Abstract
Background
The state of mild cognitive impairment (MCI) provides an optimal window for preventing progression to dementia. Combined cognitive intervention and physical exercise may yield additive and synergistic effects on cognition in older adults with MCI.
Objectives
The purpose of this study was to assess the efficacy of a combined intervention to improve cognition in older adults with MCI by comparing a control group that underwent only cognitive intervention, a control group that underwent only physical exercise, and a control group that did not undergo cognitive intervention or physical exercise.
Design
Meta-analysis of randomized controlled trials (RCTs).
Data sources
The online databases of PubMed, Web of Science, Embase, the Cochrane Library, PsycINFO, and CINAHL were systematically searched.
Review methods
The outcomes were global cognition, memory, and executive function/attention. A sensitivity analysis was conducted when the I2 statistic was > 50%.
Results
A total of 16 studies were included. The results showed that the combined intervention had positive effects on global cognition compared to the effects of the other control group [SMD = 0.27, 95% CI (0.09, 0.44), p = 0.003]. Regarding memory, the combined intervention had positive effects compared to the effects observed in the single physical exercise group [SMD = 0.25, 95% CI (0.07, 0.44), p = 0.006] and the other control group [SMD = 0.29, 95% CI (0.12, 0.47), p = 0.001]. For executive function/attention, the combined intervention had also positive effects compared to the effects of the single cognitive intervention group [SMD = 0.28, 95% CI (0.09, 0.47), p = 0.004], the single physical exercise group [SMD = 0.32, 95% CI (0.16, 0.49), p = 0.0002], and the other control group [SMD = 0.23, 95% CI (0.05, 0.41), p = 0.01].
Conclusions
The combined intervention resulted in cognitive benefits in older adults with MCI and exhibited limited superiority over the single cognitive intervention and the single physical exercise on cognitive subdomains.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
As the population ages, the number of people worldwide with dementia is expected to reach 82 million by 2030, with one new case every 3 s [1]. Dementia seriously affects the quality of life and well-being of older adults and causes a heavy burden on families and society [2]. Therefore, effective interventions are urgently needed to prevent dementia.
Mild cognitive impairment (MCI) is the intermediate phase between normal age-related cognitive decline and dementia [3]. The mean annual conversion rate of MCI to dementia is approximately 10%, which is far higher than the annual incidence (1–2%) in the general population [4, 5]. However, MCI provides an optimal window for preventing the progression to dementia [6].
Cognitive intervention and physical exercise are more suitable for improving cognitive function in older adults with MCI [7]. Many systematic reviews and meta-analyses have shown that cognitive interventions are effective for improving cognition [8,9,10]. Moreover, two previous systematic reviews reported positive effects of physical exercise on global cognition in older adults with MCI [11, 12].
Combining a single cognitive intervention with a single physical exercise intervention (referred to as a combined intervention) would greatly increase the likelihood of cognitive benefit. Meta-analyses have indicated that the combined intervention positively affect global cognition compared to no cognitive intervention or physical exercise in healthy older adults and older adults with cognitive impairments [13, 14]. However, some studies did not observe the positive effect of the combined intervention on global cognition in older adults with MCI [15, 16], while a new study has shown its effectiveness [17]. It is necessary to update the data.
Additionally, a combined intervention may yield additive and synergistic effects. However, the efficacy of the combined intervention compared to a single cognitive intervention and a single physical exercise intervention remained unclear in older adults with MCI. A recent study found that compared to a group that underwent only cognitive intervention and a group that underwent only physical exercise, the combined intervention significantly improved memory but did not significantly improve global cognition or executive function [18]. However, another new study showed positive effects of the combined intervention on global cognition and executive function compared to a group with only physical exercise [19]. However, a recent systematic review including randomized controlled trials (RCTs) focused on the effects of the combined intervention on cognition in older adults with MCI. It did not separately analyze the effects of the combined intervention, comparing to a group with only cognitive intervention and a group with only physical exercise [20]. Therefore, it is essential to reintegrate the evidence and perform quantitative analysis to clarify the cognitive efficacy of the combined intervention in older adults with MCI.
To explore the cognitive benefits of the combined intervention, the meta-analysis quantified the overall effect of the combined intervention on cognitive function (global cognition, memory, and executive function/attention). This was done by comparing the combined intervention group to the control group that received only cognitive intervention (referred to as the single cognitive intervention group), to the control group that received only physical exercise (referred to as the single physical exercise group), and to the control group that did not receive cognitive intervention or physical exercise (referred to as the other control group).
Methods
The work was reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines [21].
