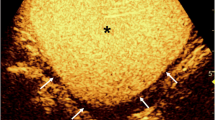
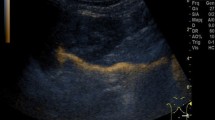
Abstract
Purpose
Although contrast-enhanced urosonography (ceVUS) has shown capable diagnostic accuracy for the diagnosis of vesicoureteral reflux (VUR) in children, the ability of ceVUS to detect intrarenal reflux (IRR) is considered limited. The purpose of our study is to assess the ability of ceVUS to detect IRR as well as its association with age, gender, and the grade of VUR.
Methods
This study included 5153 children who were referred to our clinic for ceVUS. All children underwent sonographic examinations, which were performed on a LOGIQ S8 machine equipped with dedicated software for contrast-enhanced studies with harmonic imaging. Standard ultrasound of the urinary tract was followed by bladder catheterisation and instillation of physiological normal saline and the US contrast medium (SonoVue®, Bracco).
Results
VUR was diagnosed by ceVUS in 1959 out of 5153 children (38%), of whom IRR was found in 233 of 1959 children (11.9%). A total of 285 ureteral units showing IRR were found. High grades of VUR (IV + V) with IRR were found in a total of 235 of 285 (82.81%) renal units. Bilateral IRR was found in 53 patients, usually with a high-grade VUR on both sides. Most children had VUR grade IV, predominantly those < 12 months. The younger the child, the higher the likelihood of higher-grade VUR (p = 0.02).
Conclusion
ceVUS, combined with harmonic imaging and second-generation ultrasound contrast media, enabled IRR detection in almost 12% of our patients with VUR. IRR is most commonly found in children under 1 year of age with VUR grades IV and V.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Vesicoureteral reflux (VUR) is one of the most common urinary tract abnormalities in children and is likely associated with the development of reflux nephropathy (RN) [1,2,3,4]. The presence of intrarenal reflux (IRR) acts as a risk factor for RN, because it allows urine to directly enter the renal tubular system. Although IRR findings in VUR images are not exceedingly rare, contemporary IRR reports in children are scarce [5,6,7,8]. Early studies showed an IRR incidence of < 1–10%, diagnosed by fluoroscopic voiding cystourethrography (VCUG), and this considerable variability may be due to the fleeting phenomenon of IRR and variable testing techniques [9, 10]. Contrast-enhanced voiding urosonography (ceVUS), as a radiation-free software technology, has become a widespread method in diagnosing VUR in children because of the development of second-generation ultrasound (US) contrast media with harmonic imaging [11,12,13,14]. Although a recent number of studies have indicated adequate diagnostic accuracy compared with VCUG, its presumed limited ability to detect IRR in children has been suggested as a potential drawback of ceVUS [15]. This study aims to evaluate the ability of ceVUS to detect IRR and its association with age, gender, and the grade of VUR.
Materials and methods
A total of 5153 children over 7 years (January 2013 to February 2020) referred to our clinic for VUR imaging were enrolled in this study. Indications for ceVUS were urinary tract infections, dilatation of the pelvicalyceal system and/or ureter, neurogenic bladder, and follow-up after surgical or conservative management of VUR. Both parents/guardians and older children were informed in detail of the entire procedure before the examination. They were also provided with a written leaflet describing the whole procedure. Before ceVUS, Combur9 tests (Boehringer Mannheim, Germany) and urine culture were used as urinary tract infection–free tests.
