allergy investigation can validate the tests and the allergological investigation.
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Lobera T, Audicana MT, Pozo MD, Blasco A, Fernández E, Cañada P, et al. Study of hypersensitivity reactions and anaphylaxis during anesthesia in Spain. J Investig Allergol Clin Immunol. 2008;18:350–6.
Savic LC, Kaura V, Yusaf M, Hammond-Jones A-M, Jackson R, Howell S, et al. Incidence of suspected perioperative anaphylaxis—a multicenter snapshot study. J Allergy Clin Immunol Pract. 2015:1–3.
Mertes PM, Volcheck GW, Garvey LH, Takazawa T, Platt PR, Guttormsen AB, et al. Epidemiology of perioperative anaphylaxis. Presse Med. 2016. https://doi.org/10.1016/j.lpm.2016.02.024.
•• Mertes PM, Ebo DG, Garcez T, et al. Comparative epidemiology of suspected perioperative hypersensitivity reactions. Br J Anaesth. 2019;123:e16–28 Recent revision by ISPAR group of the epidemiology of suspected perioperative hypersensitivity reactions.
Mertes PM, Alla F, Tréchot P, Auroy Y, Jougla E. Anaphylaxis during anesthesia in France: an 8-year national survey. J Allergy Clin Immunol. 2011;128:366–73.
Harper NJN, Cook TM, Garcez T, et al. Anaesthesia, surgery, and life-threatening allergic reactions: epidemiology and clinical features of perioperative anaphylaxis in the 6th National Audit Project (NAP6). Br J Anaesth. 2018;121:159–71 Epidemiology and clinical features of perioperative anaphylaxis in the 6th National Audit Project P6) in UK.
Tacquard C, Collange O, Gomis P, Malinovsky JM, Petitpain N, Demoly P, et al. Anaesthetic hypersensitivity reactions in France between 2011 and 2012: the 10th GERAP epidemiologic survey. Acta Anaesthesiol Scand. 2017;61:290–9.
Wenande E, Garvey LH. Immediate-type hypersensitivity to polyethylene glycols (PEGs): a review. Clin Exp Allergy. 2016:907–22.
•• Garvey LH, Ebo DG, Mertes P, et al. An EAACI position paper on the investigation of perioperative immediate hypersensitivity reactions. Allergy. 2019;74:1872–84 Latest European Academy of Allergy and Clinical Immunology guidelines of perioperative anaphylaxis, including currently recommended skin test concentrations for the most commonly used drugs.
Kvisselgaard AD, Krøigaard M, Mosbech HF, Garvey LH. No cases of perioperative allergy to local anaesthetics in the Danish Anaesthesia Allergy Centre. Acta Anaesthesiol Scand. 2017;61:149–55.
Takazawa T, Mitsuhata H, Mertes PM. Sugammadex and rocuronium-induced anaphylaxis. J Anesth. 2016;30:290–7.
Opstrup MS, Mosbech H, Garvey LH. Allergic sensitization to ethylene oxide in patients with suspected allergic reactions during surgery and anesthesia. J Investig Allergol Clin Immunol. 2010;20:269–70.
•• Garvey LH, Dewachter P, Hepner DL, et al. Management of suspected immediate perioperative allergic reactions: an international overview and consensus recommendations. Br J Anaesth. 2019;123:e50–64 ISPAR group management consensus of perioperative allergic reactions after a modified Delphi consensus process, which covered areas such as differential diagnosis, management during and after anaphylaxis, allergy investigations, and plans for a subsequent anaesthetic.
Garvey LH. Perioperative hypersensitivity reactions: diagnosis, Treatment and Evaluation. Curr Treat Options Allergy. 2016;3:113–28. https://doi.org/10.1007/s40521-016-0078-0.
Dewachter P, Mouton-Faivre C, Emala CW, Beloucif S. Case scenario: bronchospasm during anesthetic induction. Anesthesiology. 2011;114:1200–10.
•• Laguna J, Archilla J, Doña I, et al. Practical guidelines for perioperative hypersensitivity reactions. J Investig Allergol Clin Immunol. 2018;28:216–32 Spanish guidelines, with proposal of referral document and recommendation for drug provocation test with anaesthetics drugs.
Bonadonna P, Pagani M, Aberer W, et al. Drug hypersensitivity in clonal mast cell disorders: ENDA/EAACI position paper. Allergy Eur J Allergy Clin Immunol. 2015;70:755–63.