Search strategy
We performed a systematic literature search in two steps. First, we conducted a systematic search of six English databases: PubMed, Web of Science, Embase, the Cochrane Library, PsycINFO, and CINAHL. All databases were searched up to February 2021 based on two main concepts to identify search terms. For the first concept, we used the words related to “mild cognitive impairment” as the search terms. For the second concept, we used the words related to “combined intervention” as the search terms, including a combination of “cognitive intervention” terms, “physical exercise” terms, and combined terms, and supplementing special terms for the combined intervention. Then, the two concepts were combined to retrieve articles. Second, references of selected articles and related reviews were further screened to retrieve additional articles. The full search strategy for PubMed is presented in Supplementary Table 1.
Study selection criteria
Studies were selected according to the following inclusion criteria: (1) patients screened or diagnosed with MCI over the age of 50 years; (2) intervention group with cognitive intervention and physical exercise; (3) control group without cognitive intervention or physical exercise, with only cognitive intervention or with only physical exercise, and the intervention may include other components (e.g., omega-3 fatty acid supplementation); (4) enough information to calculate effect sizes for at least one cognitive outcome (global cognition, memory or executive function/attention); and (5) randomized controlled trial. Studies were excluded if they (1) were unpublished articles; (2) were not written in English; or (3) included patients with MCI caused by brain injury or cancer or with a history of other neurological diseases (e.g., dementia, stroke, Parkinson’s disease) or psychiatric disorders (e.g., depressive or anxiety disorders). Two reviewers screened articles based on title and abstract separately, following further full-text evaluation. If there were disagreements between the two reviewers, the discussion was conducted with a third reviewer to reach a consensus.
Risk of bias assessment
The Cochrane Collaboration Risk of Bias Tool was used for quality assessment [22]. It consisted of six domains biases: selection, performance, detection, attrition, reporting, and another bias. Each domain was rated as “low”, “high”, or “unclear” for each study by two reviewers. Differences were resolved by discussion with another reviewer.
Data extraction
We extracted five types of characteristics from the included studies, including study characteristics (author, published year, and country), sample characteristics (sample size, mean age, percentage of females, education level, and diagnosis criteria), combined intervention group characteristics (combined component, technology, mode of combination, frequency, duration and sessions, and setting), control group characteristics (the single cognitive intervention group, the single physical exercise group, the other control group), and outcome characteristics (neuropsychological tool used for measuring global cognition, memory and executive function/attention at preintervention and postintervention).
The summary statistics were the means and standard deviations (SDs) and the number of participants in each group preintervention and postintervention. When means and SDs were not available, we contacted the corresponding authors for missing data.
Two authors extracted the data individually and discussed them with another author to resolve disagreements.
Data synthesis and statistical analysis
Review Manager Version 5.0 was used for all analyses. The standardized mean difference (SMD) was calculated to measure the effect of the combined intervention from preintervention to postintervention between the combined intervention group and the control group. Pooled SMDs were calculated by averaging the effect size of all cognitive tests measuring the same outcome. The pooled SMDs were regarded as the effect size of each outcome (global cognition, memory, and executive function/attention) [23]. Pooled SMDs were weighted for the sample size of individual studies with 95% confidence interval (CI). These pooled effect sizes were ranked as small (0.2), moderate (0.5) and large (0.8) [24].
The h test was conducted by Statistic I2. The level of heterogeneity was classified as small (25%), moderate (50%), or large (75%) for I2 [23]. If I2 > 50%, the fixed-effects model was replaced by the random-effects model [23].
Given the limited number of included studies, funnel plot asymmetry examination was not performed to test publication bias [25]. When I2 > 50%, sensitivity analysis was conducted [25].
Results
Identification of studies
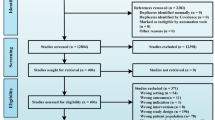
Figure 1 shows the process of study selection in detail. We identified 2569 articles initially. After removing duplicates, 1258 articles remained. Then, 1187 articles were excluded by screening titles and abstracts. Out of the remaining 71 articles screened by full text, 16 studies were included.
Figure 2 shows the results of risk-of-bias assessment. Nine studies [15, 17, 18, 26,27,28,29,30,31] used computer-generated random sequences and concealed assignment. Two studies [32, 33] performed random sequences using computers and did not specify allocation concealment. Other studies [16, 19, 34,35,36] did not introduce random sequence generation or allocation concealment in detail. Some studies had blind designs (single-blind n = 7 [16,17,18, 28, 29, 31, 33]; double-blind n = 3 [15, 26, 27]), while others did not mention it. In addition, seven studies [17, 19, 30, 31, 34,35,36] lost participants in follow-up and did not process the missing data with proper methods. One study [33] did not report the results of the mentioned measurement, which may have affected the results of the analysis. As a potential factor affecting the results, the intervention setting (e.g., supervised or unsupervised), was considered as another bias. Trained assistants or experienced therapists supervised the participants of 13 studies [15,16,17,18,19, 26, 28,29,30,31, 34,35,36] during the intervention. Other studies [27, 32, 33] did not mention the supervision.