Sonographic studies were performed on a LOGIQ S8 machine (GE Healthcare, Milwaukee, WI) equipped with dedicated software for contrast-enhanced studies with a harmonic imaging modality and low or intermediate mechanical index (MI, 0.04–0.10). The focal point was located immediately posterior to the kidneys, ureters, and bladder. A high-frequency linear transducer (7.5–10 MHz) was used for infants and small children and a low-frequency convex transducer (3.5–5 MHz) for older children. All scans were first carried out on a greyscale with the tissue-specific harmonic imaging modality. Standard US of the urinary tract in supine and prone positions was performed after bladder catheterisation and instillation of normal physiological saline and a freshly prepared suspension of the US contrast medium (SonoVue®, Bracco). The vial of the US contrast agent was prepared strictly following the manufacturer’s instructions. ceVUS was performed after instillation of 0.2–0.5% SonoVue® contrast into a 250–500 mL bag of saline, which was then infused into the bladder by gravity. The catheterisation is performed by a skilled medical practitioner. The whole examination is a teamwork of a nephrologist, a radiologist, and a skilled nurse. The bladder was progressively and homogeneously filled to the expected capacity. Bladder capacity (in millilitres) was calculated using the following formula: volume = (age + 2) × 30, where age is in years or (weight in kilograms × 7) mL for children less than 12 months of age. Continuous, alternate examination of the kidneys and urinary bladder was performed during bladder filling and voiding. Antibiotic prophylaxis was used. No sedation was required. The diagnosis of VUR was determined by the appearance of moving echogenic microbubbles from the ultrasound contrast in the upper urinary tract. The five-grade system developed by Darge and Troeger was used [16]. IRR was demonstrated if the intraparenchymal spread of contrast was detected. The appearance of contrast microbubbles outside the pelvicalyceal system’s contours or the entry of contrast into the kidney parenchyma is a diagnostic sign of IRR.
Data analysis
Data were analysed using the MedCalc Statistical Software version 15.2 (MedCalc Software bvba, Ostend, Belgium; http://www.medcalc.org; 2015). Counted data are presented as numbers and percentages. Measured data are presented as means, standard deviations (SD), and medians.
The nonparametric nature of the data was assessed using the Kolmogorov–Smirnov test. Kruskal–Wallis one-way analysis of variance was used to investigate the differences between the two groups. The nonparametric Kruskal–Wallis test was selected due to the considerable dispersion of data. Linear regression analysis was performed to assess the relationship between the nonparametric values, and p values of ≤ 0.05 were considered statistically significant.
Results
A total of 5153 children in whom ceVUS was performed were included in this study. VUR was detected in 1959 (38%) children, and in these children, IRR was found in 232 (11.9%) (Fig. 1).
Of the children with VUR and IRR, 124 were male, and 108 were female. The male-to-female ratio was 1.2:1. No male-to-female ratio predominance among children with VUR and IRR was observed (1.15 vs. 1, Chi-squared test for left IRR and right IRR p > 0.05). The median age was 11 months (2–82 months). The children were divided into three groups according to age (< 12 months, 12–24, > 24 months), in which a total of 285 ureteral units showing IRR were found. VUR grade II was found in 5 (1.75%), grade III in 44 (15.44%), grade IV in 171 (60%), and grade V in 65 (22.81%) children. VUR grade IV was the most common in children in the first year of life (113/285; 39.65%).
Unilateral left and right VURs with IRR were analysed as individual groups (left and right) and as VURs joined together regardless of their side. In all groups (individual or joined), the largest proportion of children had VUR grade IV. The majority of VUR with IRR were found in children < 12 months of age, with VUR grades IV and V (170/285; 59.65%). We also analysed VUR grades with IRR by renal units. The same high grades of VUR (IV + V) with IRR were found in a total of 235 out of 285 (82.81%) renal units (Table 1).
Bilateral VUR with IRR was found in 53 children (106 ureteral units). Children with such conditions had mostly bilateral high-grade VUR (III + IV, IV + IV, and IV + V) (Table 2).
Multiple comparison analysis showed significant differences in children with VUR and IRR between grades II and V. These differences were age-related (the youngest children are prone to a high-grade VUR) (p = 0.02). Grade IV VUR showed a substantial number of outliers and extremes, which indicate the considerable dispersion of this large group. This dispersion of data justifies using Kruskal–Wallis one-way analysis of variance (Fig. 2).
