Abstract
The reproductive success of organisms depends on the positive and negative interactions it establishes throughout its lifetime. Pollinators and mutualists increase adaptive value, while herbivory and antagonism impair plant performance. Asteraceae flowers, simultaneously interacting with pollinators, herbivores and parasitoids with the same structure (the flower head), serve as a model for studying these interactions. In this work we describe the multi-layered interactions between Proteopsis argentea,, its pollinators, endophagous herbivores and parasitoids, aiming to understand how they influence the plant's reproductive success and whether these processes are associated with its conservation status as vulnerable. We found that P. argentea produces little, but highly energetic nectar, acting in interactions with bees, hummingbirds and lepidopterans in a generalist pollination system. Antagonistic interactions were much less diverse, dominated by Xanthaciura aff. chrysura. In addition, two morphospecies of parasitoid wasps used the flower heads of P. argentea, and may have a negative impact on antagonists. We also found that P. argentea presents a reproductive insurance strategy, via autogamy, producing seeds without pollinators. The flowers of P. argentea seem to act as efficient filters, promoting interactions with potential pollinators and parasitoids and limiting interaction with endophagous herbivores. The reproductive strategies of P. argentea, with an apparently efficient floral filter and reproductive insurance in the absence of pollinators, seems to indicate that reproduction is not a limiting factor for the species. Thus, for its conversation, measures ensuring the protection of its occurrence area should be prioritized.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Interactions between organisms are ubiquitous in ecosystems and play a fundamental role in the stability of biological communities and the maintenance of biodiversity (Bronstein 2009). These interactions are diverse in nature, ranging on a continuum from mutualistic, when there is a positive balance for the species involved, to antagonistic, when those involved are impaired (Almeida-Neto et al. 2010; Rech and Brito 2012). Taken together every group of interactions established could be considered as a layer of connectivity within the interactive community (Kivela et al. 2014). In nature, plants are simultaneously exposed to these interaction layers, and they significantly influence the performance of individuals in populations (Morris et al. 2007). Understanding how traits vary between individuals and how they mediate the net effects of mutualistic and antagonistic interactions in a species throughout its lifetime is an important challenge for understanding the potential evolutionary forces operating on its genotype and phenotype (Siepielski and Benkman 2010).
In the context of mutualistic interactions, plants and their pollinators play a key role in maintaining biodiversity (Biesmeijer et al. 2006). Pollen transfer between flowers is essential for fertilization and the development of viable seeds in most flowering plants (Ollerton et al. 2011). Understanding the mutual dependence between plants and their pollinators is fundamental for the conservation of these interactions, and the loss or change in the behavior of pollinators can have negative effects on the plants relying on them (Biesmeijer et al. 2006). In this sense, we can find a myriad of structures and behaviors that different plant species may employ to attract, retain, and exploit pollinators as pollen dispersers (Pyke 2016; Gurevich and Hadany 2021).
By signaling the existence of resources, flowers also attract organisms that may not contribute to pollination or seed dispersal, but only feed on the resources offered, creating the so-called defense-apparency dilemma (Huang et al. 2022; Sasidharan et al. 2023). The presence of herbivorous organisms, such as florivores or seed parasites, represents a major cost to plant fitness (Almeida-Neto et al. 2010). Just as pollinator’s absence impairs or prevents the reproductive success of a plant, the presence of a herbivore can have a similar effect. These herbivores feed directly on the reproductive structures of plants, resulting in a decrease in floral attractiveness, viable seeds and reproductive success (Boaventura et al. 2022). In this dilemma, natural selection must favor the functioning of flowers as filters, attracting mutualists and repelling or restraining the access of antagonists (Shuttleworth and Johnson 2009; Papiorek et al. 2015).
It is important to highlight that the traditional dichotomy between mutualism and antagonism may be too simplistic to capture the complex dynamics of ecological relationships (Fontaine et al. 2011). A more realistic perspective, which recognizes the fluid and multifaceted nature of these interactions, is the objective addressing of interactive layers (Pilosof et al. 2017). This perspective recognizes that relationships between organisms are not static, but rather dynamic, varying in degrees of mutualism and antagonism over time and in different contexts (Bronstein et al. 2003; Chamberlain et al. 2014). Adopting this holistic view is fundamental to a deeper and more accurate understanding of the complex relationships that sustain the web of life in ecosystems.