Study characteristics
Table 1 summarizes the characteristics of the included studies. Sixteen studies were conducted in America (n = 3) [15, 32, 34], Australia (n = 1) [26], Belgium (n = 1) [16], China (n = 3) [18, 31, 33], France (n = 1) [35], Germany (n = 1) [36], Korea (n = 1) [30], Japan (n = 2) [17, 28], Thailand (n = 1) [29], Pakistan (n = 1) [19] and the Slovak Republic (n = 1) [27]. The total number of samples was 1337, ranging from 11 to 424. All participants were screened or diagnosed with MCI or mild neurocognitive disorders (mNCD) [29], which was a new term that had been used to replace MCI in the Diagnostic and Statistical Manual of Mental Disorders (DSM-5) by the American Psychiatric Association (APA) in 2013 [37]. Diagnostic criteria included the Mayo Criteria [26], the International Working Group (IWG) or the Key Symposium Criteria [15, 17, 28, 33, 35, 36], the National Institute of Aging-Alzheimer’s Association (NIA-AA) Criteria [18], the International Classification of Disease, Ninth Revision, Clinical Modification, MCI (ICD-9-CM 331.83) [27], the DSM-5 [29], and the criteria established by themselves [32, 33]. However, some included studies did not describe the diagnosis criteria and only mentioned that subjects were diagnosed with MCI by neurologists in hospitals [16, 19, 30, 31, 34]. The mean age of the participants was 73.33 years, and the percentage of females was 57.82%. The average level of education for each group was at least 3 years.
Out of sixteen studies, four studies [18, 26, 33, 35] used a four-group design. However, the social group in the study of Lam et al. [33] was excluded due to the mixed social component. One study [31] used three-group design. However, the health advice control group in the study of Zijun et al. [31] was excluded. The health advice control group did not contain the risk factor modification component that was included in the combined intervention. Other studies used a two-group design, including one study [30] comparing a combined intervention group to a single cognitive intervention group, four studies [19, 27, 34, 36] comparing a combined intervention group to a single physical exercise group, and six studies [15,16,17, 28, 29, 32] comparing a combined intervention group to another control group.
The combined interventions were different in regard to the combined components, mode, frequency, duration, and setting. For the cognitive component of the combined intervention, 11 studies specifically described the cognitive domain of intervention, such as memory (n = 7) [16, 19, 26, 27, 29, 30, 36] and executive function/attention (n = 9) [15, 16, 26, 27, 29, 30, 32, 34, 35]. For the physical component of the combined intervention, seven studies [17, 19, 28, 32, 34,35,36] involved aerobic exercise, one study [26] involved resistance training, six studies [15,16,17,18, 27, 28] involved balance training, five studies [17, 18, 27, 28, 30] involved strengthening, and three studies [29, 31, 33] involved mind–body exercise. In addition, four studies [16, 18, 31, 36] included other components (e.g., usual care, omega-3 FA, and nurse-led risk factor modification). The combined modes of cognitive intervention and physical exercise varied, including sequential combination (n = 4) [26, 31, 33, 36], simultaneous combination (six studies [15, 16, 19, 31, 32, 34] for exergame and two studies [18, 35] for dual-task) and mixed combination (n = 4) [17, 27,28,29]. Exergame used different technologies (e.g., Kinect, recumbent stationary bike, wireless remote device, BioRescue platform, inertial sensors, touchscreen monitor, grip air bulb, joysticks, attachments) to control progress of virtual world or attainment of goals. The frequencies varied from one session/week [17, 27, 32] to seven sessions/week [27], with two sessions/week mostly [15, 16, 28, 29, 35, 36]. The durations varied from 4 weeks [15] to 40 weeks [17], with 24 weeks mostly [26, 28, 32, 34]. Eight studies [15, 18, 19, 26,27,28,29, 31] were conducted in a medical setting. Three studies [17, 32, 33] were conducted in a social setting. Four studies [15, 19, 30, 34] were conducted in the form of individuals. Seven studies [18, 26, 28, 29, 32, 33, 35] were conducted in the form of a group. Two studies [31, 36] were conducted in the form of individuals and groups. Thirteen studies [15,16,17,18,19, 26, 28,29,30,31, 34,35,36] were conducted under professional supervision. Other studies do not specify the setting.
For the control group, five studies [26, 27, 33,34,35] used the same single cognitive intervention and single physical exercise as those used for the combined intervention. Four studies [18, 19, 30, 36] used a single cognitive intervention and a single physical exercise that was different from the combined intervention. Another control group (e.g., healthy education, watching videos, no training, usual lifestyle, usual care, and nurse-led risk factor modification) was used in ten studies [15,16,17,18, 26, 28, 29, 31, 32, 35].