Regression analysis showed that the younger the child, the more likely they have high-grade VUR with IRR. Vice versa, older children are prone to have lower-grade VUR with IRR (Fig. 3).
Discussion
Most reports on VUR with IRR were published almost half a century ago [2, 3, 10, 17]. The lack of reports over recent decades is probably due to the axiom that IRR does not play a role in the decision-making regarding the management of VUR, which is primarily based on reflux grade [18]. However, IRR has been identified as important in understanding the pathophysiology of reflux nephropathy [17, 19, 20]. Concave papillae, which are typically most frequent in the polar kidney area, are prerequisites for developing IRR and renal scars. Children with VUR, accompanied by IRR, are generally considered to be at the more severe end of the clinical spectrum [7, 21]. Baseline Tc-99 m dimercaptosuccinic acid (DMSA) scans were found to be worse in an IRR group than in a non-IRR group of children. However, a similar incidence of urinary tract infections and reflux resolution were observed in both groups with medical treatment [18]. A retrospective study with 50 patients showed that cortical defects on Tc-99 m DMSA correlated well with IRR sites, and these sites tended to progress to renal scarring [21]. Other studies confirmed that renal scarring distribution is almost identical to that of renal impairment in children with IRR [1, 7, 20, 22]. Some authors suggest that ureteral reimplantation interventions should be considered sooner in children with IRR than in those without [21, 23]. As IRR is not a stable entity but rather a fleeting phenomenon, fluoroscopic VCUG depends on timely performed imaging to detect IRR [24]. Therefore, IRR might be missed by VCUG even in high-grade VUR because of the variable techniques used in VCUG, possible inadequate bladder filling, the dilution of a small amount of radiographic contrast in the already-dilated collecting system, obscuration by overlying bowel shadow, low image quality, and too narrow collimation of the X-ray field, focusing only on the bladder and urethra during micturition. These technical variabilities may explain the wide range of IRR incidence and may contribute to the lower sensitivity of VUR detection by VCUG. Although an incidence of < 2% has been found in most studies, a recent study showed that using a standardised four-phase VCUG with pulsed fluoroscopy and spot films with optimal imaging during voiding allows detection of up to 11% IRR in patients with VUR [5].
Developing tissue in children is more sensitive to the effects of radiation. As life expectancy becomes considerably longer, the potential oncogenic impact of radiation can accumulate over time [25,26,27,28]. Recent advances in tissue harmonic and contrast-specific imaging techniques, together with the development of second-generation contrast agents, have improved the potential of ultrasonography in both the diagnosis and grading of VUR in children. This advancement in ultrasound technology has improved the detection of contrast bubbles in renal tissue, resulting in better detection of IRR [6].
More than 70% of IRR is detected during the voiding phase. Since IRR is a fleeting phenomenon, ceVUS increases the chances of catching IRR as it is often seen at a later stage of the voiding phase of micturition when the child is asked to urinate. Proper bladder filling avoids overlooking IRR, but overfilling the bladder is also not recommended [29, 30]. For accurate IRR detection, voiding at maximal intrapelvic pressure is crucial. As some children are prone to void during the examination before their bladder is full, repetition of contrast filling of the bladder in such cases may be unavoidable [31]. Repeated filling of the bladder may also be necessary if there is a negative result during the initial examination, mainly if a marked discrepancy is found in kidney size and intermittent dilatation of the ureters on US [14]. Differences in VUR detection and its grade have been reported on repeated VCUG and radionuclide cystography [31, 32]. Such occurrences can be avoided by cyclic ceVUS filling, which increases the VUR detection rate [33,34,35]. An additional advantage of ceVUS as a repeatable method of real-time imaging is that it allows extended continuous scanning, which increases the possibility of detecting IRR as a fleeting phenomenon.
As IRR depends on the papillary duct’s opening, which is rarely seen in older children, IRR is rarely found in children > 5 years. Our results confirm this opinion, because IRR is more commonly found in younger children (≤ 1 year) and high-grade VUR (IV and V). As was previously mentioned, if bilateral VUR with IRR is found, higher grades of VUR are detected on both sides.
