Abstract
The monitoring of wetlands is essential as they provide significant ecosystem services to our biosphere. Diatoms are widely distributed, species rich community and respond quickly to changes in water quality parameters, and may be used for monitoring of water quality of any aquatic ecosystem. There is a scarcity of available information on diatom community structure of wetland of North India. Therefore, this study is first report, to determine the effect of physico–chemical parameters on diatom community composition of Ropar wetland of Punjab (India). In this study benthic, epiphytic, planktonic and epilithic diatom samples were collected from eight selected sites (RW1–RW8) from littoral zone and were analyzed according to standard methodologies. Principal component analysis (PCA) and canonical correspondence analysis (CCA) were employed to determine the response of diatom community structure to spatial change in physico–chemical parameters of study area. A total of 63 diatom taxa representing 39 genera, 25 families and 14 orders were identified from which 58 taxa are reported for first time from Punjab state of India. Results revealed that this wetland had lower diatom diversity with Navicula, Cymbella, Gomphonema and Nitzschia being the dominant genera. Further, PCA and CCA analysis revealed that total dissolved salts, total hardness, phosphate and nitrate are key physico–chemical parameters driving the diatom community structure of the study area. The information obtained from our study will be helpful to understand the ecological conditions of this wetland ecosystem and to develop the conservation and management strategies for similar habitats.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Wetlands are one of the important aquatic ecosystems, which not only play a crucial role in the survival of macro and microorganisms but also govern their productivity (Lawson 2011; Batanero et al. 2022). It also renders useful services to the human beings in the form of many natural resources by ultimately catering their water, food and other survival requirements. Being an important source of freshwater, these are quite vulnerable to the various external factors affecting water quality viz. industrial effluents, urbanization, agricultural runoff, domestic sewage, anthropogenic pressure etc. (Erwin 2009; Kock et al. 2019). The quality of water depends on its physical, chemical and biological properties. Even a slight change in these properties of water caused by external influencing factors, can have an adverse impact on the biodiversity as a whole (Li et al. 2010; Akhter and Brraich 2020a). Although, there are many biomonitoring methods for assessment of water quality are in practice, but diatoms-based monitoring has recognized as one of the important water qualities defining parameters around the world (Pandey et al. 2017; Kock et al. 2019).
Diatoms are unicellular and microscopic organisms having cell walls composed of silica, and are important primary producers of aquatic ecosystems (Antonelli et al. 2017; Kumar and Singh 2017). As one of the most abundant and diverse photosynthetic groups, the net primary production of these organisms is higher than all the worlds’ tropical rainforests (Joseph and Francis 2019). These organisms play an important role in geochemical cycling of naturally occurring elements particularly in carbon and silicon at various suitable habitats (Benoiston et al. 2017). Due to long persistence of their siliceous cell walls they are favoured to be use in paleolimnological or archaeological studies (Morin et al. 2020). The occurrence of diatom species mainly depends on environmental conditions and water chemistry of a particular habitat (Muñoz-López and Rivera-Rondón 2022). Globally, these organisms are recognized as useful biological indicator for the assessment of water quality, due to their quick response to physico-chemical changes of the habitat (Kock et al. 2019; Mbao et al. 2022). The diatom community structure is mainly influenced by range of factors like physicochemistry (pH, EC, TDS, phosphate, TH), habitat type, inflow of different pollution sources etc. (Michelutti et al. 2013; Soeprobowati et al. 2016). The diatom communities provide holistic and integrated approach towards the integrity of lotic and lentic ecosystems due to these responses (Dalu et al. 2016; Shibabaw et al. 2021).
Currently, due to the insufficient knowledge about the diatom diversity from Indian water bodies these organisms are generally ignored in environmental monitoring programme (Murulidhar and Murthy 2014). According to the available literature, aquatic habitats of central and southern regions of India has been explored by various workers (Verma and Nautiyal 2010, 2016; Karthick 2010; Karthick et al. 2013; Thacker and Karthick 2022), but there is a general paucity of information on diatom diversity from various aquatic habitats including wetlands of Punjab and other states of North India. Although, the water quality conditions and phytoplankton diversity of wetlands of Punjab has been explored by various workers (Sharma et al. 2013, 2017; Brraich and Kaur 2015; Brraich and Saini 2015; Singh et al. 2020, 2022; Brraich et al. 2021; Komal et al. 2021). But, to the best of our knowledge only few reports are available in literature on diatoms diversity from water bodies of Punjab (Thakar and Singh 2010; Pareek et al. 2018; Sharma et al. 2019).
Punjab has around 40 important wetlands, covering an area of 76,476 ha, out of which six wetlands namely Beas Conservation Reserve, Harike, Kanjli, Keshopur Miani, Nangal and Ropar are recognized as Ramsar sites. Ropar wetland is not only an important aquatic ecosystem with profuse biodiversity and ecological properties but also, one of the significant recreational, socio-economic and scientific development centers (Akhter and Brraich 2020a; Singh et al. 2022). The partial water sampling and monitoring of this important wetland is of a concern as it is threatened by the industrial expansion, indiscriminate use of agricultural land, human interferences and recreational activities (Brraich et al. 2021; Komal et al. 2021). The present study has been carried out with the objective to investigate and document the diversity of diatoms as well as their response to the spatial variation in selected physico-chemical parameters of Ropar wetland of Punjab.