Asteraceae is one of the largest and most diverse plant families, presenting a wide variety of species all over the world. In Brazil, the Asteraceae are represented by 335 genera, with 2228 species, of which 1369 are endemic (Flora do Brasil 2024). Furthermore, it is a highly suitable study model for investigating mutualistic and antagonistic interactions simultaneously (Almeida et al. 2006; Theis et al. 2007; Perre et al. 2011; Amorim et al. 2021; Lewinsohn et al. 2022). Theis et al. (2007) revealed that plants of this family, such as the Cirsium species investigated, can adjust the emission of fragrance to attract pollinators during periods of insect activity and minimize the attraction of floral herbivores. This is because the flower-head inflorescence (capitulum), where the flowers and fruits (achenes) are located, interact concomitantly with pollinators and herbivores (flower-head endophages) (Theis et al. 2007). This dual attraction of different groups of organisms with different net effects on fitness makes the Asteraceae an exemplary model for examining the complex relationships between plants and the various organisms they interact with (Romero and Vasconcellos-Neto 2005). It also allows us to understand the evolutionary mechanisms resulting from this balance of ecological effects on propagule production (Cosmo et al. 2023). Although the Asteraceae family has been considered in ecological studies focused on community assembly (Almeida et al. 2006; Nascimento et al. 2014; Bergamini et al. 2017), detailed analyses of the antagonistic forces operating with the organisms attracted to floral structures are needed. The intricate network of relationships between plants in the family, pollinators, endophages and their parasitoids, raises the need for a thorough investigation into the floral functioning of Asteraceae.
In this scenario, Proteopsis argentea Mart. & Zucc. ex Sch.Bip. (Asteraceae) stands out as an herb endemic to the campos rupestres classified as vulnerable to extinction (Ministério do Meio Ambiente 2022; Loeuille 2024), which is aggravated by its occurrence in geographically isolated populations (Jesus et al. 2001), many of them outside protected areas (Martinelli and Moraes 2013), which can lead to a loss of genetic variability. This spatial fragmentation, mediated by habitat loss and anthropogenic pressures, is one of the main threats to the persistence of this species (Flora do Brasil 2024). Curiously, despite its critical position in the context of conservation, the natural history of this species remains largely unknown as it seems to be the case for endangered Asteraceae in general. It is in this context that our case study is inserted, aiming to fill this gap with a comprehensive investigation of the interactions occurring in the flower heads of P. argentea. We also sought to understand what are the potential consequences of these interactions to the conservation efforts of this species. Our primary objective was to evaluate seed production in P. argentea to determine the role of pollination and herbivory in its reproductive success. We particularly pursued how these factors influence the persistence of P. argentea and, based on this understanding, help develop more effective conservation strategies to protect vulnerable plant species.
2 Materials and methods
2.1 Study area
Our study was conducted between March and April 2023, in a natural area of campos rupestres belonging to the Universidade Federal dos Vales do Jequitinhonha e Mucuri (UFVJM – Fig. 1). Campos rupestres are ancient ecosystems found at altitudes above 900 m, characterized by rocky outcrops and shallow, acidic, nutrient-poor soils (Alves et al. 2014).
Most campos rupestres are distributed in the Espinhaço mountain range, in the states of Bahia and Minas Gerais. These are megadiverse environments, concentrating almost 15% of the Brazilian flora in less than 1% of its national territory, with a large number of endemic species (Silveira et al. 2016). We sampled 22 Proteopsis argentea individuals, which produced a total of 54 glomeruli. Each glomerulus, in turn, produced on average 13 ± 2.2 inflorescences. Proteopis argentea only occurs in campos rupestres, within occurrence sites smaller than 500 km2. Despite their importance and the little we know about how they function, campos rupestres have faced significant threats due to human activity.
2.2 Flower morphology
We measured the tube length of P. argentea flowers from nine different individuals. For each individual, we selected two flowers from different inflorescences. We measured floral tubes using photographs shot, with the Sony Handycam HDR-CX405 Full HD NTSC/PAL camera, against 10-mm-scaled paper (Schneider et al. 2012) and the ImageJ software.
