Abstract
The aim of this work is to investigate the global dynamical behaviors of two human immunodeficiency virus infection models with cure of infected cells in eclipse stage and Cytotoxic T Lymphocytes (CTL) immune response. The first model is formalized by ordinary differential equations and the second is described by partial differential equations. By constructing appropriate Lyapunov functionals, the global stability of both models is established and characterized by two threshold parameters that are the basic reproduction number \(R_{0}\) and the CTL immune response reproduction number \(R_{1}\). Furthermore, the models and results presented in many previous studies are extended and improved.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
During human immunodeficiency virus (HIV) infection, a part of infected cells in eclipse stage returns to the uninfected state. In addition, Cytotoxic T Lymphocytes (CTL) cells play an important role in antiviral defense by killing the productive infected cells. Therefore, many mathematical models has been proposed to model the role of CTL immune response in HIV infection. One of the earliest of these models was proposed by Nowak and Bangham [1] in order to describe the interaction between susceptible cells, infected cells, viruses and CTL cells. Further, the model of [1] was improved by many researchers (see for example, [2–5]). All the above models have not considered the cure of infected cells in eclipse stage. For this reason, Rong et al. [6] constructed an HIV infection model with bilinear incidence rate and cure of infected cells in eclipse stage, but they not considered the CTL immune response and they only established the local stability of equilibria. The global stability was investigated by Buonomo and Vargas-De-L?on [7]. To improve the model [6], Hu et al. [8] replaced the bilinear incidence rate by a saturated incidence rate and they investigated the global stability of the improved model. In 2015, Wang et al. [9] replaced the saturated incidence rate by the Beddington–Deangelis functional response and they established the local and global stability of equilibria. In addition, Maziane et al. [10] replaced the saturated incidence rate by the Hattaf’s [11] incidence rate and they extended the ODE model to a system with partial differential equations in order to take into account the mobility of the virus. The global stability of both models is investigated by constructing suitable Lyapunov functionals. In [12], Lv et al. replaced the bilinear incidence rate by the Beddington–DeAngelis functional response and they included the CTL immune response into the model [6]. By constructing suitable Lyapunov functionals, they investigated the global stability of equilibria in the terms of the basic reproduction number and the immune response reproduction number.
To extend the model of Lv et al. [12] and improve the ODE models presented in [6–10] by considering the effect of CTL immune response, we propose the following model
where T(t), E(t), I(t), V(t) and C(t) denote the densities of uninfected CD\(4^{+}\) T cells, infected cells in the eclipse stage (unproductive infected cells), productive infected cells, free virus particles and CTL cells at time t, respectively. The parameter \(\lambda \) is the production rate of the uninfected cells, \(\mu _{T}\) is their death rate and f(T, V) is the rate of uninfected cells to become infected by virus. The parameters \(\mu _{E}\), \(\mu _{I}\) and \(\mu _{V}\) denote the death rates of unproductive infected cells, productive infected cells and virus, respectively. The constant \(\rho \) is the rate at which the unproductive infected cells return to the uninfected cells. The constant \(\gamma \) is the rate at which infected cells in the eclipse stage become productive infected cells and k is the production rate of virions by infected cells. The productive infected cells are killed by CTL cells at rate p while a denotes the proliferation rate of CTL cells. Here, we consider the Hattaf’s [11] incidence rate of the form \(f(T,V)=\frac{\beta T}{1+\alpha _{1}T+\alpha _{2}V+\alpha _{3}TV}\), where \(\alpha _1\), \(\alpha _2\), \(\alpha _3\ge 0\) are the saturation factors measuring the psychological or inhibitory effect and \(\beta >0\) is the infection coefficient. We recall that this incidence rate includes the common types such as the bilinear incidence (or mass action incidence) when \(\alpha _1=\alpha _2=\alpha _3=0\), the saturated incidence rate when \(\alpha _1=\alpha _3=0\) or \(\alpha _2=\alpha _3=0\), the Beddington-DeAngelis functional response introduced in [13, 14] when \(\alpha _3=0\), and Crowley-Martin functional response presented in [15] and used in [16, 17] when \(\alpha _3=\alpha _1\alpha _2\). It is important to note that the model of Lv et al. [12] is a special case of system (1) when \(\alpha _3=0\). The first aim of this study is the generalization of the main results presented in [12].