Imaging studies have shown that ceVUS has the advantage of detecting VUR when compared with previous ultrasound urosonography methods [12,13,14, 29, 30]. Harmonic imaging techniques, which use nonlinear tissue and contrast agent properties, can partially reduce a certain number of artifacts observed by previous imaging methods. Contrast-specific harmonic imaging mode, together with the subtraction technique, further increases the conspicuity of the microbubble. Setting a mechanical index (MI) below 0.10 is essential to break the microbubbles of the second-generation USCA. With an increase in MI, the rate of microbubbles destruction increases. Contrast-enhanced harmonic ultrasonography with low MI caused by fair detail resolution, image quality, margin sharpness, and penetration can facilitate a more precise IRR diagnosis. Improvements in sonographic techniques have reduced the disadvantages of the previous method of ceVUS detection.
We are fully aware of the difficulty of performing ceVUS. A trained and skilled operator must move the probe quickly and accurately and record IRR from one kidney to another. Therefore, IRR detection depends not only on the equipment but also on the operator’s training, experience, and education. Judging from our experience, IRR lasts sufficiently long to be detected in realtime on both kidneys. The main advantage of ceVUS lies in the sufficient detection time of VUR (20–30 min). During that time, the fleeting IRR phenomenon is more likely to be detected or, if missed, even repeated on the same spot.
In our country, the Health Insurance Fund covers full reimbursement of ceVUS radiological examination. We believe that the refund of ceVUS is essential for more widespread use of ceVUS.
Conclusion
ceVUS, combined with harmonic imaging and second-generation ultrasound contrast media, enabled IRR detection in almost 12% of children with VUR. The majority of children with IRR were younger than 12 months and had VUR grades IV and V. The younger the child, the more likely they have higher-grade VUR with IRR. The continuous scanning in which ceVUS detects IRR fits well to IRR’s intermittent nature as a fleeting phenomenon.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Hannerz L, Wikstad I, Johansson L, Broberger O, Aperia A (1987) Distribution of renal scars and intrarenal reflux in children with a past history of urinary tract infection. Acta radiologica (Stockholm, Sweden: 1987) 28(4):443–446
Hodson CJ, Edwards D (1960) Chronic pyelonephritis and vesico-ureteric reflex. Clin Radiol 11:219–231. https://doi.org/10.1016/s0009-9260(60)80047-5
Hodson CJ (1981) Neuhauser lecture. Reflux nephropathy: a personal historical review. AJR Am J Roentgenol 137(3):451–462. https://doi.org/10.2214/ajr.137.3.451
Bailey RR (1973) The relationship of vesico-ureteric reflux to urinary tract infection and chronic pyelonephritis-reflux nephropathy. Clin Nephrol 1(3):132–141
Schneider KO, Lindemeyer K, Kammer B (2019) Intrarenal reflux, an overlooked entity—retrospective analysis of 1166 voiding cysturethrographies in children. Pediatr Radiol 49(5):617–625. https://doi.org/10.1007/s00247-018-04330-z
Colleran GC, Barnewolt CE, Chow JS, Paltiel HJ (2016) Intrarenal reflux: diagnosis at contrast-enhanced voiding urosonography. J Ultrasound Med Off J Am Inst Ultrasound Med 35(8):1811–1819. https://doi.org/10.7863/ultra.15.09056
Fukui S, Watanabe M, Yoshino K (2013) Intrarenal reflux in primary vesicoureteral reflux. Int J Urol Off J Jpn Urol Assoc 20(6):631–636. https://doi.org/10.1111/iju.12015