2.3 Nectar soluble sugar production and concentration
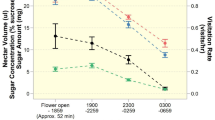
We quantified nectar production and concentration in 10 individuals of P. argentea. We measured the sugar concentration in the nectar in all of them, but in only 8 its was possible to measure nectar volume. The nectar sugar concentration was measured using a portable refractometer (Hilitand) with a measuring range of 0–50% Brix and a resolution of 0.1%. To measure nectar volume, we bagged the inflorescences with organza bags at sunset the day before data collection. The following day, between 8 and 10 am, we measured the nectar volume using a microcapillary (1 µL) in a flower at anthesis per inflorescence. The nectar was absorbed by the microcapillary tubes due to capillarity. The amount of nectar withdrawn was then measured by the height of the nectar column inside the tube, and the filled proportion of the entire column (1 µL) was calculated. Subsequently, the nectar in the microcapillary tubes was washed with distilled water (Morrant et al. 2009). The number of inflorescences evaluated per plant varies between one and five (30 per volume and 36 per concentration) depending on the availability of inflorescences on the plant at the time of data collection.
2.4 Seed production
We selected 14 plants to quantify and qualify their seed production. The average number of inflorescences per individual was 5 (± 2.5). Following flower senescence, we bagged the inflorescences to prevent damage and protect the developing seeds. Once mature, seeds were collected, counted and separated into viable and non-viable based on morphological viability criteria. We considered full seeds (i.e., containing endosperm) as viable and empty seeds (without endosperm) as non-viable (Feitosa et al. 2009).
2.5 Dependence on biotic pollination for seed production
To assess pollinator dependence, we conducted an experiment with eight individuals, each subjected to two conditions: In the treatment, the flower buds (n = 31) were isolated from floral visitors with organza bags. In the control group, the same number of flower buds on each plant were marked with cotton strings, with free access to pollinators. Following the fruiting period, once the seeds were ripe, we counted and compared the total number of seeds produced in both groups.
2.6 Floral visitors
We observed 19 Proteopsis argentea individuals during the flowering period between March and April 2023. During this period, we collected a total of 31 h in data on interactions between plants and their floral visitors. We recorded the number of open flowers and the number observed per plant at each moment. In addition, we carried out focal observations at regular intervals of 30 min for each individual, taking notes and identifying the species of floral visitors that visited them.
2.7 Flower-head endophages
To assess the incidence and abundance of herbivory by flower-head endophages and abundance of parasitoids, we collected P. argentea inflorescences at the moment they entered senescence following standard methodology (Almeida et al. 2006). After collection, we placed the flower heads in plastic jars covered with organza to allow air to pass through and ensure the emergence of insects from inside the flower structures.
Every week, the emerged insects were euthanized with carbon dioxide and by freezing. This approach ensured that their morphological features were preserved for later taxonomic identification at the lowest possible level.
2.8 Data analysis
Reproductive Success Analysis: To analyze reproductive success, we used a generalized linear mixed model (GLMM) with a binomial distribution and logit link function. The model was fitted using the glmer function from the lme4 package in the R programming environment. The model formula was defined as the count of "Feasible" and "Infeasible" events relative to a constant predictor, with a random term for the effect of the individual. Akaike (AIC) and Bayes (BIC) information criteria, as well as deviation and log-likelihood estimates were recorded to assess the fit of the model to the data.
Furthermore, we calculated the average probability of success using the emmeans package and its function emmeans. We specified the response type as "response" to obtain the mean probability estimates and their 95% confidence intervals.
Pollinator Dependency Analysis: To assess pollinator dependence on seed production, we fit a linear mixed model (LMM) using the lmer function from the lme4 package. The model formula included the number of seeds as the response variable, with treatment as a fixed predictor and a random term for the individual effect. We used the restricted maximum likelihood (REML) estimation method to fit the model. We recorded the deviations and parameters of the random and fixed effects.
Furthermore, we performed mean contrasts between the "With pollinator" and "Without pollinator" treatments using the emmeans package and its emmeans() function. We calculated the estimated means for each treatment along with their 95% confidence intervals. The estimated difference between treatments was recorded, along with the p-value associated with the contrast, to assess the statistical significance of the differences.
3 Results
3.1 Floral and phenological aspects and nectar soluble sugar production and concentration
Proteopsis argentea flowers present a tubular structure with an average size of 1.6 cm (SD ± 0.1 cm) and a lilac color. They are arranged in flower-head inflorescences (capitula), organized in glomeruli. In these inflorescences, flower heads flower asynchronously. We observed an average production of accumulated nectar per flower of approximately 0.7 ± 0.2 µL, composed of around 43 ± 0.5% SD of sugars.