2 Materials and methods
Study area
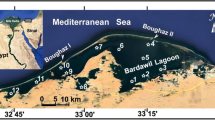
– Punjab is one of the agrarian states of India with about 84% land in under cultivation and has nearly 40 important wetlands covering nearly 1% of the total geographical area (Ladhar 2002; Kumar and Singh 2020; Singh et al. 2022). These wetlands play an important role as source of irrigation, power generation and as suitable habitats for number of plants and animals. Six wetlands (Beas Conservation Reserve, Harike, Kanjli, Keshopur-Miani, Nangal and Ropar) are having the tag of Ramsar site and other are declared as the wetlands of national importance. The Ropar wetland, is manmade fresh water riverine and lacustrine wetland, located at 31° 01ʹ North and 76° 30ʹ East (Fig. 1) on Sutlej River at an elevation of 275 m above sea level. Climatically this area falls under semi-arid zone of Punjab with mean annual rainfall of 1518 mm (Brraich and Akhter 2019). It came into existence in 1952 with the construction of water headwork on Sutlej River to supply and divert water for irrigation through canals, industrial and drinking water supply (Ladhar 2002). This wetland covers an area of 1365 ha including 800 ha area under water (ENVIS 2015). The evergreen forest and marshy area of this wetland are important and vital habitats for the migratory birds. Ropar wetland is recognized as Ramsar site in the year 2002, as it supports habitat for a number of threatened and endangered species (Ladhar 2005).
Water sampling and physico-chemical analysis
– Water samples (n = 24, three from each site) were collected in clean and sterilized plastic bottles of five-liter capacity from eight selected sites (RW1–RW8) during October, 2020 from Ropar wetland (Ramsar site) of Punjab (India). A total of seventeen parameters were analyzed to study the physicochemical properties of this wetland. The water temperature (WT, ℃) (Perfit), pH (Hanna Instruments, India), electrical conductivity (EC, µS/cm) (Hanna Instruments, India) and total dissolve salts (TDS, mg/l) (HM digital, Inc., CA, USA) were noted down on the spot at the time of sampling. Whereas, rest of the parameters including carbonate (CO3, mg/l), bicarbonate (HCO3, mg/l), total alkalinity (TA, mg/l), total hardness (TH, mg/l), calcium (Ca, mg/l), magnesium (Mg, mg/l), chloride (Cl, mg/l), phosphate (PO4, mg/l), ammonium (NH4, mg/l), sulphate (SO4, mg/l), nitrate (NO3, mg/l), sodium (Na, mg/l), potassium (K, mg/l) were analyzed within the 24 h of sampling by following standard procedure (APHA 2012).
Sampling and identification of diatoms
– To study the relationship between diatom and physico-chemical parameters of Ropar wetland, a total of 32 samples were collected from eight selected sampling sites (RW1–RW8) according to different microhabitats of diatoms. As Ropar wetland is a Ramsar site, maximum care was taken before sampling. Diatom samples were collected not only from point of view of taxonomists but also as perception of ecologist’s without disturbing the other biota of wetland ecosystem. The benthic, epilithic, epiphytic and planktonic diatom samples were collected separately from each sampling site. The benthic, epilithic and epiphytic samples were collected with the help of pipette/dropper/forceps/knife etc., whereas concentrated 100 ml planktonic samples were collected with plankton net (20 μm) by filtering 25 L water. All the collected samples were transferred to sterilized glass bottle (100 ml) containing the preservative solution (4% formaldehyde) and brought to the laboratory for further analysis. The organic matter present in the samples was digested by boiling the samples with concentrated nitric acid. The cleaned samples were then transferred to centrifuge tubes and centrifuged at 4000 rpm for 10 min and rinsed five times with distilled water to remove the excess acid; subsequently a drop of diatom samples from a pipette was put on a cover slip and allowed to dry overnight at room temperature in a clean chamber (Karthick et al. 2013). Permanent slides were prepared using Naphrax and light microscopy (Leica) observations were done at 10X, 40X, 100X combination of lenses using immersion oil. Plates of microphotographs were prepared using Inkscape 0.92.3 software. Identification of diatom taxa was done on the basis of their morphological characteristics by following Karthick (2010), Karthick et al. (2013) and Cantonati et al. (2017). The relative abundance of diatom taxa was determined by using drop count method (Trivedy and Goel1984), whereas evenness was measured on the basis of presence and absence of each identified organism in each collected sample (Bak et al. 2004).