Table 2 summarized the outcome measures. For assessing global cognition, the included studies used the Mini-Mental State Examination (MMSE) (n = 5) [17, 19, 27, 28, 33], Montreal Cognitive Assessment (MoCA) (n = 5) [15, 16, 19, 30, 31], Alzheimer’s Disease Assessment Scale-Cognitive subscale (ADAS-Cog) (n = 4) [26, 28, 31, 33], Neurobehavioral Cognitive Status Examination (NCSE) (n = 1) [18], and Computerized Assessment of Mild Cognitive Impairment (CAMCI) (n = 1) [32]. To measure memory, the List Learning subsection of the ADAS-Cog (n = 2) [26, 34], List Learning Delayed Recall test for episodic memory (n = 1) [33], Rey Auditory Verbal Learning Test (n = 3) [17, 27, 36], Consortium to Establish a Registry for Alzheimer’s Disease (CERAD) Word-List Learning Test (n = 1) [29], Verbal Learning Test (n = 1) [18], Wechsler Memory Scale-Revised Logical Memory II (n = 1) [17], Auditory Logical Memory I and II subtests of the Wechsler Memory Scale-Third Edition (n = 2) [26, 28], and visual via Benton Visual Retention Test-Revised Fifth Edition (n = 1) [26] were used. Executive function/attention was measured by the Trial Making Test (n = 9) [15, 17,18,19, 27, 29, 30, 33, 36], Stroop test (n = 4) [27, 34,35,36], Digit span (n = 6) [29, 30, 33,34,35,36], Symbol Digit Modalities Test (n = 1) [26], Verbal Fluency Test (n = 4) [17, 26, 29, 33], Matrix Reasoning test (n = 1) [35], Matrices and Similarities subtests of the Wechsler Adult Intelligence Scale third Edition (n = 1) [26], Color Trials (n = 1) [34], Disjunctive Reaction Time (n = 1) [27], Block Design (n = 1) [29], and Tracking A and B (n = 1) [32].
Effects analyses of combined intervention
We summarized the results of all forest plots (the number of studies, SMD, 95% CI, p, I2 and p for I2) in Table 3. All forest plots can be seen in the supplementary material.
Effects of combined intervention on global cognition
As Table 3 shows, compared to the single cognitive intervention and the single physical exercise, the effects of the combined intervention on global cognition were not significant, with high heterogeneity [SMD = 0.81, 95% CI ( – 0.09, 1.71), p = 0.08; I2 = 90%, p for I2 < 0.001]; [SMD = 0.41, 95% CI ( – 0.06, 0.89), p = 0.09; I2 = 79%, p for I2 < 0.001]. Compared to the other control group, the effect of the combined intervention on global cognition was significant, with low heterogeneity [SMD = 0.27, 95% CI (0.09, 0.44), p = 0.003; I2 = 0%, p for I2 = 0.68].
Effects of combined intervention on memory
As Table 3 shows, compared to the single cognitive intervention, the effect of the combined intervention on memory was not significant, with moderate heterogeneity [SMD = 0.00, 95% CI ( – 0.20, 0.21), p = 0.97; I2 = 44%, p for I2 = 0.17]. Compared to the single physical exercise group and the other control group, the effects of the combined intervention on memory were significant, with low heterogeneity [SMD = 0.25, 95% CI (0.07, 0.44), p = 0.006; I2 = 11%, p for I2 = 0.35]; [SMD = 0.29, 95% CI (0.12, 0.47), p = 0.001; I2 = 0%, p for I2 = 0.55].
Effects of combined intervention on executive function/attention
As Table 3 shows, compared to the single cognitive intervention group, the single physical exercise group, and the other control group, the effects of the combined intervention on executive function/attention were significant, with low heterogeneity [SMD = 0.28, 95% CI (0.09, 0.47), p = 0.004; I2 = 0%, p for I2 = 0.43]; [SMD = 0.32, 95% CI (0.16, 0.49), p = 0.0002; I 2 = 0%, p for I2 = 0.72]; [SMD = 0.23, 95% CI (0.05, 0.41), p = 0.01; I2 = 0%, p for I2 = 0.70].
Sensitivity analysis
Compared to the single cognitive intervention group and the single physical exercise group, the effects of the combined intervention on global cognition exhibited high heterogeneity (I2 = 90%, p for I2 < 0.001; I2 = 79%, p for I2 < 0.001). Thus, sensitivity analysis was conducted to evaluate the effects of each included study on the heterogeneity. As Table 4 shows, the I2 values and p for I2 were obviously changed when the study of Park et al. (2020) was omitted. As Table 5 shows, the I2 values and p for I2 were almost unchanged when each included study was omitted, which indicated the stability of our meta-analysis results.
Discussions
This meta-analysis included 16 RCTs and explored the effect of the combined intervention on cognitive function (global cognition, memory, and executive function/attention) in older adults with MCI by comparing it to the single cognitive intervention group, the single physical exercise group, and the other control group. We found that the combined intervention has small-to-medium effect on global cognition compared to the other control group, and a small-to-medium effect on memory compared to the single physical exercise group and the other control group, and moreover, a small-to-medium effect on executive function/attention compared to the single cognitive intervention group, the single physical exercise group, and the other control group.