3.2 Seed production and dependence on pollinators
Our results indicate a high probability of reproductive success among the individuals studied, with an overall estimate of approximately 95.1%. Furthermore, with a 95% confidence level, we can state that the proportion of viable seeds is between 89% and 97.9%. The adjusted model presented reasonable AIC and BIC values, indicating an adequate fit to the data. Finally, the random effects included in the model were considered, recognizing the possibility of variation not explained by the explanatory variables included.
Our results show similar estimated means for the number of seeds produced in both treatments in P. argentea. With pollinators the estimated average was 105.6 seeds, with a confidence interval of 92.3 to 119. Without pollinators, the estimated average was slightly lower, totaling 97.3 seeds, with a confidence interval of 84.0 to 111. When comparing these treatments, the estimated difference between them was 8.23 seeds and not significant (p = 0.2, Fig. 2).
3.3 Floral visitors
We observed a rich diversity of pollinators visiting the inflorescences of P. argentea. Bees stood out as the most representative group of interactions (55.3%), with the species Bombus morio, Agapostemon sp. Centris tarsata and Megachilidae sp.1 (Fig. 3) as the most abundant in this group, with 232, 153, 121 and 100 interactions, respectively. In addition, the hummingbirds Augastes scutatus and Chlorostilbon lucidus also played an important role in visitation, with 96 and 44 interactions respectively, together accounting for 12% of the 1136 interactions observed involving all visitors (Fig. 3).
Floral visitors of Proteopsis argentea. A Halictidae sp. B Centris tarsata. C Bombus morio. D Sarbia damippe. E Chlorostilbon lucidus. F Augastes scutatus. G Dielis trifasciata. H Synargis paulistinha. The image board was created with Microsoft Office Professional Plus 2019 software (version 2203) for Windows 64 bits
We also observed dipterans (9.2%) visiting the flowers of Proteopsis argentea, represented by 11 different morphotypes, totaling 105 interactions. In addition, we recorded other groups of visitors, such as beetles (9 morphotypes with 0.8% of interactions), butterflies including Sarbia damippe (25 visits) and Synargis paulistina (2 visits) which accounted for 2.4% of visits and a wasp, Dielis trifasciata, with a single interaction record (Fig. 3). The visit frequency distribution curve showed an inverted J shape with many rare species and few frequent ones (For a more detailed analysis, please refer to Supplementary Fig. 4).
3.4 Flower-head endophages
With 42 individuals collected, Xanthaciura aff. chrysura was the main herbivore of Proteopsis argentea. In addition, a Tortricid was identified as a herbivore, with 14 occurrences. We also observed the morphospecies Podagrion sp. (Spinola, 1811) and another torymid, both parasitoid wasps, with one occurrence each.
4 Discussion
Proteopsis argentea is an autogamous species that attracts a large number of floral visitors and has good reproductive success, as measured by the production of viable seeds in the wild. The generalization of interaction with pollinators contrasts with its restriction when it comes to endophagous herbivores. This dichotomy suggests an adaptive strategy involving a balance between dependence on pollinators and the need for self-defense against herbivores, reflecting an evolutionary response to environmental fluctuations (Zhang et al. 2022). While the floral traits of this species attract a wide range of pollinators, the concomitant repulsion mechanism directed at endophagous herbivores can be considered a form of specialized defense, resulting in the preservation of floral and progeny integrity and, consequently, the plant's reproductive success. In this sense, P. argentea seems to have effective floral filters to ensure that pollinators are attracted but not antagonistic herbivores.
While nectar production suggests a highly effective floral reward strategy to attract and maintain a variety of visitors, the fruiting rate did not differ between pollinated and bagged flowers, suggesting a reproductive security strategy in the absence of pollinators. Likewise, we can see a morphological specialization in the floral tubes, which allows legitimate access to nectar only for pollinators with long mouthparts. Moreover germination tests are required to define the quality of each group of seeds produced.
The high frequency of hummingbird visits to Proteopsis argentea indicates that these birds benefit from highly energetic nectar produced (Johnson & Nicolson 2008; Van der Kooi et al. 2021). Nectar production is a costly activity for plants (Pyke and Ren 2023). Therefore, it is likely that this activity has some benefit for plant reproduction, such as genetic diversity. In fact, nectar attracts many visitors, which can increasing the chances of pollination, which has also been reported in several other species, even in families predominantly composed of plants that offer pollen as a reward (Brito et al. 2017).