System (1) assumes that cells and viruses are well mixed, and ignores the mobility of cells and viruses. Motivated by the works [18–21] and make the model more realistic by considering the mobility of cells and viruses, we propose the following diffusive HIV infection model
where T(x, t), I(x, t), E(x, t), V(x, t) and C(x, t) are the densities of uninfected CD4\(^{+}\) T cells, infected cells in the eclipse stage, productive infected cells, free virus particles and CTL cells at location x and time t, respectively. The positive constant d is the diffusion coefficient of virus, and the other positive constant parameters have same meanings as in ODE model (1).
In this study, we consider our model (2) with homogenous Neumann boundary condition
and initial conditions
where \(\Omega \) is a bounded domain in \(\hbox {I R}^{n}\) with smooth boundary \(\partial \Omega \), \(\Delta =\displaystyle {\sum \nolimits _{i=1}^{n}}\frac{\partial ^{2}}{\partial x_{i}^{2}}\) is the Laplacian operator and \(\frac{\partial V}{\partial \nu }\) is the outward normal derivative on \(\partial \Omega \). Biologically speaking, the Neumann boundary condition means that the free virus particles do not move across the boundary \(\partial \Omega \).
The organization of this paper is as follows. In the next section, we well first determine the equilibria of our ODE model and derive two threshold parameters for viral infection \(R_{0}\) and for CTL immune response \(R_{1}\). After, we establish the global stability of these equilibria that are the infection-free equilibrium, the immune-free infection equilibrium and the chronic infection equilibrium. Further, we focus on the global dynamics of our PDE model in Sect. 3. In order to validate our theoretical results, numerical simulations are presented in Sect. 4. Finally, a brief conclusion and discussion are given in Sect. 5.
2 Global stability of the ODE model
System (1) has always an infection-free equilibrium of the form \(Q_{0}(\displaystyle \frac{\lambda }{\mu _{T}},0,0,0,0)\).
Hence, the basic reproduction number of (1) is given by
We recall that \(R_{0}\) represents the number of secondary infections produced by one productive infected cell during the period of infection when all cells are uninfected.
The other equilibria of system (1) are solutions of the following system
By (5), we get \(C=0\) or \(I=\frac{\mu _{C}}{a}.\)
If \(C=0\), using (6)–(4), we obtain \(E=\frac{\lambda -\mu _{T}T}{\mu _{E}+\gamma }\), \(I=\frac{\gamma (\lambda -\mu _{T}T)}{\mu _{I}(\mu _{E}+\gamma )}\), \(V=\frac{k\gamma (\lambda -\mu _{T}T)}{\mu _{I}\mu _{V}(\mu _{E}+\gamma )}\) and
Since \(E=\frac{\lambda -\mu _{T}T}{\mu _{E}+\gamma }\ge 0\), we have \(T\le \frac{\lambda }{\mu _{T}}\). So, there is no biological equilibrium when \(T>\frac{\lambda }{\mu _{T}}\). Define the function \(g_{1}\) on the interval \(\big [0,\frac{\lambda }{\mu _{T}}\big ]\) by
We have \(g_{1}(0)=-\frac{(\rho +\mu _{E}+\gamma )\mu _{V}\mu _{I}}{k\gamma }<0\), \(g_{1}(\frac{\lambda }{\mu _{T}})=\frac{(\rho +\mu _{E}+\gamma )\mu _{V}\mu _{I}}{k\gamma }(R_{0}-1)\) and
If \(R_{0}>1\), there exist an other biological equilibrium \(Q_{1}(T_{1},E_{1},I_{1},V_{1},0)\) with \(T_{1}\in (0,\frac{\lambda }{\mu _{T}})\), \(E_{1}=\frac{\lambda -\mu _{T}T_{1}}{\mu _{E}+\gamma }\), \(I_{1}=\frac{\gamma (\lambda -\mu _{T}T_{1})}{\mu _{I}(\mu _{E}+\gamma )}\) and \(V_{1}=\frac{k\gamma (\lambda -\mu _{T}T_{1})}{\mu _{I}\mu _{V}(\mu _{E}+\gamma )}\). This equilibrium correspond to positive levels of healthy cells, unproductive infected cells, productive infected cells and virus, but no CTL immune response.