Statistical analysis
– To illustrate the variation in parameters, descriptive statistics, viz. mean and standard deviation were calculated. One-way analysis of variance (ANOVA) was applied to find out the significant differences in the water chemistry among different sampling sites. Principal Component Analysis (PCA) was used to find out the spatial pattern of water quality parameters and relationships among sampling site on the basis their water chemistry by using log (x + 1) transformed data. Canonical Correspondence Analysis (CCA) was employed on the basis of calculated relative abundance to find out the influence of different physico-chemical parameters on the distribution of diatoms. The diatoms taxa of having > 50% relative abundance (RA) were taken in consideration for CCA. The software used for statistical analysis includes Microsoft Office Excel 2010, SPSS statistics 18 and XLSTAT version 2020.5.
3 Results
Spatial variation in water chemistry
– During this study, a total of 17 physico-chemical parameters of water were analyzed from eight selected sampling sites (RW1–RW8) to understand the spatial variation in water chemistry of Ropar wetland. The variations (mean, standard deviation and range) in the various physico-chemical parameters have been presented in Table 1. All the physico-chemical parameters showed significant variations among sampling sites (p < 0.05) (Table S1). At time of sampling, the water temperature of Ropar wetland was ranged from 14.0 ± 0.20 ℃ (RW1) to 16.02 ± 0.20 ℃ (RW6). The water of Ropar wetland was alkaline in nature and showed slight variations in different sites with a range of 7.80 ± 0.05 (RW2) to 8.20 ± 0.20 (RW8). Significant variation was observed in the values of electrical conductivity (EC) among different sampling sites. The maximum value (339.56 ± 19.68 µS cm−1) of EC was recorded from RW8, whereas minimum value (257.12 ± 3.17 µS cm−1) was observed from RW4 (Table 1).
– The amount of TDS in all sampling sites of study area was ranged in between 200.00 ± 2.00 mg l−1 (RW5) to 279.00 ± 3.00 mg l−1 (RW8). The concentration of carbonate in water of all sampling sites was ranged from 22.49 ± 0.62 mg l−1 (RW4) to 32.36 ± 1.52 mg l−1 (RW8), whereas the values of bicarbonate were ranged in between 97.35 ± 3.79 mg l−1 (RW1) to 131.87 ± 1.05 mg l−1 (RW8). The values of total alkalinity showed significant variations (p < 0.05) among different sampling sites and varied from 25.37 ± 1.11 mg l−1 at site RW4 to 36.85 ± 1.09 mg l−1 at site RW8. In present study the maximum value (142.49 ± 0.58 mg l−1) of total hardness was recorded from sampling site RW8, whereas minimum (119.97 ± 1.00 mg l−1) from site RW2. The chloride content in water of study area was varied from 23.30 ± 0.12 mg l−1 (RW5) to 29.14 ± 0.06 mg l−1 (RW8). The concentration of sulphate was ranged from 0.61 ± 0.007 mg l−1 (RW5) to 0.91 ± 0.01 mg l−1 (RW8) in water all sampling sites of study area. The maximum phosphate (0.22 ± 0.008 mg l−1) and nitrate (0.36 ± 0.01 mg l−1) content was estimated from the water samples collected from site RW8, whereas minimum phosphate (0.06 ± 0.006 mg l−1) and nitrate (0.18 ± 0.02 mg l−1) from site RW5 (Table 1).
Principal component analysis (PCA)
– PCA analysis revealed that the first two principal components (PCs) explained more than 76.69% of the total variance in analyzed physico-chemical parameters (Table 2; Fig. 2). PC1 had high correlation with EC (0.94), TDS (0.80), PO4, (0.93), NO3 (0.94), SO4 (0.94), Mg (0.90), NH4 (0.80), TA (0.82), Cl (0.73), while TH (0.93), HCO3 (0.93), Ca (0.89), WT (0.79) and pH (0.64) were highly correlated with PC2. The PC1 represents the parameters which would correspond to natural weathering of rocks from the surrounding area and waste water discharges to the wetland. PCA identified three groups on the basis of individual physico-chemical parameters. Group I include two sites RW1 and RW8, were distinguished from others by their relatively higher values of EC, TA and TDS and PO4, NO3, SO4, Mg, NH4, and Cl content. Group II includes another two sites RW2 and RW6, may be grouped together based on their similar sodium and potassium content. Site RW7 may be distinct from rest of the sites and represent third PCA group due to relatively higher values of pH and carbonate and calcium content. While sites RW3, RW4 and RW5 were independent due to intermediate concentration of the water quality parameters.