Mutualistic interactions predominated, with bees standing out as the main floral visitors. This observation corroborates the relevance of these insects as key pollinators in natural ecosystems (Pinheiro et al. 2014). In addition, previous studies, such as Monteiro et al. (2021), showed that bee pollination was predominant in a large sample of 636 plant species studied, totaling 56% of the interactions recorded in campos rupestres. Larger bees such as Bombus morio and Centris tarsata stood out as the species that interacted most with the flower heads of P. argentea. In addition, hummingbirds Augastes scutatus and Chlorostilbon lucidus also played an important role in visitation, demonstrating the importance of these birds as pollinators. The occurrence of P. argentea in small populations scattered throughout campos rupestres highlights the importance of pollinators with large body sizes, such as bees and hummingbirds, capable of flying long distances and promoting the exchange of genetic material between distant populations. This contributes to genetic diversity and the conservation of the species (Monteiro et al. 2021).
Our results show that Proteopsis argentea is autogamous, resulting in a reproductive security mechanism (Rech et al. 2018). This reproductive flexibility can be interpreted as an advantageous adaptation to environments with a shortage or fluctuations in the availability of effective pollinators, as well as being beneficial for small, dispersed populations subject to recurrent local extinction events. However, although autogamy can represent a reproductive security mechanism, this strategy can also be associated with costs, such as the loss of genetic variability and increased inbreeding (Jesus et al. 2001; Herlihy & Eckert 2002). Therefore, the studied P. argentea population seems to be benefiting from a balance between cross-pollination by animals and autogamy to optimize its reproductive efficiency under different ecological scenarios.
We observed a low diversity of antagonists, which, together with the presence of natural enemies, are integrated with the positive effects of pollinators and even autogamy. Such interactions can play a crucial role in population regulation and behavior of endophagous, exerting an indirect influence on plant reproduction. The relevance of these interactions is supported by studies such as Nunes et al. 2018, who investigated a scenario in which weevils pollinated orchids before their larvae feed on the growing fruit. The results revealed that this relationship has an adverse impact on the plant, since the endophagous larvae often consume the entire infested fruit. However, parasitoid wasps often rescue part of the infested fruit, eliminating the endophagous larvae before the entire fruit content is consumed. Fruits that are rescued exhibit seed viability and biomass comparable to that of uninfested fruits, thus highlighting the fundamental relevance of parasitoids in mitigating the effects of the interaction between the plant and its parasitic pollinator on the plant's fitness.
We conclude that the reproductive ecology of Proteopsis argentea is truly complex, showing a multilayered network of interactions between pollinators, endophages, parasitoids and the plant itself. The filter function performed by the flowers, the dual mechanism of reproductive security, as well as the presence of long-flight pollinators such as bees and hummingbirds, play vital roles in the efficient transfer of pollen between small and distant populations of P. argentea in the mosaic-like environment of campos rupestres. Our results seem to indicate that the vulnerable threatened status of P. argentea is not associated with its ability to produce seeds, but with other processes acting on germination, recruitment, establishment, and maintenance of viable populations in its potential area of occurrence. In addition, many of these populations are located outside protected areas, making them vulnerable to human activities and environmental degradation, which further accentuates the challenges for the preservation of P. argentea and its complex reproductive ecology in campos rupestres. Urban expansion, eucalyptus agriculture and silviculture, mining and other anthropogenic processes have contributed to the reduction of these natural areas, threatening the survival of various species, and negatively impacting biodiversity in the campos rupestres (Alves et al. 2014; Fernandes 2016; Fernandes et al. 2018).