If \(C\ne 0\), then \(I=\frac{\mu _{C}}{a}\). Using (6)–(4), we deduce \(E=\frac{\lambda -\mu _{T}T}{\mu _{E}+\gamma }\), \(V=\frac{k\mu _{C}}{a\mu _{V}}\) and
Since \(C=\frac{a \gamma (\lambda -\mu _{T}T)-\mu _{I} \mu _{C} (\mu _{E}+\gamma )}{p\mu _{C}(\mu _{E}+\gamma )}\ge 0\), we have \(T\le \frac{\lambda }{\mu _{T}}-\frac{\mu _{I}\mu _{C}(\mu _{E} + \gamma )}{a \gamma \mu _{T}}\). Then there exists no equilibrium when \(T>\frac{\lambda }{\mu _{T}}-\frac{\mu _{I}\mu _{C}(\mu _{E} + \gamma )}{a \gamma \mu _{T}}\) or \(\frac{\lambda }{\mu _{T}}-\frac{\mu _{I}\mu _{C}(\mu _{E} + \gamma )}{a \gamma \mu _{T}}\le 0\).
Define the function \(g_{2}\) on the interval \(\big [0,\frac{\lambda }{\mu _{T}}-\frac{\mu _{I}\mu _{C}(\mu _{E} + \gamma )}{a \gamma \mu _{T}}]\) by
We have \(g_{2}(0)=-\frac{\lambda a\mu _{V}(\mu _{E}+\rho +\gamma )}{k\mu _{C}(\mu _{E}+\gamma )}<0\) and \(g_{2}'(T)=\frac{\partial f}{\partial T}+\frac{\mu _{T}a\mu _{V}(\mu _{E}+\rho +\gamma )}{k\mu _{C}(\mu _{E}+\gamma )}>0.\)
In addition to \(R_{0}\), we define the CTL immune response reproduction number \(R_{1}\) of our ODE model by
where \(\frac{1}{\mu _{C}}\) represents the average life expectancy of CTL cells, and \(I_{1}\) is the number of productive infected cells at \(Q_{1}\). Hence, \(R_{1}\) represents the average number of CTL cells activated by the productive infected cells when viral infection is successful.
If \(R_{1}<1\), then \(I_{1}<\frac{\mu _{C}}{a}\), \(T_{1}>\frac{\lambda }{\mu _{T}}-\frac{\mu _{I}\mu _{C}(\mu _{E} + \gamma )}{a \gamma \mu _{T}}\) and
Hence, \(g_{2}(\frac{\lambda }{\mu _{T}}-\frac{\mu _{I}\mu _{C}(\mu _{E} + \gamma )}{a \gamma \mu _{T}})<0\). Then there is no equilibrium when \(R_{1}<1\).
If \(R_{1}>1\), then \(I_{1}>\frac{\mu _{C}}{a}\), \(T_{1}<\frac{\lambda }{\mu _{T}}-\frac{\mu _{I}\mu _{C}(\mu _{E} + \gamma )}{a \gamma \mu _{T}}\) and \(g_{2}(\frac{\lambda }{\mu _{T}}-\frac{\mu _{I}\mu _{C}(\mu _{E} + \gamma )}{a \gamma \mu _{T}})>0\). Therefore, if \(R_{1}>1\), there exists an infection equilibrium \(Q_{2}(T_{2},E_{2},I_{2},V_{2},C_{2})\) with \(T_{2}\in (0,\frac{\lambda }{\mu _{T}}-\frac{\mu _{I}\mu _{C}(\mu _{E} + \gamma )}{a \gamma \mu _{T}})\), \(E_{2}=\frac{\lambda -\mu _{T}T_{2}}{\mu _{E}+\gamma }\), \(I_{2}=\frac{\mu _{C}}{a}\), \(V_{2}=\frac{k\mu _{C}}{a\mu _{V}}\) and \(C_{2}=\frac{a \gamma (\lambda -\mu _{T}T_{2})-\mu _{I} \mu _{C} (\mu _{E}+\gamma )}{p\mu _{C}(\mu _{E}+\gamma )}\). This equilibrium denotes the state in which both the virus and the CTL immune response are present.