Interpretation of results and comparison with previous research
To our knowledge, this is the first meta-analysis examining the effect of the combined intervention on cognition in older adults with MCI by comparing it to three different types of control groups. Zhu et al. [13] investigated the effect of the combined intervention on cognition in healthy older adults in a meta-analysis. Twenty studies were included, of which 14 studies were RCTs. The meta-analysis showed positive effects of the combined intervention on global cognition, memory, executive function, and attention when comparing the combined intervention to the other control group, which was consistent with our findings. Zhu et al. found a small-to-medium effect of the combined intervention on global cognition when comparing the combined intervention to a single physical exercise, which was in contrast with our current data. We thought that Zhu et al. [13] should be more cautious in interpreting their results, as the results are based on only one article. In addition, the methods used to measure cognition varied across studies, which limited their comparability.
Karssemeijer et al. [14] conducted a meta-analysis examining the efficacy of the combined intervention on global cognition, memory, and executive function/attention in older adults with MCI or dementia compared to the other control group. Ten RCTs were included, of which only three studies were conducted in older adults with MCI. The meta-analysis showed a positive small-to-medium effect of the combined intervention on global cognition, but no significant effect of the combined intervention on memory and executive function/attention, which was different from our results that the small-to-medium effect of the combined intervention on memory and executive function/attention compared to the other control group. The results of Karssemeijer et al. [14] were influenced by dementia. Perhaps the combined intervention has difficulty improving the cognition of dementia, which has more seriously impaired cognition than MCI.
A recent systematic review conducted by Yang et al. [20] reviewed the effectiveness of the combined intervention in older adults with MCI based on ten RCTs. Most studies reported a significant improvement in global cognition, memory, executive function, and attention. The preliminary results showed positive effects of the combined intervention on cognition in older adults at risk of developing dementia. This meta-analysis added new evidence to the qualitative review of Yang et al., and we were able to quantify the magnitude of the overall effect, confirming the efficacy of the combined intervention in older adults with MCI.
Studies have shown that cognitive intervention and physical exercise are clinically effective for cognitive improvement [8,9,10,11,12]. Neural plasticity is a probable explanation for positive training effects [38]. Brain plasticity refers to the brain’s capacity to change and adapt, physically and functionally, including the potential of neurons to change their synaptic connections [39]. Animal experiments have revealed that physical exercise can facilitate neuronal proliferation and that cognitive intervention can increase the survival of these proliferating neurons and guide these neurons to integrate into the working brain network by activity-dependent synaptic adaptation to maintain the last positive plastic changes [40,41,42]. Therefore, we assumed that the combined intervention was more effective than the single-component intervention on cognition. However, our results were contrary to our hypothesis. Lam et al. [33] observed the same negative results. Perhaps the combined intervention meant double challenges, which may cause excessive stress and lead to weakened cognitive benefits of single cognitive intervention and single physical exercise in the combined intervention. The interaction between combined components may inhibit rather than promote neural plasticity, making unobserved cognitive enhancement effects of the combined intervention [26].
Interestingly, a significant improvement in memory with the combined intervention was observed when compared to single physical exercise but was not observed when compared to single cognitive intervention. We hypothesized that a single cognitive intervention would improve memory more than a single physical exercise during MCI [38]. The effectiveness of a single physical exercise on memory was too small to offset the negative effect of the combination of the two components, so the combined intervention had a less positive effect on memory than a single cognitive intervention. In contrast, the effectiveness of a single cognitive intervention on memory was large enough to offset the negative effect of the combination of the two components so that the combined intervention had a more positive effect on memory than a single physical exercise. MCI is a critical period during which cognitive restructuring and neuroplasticity such as compensation still occur [38]. Perhaps single cognitive intervention is meaningful for MCI to maintain the survival of existing neurons and increase plasticity, rather than the effect of single physical exercise on increasing new neurons.
Although the effectiveness of the combined intervention on executive function/attention supported our hypothesis, it still did not determine the separate effects of the combined intervention on executive function and attention. Moreover, executive function and attention are very complex cognitive subdomains [43]. Executive function includes planning, decision-making, working memory, responding to feedback/error correction, overriding habits/inhibition, and mental flexibility [44]. Attention includes sustained attention, divided attention, selective attention, and processing speed [44]. Patients with AD often perform poor selective and divided attention, failed inhibition of interfering stimuli, and poor manipulation skills. Therefore, it is important to identify the role of the combined intervention in improving executive function, attention, and their subdomains to decrease the likelihood of AD progression during MCI.
Strengths and limitations
The most significant advantage of the present study was that the included studies were all RCTs. The characteristics of each included study were summarized from five perspectives (study, sample, combined intervention group, control group, outcome). Furthermore, focusing on the cognitive domains (global cognition, memory, and executive function) was more important for MCI. In addition, the cognitive benefits of the combined intervention on cognitive function were further clarified by comparing the combined intervention group to the single-component intervention group.