Diversity of diatom in Ropar wetland
– A total of 63 diatom taxa belonging to 39 genera, 25 families of 14 orders were identified on the basis of their morphological characteristics from study area (Table 3; Fig. 3a–c). In this wetland, most of the reported diatom taxa were benthic (39) followed by epiphytic (12), planktonic (09), and epilithic (03) in nature. The diatom community was mainly dominated by biraphid diatoms (viz. Navicula, Nitzschia, Gomphonema, Cymbella, Tryblionella etc.), followed by araphids (viz. Ulnaria, Diatoma, Staurosira and Staurosirella) and centric (Cyclotella, Melosira). In terms of number of species, the genus Navicula with 05 species dominates this wetland followed by the Cymbella, Nitzschia and Gomphonema with 4 species each. The highest relative abundance was achieved by Cocconeis pediculus (Ehrenberg) 3.19% which had evenness value of 53.12% followed by Cocconeis placentula ‘Ehrenberg’ in Murobuse (RA 2.64%, evenness 56.25%), Cyclotella meneghiniana Kützing (RA 2.64%, evenness 78.12%), Craticula ambigua (Ehrenberg) (RA 2.21%, evenness 28.12%), Frustulia vulgaris (Thwaites) De Toni (RA 2.14%, evenness 37.5%) (Table 3). The least dominant taxa were Planothidium biporomum (M.H. Hohn & Hellerman) Lange-Bertalot (RA 0.90%, evenness 28.12%), Navicula witkowskii (Lange-Bertalot, Iserentant & Metzeltin) (RA 0.93%, evenness 53.12%), Entomoneis paludosa (W. Smith) Reimerss (RA 0.97%, evenness 12.5%), Nitzschia solgensis Cleve-Euler 34.37 (RA 1.09%, evenness 34.37%) (Table 3). The diatom species that are found to be frequent in over 50% of the sampling sites includes taxa like Navicula rostellata (Kützing), Navicula capitatoradiata (Germain), Navicula erifuga (Lange-Bertalot), Sellaphora pseudopupula (Krasske) Lange-Bertalot, Sellaphora mutatoides (Lange-Bertalot & Metzeltin), Diadesmis confervacea (Kützing), Diploneis calcilacustris (Lange-Bertalot & A. Fuhrmann), Staurosirella pinnata (Ehrenberg) D. M. Williams & Round, Cymbella parva (W. Smith) Kirchner (Table 3).
a Micro-photograph of diatom species identified from study area. (a): Navicula rostellata; (b): Navicula capitatoradiata; (c): Navicula erifuga; (d): Navicula witkowskii; (e): Navicula viridula; (f): Caloneis aequatorialis; (g): Caloneis silicula; (h): Fallacia pygmea; (i): Sellaphora pseudopupula; (j): Sellaphora mutatoides; (k): Frustulia vulgaris; (l): Halamphora coffeiformis; (m): Halamphora montana; (n): Diadesmis confervacea; (o): Luticola saprophila; (p): Gyrosigma obtusatum; (q): Craticula cuspidata; (r): Craticula ambigua; (s): Pleurosigma salinarum; (t): Pinnularia amabilis; (u): Pinnularia neomajor; (v): Diploneis calcilacustris. (Scale = 10 µm). b Micro-photograph of diatom species identified from study area. (a): Ulnaria ulna: (b): Ulnaria ulna var. repanda; (c): Staurosira construens; (d): Staurosirella pinnata; (e): Iconella angusta; (f): Iconella capronii; (g): Surirella librile; (h): Entomoneis paludosa; (i): Cymbella tumida; (j): Cymbella hustedtii; (k): Cymbella neoleptoceros; (l): Cymbella parva; (m): Cymbopleura frequens; (n): Gomphonema olivaceum; (o): Gomphonema balticum; (p): Gomphonema affine; (q): Gomphonema clavatum; (r): Encyonema lacustre; (s): Placoneis clementispronina; (t): Nitzschia obtusa; (u): Nitzschia solgensis; (v): Nitzschia tabellaria; (w): Nitzschia clausii; (x): Hantzschia calcifuga. (Scale = 10 µm). c Micro-photograph of diatom species identified from study area. (a): Hantzschia amphioxys; (b): Tryblionella constricta; (c): Tryblionella hungarica; (d): Diatoma vulgaris; (e): Cyclotella meneghiniana; (f): Cyclotella ocellata; (g): Spicaticribra kodaikanaliana; (h): Amphora copulata; (i): Aulacoseira granulata; (j): Epithemia gibba; (k): Achnanthes inflata; (l): Cocconeis placentula; (m): Cocconeis pediculus; (n): Lemnicola hungarica; (o): Planothidium biporomum; (p): Melosira varians; (q): Pleurosira laevis. (Scale = 10 µm)
Effect of spatial variation in water chemistry on diatom diversity
– Canonical correspondence analysis (CCA) revealed that the spatial variation in water chemistry of the study area influence the diatom community structure. The first two axes (Axis 1 & 2) of CCA biplot (λ1 = 0.19, λ2 = 0.15) explained 56.11% of the total variance (Table 4, Fig. 4). Axis 1 is negatively correlated to pH (−0.04) and sites include RW1, RW2, RW4-RW6 while positively correlated with TDS (0.14), WT (0.15), PO4 (0.06), NO3 (0.07) Na (0.38) and sites viz. RW3, RW7 and RW8. Diatom taxa including Surirella capronioides (0.95), Hantzschia amphioxys (0.95), Aulacoseira granulata (0.68), Sellaphora pseudopupula (0.68), Halamphora coffeiformis (0.51), Ulnaria ulna var. repanda (0.49), Navicula rostellata (0.46) showed positive correlation with the axis 1, whereas Fallacia pygmea (−0.99), Cyclotella ocellata (−0.77), Tryblionella hungarica (−0.76), Achnanthes inflata (−0.71) etc. showed negative correlation. However, axis 2 showed positive loadings with pH (0.10), NO3 (0.007), sites such as RW1, RW3, RW5 and RW7, whereas had negative loadings with TDS (−0.10), WT (−0.08), Na (−0.08), PO4 (−0.03), sites RW2, RW4, RW6 and RW8. The highly positive correlated taxa on the same axis were Halamphora montana (0.48), Pinnularia amabilis (0.47), Tryblionella constricta (0.41), Navicula rostellata (0.40), Craticula ambigua (0.40), Navicula capitatoradiata (0.40) etc. while some of the taxa revealed highly negative correlation on axis 2 including Placoneis clementispronina (−0.98), Caloneis silicula (−0.73) and Gomphonema clavatum (−0.61) etc.