Summarizing the above discussions in the following result.
Theorem 2.1
-
(i)
When \(R_0 \le 1\), the system (1) has always an infection-free equilibrium of the form \(Q_{0}(\displaystyle \frac{\lambda }{\mu _{T}},0,0,0,0)\).
-
(ii)
When \(R_0>1\), the system (1) has an immune-free infection equilibrium of the form \(Q_{1}(T_{1},E_{1},I_{1},V_{1},0)\) with \(T_{1}\in (0,\displaystyle \frac{\lambda }{\mu _{T}})\), \(E_{1}=\frac{\lambda -\mu _{T}T_{1}}{\mu _{E}+\gamma }\), \(I_{1}=\frac{\gamma (\lambda -\mu _{T}T_{1})}{\mu _{I}(\mu _{E}+\gamma )}\) and \(V_{1}=\frac{k\gamma (\lambda -\mu _{T}T_{1})}{\mu _{I}\mu _{V}(\mu _{E}+\gamma )}\).
-
(iii)
When \(R_1>1\), the system (1) has an infection equilibrium of the form \(Q_{2}(T_{2},E_{2},I_{2},V_{2},C_{2})\) with \(T_{2}\in (0,\frac{\lambda }{\mu _{T}}-\frac{\mu _{I}\mu _{C}(\mu _{E} + \gamma )}{a \gamma \mu _{T}})\), \(E_{2}=\frac{\lambda -\mu _{T}T_{2}}{\mu _{E}+\gamma }\), \(I_{2}=\frac{\mu _{C}}{a}\), \(V_{2}=\frac{k\mu _{C}}{a\mu _{V}}\) and \(C_{2}=\frac{a \gamma (\lambda -\mu _{T}T_{2})-\mu _{I} \mu _{C} (\mu _{E}+\gamma )}{p\mu _{C}(\mu _{E}+\gamma )}\).
Now, we focus on the global stability of the three equilibria of system (1). At first, we have
Theorem 2.2
If \(R_{0}\le 1\), then the infection-free equilibrium \(Q_{0}\) is globally asymptotically stable.
Proof
Consider the following Lyapunov functional
where \(T_{0}=\frac{\lambda }{\mu _{T}}\).
Calculating the time derivative of \(W_{0}\) along the positive solutions of system (1), we obtain
Noting that \(\lambda =\mu _{T}T_{0}\), we get
Therefore, \(\frac{dW_{0}}{dt}\le 0\) if \(R_{0}\le 1\). Further, \(\frac{dW_{0}}{dt}=0\) if and only if \(T=\frac{\lambda }{\mu _{T}}\), \(E=0\), \(I=0\), \(V=0\) and \(C=0\). Hence, the largest compact invariant set in \(\{(T,E,I,V,C)|\frac{dW_{0}}{dt}=0\}\) is just the singleton \(\{Q_{0}\}\). Thus, the global stability of the infection-free equilibrium \(Q_{0}\) follows from LaSalle’s invariance principle [22]. \(\square \)
Theorem 2.3
The immune-free infection equilibrium \(Q_{1}\) of system (1) is globally asymptotically stable if \(R_{1}\le 1<R_{0}\) and
Proof
Construct a Lyapunov functional \(W_{1}\) as follows
where \(\Phi (x)=x-1-\ln (x)\).
The time derivative of \(W_{0}\) along the positive solutions of system (1) satisfies
By \(\lambda =\mu _{T} T_{1}+f(T_{1},V_{1})V_{1}-\rho E_{1}\), we get
Using the arithmetic-geometric inequality, we get
Therefore, \(\frac{dW_{1}}{dt}\le 0\) if \(R_{1}\le 1\) and \(\rho E_{1}\leqslant \mu _{T} T_{1}\).