However, there were still deficiencies in our research, which limited the interpretation of the results. First, MCI diagnostic criteria are important for the definition of the study populations. The Mayo criteria, the IWG criteria, the NIA-AA criteria, ICD-9-CM 331.83, and DSM-5 were conceptually similar [37], but some included studies did not mention MCI diagnostic criteria, which makes it impossible to interpret the findings better. Second, due to the small number of included studies, publication bias analysis was not performed, and subgroup analysis was not conducted on the factors affecting the combined intervention, such as combined mode and the level of exposure to the intervention. Sensitivity analysis showed that Park et al. [30] caused the large heterogeneity when comparing the effect of the combined intervention to the single cognitive intervention group on global cognition. Perhaps the combined mode of cognitive intervention and physical exercise in the study of Park et al. [30] was virtual reality-based exergame, which was different from the other three studies. However, the limited included studies make cautious in interpretating the heterogeneity. Third, we did not explain the efficacy of the combined intervention based on subdomains of memory, executive function, and attention. Fourth, some articles were excluded due to incomplete data and language restrictions.
Implications for future research
In the future, more RCTs with rigorous designs, such as multiarm designs, will be needed to provide high-quality evidence for exploring the effects of combined interventions, since our results only partially supported the hypothesized superiority of the combined interventions. In addition, using standardized tools with high sensitivity and specificity to assess cognition will increase the credibility of the results. Moreover, evaluating the maintaining effect of combined intervention on cognitive function will be meaningful. Finally, some researchers [45, 46] proposed that simultaneous cognitive intervention and physical exercise might be crucial for interaction effects between cognitive and physical components. Thus, comparing different combined interventions may help understand the factors that influence combined interventions to develop optimally combined intervention programs.
Conclusion
The results of this meta-analysis showed that combined interventions effectively improved global cognition, memory, and executive function/attention in older adults with MCI. However, combined interventions demonstrated superiority over single physical exercise on memory and executive function/attention and superiority over single cognitive intervention on executive function/attention. In this meta-analysis, the number of included studies was limited and showed a large methodological heterogeneity in intervention characteristics. Thus, the current results should be interpreted with caution. In the future, there is the need for well-designed RCTs with multiple arms, including combined intervention control groups and standardized tools assessing various cognitive domains, to explore further the immediate and long-term effect of combined intervention on cognitive function.
References
Alzheimer's Disease International. World Alzheimer Report 2018: The state of the art of dementia research: New frontiers [EB/OL]. [2018–09–01]. https://www.alz.co.uk/research/WorldAlzheimerReport2018.pdf
Global action plan on the public health response to dementia 2017–2025 [EB/OL]. [2019–02–10]. https://www.who.int/mental_health/neurology/dementia/action_plan_2017_2025/en/
Hampel H, Lista S (2016) Dementia: The rising global tide of cognitive impairment. Nat Rev Neurol 12:131–132. https://doi.org/10.1038/nrneurol.2015.250
Roberts RO, Knopman DS, Mielke MM et al (2014) Higher risk of progression to dementia in mild cognitive impairment cases who revert to normal. Neurology 82:317–325. https://doi.org/10.1212/WNL.0000000000000055
Petersen RC, Lopez O, Armstrong MJ et al (2018) Author response: Practice guideline update summary: Mild cognitive impairment: Report of the Guideline Development, Dissemination, and Implementation Subcommittee of the American Academy of Neurology. Neurology 91:373–374. https://doi.org/10.1212/WNL.0000000000006042
Simon SS, Yokomizo JE, Bottino CM (2012) Cognitive intervention in amnestic Mild Cognitive Impairment: a systematic review. Neurosci Biobehav Rev 36:1163–1178. https://doi.org/10.1016/j.neubiorev.2012.01.007
Huckans M, Hutson L, Twamley E et al (2013) Efficacy of cognitive rehabilitation therapies for mild cognitive impairment (MCI) in older adults: working toward a theoretical model and evidence-based interventions. Neuropsychol Rev 23:63–80. https://doi.org/10.1007/s11065-013-9230-9
Liang JH, Shen WT, Li JY et al (2019) The optimal treatment for improving cognitive function in elder people with mild cognitive impairment incorporating Bayesian network meta-analysis and systematic review. Ageing Res Rev 51:85–96. https://doi.org/10.1016/j.arr.2019.01.009