CCA biplot showing relationship between physico–chemical parameters and occurrence of diatom taxa at different sampling sites. RW1: Roper wetland site 1, RW2: Roper wetland site 2, RW3: Roper wetland site 3, RW4: Roper wetland site 4, RW5: Roper wetland site 5, RW6: Roper wetland site 6, RW7: Roper wetland site 7, RW8: Roper wetland site 8. Aci: Achnanthes inflata, Amc: Amphora copulata, Aug: Aulacoseira granulata, Caa: Caloneis aequatorialis, Cas: Caloneis silicula, Cop: Cocconeis placentula, Cope: Cocconeis pediculus, Cra: Craticula ambigua, Crc: Craticula cuspidata, Cyh: Cymbella hustedtii, Cym: Cyclotella meneghiniana, Cyo: Cyclotella ocellata, Cyt: Cymbella tumida, Div: Diatoma vulgaris, Fap: Fallacia pygmea, Frv: Frustulia vulgaris, Goa: Gomphonema affine, Gocc: Gomphonema clavatum, Goo: Gomphonema olivaceum, Gyo: Gyrosigma obtusatum, Haa: Hantzschia amphioxys, Hac: Halamphora coffeiformis, Ham: Halamphora montana, Mev: Melosira varians, Nac: Navicula capitatoradiata, Nae: Navicula erifuga, Nar: Navicula rostellata, Nav: Navicula viridula, Pia: Pinnularia amabilis, Plc: Placoneis clementispronina, Plle: Pleurosira laevis, Pls: Pleurosigma salinarum, Sep: Sellaphora pseudopupula, Spk: Spicaticribra kodaikanaliana, Suc: Surirella capronioides, Sul: Cymatopleura solea, Trc: Tryblionella constricta, Trh: Tryblionella hungarica, Ulu: Ulnaria ulna, Ulur: Ulnaria ulna var. repanda. NO3: nitrate, PO4: phosphate, TDS: total dissolved salts, TH: total hardness, K: potassium
4 Discussion
The wetlands are generally considered as sink for pollutant and have potential to purify water but at the same time they are susceptible to nutrient enrich environment. The present and previous studies on aquatic sampling indicate that Ropar wetland is currently under high natural and anthropogenic pressure (Akhter and Brraich 2020a, b; Brraich et al. 2021; Komal et al. 2021; Singh et al. 2022). Due to this high nutrient load increasing day by day on this wetland may lead to water quality degradation. The diatoms are recognized as bioindicator of water quality, due to their sensitivity and quick response to change in water chemistry of an aquatic ecosystem. Many diatom species are known to grow well in high nutrient concentration and their presence in water indicates degraded water quality. Few diatom species including Navicula rostellata, Melosira varians, Cyclotella meneghiniana, Gomphonema olivaceum and Entomoneis paludosa reported from around the world from nutrient rich eutrophic water were also recorded in current study from Roper wetland.
In present study, the diatoms and water samples were collected from selected sites of Ropar wetland to investigate the diversity of diatoms as well as their response to the spatial variation in water chemistry. The physico-chemical analysis of selected parameters revealed significant spatial variation in water chemistry of Ropar wetland (Table 1). The water of Ropar wetland was found alkaline in nature with significant variation in pH and water temperature of selected sites at time of sample collection. The alkaline nature of water promotes the growth of the phytoplanktons which generally helps to increase in the primary productivity of an aquatic ecosystem (Kumar and Prabhahar 2012; Sharma et al. 2013). However, all the sampling sites showed variation in EC and TDS values. Electrical conductivity and TDS have a direct correlation as more is the EC, greater will be the TDS of water body (Rusydi 2018). The higher values of EC and TDS were observed at sampling site RW8 may be due to accumulation of the dissolved salts, chlorides, sulphate, sodium and potassium from wastewater, agricultural runoff, and anthropogenic activities (household waste, organic matters, agricultural and industrial run off) (Mahananda et al. 2010; Kataria et al. 2011; Reyes-Toscano et al. 2020; Singh et al. 2020). However, the weathering of rocks, suspension of deposited salts, wastewater and agricultural runoff may be responsible for the higher content of chloride. The gypsum, sulphate-containing fertilizers and other minerals in agricultural runoff are the main sources of sulphate in water bodies with agriculture catchment area (Pande and Moharir 2018). Moreover, the high content of nitrate and phosphate in aquatic ecosystems results in eutrophication which favours the luxuriant growth of phytoplankton. The phosphate and nitrate content in water is mainly increased with the addition of untreated sewage, industrial effluent and runoff from adjoining agricultural field in to the water bodies (Singh 2013; Singh and Ahluwalia 2013; Jindal et al. 2015). The overall trend of physico-chemical parameters in the study area revealed that water of sampling site RW8 is most degraded followed by site RW1 which may be due to the high pollution load from natural as well as anthropogenic processes.