Obviously, the condition \(\rho E_{1}\leqslant \mu _{T} T_{1}\) is equivalent to
In addition, \(\frac{dW_{1}}{dt}=0\) if and only if \(T=T_{1}\), \(E=E_{1}\), \(I=I_{1}\), \(V=V_{1}\) and \(C=0\). Hence, the largest compact invariant set in \(\{(T,E,I,V,C)|\frac{dW_{1}}{dt}=0\}\) is the singleton \(\{Q_{1}\}\). This proves the global stability of \(Q_{1}\) by using LaSalle invariance principle. \(\square \)
Remark 2.4
If \(\alpha _{3}=0\), then the condition (12) becomes
which is the condition given by Lv et al. [12] that ensures the global asymptotic stability of the immune-free infection equilibrium.
It is important to see that
According to theorem 2.3, we get the following result
Corollary 2.5
-
(i)
The immune-free infection equilibrium \(Q_{1}\) is globally asymptotically stable if \(R_{1}\le 1<R_{0}\) and \(\rho \) sufficiently small.
-
(ii)
The immune-free infection equilibrium \(Q_{1}\) is globally asymptotically stable if \(R_{1}\le 1<R_{0}\) and \(\gamma \) sufficiently large.
Finally, we investigate the global stability of the third equilibrium \(Q_{2}\).
Theorem 2.6
The chronic infection equilibrium with immune response \(Q_{2}\) is globally asymptotically stable if \(R_{1}>1\) and
Proof
Define a Lyapunov functional \(W_{2}\) as follows
Calculating the derivative of \(W_{2}\) along the positive solutions of the system (1), we have
Applying the equality \(\lambda =\mu _{T} T_{2}+f(T_{2},V_{2})V_{2}-\rho E_{2}\), we get
Stability of the infection-free equilibrium \(Q_{0}\) for system (1)
From (13), we deduce that \(\frac{dW_{2}}{dt}\le 0\) if \(R_{1}>1\) and \(\rho E_{2}\leqslant \mu _{T} T_{2}\).
However, this last condition is equivalent to
Note that \(\frac{dW_{1}}{dt}=0\) if and only if \(T=T_{2}\), \(E=E_{2}\), \(I=I_{2}\), \(V=V_{2}\) and \(C=C_{2}\). Hence, the largest compact invariant set in \(\{(T,E,I,V,C)|\frac{dW_{2}}{dt}=0\}\) is the singleton \(\{Q_{2}\}\). Based on the LaSalle invariance principle, we deduce that \(Q_{2}\) is globally asymptotically stable. This completes the proof. \(\square \)
We remark that the condition (14) holds for \(\rho \) sufficiently small, or \(\gamma \) sufficiently large. Hence, we have the following corollary.
Corollary 2.7
-
(i)
The chronic infection equilibrium \(Q_{2}\) is globally asymptotically stable if \(R_{1}>1\) and \(\rho \) sufficiently small.
-
(ii)
The chronic infection equilibrium \(Q_{2}\) is globally asymptotically stable if \(R_{1}>1\) and \(\gamma \) sufficiently large.
In addition, we have the following remark.
Remark 2.8
If \(\alpha _{3}=0\), then the condition (14) becomes
which is the condition given by Lv et al. [12] that ensures the global asymptotic stability of the chronic infection equilibrium.
Stability of the infection-free equilibrium \(Q_{0}\) for system (2)
Stability of the immune-free infection equilibrium \(Q_{1}\) for system (1)
3 Global stability of the PDE model
It is not hard to see that the PDE model (2) has the same three equilibria as the ODE model (1), namely, the infection-free equilibrium \(Q_{0}\), the immune-free infection equilibrium \(Q_{1}\) which exists whenever \(R_{0}>1 \)and the chronic infection equilibrium \(Q_{2}\) which exists if \(R_{1}>1\).
Now, we investigate the global stability of problem (2)–(4) by constructing appropriate Lyapunov functionals. The construction of these Lyapunov functionals is based on the method proposed by Hattaf and Yousfi [23].