Hong YJ, Jang EH, Hwang J et al (2015) The efficacy of cognitive intervention programs for mild cognitive impairment: a systematic review. Curr Alzheimer Res 12:527–542. https://doi.org/10.2174/1567205012666150530201636
Yang HL, Chan PT, Chang PC et al (2018) Memory-focused interventions for people with cognitive disorders: a systematic review and meta-analysis of randomized controlled studies. Int J Nurs Stud 78:44–51. https://doi.org/10.1016/j.ijnurstu.2017.08.005
Öhman H, Savikko N, Strandberg TE et al (2014) Effect of physical exercise on cognitive performance in older adults with mild cognitive impairment or dementia: a systematic review. Dement Geriatr Cogn Disord 38:347–365. https://doi.org/10.1159/000365388
Song D, Yu D, Li P et al (2018) The effectiveness of physical exercise on cognitive and psychological outcomes in individuals with mild cognitive impairment: a systematic review and meta-analysis. Int J Nurs Stud 79:155–164. https://doi.org/10.1016/j.ijnurstu.2018.01.002
Zhu X, Yin S, Lang M et al (2016) The more the better? A meta-analysis on effects of combined cognitive and physical intervention on cognition in healthy older adults. Ageing Res Rev 31:67–79. https://doi.org/10.1016/j.arr.2016.07.003
Karssemeijer E, Aaronson JA, Bossers WJ et al (2017) Positive effects of combined cognitive and physical exercise training on cognitive function in older adults with mild cognitive impairment or dementia: A meta-analysis. Ageing Res Rev 40:75–83. https://doi.org/10.1016/j.arr.2017.09.003
Schwenk M, Sabbagh M, Lin I et al (2016) Sensor-based balance training with motion feedback in people with mild cognitive impairment. J Rehabil Res Dev 53:945–958. https://doi.org/10.1682/JRRD.2015.05.0089
Delbroek T, Vermeylen W, Spildooren J (2017) The effect of cognitive-motor dual task training with the biorescue force platform on cognition, balance and dual task performance in institutionalized older adults: a randomized controlled trial. J Phys Ther Sci 29:1137–1143. https://doi.org/10.1589/jpts.29.1137
Shimada H, Makizako H, Doi T et al (2018) Effects of combined physical and cognitive exercises on cognition and mobility in patients with mild cognitive impairment: a randomized clinical trial. J Am Med Dir Assoc 19:584–591. https://doi.org/10.1016/j.jamda.2017.09.019
Law L, Mok V, Yau M (2019) Effects of functional tasks exercise on cognitive functions of older adults with mild cognitive impairment: a randomized controlled pilot trial. Alzheimers Res Ther 11:98. https://doi.org/10.1186/s13195-019-0548-2
Amjad I, Toor H, Niazi IK et al (2019) Xbox 360 kinect cognitive games improve slowness, complexity of EEG, and cognitive functions in subjects with mild cognitive impairment: a randomized control trial. Games Health J 8:144–152. https://doi.org/10.1089/g4h.2018.0029
Yang C, Moore A, Mpofu E et al (2019) Effectiveness of combined cognitive and physical interventions to enhance functioning in older adults with mild cognitive impairment: a systematic review of randomized controlled trials. Gerontologist. https://doi.org/10.1093/geront/gnz149
Moher D, Liberati A, Tetzlaff J et al (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med 151:264–269. https://doi.org/10.7326/0003-4819-151-4-200908180-00135
Higgins JP, Altman DG, Gøtzsche PC et al (2011) Cochrane bias methods group, & cochrane statistical methods group. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 343:d5928. https://doi.org/10.1136/bmj.d5928
Borrenstein M, Hedges LV, Higgins JP et al (2009) Introduction to Meta-Analysis. John Wiley & Sons, United Kingdom . https://doi.org/10.1002/9780470743386
Cohen J (1992) A power primer. Psychol Bull 112:155–159. https://doi.org/10.1037/0033-2909.112.1.155
Higgins JP, Green S (Eds.) (2011) Cochrane Handbook for Systematic Reviews of Interventions. John Wiley & Sons 4 http://refhub.elsevier.com/S0020-7489(18)30002-6/sbref0110
Fiatarone Singh MA, Gates N, Saigal N et al (2014) The study of mental and resistance training (SMART) study-resistance training and/or cognitive training in mild cognitive impairment: a randomized, double-blind, double-sham controlled trial. J Am Med Dir Assoc 15:873–880. https://doi.org/10.1016/j.jamda.2014.09.010
Hagovska M, Nagyova I (2016) The transfer of skills from cognitive and physical training to activities of daily living: a randomised controlled study. Eur J Ageing 14:133–142. https://doi.org/10.1007/s10433-016-0395-y
Suzuki T, Shimada H, Makizako H et al (2013) A randomized controlled trial of multicomponent exercise in older adults with mild cognitive impairment. PLoS ONE 8:e61483. https://doi.org/10.1371/journal.pone.0061483
Griffiths J, Thaikruea L, Wongpakaran N et al (2020) Effects of combined physical movement activity and multifaceted cognitive training in older people with mild neurocognitive disorder in a rural community: a randomized control trial. Dement Geriatr Cogn Disord 49:194–201. https://doi.org/10.1159/000507922