Principal component analysis (PCA) has been employed to find out the variation patterns in water chemistry on the basis of analyzed physico-chemical parameters. PCA differentiated the data set into three groups on the basis of spatial variation in water chemistry of selected sites of Ropar wetland (Table 2; Fig. 2). Group I includes sites RW1, RW7 and RW8 with higher content of EC, TA and TDS and PO4, NO3, SO4, Mg, NH4, and Cl may be due to their exposure to pollutants from domestic waste, siltation, fishing and industrial effluents (Kour et al. 2022; Pratiwi et al. 2023). Group II is formed by sampling sites RW2 and RW6 with high content of sodium and potassium. Accumulation of sodium and potassium content into these sites of wetland may due to surface runoff, weathering of rocks, minerals and sewage discharges from adjoining areas into the water (Bhateria and Jain 2016; Akhtar et al. 2021). While rest of the sampling sites (RW3, RW4 and RW5) represents group III which showed somewhat less content of nutrients in comparison to other sites may be due to less anthropogenic activities and entries of fresh waters near these sites which may dilute the concentration of nutrient contents. Different magnitude of anthropogenic pressure on these selected sites may be the reason for their different water quality (Akhtar et al. 2021; Singh et al. 2022; de Almeida et al. 2023). Water quality at these sites may be mainly influences by agricultural runoff, industrial effluents, and discharges through anthropogenic activities from the catchment areas (Singh et al. 2020, 2022; Siriwardhana et al. 2023).
In present study, the morpho-taxonomic analysis of selected diatom samples revealed that the study area exhibit significant of diatom diversity. However, among 63 diatom taxa identified during current study, earlier only five species have been reported from Punjab (Diatoma vulgaris Bory, Cocconeis placentula Ehrenberg, Craticula cuspidata (Kutzing) D.G.Mann, Gomphonema olivaceum (Hornemann) Ehrenberg and Ulnaria ulna (Nitzsch) P. Compère (Thakar and Singh 2010) (Table 3). While analyzing the diversity of phytoplankton in relation to physico-chemical parameters of water or for the forensic purpose’s taxa such as Achnanthes, Ulnaria, Navicula, Gyrosigma, Pinnularia, Nitzschia, Cymbella, Gomphonema, Amphora, Cavinula, Neidium, Cyclotella, Surirella, Melosira, Cocconeis etc. were previously recorded from the different parts of Punjab but only at genus level (Thakar and Singh 2010; Brraich and Kaur 2015; Brraich and Saini 2015; Marbaniang and Paul 2015). Thus, during present study 58 taxa are reported for the first time from Ropar Wetland as well as from Punjab state of India.
In present study, Cymbella, Navicula, Ulnaria, Nitzschia, Cocconeis, Halamphora and Cyclotella were observed as the most frequent taxa, while Amphora and Diatoma as rare diatom taxa from the study area. Similar observations have also been reported by Sharma et al. (2013) from River Sutlej, Punjab state of India. The present investigation also indicated that, biraphid diatoms (viz. Navicula, Nitzschia, Gomphonema, Cymbella, Tryblionella etc.) dominated the study area, which was followed by araphids (viz. Ulnaria, Diatoma, Staurosira and Staurosirella) and rare one was centric diatoms (Cyclotella and Melosira). Biraphids have also been recorded earlier from Himalayan Rivers (Nautiyal et al. 2004; Sharma et al. 2007) and Kumaun region (Verma and Nautiyal 2009) as dominant diatom taxa. Biraphid diatoms (Navicula, Nitzschia, Cymbella and Achnanthidium) were also observed as dominant genera in Western Ghats and peninsular regions of India (Karthick et al. 2013). Nautiyal and Verma (2009) have recorded 293 diatom species from two different biogeographic regions of India, the Himalaya and the Central Highlands (Vindhya region) and observed that the number of diatom species in Vindhya (293 taxa) region was more than the Himalaya (189 taxa). On the basis of number of species, the genus Navicula was found to be the dominant genera with maximum number of species (05) followed by the Cymbella, Nitzschia and Gomphonema with 4 species each (Table 3). In present study, most of the reported diatom taxa were benthic (39) followed by epiphytic (12), planktonic (09), and epilithic (03) in nature. The dominance of benthic forms over planktonic may be due to the flowing nature of water and absence of deep stagnant parts of this wetland. Similar observations have been made by different workers from different parts of India as well as from other countries (Bella et al. 2007; Paul et al. 2016). The Synedra ulna var. repanda which was earlier reported from China (Qingmin et al. 2008) has been reported for the first time from India during this study. Taxon Ulnaria ulna var. repanda is quite similar to Ulnaria ulna in terms of density of striae and frustules, the main characteristic feature which makes it distinct from other species of Ulnaria is the undulate valves having a straight pseudoraphe. The length of the Ulnaria ulna var. repanda was 350.2–371.2 µm with 8–8.12 µm breadth, striae are parallel in the middle with density varies from 8 to 9 µm and radiate in the apices (Fig. 3b).