Theorem 3.1
For all diffusion coefficients, we have
-
(i)
The infection-free equilibrium \(Q_{0}\) of problem (2)–(4) is globally asymptotically stable if \(R_{0}\le 1\).
-
(ii)
The immune-free infection equilibrium \(Q_{1}\) of problem (2)–(4) is globally asymptotically stable if \(R_{1}\le 1<R_{0}\) and the condition (12) holds .
-
(iii)
The chronic infection equilibrium \(Q_{2}\) of problem (2)–(4) is globally asymptotically stable if \(R_{1}>1\) and the condition (14) holds.
Proof
Let \(u(t,x)=\big (T(t,x),I(t,x),V(t,x),E(t,x),C(t,x)\big )\) be a solution of (2)–(4), and we put
where \(W_{i}\) is the Lyapunov functional for the ODE model (1) at \(Q_{i}\), \(i=0,1,2\).
It is easy to check that \(W_{i}\) satisfies the condition (15) given in [23], for all \(i=0,1,2\). From Proposition 2.1 given in Ref. [23], we deduce that \(L_{i}\) is a Lyapunov functional for problem (2)–(4) at \(Q_{i}\). This completes the proof. \(\square \)
Stability of the immune-free infection equilibrium \(Q_{1}\) for system (2)
Stability of the chronic infection equilibrium \(Q_{2}\) for system (1)
4 Numerical simulations
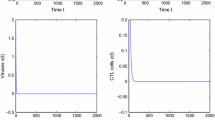
In this section, we present some numerical simulations to validate our theoretical results. Firstly, we consider parameter values \(\Lambda =10\) cells \(\mu l^{-1}\) day\(^{-1}\), \(\mu _{T}=0.0139\) day\(^{-1}\), \(\beta =2.4\times 10^{-5}\) \(\mu l\) virion\(^{-1}\) day\(^{-1}\), \(\alpha _{1}=0.1\), \(\alpha _{2}=0.01\), \(\alpha _{3}=0.00001\), \(\rho =0.01\) day\(^{-1}\), \(\gamma =0.01\) day\(^{-1}\), \(\mu _{I}=0.27\) day\(^{-1}\), \(\mu _{E}=0.0347\) day\(^{-1}\), \(p=0.001\) cell\(^{-1}\) \(\mu l\) day\(^{-1}\), \(k=1200\) virion cell\(^{-1}\) day\(^{-1}\), \(\mu _{V}=3\) day\(^{-1}\), \(a=0.002\) cell\(^{-1}\) \(\mu l\) day\(^{-1}\), \(\mu _{C}=0.1\) day\(^{-1}\) and \(d=0.1\). Here, the values p, a and \(\mu _{C}\) are taken from [1, 5], while the other parameter values are chosen from [10]. For these set of parameter values, we have \(R_{0}=0.1141<1\). Hence, systems (1) and (2) have an infection-free equilibrium \(Q_{0}(719,4245,0,0,0,0)\) which is globally asymptotically stable according to Theorems 2.2 and 3.1 (i). Figures 1 and 2 illustrate this result.
Secondly, we choose \(\beta =0.0012\) \(\mu l\) virion\(^{-1}\) day\(^{-1}\) and do not change the other parameter values. By calculation, we have \(R_{0}=3.2055>1\), \(R_{1}=0.1427<1\) and \(\frac{[\mu _{T}\mu _{I}\mu _{V}(\mu _{E}+\gamma )+\alpha _{2}\mu _{T}\lambda k\gamma ](\mu _{E}+\rho +\gamma )+\rho \alpha _{3}k\gamma \lambda ^{2}}{\rho \mu _{I}\mu _{V}(\mu _{E}+\rho +\gamma )(\mu _{T}+\alpha _{1}\lambda )}= 2.2725\). In this case, both systems have an immune-free equilibrium \(Q_{1}(138.53,183.16,6.80,2724.2,0)\). By Theorems 2.3 and 3.1 (ii), \(Q_{1}\) is globally asymptotically stable (see Figs. 3 and 4). We see that in the absence of CTL cells, the number of CD4\(^{+}\) T cells decreases to the value 138.53, which means that the patient enters in the phase AIDS (< 200 cell mm\(^{-3}\)).