Park JS, Jung YJ, Lee G (2020) Virtual reality-based cognitive-motor rehabilitation in older adults with mild cognitive impairment: a randomized controlled study on motivation and cognitive function. Healthcare (Basel) 8:335. https://doi.org/10.3390/healthcare8030335
Xu Z, Zhang D, Lee ATC et al (2020) A pilot feasibility randomized controlled trial on combining mind-body physical exercise, cognitive training, and nurse-led risk factor modification to reduce cognitive decline among older adults with mild cognitive impairment in primary care. PeerJ 8:9845. https://doi.org/10.7717/peerj.9845
Hughes TF, Flatt JD, Fu B et al (2014) Interactive video gaming compared to health education in older adults with MCI: a feasibility study. Int J Geriatr Psych 29:890–898. https://doi.org/10.1002/gps.4075
Lam LC, Chan WC, Leung T et al (2015) Would older adults with mild cognitive impairment adhere to and benefit from a structured lifestyle activity intervention to enhance cognition?: a cluster randomized controlled trial. PLoS ONE 10:e0118173. https://doi.org/10.1371/journal.pone.0118173
Anderson-Hanley C, Barcelos NM, Zimmerman EA (2018) The Aerobic and cognitive exercise study (ACES) for community-dwelling older adults with or at-risk for mild cognitive impairment (MCI): neuropsychological, neurobiological and neuroimaging outcomes of a randomized clinical trial. Front Aging Neurosci 10:76. https://doi.org/10.3389/fnagi.2018.00076
Laure CD, Alexandra P, Sylvie B et al (2018) Effects of simultaneous aerobic and cognitive training on executive functions, cardiovascular fitness and functional abilities in older adults with mild cognitive impairment. Ment Health Phys Act. https://doi.org/10.1016/j.mhpa.2018.06.001
Köbe T, Witte AV, Schnelle A et al (2016) Combined omega-3 fatty acids, aerobic exercise and cognitive stimulation prevents decline in gray matter volume of the frontal, parietal and cingulate cortex in patients with mild cognitive impairment. Neuroimage 131:226–238. https://doi.org/10.1016/j.neuroimage.2015.09.050
Sachdev PS, Blacker D, Blazer DG et al (2014) Classifying neurocognitive disorders: the DSM-5 approach. Nat Rev Neurol 10:634–642. https://doi.org/10.1038/nrneurol.2014.181
Bherer L (2015) Cognitive plasticity in older adults: effects of cognitive training and physical exercise. Ann N Y Acad Sci 1337:1–6. https://doi.org/10.1111/nyas.12682
Foster PP, Rosenblatt KP, Kuljiš RO (2011) Exercise-induced cognitive plasticity, implications for mild cognitive impairment and Alzheimer’s disease. Front Neurol 2:28. https://doi.org/10.3389/fneur.2011.00028
Fabel K, Kempermann G (2008) Physical activity and the regulation of neurogenesis in the adult and aging brain. Neuromolecular Med 10:59–66. https://doi.org/10.1007/s12017-008-8031-4
Fabel K, Wolf SA, Ehninger D et al (2009) Additive effects of physical exercise and environmental enrichment on adult hippocampal neurogenesis in mice. Front Neurosci 3:50. https://doi.org/10.3389/neuro.22.002.2009
Kempermann G, Fabel K, Ehninger D et al (2010) Why and how physical activity promotes experience-induced brain plasticity. Front Neurosci 4:189. https://doi.org/10.3389/fnins.2010.00189
Wang Z, Dong B (2018) Screening for cognitive impairment in geriatrics. Clin Geriatr Med 34:515–536. https://doi.org/10.1016/j.cger.2018.06.004
American Psychiatric Association (1994) Diagnostic and statistical manual of mental disorders (DSM-IV). Washington, DC: American Psychiatric Association http://refhub.elsevier.com/S0749-0690(18)30997-2/sref2
Law LL, Barnett F, Yau MK et al (2014) Effects of combined cognitive and exercise interventions on cognition in older adults with and without cognitive impairment: a systematic review. Ageing Res Rev 15:61–75. https://doi.org/10.1016/j.arr.2014.02.008
McEwen SC, Siddarth P, Rahi B et al (2018) Simultaneous aerobic exercise and memory training program in older adults with subjective memory impairments. J Alzheimer’s Dis 62:795–806. https://doi.org/10.3233/JAD-170846
Acknowledgements
This work was supported by the Health and Family Planning Committee of Jilin Province, China [grant numbers 2020J036] and the Education Department of Jilin Province, China [grant numbers 2015J505]. I would like to thank the organizations for funding my research.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
None.
Statement of human and animals rights
This review reports no participant data or original research findings that require ethics approval.
Informed consent
For this type of study, formal consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Meng, Q., Yin, H., Wang, S. et al. The effect of combined cognitive intervention and physical exercise on cognitive function in older adults with mild cognitive impairment: a meta-analysis of randomized controlled trials. Aging Clin Exp Res 34, 261–276 (2022). https://doi.org/10.1007/s40520-021-01877-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40520-021-01877-0