Johansson SGO, Bieber T, Dahl R, Friedmann PS, Lanier BQ, Lockey RF, et al. Revised nomenclature for allergy for global use: report of the Nomenclature Review Committee of the World Allergy Organization, October 2003. J Allergy Clin Immunol. 2004;113:832–6.
Dong S, Acouetey DS, Guéant-Rodriguez R-M, Zmirou-Navier D, Rémen T, Blanca M, et al. Prevalence of IgE against neuromuscular blocking agents in hairdressers and bakers. Clin Exp Allergy. 2013;43:1256–62.
Florvaag E, Johansson SGO, Oman H, Venemalm L, Degerbeck F, Dybendal T, et al. Prevalence of IgE antibodies to morphine. Relation to the high and low incidences of NMBA anaphylaxis in Norway and Sweden, respectively. Acta Anaesthesiol Scand. 2005;49:437–44.
Baldo BA, Pham NH. Histamine-releasing and allergenic properties of opioid analgesic drugs: resolving the two. In: Anaesth. Intensive Care; 2012. p. 216–35.
McNeil BD, Pundir P, Meeker S, Han L, Undem BJ, Kulka M, et al. Identification of a mast-cell-specific receptor crucial for pseudo-allergic drug reactions. Nature. 2014;519:237–41.
• Takazawa T, Sabato V, Ebo DG. In vitro diagnostic tests for perioperative hypersensitivity, a narrative review: potential, limitations, and perspectives. Br J Anaesth. 2019;123:117–25 ISPAR group narrative review of in vitro test for diagnosis perioperative hypersensitivity reactions.
Dewachter P, Mouton-Faivre C, Hepner DL. Perioperative anaphylaxis: what should be known? Curr Allergy Asthma Rep. 2015;15:21.
Mertes PM, Malinovsky JM, Jouffroy L, Aberer W, Terreehorst I, Brockow K, et al. Reducing the risk of anaphylaxis during anesthesia: 2011 updated guidelines for clinical practice. J Investig Allergol Clin Immunol. 2011;21:442–53.
Kroigaard M, Garvey LH, Gillberg L, Johansson SG, Mosbech H, Florvaag E, et al. Scandinavian clinical practice guidelines on the diagnosis, management and follow-up of anaphylaxis during anaesthesia. Acta Anaesthesiol Scand. 2007;51:655–70.
Harper NJN, Dixon T, Dugué P, Edgar DM, Fay A, Gooi HC, et al. Suspected anaphylactic reactions associated with anaesthesia. Anaesthesia. 2009;64:199–211.
Dewachter P, Mouton-Faivre C, Emala CW. Anaphylaxis and anesthesia: controversies and new insights. Anesthesiology. 2009;111:1141–50.
Leysen J, Witte L De, Bridts CH, Ebo DG (2013) Anaphylaxis during general anaesthesia: a 10-year survey 1 at the University Hospital of Antwerp. Proceedings of the Belgian Royal Academics of Medicine 2:88–100.
Malinovsky J-M, Decagny S, Wessel F, Guilloux L, Mertes PM. Systematic follow-up increases incidence of anaphylaxis during adverse reactions in anesthetized patients. Acta Anaesthesiol Scand. 2008;52:175–81.
Lafuente A, Javaloyes G, Berroa F, Goikoetxea MJ, Moncada R, Sanz ML, et al. Early skin testing is effective for diagnosis of hypersensitivity reactions occurring during anesthesia. 2013;68:820–2.
Ring JMK. Incidence of anaphylactoid reactions following infusion with colloid volume substitutes. Lancet. 1977;1:466–9.
Mirone C, Preziosi D, Mascheri A, et al. Identification of risk factors of severe hypersensitivity reactions in general anaesthesia. Clin Mol Allergy. 2015;13:11.
•• Harper NJN, Cook TM, Garcez T, et al. Anaesthesia, surgery, and life-threatening allergic reactions: management and outcomes in the 6th National Audit Project (NAP6). Br J Anaesth. 2018;121:172–88 Management life-threatening perioperative anaphylaxis reactions allergic reaction in the 6th National Audit Project (NAP6) in UK.
• Baretto RL, Beck S, Heslegrave J, Melchior C, Mohamed O, Ekbote A, et al. Validation of international consensus equation for acute serum total tryptase in mast cell activation: a perioperative perspective. Allergy. 2017;72:2031–4 Validation of the algorithm for interpretation of serum tryptase with a comparison of acute levels with patients own baseline level.
Fisher MM, Baldo BA. Mast cell tryptase in anaesthetic anaphylactoid reactions. Br J Anaesth. 1998;80:26–9.