Stability of the chronic infection equilibrium \(Q_{2}\) for system (2)
Stability of the immune-free infection equilibrium \(Q_{1}\) with condition (12) not satisfied
Thirdly, we change one parameter which is \(a=0.065\) cell\(^{-1}\) \(\mu l\) day\(^{-1}\). By calculation, we have \(R_{1}=4.6379>1\). Then systems (1) and (2) have a chronic infection equilibrium \(Q_{2}(370.56,106.97,1.57,629.65,409.12)\). Therefore, by Theorems 2.6 and 3.1 (iii), \(Q_{2}\) is globally asymptotically stable (see Figs. 5 and 6). In this case, we see that in the presence of CTL cells, the number of CD4\(^{+}\) T cells increases to the value \(252.0792>200\) cell mm\(^{-3}\), which means that the patient is no longer in the phase AIDS.
Finally, in Fig. 7, we see that the dynamics of HIV infection converges to steady state \(Q_{1}\) for all initial conditions. However, the condition (12) is not satisfied with \(R_{0}=4.1501>1\), \(R_{1}=0.0999<1\) and
Similarly, in Fig. 8, we see that the dynamics of HIV infection converges to steady state \(Q_{2}\) for all initial conditions. However, the condition (14) is not satisfied with \(R_{1}=3.4338\) and \(\alpha _{1}\lambda \rho a\mu _{V}+\mu _{T}(\rho +\mu _{E}+\gamma )(\alpha _{2}k\mu _{C}+a\mu _{V})+\alpha _{3}\rho \lambda k\mu _{C}-k\beta \mu _{C}\rho =-0.0041<0\). Therefore, the conditions (12) and (14) are not necessary for the global stability of \(Q_{1}\) and \(Q_{2}\).
Stability of the chronic infection equilibrium \(Q_{2}\) with condition (14) not satisfied
5 Conclusion and discussion
In this paper, we have proposed two HIV infection models. The first is an ODE model with cure of infected cells in eclipse stage, CTL immune response and Hattaf’s incidence rate which includes the traditional bilinear incidence rate, the saturated incidence rate, the Beddington–DeAngelis functional response and the Crowley-Martin functional response. The second is a PDE model that extends the first one by taking into account the diffusion of virus. The two models admits three equilibria, namely, the infection-free equilibrium \(Q_{0}\), the immune-free infection equilibrium \(Q_{1}\) which exists whenever \(R_{0}>1 \) and the chronic infection equilibrium \(Q_{2}\) which exists if \(R_{1}>1\). The global stability of these three equilibria have obtained in terms of the basic reproduction number \(R_{0}\) and the CTL immune response reproduction number \(R_{1}\). It is shown that \(Q_f\) is globally asymptotically stable when \(R_{0}\le 1\), \(Q_1\) is globally asymptotically stable when \(R_{1}\le 1<R_{0}\) and the condition (12) holds, and \(Q_{2}\) is globally asymptotically stable when \(R_{1}>1\) and the condition (14) holds.
In addition, we remark that if the cure rate \(\rho \) is sufficiently small or the value of \(\gamma \) is sufficiently large, the conditions (12) and (14) are satisfied. From the numerical simulations (Figs. 7 and 8), we see that both equilibria \(Q_{1}\) and \(Q_{2}\) remain globally asymptotically stable without the conditions (12) and (14) are satisfied.
Observing that the basic reproduction number \(R_{0}\) is independent of the CTL immune parameters. Further, by comparing the components of healthy cells, infected cells in the eclipse stage, productive infected cells and viral load before and after the activation of CTL response, we have \(T_{2}\)>\(T_{1}\), \(E_{2}<E_{1}\), \(I_{2}<I_{1}\) and \(V_{2}<V_{1}\) when \(R_{1}>1\). Therefore, we deduce that the activation of CTL immune response is unable to eliminate the virus in the host population, but plays an important role in HIV infection by reducing the viral load, increasing the healthy cells and decreasing the two classes of infected cells.