References
Almeida AM, Fonseca CR, Prado PI, Almeida-Neto M, Diniz S, Kubota U, Braun MR, Raimundo RLG, Anjos LA, Mendonça TG, Futada SM, Lewinsohn TM (2006) Assemblages of endophagous insects on Asteraceae in São Paulo cerrados. Neotrop Entomol 35:1–22. https://doi.org/10.1590/2175-7860202374021
Almeida-Neto M, Kubota U, Braun MR, Lewinsohn TM (2010) The impact of flower head endophages on seed set of a native population of Chromolaena odorata (L.) King & Robinson (Asteraceae). Bioikos 24:105–112
Alves RJV, Silva NG, Oliveira JA, Medeiros D (2014) Circumscribing campo rupestre – megadiverse Brazilian rocky montane savannas. Braz J Biol 74:355–362. https://doi.org/10.1590/1519-6984.23212
Amorim MD, Costa DS, Krahl DRP, Fischer E, Rech AR (2021) Gongylolepis martiana, an Asteraceae pollinated by bats in the Amazon. Plant Biol 23:728–734. https://doi.org/10.1111/plb.13283
Bergamini LL, Lewinsohn TM, Jorge LR, Almeida-Neto M (2017) Manifold influences of phylogenetic structure on a plant–herbivore network. Oikos 126:703–712. https://doi.org/10.1111/oik.03567
Biesmeijer JC, Roberts SPM, Reemer M, Ohlemüller R, Edwards M, Peeters T, Schaffers AP, Potts SG, Kleukers R, Thomas CD, Settele J, Kunin WE (2006) Parallel declines in pollinators and insect-pollinated plants in Britain and the Netherlands. Science 313:351–354. https://doi.org/10.1126/science.112786
Boaventura MG, Villamil N, Teixido AL, Tito R, Vasconcelos HL, Silveira FAO, Cornelissen T (2022) Revisiting florivory: an integrative review and global patterns of a neglected interaction. New Phytol 233:132–144. https://doi.org/10.1111/nph.17670
Brito VLG, Rech AR, Ollerton J, Sazima M (2017) Nectar production, reproductive success and the evolution of generalised pollination within a specialised pollen-rewarding plant family: a case study using Miconia theizans. Plant Syst Evol 303:709–718. https://doi.org/10.1007/s00606-017-1405-z
Bronstein JL (2009) The evolution of facilitation and mutualism. J Ecol 97:1160–1170. https://doi.org/10.1111/j.1365-2745.2009.01566.x
Bronstein JL, Wilson WG, Morris WF (2003) Ecological dynamics of mutualist/antagonist communities. Am Nat 162:S24-39. https://doi.org/10.1086/378645
Chamberlain SA, Bronstein JL, Rudgers JA, Etienne R (2014) How context dependent are species interactions? Ecol Lett 17:881–890. https://doi.org/10.1111/ele.12279
Cosmo LG, Assis AP, de Aguiar MA, Pires MM, Valido A, Jordano P, Thompson JN, Bascompte J, Guimarães PR Jr (2023) Indirect effects shape species fitness in coevolved mutualistic networks. Nature 619:788–792. https://doi.org/10.1038/s41586-023-06319-7
Feitosa SS, Davide AC, Tonetti OAO, Fabricante JR, Lui JJ (2009) Estudos de viabilidade de sementes de candeia Eremanthus erythropappus (DC.) MacLeish por meio de testes de germinação e raios X. Floresta 39:393–399. https://doi.org/10.5380/rf.v39i2.14565
Fernandes GW, Barbosa NPU, Alberton B, Barbieri A, Dirzo R, Goulart F, Guerra TJ, Morellato LPC, Solar RRC (2018) The deadly route to collapse and the uncertain fate of Brazilian rupestrian grasslands. Biodivers Conserv 27:2587–2603. https://doi.org/10.1007/s10531-018-1556-4
Fernandes GW (2016) The shady future of the rupestrian grassland: major threats to conservation and challenges in the Anthropocene. In: Fernandes G (ed) Ecology and conservation of mountaintop grasslands in Brazil. Springer, Cham, pp 545–561. https://doi.org/10.1007/978-3-319-29808-5_23
Flora e Funga do Brasil (2024) Asteraceae. Jardim Botânico do Rio de Janeiro. Disponível em: https://floradobrasil.jbrj.gov.br/FB55. Acesso em: 24 fev. 2024.