Dewachter P. Perioperative management of patients with mastocytosis. Anesthesiology. 2014;120:753–9.
Laroche D, Gomis P, Gallimidi E, Malinovsky JM, Mertes PM. Diagnostic value of histamine and tryptase concentrations in severe anaphylaxis with shock or cardiac arrest during anesthesia. Anesthesiology. 2014;121:272–9.
Schulberg EM, Webb AR, Kolawole H. Early skin and challenge testing after rocuronium anaphylaxis. Anaesth Intensive Care. 2016;44:425–7.
Soetens F, Rose M, Fisher M. Timing of skin testing after a suspected anaphylactic reaction during anaesthesia. Acta Anaesthesiol Scand. 2012;56:1042–6.
Opstrup MS, Mosbech H, Skov PS, Poulsen LK. Standardized testing with chlorhexidine in perioperative allergy – a large single-centre evaluation. 2014;69:1390–6.
Mayorga C, Celik G, Rouzaire P, et al. In vitro tests for drug hypersensitivity reactions: an ENDA/EAACI Drug Allergy Interest Group position paper. Allergy Eur J Allergy Clin Immunol. 2016. https://doi.org/10.1111/all.12886.
Demoly P, Adkinson NF, Brockow K, et al. International consensus on drug allergy. Allergy Eur J Allergy Clin Immunol. 2014;69:420–37.
Ebo DG, Faber M, Elst J, Van Gasse AL, Bridts CH, Mertens C, et al. In vitro diagnosis of immediate drug hypersensitivity during anesthesia: a review of the literature. J Allergy Clin Immunol Pract. 2018;6:1176–84.
Ebo DG, Bridts CH, Hagendorens MM, Mertens CH, De Clerck LS, Stevens WJ. Flow-assisted diagnostic management of anaphylaxis from rocuronium bromide. Allergy. 2006;61:935–9.
Sabato V, Ebo DG. Hypersensitivity to neuromuscular blocking agents: can skin tests give the green light for re-exposure? J Allergy Clin Immunol Pract. 2018;6:1690–1.
Salas M, Fernández-Santamaría R, Mayorga C, et al. Use of the basophil activation test may reduce the need for drug provocation in amoxicillin-clavulanic allergy. J Allergy Clin Immunol Pract. 2018;6:1010–1018.e2.
Laguna JJ, Bogas G, Salas M, et al. The basophil activation test can be of value for diagnosing immediate allergic reactions to omeprazole. J Allergy Clin Immunol Pract. 2018;6:1628–1636.e2.
Van Gasse AL, Elst J, Bridts CH, Mertens C, Faber M, Hagendorens MM, et al. Rocuronium hypersensitivity: does off-target occupation of the MRGPRX2 receptor play a role? J Allergy Clin Immunol Pract. 2019;7:998–1003.
•• Garvey LH, Ebo DG, Krøigaard M, et al. The use of drug provocation testing in the investigation of suspected immediate perioperative allergic reactions: current status. Br J Anaesth. 2019;123:e126–34 A consensus of ISPAR group of drug provocation tests in perioperative anaphylaxis.
Aberer W, Bircher A, Romano A, Blanca M, Campi P, Fernandez J, et al. Drug provocation testing in the diagnosis of drug hypersensitivity reactions: general considerations. Allergy. 2003;58:854–63.
Bousquet P-J, Gaeta F, Bousquet-Rouanet L, Lefrant J-Y, Demoly P, Romano A. Provocation tests in diagnosing drug hypersensitivity. Curr Pharm Des. 2008;14:2792–802.
ASA Physical Status Classification System | American Society of Anesthesiologists (ASA). https://www.asahq.org/standards-and-guidelines/asa-physical-status-classification-system. Accessed 5 Dec 2019.
Gibbs NM, Sadleir PH, Clarke RC, Platt PR. Survival from perioperative anaphylaxis in Western Australia 2000-2009. Br J Anaesth. 2013;111:589–93.
Garvey LH, Belhage B, Krøigaard M, Husum B, Malling HJ, Mosbech H. Treatment with epinephrine (adrenaline) in suspected anaphylaxis during anesthesia in Denmark. Anesthesiology. 2011;115:111–6.
Harper NJN, Dixon T, Dugué P, et al. Guidelines suspected anaphylactic reactions associated with anaesthesia. Anaesthesia. 2009;64:199–211.
•• Kolawole H, Marshall SD, Crilly H, Kerridge R, Roessler P. Special article Australian and New Zealand Anaesthetic allergy group/Australian and New Zealand College of Anaesthetists perioperative anaphylaxis management guidelines. Anaesth Intensive Care. 2017;45:151–9 Australian and New Zealand perioperative anaphylaxis guiedelines.