References
Nowak MA, Bangham CRM (1996) Population dynamics of immune responses to persistent viruses. Science 272:74–79
Zhou X, Shi X, Zhang Z, Song X (2009) Dynamical behavior of a virus dynamics model with CTL immune response. Appl Math Comput 213(2):329–347
Wang X, Tao Y, Song X (2011) Global stability of a virus dynamics model with Beddington–DeAngelis incidence rate and CTL immune response. Nonlinear Dyn 66:825–830
Hattaf K, Yousfi N, Tridane A (2012) Global stability analysis of a generalized virus dynamics model with the immune response. Can Appl Math Q 20(4):499–518
Wang Y, Zhou Y, Brauer F, Heffernan JM (2013) Viral dynamics model with CTL immune response incorporating antiretroviral therapy. J Math Biol 67:901–934
Rong L, Gilchrist MA, Feng Z, Perelson AS (2007) Modeling within-host HIV-1 dynamics and the evolution of drug resistance: trade-offs between viral enzyme function and drug susceptibility. J Theoret Biol 247:804–818
Buonomo B, Vargas-De-Léon C (2012) Global stability for an HIV-1 infection model including an eclipse stage of infected cells. J Math Anal Appl 385:709–720
Hu Z, Pang W, Liao F, Ma W (2014) Analysis of a CD4\(^{+}\) T cell viral infection model with a class of saturated infection rate. Discrete Continuous Dyn Syst Ser B 19:735–745
Wang J, Lang J, Liu X (2015) Global dynamics for viral infection model with Beddington–Deangelis functional response and an eclipse stage of infected cells. Discrete and Continuous Dyn Syst Ser B 20(9):3215–3233
Maziane M, Lotfi E, Hattaf K, Yousfi N (2015) Dynamics of a class of HIV infection models with cure of infected cells in eclipse stage. Acta Biotheor 63:363–380
Hattaf K, Yousfi N, Tridane A (2013) Stability analysis of a virus dynamics model with general incidence rate and two delays. Appl Math Comput 221:514–521
Lv C, Huang L, Yuan Z (2014) Global stability for an HIV-1 infection model with Beddington–DeAngelis incidence rate and CTL immune response. Commun Nonlinear Sci Numer Simul 19:121–127
Beddington JR (1975) Mutual interference between parasites or predators and its effect on searching efficiency. J Anim Ecol 44:331–341
DeAngelis DL, Goldsten RA, Neill R (1975) A model for trophic interaction. Ecology 56:881–892
Crowley PH, Martin EK (1989) Functional responses and interference within and between year classes of a dragonfly population. J North Am Benthol Soc 8:211–221
Zhou X, Cui J (2011) Global stability of the viral dynamics with Crowley-Martin functional response. Bull Korean Math Soc 48(3):555–574
Liu XQ, Zhong SM, Tian BD, Zheng FX (2013) Asymptotic properties of a stochastic predator–prey model with Crowley-Martin functional response. J Appl Math Comput 43:479–490
Brauner C-M, Jolly D, Lorenzi L, Thiebaut R (2011) Heterogeneous viral environment in a HIV spatial model. Discrete Cont Dyn B 15:545–572
Wang K, Wang W (2007) Propagation of HBV with spatial dependence. Math Biosci 210:78–95
Hattaf K, Yousfi N (2015) Global dynamics of a delay reaction–diffusion model for viral infection with specific functional response. Comput Appl Math 34(3):807–818
Hattaf K, Yousfi N (2015) A generalized HBV model with diffusion and two delays. Comput Math Appl 69(1):31–40
LaSalle JP (1976) The stability of dynamical systems. In: Regional conference series in applied mathematics, SIAM, Philadelphia
Hattaf K, Yousfi N (2013) Global stability for reaction–diffusion equations in biology. Comput Math Appl 66:1488–1497
Acknowledgments
We would like to express their gratitude to the editor and the anonymous referees for their constructive comments and suggestions, which have improved the quality of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maziane, M., Hattaf, K. & Yousfi, N. Global stability for a class of HIV infection models with cure of infected cells in eclipse stage and CTL immune response. Int. J. Dynam. Control 5, 1035–1045 (2017). https://doi.org/10.1007/s40435-016-0268-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40435-016-0268-4