Canonical correspondence analysis (CCA) has been used to analyze the effect of spatial variation in water chemistry on distribution of diatoms in Ropar Wetland. The resultant CCA biplot showed distribution of diatom taxa, physico-chemical parameters and sampling sites into two groups along both the axes of biplot on the basis different nutrient requirements of different diatom taxa (Fig. 4). Group I include Cocconeis pediculus, Cyclotella meneghiniana, Pleurosigma salinarum, Cymbella tumida, Navicula viridula, Ulnaria ulna, Nitzschia obtusa and Caloneis silicula which shows their close relationships with mesotrophic or nutrient rich conditions (high TDS, TH, NO3 and PO4 content). Kennedy and Buckley (2021) have also reported similar observation from Irish lakes. These diatom taxa represent Group I may be the nutrient tolerant taxa and act as indicator of change in the content of TDS, TH, NO3 and PO4 in water of Ropar wetland and other similar aquatic habitats (Barinova and Mamanazarova 2021). The high contents of TDS, TH, NO3 and PO4 in water of Ropar wetland may attributed to the addition of various salts resultant of untreated discharge of industrial effluent and agricultural runoff etc. (Singh et al. 2017, 2022; Reyes-Toscano et al. 2020). The Group II is represented by the diatom taxa having high abundance in sampling sites with oligotrophic water conditions. From CCA plot, it is evident that the abundance of diatom taxa in Ropar wetland depends on the availability of nutrients. From CCA biplot, it is clear that the majority of dominant species have high affinity to TDS, TH, PO4 and NO3 (Fig. 4). This reveals that these parameters influence the distribution and occurrence of diatoms in this wetland. Kock et al. (2019) also observed the influence of environmental variables on the distribution and occurrence of diatoms in lake ecosystem. The different environmental factors like pH, EC, alkalinity, total hardness, phosphate and nitrate in any aquatic ecosystem play an important role in the distribution of diatom community structure (Dibong and Ndjouondo 2014; Dimowo et al. 2018; Chintapenta et al. 2018; Silva-Lehmkuhl et al. 2019; Maraşlıoğlu et al. 2020).
In present study, Navicula rostellata was abundantly found on the sites RW1, RW7 and RW8 having high nutrient concentration than other selected sampling sites. Melosira varians in case of our study was mainly observed on the sites RW1, RW7 and RW8. Melosira varians has been reported to found in eutrophic water conditions and can be an indicator of high trophic level (Dimowo et al. 2018). According to Lacerda et al. (2004) Cyclotella meneghiniana had known to be significantly correlated with total alkalinity, hardness and phosphate. The abundance of Gomphonema olivaceum increases with the increase in concentration of phosphate and can be act as good indicator of high phosphate content in water (Maraşlıoğlu et al. 2020). The species of genera like Amphora, Cyclotella, Cocconeis, Cymbella, Navicula, Cymatopleura and Nitzschia are known to occur in calcareous and alkaline water (Kivrak and Uygun 2012; Cantonati et al. 2017). Entomoneis paludosa observed during the present study is strictly found in saline water as well as salt-contaminated water. The higher abundance this taxon in water indicates the high salinity in aquatic ecosystem (Cantonati et al. 2017). These taxa have also been commonly recorded from the nutrient rich water with high electrical conductivity and considered as an indicator of oligo-eutrophic status of water. Although, the concentration of physico-chemical parameters measured during present study are lower but the presence of these high nutrient tolerant diatom taxa indicates that this wetland is under the threat of eutrophication.
Our study concludes that water chemistry of Ropar wetland showed spatial variations which also affect the diversity and distribution of diatoms. This wetland harbors unique assemblage of benthic, epiphytic, planktonic and epilithic diatom taxa with varied distributional pattern and occurrence. One of the features of this study is that, among 63 identified diatom taxa, 58 taxa of which are new records for Punjab (India), whereas Ulnaria ulna var. repanda is the first report from India. The CCA depict that the TDS, TH, PO4 and NO3 are the key physico–chemical parameters mainly influencing the occurrence and distribution of diatom taxa in Ropar wetland. This study highlights the unexplored diatom diversity, their relationships with the current physico–chemical condition and provides information, to use diatoms in the biomonitoring studies in similar habitats. Further, recommend that there is an urgent need to develop programs, taking into account the diatoms and all other aquatic communities, for conservation and management of biodiversity of this Ramsar site (Ropar wetland) and all other aquatic ecosystems of this region.
Data availability
All necessary data is included in this article.