Darge K, Trusen A, Gordjani N, Riedmiller H (2003) Intrarenal reflux: diagnosis with contrast-enhanced harmonic US. Pediatr Radiol 33(10):729–731. https://doi.org/10.1007/s00247-003-1050-2
Gotoh T, Asano Y, Nonomura K, Togashi M, Koyanagi T (1991) Intrarenal reflux in children with vesicoureteral reflux. Nihon Hinyokika Gakkai Zasshi 82(9):1480–1486
Cremin BJ (1979) Observations on vesico-ureteric reflux and intrarenal reflux: a review and survey of material. Clin Radiol 30(6):607–621. https://doi.org/10.1016/s0009-9260(79)80003-3
Ključevšek D, Battelino N, Tomažič M, Kersnik Levart T (2012) A comparison of echo-enhanced voiding urosonography with X-ray voiding cystourethrography in the first year of life. Acta paediatrica (Oslo, Norway: 1992) 101(5):e235–e239. https://doi.org/10.1111/j.1651-2227.2011.02588.x
Darge K (2002) Diagnosis of vesicoureteral reflux with ultrasonography. Pediatr Nephrol (Berlin, Germany) 17(1):52–60. https://doi.org/10.1007/s004670200010
Papadopoulou F, Anthopoulou A, Siomou E, Efremidis S, Tsamboulas C, Darge K (2009) Harmonic voiding urosonography with a second-generation contrast agent for the diagnosis of vesicoureteral reflux. Pediatr Radiol 39(3):239–244. https://doi.org/10.1007/s00247-008-1080-x
Kis E, Nyitrai A, Várkonyi I, Máttyus I, Cseprekál O, Reusz G, Szabó A (2010) Voiding urosonography with second-generation contrast agent versus voiding cystourethrography. Pediatr Nephrol (Berlin, Germany) 25(11):2289–2293. https://doi.org/10.1007/s00467-010-1618-7
von Rohden L, Bosse U, Wiemann D (1995) Reflux sonography in children with ultrasound contrast media in comparison to voiding cystourethrography (in German). Paediatr Prax 49:49–58
Darge K, Troeger J (2002) Vesicoureteral reflux grading in contrast-enhanced voiding urosonography. Eur J Radiol 43(2):122–128. https://doi.org/10.1016/s0720-048x(02)00114-6
Ransley PG (1976) Opacification of the renal parenchyma in obstruction and reflux. Pediatr Radiol 4(4):226–232. https://doi.org/10.1007/BF02461530
Boubnova J, Sergent-Alaoui A, Deschênes G, Audry G (2011) Evolution and prognosis value of intrarenal reflux. J Pediatr Urol 7(6):638–643. https://doi.org/10.1016/j.jpurol.2010.09.015
Hellström M, Jacobsson B, Mårild S, Jodal U (1989) Voiding cystourethrography as a predictor of reflux nephropathy in children with urinary-tract infection. AJR Am J Roentgenol 152(4):801–804. https://doi.org/10.2214/ajr.152.4.801
Rolleston GL, Maling TM, Hodson CJ (1974) Intrarenal reflux andthe scarred kidney. Arch Dis Child 49:531–539
Kim SW, Im YJ, Hong CH, Han SW (2010) The clinical significance of intrarenal reflux in voiding cystourethrography (VCUG). Korean J Urol 51(1):60–63. https://doi.org/10.4111/kju.2010.51.1.60
Rose JS, Glassberg KI, Waterhouse K (1975) Intrarenal reflux and its relationship to renal scarring. J Urol 113(3):400–403. https://doi.org/10.1016/s0022-5347(17)59492-6
Kanumakala S, Kalidasan V, Kenney I (2004) Intra-renal reflux. Arch Dis Child 89(7):692. https://doi.org/10.1136/adc.2003.041517
Lebowitz RL (1986) The detection of vesicoureteral reflux in the child. Invest Radiol 21(7):519–531. https://doi.org/10.1097/00004424-198607000-00003
Perisinakis K, Raissaki M, Damilakis J, Stratakis J, Neratzoulakis J, Gourtsoyiannis N (2006) Fluoroscopy-controlled voiding cystourethrography in infants and children: are the radiation risks trivial? Eur Radiol 16(4):846–851. https://doi.org/10.1007/s00330-005-0072-6
Ward VL (2006) Patient dose reduction during voiding cystourethrography. Pediatr Radiol 36(Suppl 2):168–172. https://doi.org/10.1007/s00247-006-0213-3