Fontaine C, Guimaraes PR Jr, Kefi S, Loeuille N, Memmott J, Van Der Putten WH, Van Veen FJF, Thebault E (2011) The ecological and evolutionary implications of merging different types of networks. Ecol Lett 14:1170–1181. https://doi.org/10.1111/j.1461-0248.2011.01688.x
Gurevich Y, Hadany L (2021) Floral complexity can help maintain plant diversity by inducing pollinator specialization. J Ecol 109:2897–2908. https://doi.org/10.1111/1365-2745.13690
Herlihy CR, Eckert CG (2002) Genetic cost of reproductive assurance in a self-fertilizing plant. Nature 416:320–323. https://doi.org/10.1038/416320a
Huang L, Liu Y, Dou L, Pan S, Li Z, Zhang J, Li J (2022) Mutualist- and antagonist-mediated selection contribute to trait diversification of flowers. PeerJ. https://doi.org/10.7717/peerj.14107
Jesus FF, Solferini VN, Semir J, Prado PI (2001) Local genetic differentiation in Proteopsis argentea (Asteraceae), a perennial herb endemic in Brazil. Plant Syst Evol 226:59–68. https://doi.org/10.1007/s006060170073
Johnson SD, Nicolson SW (2008) Evolutionary associations between nectar properties and specificity in bird pollination systems. Biol Lett 4:49–52. https://doi.org/10.1098/rsbl.2007.0496
Kivelä M, Arenas A, Barthelemy M, Gleeson JP, Moreno Y, Porter MA (2014) Multilayer networks. J Complex Netw 2:203–271. https://doi.org/10.1093/comnet/cnu016
Lewinsohn TM, Almeida-Neto M, Almeida AM, Prado PI, Jorge LR (2022) From insect-plant interactions to ecological networks. Biota Neotrop 22:1–15. https://doi.org/10.1590/1676-0611-BN-2022-1399
Loeuille BFP (2024) Proteopsis in Flora e Funga do Brasil. Jardim Botânico do Rio de Janeiro. Disponível em: https://floradobrasil.jbrj.gov.br/FB25250. Acesso em: 24 fev. 2024
Martinelli G, Moraes MA (2013) Livro vermelho da flora do Brasil. Instituto de Pesquisas Jardim Botânico do Rio de Janeiro, Rio de Janeiro
Ministério do Meio Ambiente (2022) Lista Nacional de Espécies Ameaçadas de Extinção. Disponível em: https://www.icmbio.gov.br/cepsul/destaques-e-eventos/704-atualizacao-da-lista-oficial-das-especies-ameacadas-de-extincao.html. Acesso em: 28 jun. 2023
Monteiro BL, Camargo MGG, Loiola PP, Carstensen DW, Gustafsson S, Morellato PLC (2021) Pollination in the campo rupestre: a test of hypothesis for an ancient tropical mountain vegetation. Biol J Linn Soc 133:512–530. https://doi.org/10.1093/biolinnean/blaa205
Morrant DS, Schumann R, Petit S (2009) Field methods for sampling and storing nectar from flowers with low nectar volumes. Ann Bot 103:533–542. https://doi.org/10.1093/aob/mcn241
Morris WF, Hufbauer RA, Agrawal AA, Bever JD, Borowicz VA, Gilbert GS, Maron JL, Mitchell CE, Parker IM, Power AG, Torchin ME, Vazquez DP (2007) Direct and interactive effects of enemies and mutualists on plant performance: a meta-analysis. Ecology 88:1021–1029. https://doi.org/10.1890/06-0442
Nascimento AR, Almeida-Neto M, Almeida AM, Fonseca CR, Lewinsohn TM, Penteado-Dias AM (2014) Parasitoid wasps in flower heads of Asteraceae in the Brazilian Cerrado: taxonomical composition and determinants of diversity. Neotrop Entomol 43:298–306. https://doi.org/10.1007/s13744-014-0218-9
Nunes CEP, Maruyama PK, Azevedo-Silva M, Sazima M (2018) Parasitoids turn herbivores into mutualists in a nursery system involving active pollination. Curr Biol 28:980–986. https://doi.org/10.1016/j.cub.2018.02.013
Ollerton J, Winfree R, Tarrant S (2011) How many flowering plants are pollinated by animals? Oikos 120:321–326. https://doi.org/10.1111/j.1600-0706.2010.18644.x
Papiorek S, Junker RR, Alves-dos-Santos I, Melo GAR, Amaral-Neto LP, Sazima M, Wolowski M, Freitas L, Lunau K (2015) Bees, birds and yellow flowers: pollinator-dependent convergent evolution of UV patterns. Plant Biol 18:46–55. https://doi.org/10.1111/plb.12322
Perre P, Loyola RD, Lewinsohn TM, Almeida-Neto M (2011) Insects on urban plants: contrasting the flower head feeding assemblages on native and exotic hosts. Urban Ecosyst 14:711–722. https://doi.org/10.1007/s11252-011-0179-2
Pilosof S, Porter MA, Pascual M, Kéfi S (2017) The multilayer nature of ecological networks. Nat Ecol Evol 1:1–9. https://doi.org/10.1038/s41559-017-0101
Pinheiro M, Gaglianone MC, Nunes CEP, Sigrist MR, Santos IA (2014) Polinização por abelhas. In: Rech ARR, Agostini K, Oliveira PE, Machado IC (eds) Biologia da polinização, 1ª ed. Editora Projeto Cultural, pp 205–233.