Garvey LH. Practical aspects of perioperative anaphylaxis. Trends Anaesth Crit Care. 2013;3:320–6.
Sadleir PHM, Clarke RC, Bozic B, Platt PR. Consequences of proceeding with surgery after resuscitation from intra-operative anaphylaxis. Anaesthesia. 2018;73:32–9.
Scolaro RJ, Crilly HM, Maycock EJ, McAleer PT, Nicholls KA, Rose MA, et al. Australian and New Zealand anaesthetic allergy group perioperative anaphylaxis investigation guidelines. Anaesth Intensive Care. 2017;45:543–55.
Garvey L. Old, new and hidden causes of perioperative hypersensitivity. Curr Pharm Des. 2016;22:6814–24.
•• Dewachter P, Kopac P, Laguna JJ, Mertes PM, Sabato V, Volcheck GW, et al. Anaesthetic management of patients with pre-existing allergic conditions: a narrative review. Br J Anaesth. 2019;123:e65–81 ISPAR group narrative review of anaesthetic management of patients with pre-existing allergic conditions.
Molina-Infante J, Arias A, Vara-Brenes D, Prados-Manzano R, Gonzalez-Cervera J, Alvarado-Arenas M, et al. Propofol administration is safe in adult eosinophilic esophagitis patients sensitized to egg, soy, or peanut. Allergy Eur J Allergy Clin Immunol. 2014;69:388–94.
American Academy of Allergy Asthma and Immunology. Soy-allergic and Egg-allergic can Receive Anesthesia | AAAAI. https://www.aaaai.org/conditions-and-treatments/library/allergy-library/soy-egg-anesthesia. Accessed 4 Dec 2019.
Blumenthal KG, Ryan EE, Li Y, Lee H, Kuhlen JL, Shenoy ES. The impact of a reported penicillin allergy on surgical site infection risk. Clin Infect Dis. 2018;66:329–36.
Blumenthal KG, Shenoy ES, Varughese CA, Hurwitz S, Hooper DC, Banerji A. Impact of a clinical guideline for prescribing antibiotics to inpatients reporting penicillin or cephalosporin allergy. Ann Allergy Asthma Immunol. 2015;115:294–300.e2.
Sadleir PH, Platt PR. Chlorhexidine anaphylaxis: again and again. Anaesth Intensive Care. 2019;47:13–5.
CDC (2019) Centers for Disease Control and Prevention. Available from: https://www.cdc.gov/infectioncontrol/guidelines/cauti/index.html. Accessed 22 Dec 2019.
Renz C, Lynch J, Thurn J, Moss J. Histamine release during rapid vancomycin administration. Inflamm Res. 1998;47:69–70.
Leysen J, Bridts CH, De Clerck LS, Vercauteren M, Lambert J, Weyler JJ, et al. Allergy to rocuronium: from clinical suspicion to correct diagnosis. Allergy. 2011;66:1014–9.
• Hopkins PM, Cooke PJ, Clarke RC, et al. Consensus clinical scoring for suspected perioperative immediate hypersensitivity reactions. Br J Anaesth. 2019;123:e29–37 ISPAR group proposal of a clinical scoring for suspected perioperative immediate hypersensitivity reactions to after Delphi consensus process involving a panel of 25 international multidisciplinary experts.
Kolawole H, Guttormsen AB, Hepner DL, Kroigaard M, Marshall S. Use of simulation to improve management of perioperative anaphylaxis: a narrative review. Br J Anaesth. 2019;123:104–9.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Jose Julio Laguna, Cosmin Boteanu, Esther Moreno, Javier Dionicio, Sonsoles Martin, Maria Aranzazu Jimenez-Blanco, Gador Bogas, and Joaquin Archilla declare no conflicts of interest relevant to this manuscript.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Drug Allergy
Laguna JJ, Boteanu C, Moreno E, Dionicio J, Jimenez-Blanco M, and Bogas G participated in the ISCIII RETIC ARADyAL: RD16/0006/0033, RD16/0006/0019, and RD16/0006/0001.
Rights and permissions
About this article
Cite this article
Laguna, J.J., Boteanu, C., Moreno, E. et al. Recommendations for Diagnosing and Management of Patients with Perioperative Drug Reactions. Curr Treat Options Allergy 7, 181–197 (2020). https://doi.org/10.1007/s40521-020-00253-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40521-020-00253-z