Ward VL, Barnewolt CE, Strauss KJ, Lebowitz RL, Venkatakrishnan V, Stehr M, McLellan DL, Peters CA, Zurakowski D, Dunning PS, Taylor GA (2006) Radiation exposure reduction during voiding cystourethrography in a pediatric porcine model of vesicoureteral reflux. Radiology 238(1):96–106. https://doi.org/10.1148/radiol.2381041433
Schneider K, Krüger-Stollfuss I, Ernst G, Kohn MM (2001) Paediatric fluoroscopy–a survey of children’s hospitals in Europe. I. Staffing, frequency of fluoroscopic procedures and investigation technique. Pediatr Radiol 31(4):238–246. https://doi.org/10.1007/s002470100429
Darge K (2008) Voiding urosonography with ultrasound contrast agents for the diagnosis of vesicoureteric reflux in children. I. Procedure. Pediatr Radiol 38(1):40–53. https://doi.org/10.1007/s00247-007-0529-7
Darge K (2008) Voiding urosonography with US contrast agents for the diagnosis of vesicoureteric reflux in children. II. Comparison with radiological examinations. Pediatr Radiol 38(1):54–127. https://doi.org/10.1007/s00247-007-0528-8
Jequier S, Jequier JC (1989) Reliability of voiding cystourethrography to detect reflux. AJR Am J Roentgenol 153(4):807–810. https://doi.org/10.2214/ajr.153.4.807
Joaquim AI, de Godoy MF, Burdmann EA (2015) Cyclic direct radionuclide cystography in the diagnosis and characterization of vesicoureteral reflux in children and adults. Clin Nucl Med 40(8):627–631. https://doi.org/10.1097/RLU.000000000000079
Papadopoulou F, Tsampoulas C, Siomou E, Tzovara J, Siamopoulou A, Efremidis SC (2006) Cyclic contrast-enhanced harmonic voiding urosonography for the evaluation of reflux. Can we keep the cost of the examination low? Eur Radiol 16(11):2521–2526
Kenda RB, Kenig A, Novljan G, Ponikvar R, Ponikvar JB (2001) Cyclic voiding urosonography for detecting vesicoureteric reflux in renal transplant recipients. Nephrol Dial Transpl 16(11):2229–2231
Novljan G, Kenig A, Rus R, Kenda RB (2003) Cyclic voiding urosonography in detecting vesicoureteral reflux in children. Pediatr Nephrol (Berlin, Germany) 18(10):992–995. https://doi.org/10.1007/s00467-003-1228-8
Funding
The authors declare that they did not receive any funding for this manuscript.
Author information
Authors and Affiliations
Contributions
AC-R was responsible for data, conceptualized the study, carried out analyses, drafted the initial manuscript, revised, and approved the final manuscript. DT carried out the statiscial analysis and interpretation of data, revised, and approved the final manuscript. DM conceptualized the study, revised and approved the final manuscript. IP collected and revised the data. GR conceptualized and designed the study, carried out the analyses, drafted the initial manuscript and approved the final manuscript as submitted.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This retrospective study has been approved by the Ethics committee of the Helena Clinic for Pediatric Medicine which waved the requirement for informed consent.
Consent to participate
Both parents/guardians and older children were informed in detail of the entire procedure before the examination. They were also provided with a written leaflet describing the whole procedure.
Consent for publication
The authors agree to publish this manuscript in the Journal of Ultrasound.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cvitkovic-Roic, A., Turudic, D., Milosevic, D. et al. Contrast-enhanced voiding urosonography in the diagnosis of intrarenal reflux. J Ultrasound 25, 89–95 (2022). https://doi.org/10.1007/s40477-021-00568-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40477-021-00568-w