Pyke GH (2016) Floral nectar: pollinator attraction or manipulation? Trends Ecol Evol 31:339–341. https://doi.org/10.1016/j.tree.2016.02.013
Pyke GH, Ren ZX (2023) Floral nectar production: what cost to a plant? Biol Rev 98:2078–2090. https://doi.org/10.1111/brv.12997
Rech AR, Brito VLG (2012) Mutualismos extremos de polinização: história natural e tendências evolutivas. Oecol Australis 16:297–310. https://doi.org/10.4257/oeco.2012.1602.08
Rech AR, Jorge LR, Ollerton J, Sazima M (2018) Pollinator availability, mating system and variation in flower morphology in a tropical savanna tree. Acta Bot Bras 32:462–472. https://doi.org/10.1590/0102-33062018abb0220
Romero GQ, Vasconcellos Neto J (2005) Flowering phenology, seed set and arthropod guilds in Trichogoniopsis adenantha (DC) (Asteraceae) in south-east Brazil. Rev Bras Bot 28:171–178
Sasidharan R, Junker RR, Eilers EJ, Müller C (2023) Floral volatiles evoke partially similar responses in both florivores and pollinators and are correlated with non-volatile reward chemicals. Ann Bot 132:1–14. https://doi.org/10.1093/aob/mcad064
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9:671–675. https://doi.org/10.1038/nmeth.2089
Shuttleworth A, Johnson SD (2009) The importance of scent and nectar filters in a specialized wasp-pollination system. Funct Ecol 23:931–940. https://doi.org/10.1111/j.1365-2435.2009.01573.x
Siepielski AM, Benkman CW (2010) Conflicting selection from an antagonist and a mutualist enhances phenotypic variation in a plant. Evolution 64:1120–1128. https://doi.org/10.1111/j.1558-5646.2009.00867.x
Silveira FAO, Negreiros D, Barbosa NPU, Buisson E, Carmo FF, Carstensen DW, Conceição AA, Cornelissen TG, Echternacht L, Fernandes GW, Garcia QS, Guerra TJ, Jacobi CM, Lemos-Filho JP, Le Stradic S, Morellato LPC, Neves FS, Oliveira RS, Schaefer CE, Viana PL, Lambers H (2016) Ecology and evolution of plant diversity in the endangered campo rupestre: a neglected conservation priority. Plant Soil 403:129–152. https://doi.org/10.1007/s11104-015-2637-8
Theis N, Lerdau M, Raguso RA (2007) The challenge of attracting pollinators while evading floral herbivores: Patterns of fragrance emission in Cirsium arvense and Cirsium repandum (Asteraceae). Int J Plant Sci 168:587–601. https://doi.org/10.1086/513481
Van der Kooi CJ, Vallejo-Marín M, Leonhardt SD (2021) Mutualisms and (a)symmetry in Plant-Pollinator Interactions. Curr Biol 31:91–99. https://doi.org/10.1016/j.cub.2020.11.020
Zhang W, Jiang Y, Chen F, Guan Z, Wei G, Chen X, Zhang C, Köllner TG, Chen S, Chen F, Chen F (2022) Dynamic regulation of volatile terpenoid production and emission from Chrysanthemum morifolium capitula. Plant Physiol Biochem 182:11–21. https://doi.org/10.1016/j.plaphy.2022.03.039
Acknowledgements
Fundação de Amparo à Pesquisa do Estado de Minas Gerais—FAPEMIG (APQ-00932-21, APQ-03100-21, RED-00253-16, APQ-02806-22, APQ-03364-21, APQ-01151-22, APQ-01822-21), Conselho Nacional de Desenvolvimento Científico e Tecnológico—CNPq (311665/2022-5, 400904/2019-5, 423939/2021-1), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—CAPES (Finantial Code 001, 88887.837988/2023-00), Fundação de Amparo à Pesquisa do Estado de São Paulo—FAPESP nº 2021/09247-5.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by JPRB, IAOF, DNS and CSN. The first draft of the manuscript was written by JPRB. ACPM, GJB, LRJ and ARR reviewed and commented on previous versions of the manuscript. All authors read and approved the final manuscript. The authors have no confict of interest to declare.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Borges, J.P.R., Ferreira, I.A.O., Silva, D.N. et al. Unveiling the reproduction of Proteopsis argentea (Asteraceae): how the complex pollination system might influence its conservation status. Braz. J. Bot (2024). https://doi.org/10.1007/s40415-024-01025-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40415-024-01025-8