References
Akhtar N, Ishak MIS, Bhawani SA, Umar K (2021) Various natural and anthropogenic factors responsible for water quality degradation: a review. Water 13:2660. https://doi.org/10.3390/w13192660
Akhter S, Brraich OS (2020a) Physico-chemical analysis of fresh water of Ropar Wetland (Ramsar site), India. Curr World Environ 15:117–126. https://doi.org/10.12944/CWE.15.1.15
Akhter S, Brraich OS (2020b) Spatial and temporal distribution of phytoplankton from Ropar wetland (Ramsar Site) Punjab, India. Appl Ecol Environ Sci 8:25–33. https://doi.org/10.12691/aees-8-1-4
Antonelli M, Carlos EW, Luc E, Teuling AJ, Laurent P (2017) On the potential for terrestrial diatom communities and diatom indices to identify anthropogenic disturbance in soils. Ecol Indic 75:73–81. https://doi.org/10.1016/j.ecolind.2016.12.003
APHA (2012) Standard methods for the examination of water and wastewater, 22nd edn. American Public Health Association, Washington
Bak M, Witkowski A, Lange-Bertalot H, Dadala A (2004) Ecology of the Szczecin Lagoon diatom flora with reference to the utility of diatom indices in assessing water quality. Diatom 20:23–31. https://doi.org/10.11464/diatom1985.20.0_23
Barinova S, Mamanazarova K (2021) Diatom algae-indicators of water quality in the lower Zarafshan river. Uzbekistan Water 13:358. https://doi.org/10.3390/w13030358
Batanero GL, Green AJ, Amat JA, Vittecoq M, Suttle CA, Reche I (2022) Patterns of microbial abundance and heterotrophic activity along nitrogen and salinity gradients in coastal wetlands. Aquat Sci 84:22. https://doi.org/10.1007/s00027-022-00855-6
Bella VD, Puccinelli C, Marchegiani S, Mancini L (2007) Benthic diatom communities and their relationship to water chemistry in wetlands of Central Italy. Ann Limnol-Int J Lim 43:89–99. https://doi.org/10.1051/limn/2007021
Benoiston AS, Ibarbalz FM, Bittner L, Guidi L, Jahn O, Dutkiewicz S, Bowler C (2017) The evolution of diatoms and their biogeochemical functions. Philos Trans R Soc B 372:20160397. https://doi.org/10.1098/rstb.2016.0397
Bhateria R, Jain D (2016) Water quality assessment of lake water: a review. Sustain Water Resour Manag 2:161–173. https://doi.org/10.1007/s40899-015-0014-7
Brraich OS, Akhter S (2019) Diversity and distribution of zooplankton in Ropar Wetland (Ramsar Site) Punjab, India. Nat Environ Pollut Technol 18:451–458
Brraich OS, Kaur R (2015) Phytoplankton community structure and species diversity of Nangal Wetland, Punjab, India. Int J Biol Sci 4:76–83
Brraich OS, Saini SK (2015) Phytoplankton abundance and species diversity in Ranjit Sagar wetland, Punjab (India). Curr World Environ 10:215–221. https://doi.org/10.12944/CWE.10.1.25
Brraich OS, Kaur N, Akhter S (2021) Assessment of limnological parameters and water quality indices of Harike wetland (Ramsar Site), Punjab (India). Appl Ecol Environ Sci 9:591–598. https://doi.org/10.12691/aees-9-6-3
Cantonati M, Kelly MG, Lange-Bertalot H (2017) Freshwater benthic diatoms of central europe: over 800 common species used in ecological assessment. Koeltz Botanical Books, Schmitten-Oberreifenberg
Chintapenta KL, Coyne KJ, Pappas A, Lee K, Dixon C, Kalavacharla V, Ozbay Z (2018) Diversity of diatom communities in Delaware Tidal wetland and their relationship to water quality. Front Environ Sci 6:1–15. https://doi.org/10.3389/fenvs.2018.00057
Dalu T, Bere T, Froneman PW (2016) Assessment of water quality based on diatom indices in a small temperate river system, Kowie river, South Africa. Water SA 42:183–193. https://doi.org/10.4314/wsa.v42i2.2
de Almeida RGB, Lamparelli MC, Dodds WK, Cunha DGF (2023) Sampling frequency optimization of the water quality monitoring network in São Paulo State (Brazil) towards adaptive monitoring in a developing country. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-023-29998-1
Dibong SD, Ndjouondo P (2014) Floristic inventory and ecology of algae of the Kambo and Longmayagui rivers of the Douala wetland (Cameroon). Int J Biol Chem Sci 8:2560–2577. https://doi.org/10.4314/jab.v80i1.15
Dimowo BO, Gbadebo AM, Taiwo AM, Omoniyi IT (2018) Planktonic diatoms as bio-indicators of ecological integrity of lower Ogun River, Abeokuta, Southwestern, Nigeria. Int J Aquacult 8:1–14
ENVIS (2015) Wetland of Punjab, Punjab ENVIS centre
Erwin K (2009) Wetlands and global climate change: the role of wetland restoration in a changing world. Wetl Ecol Manag 17:71–84. https://doi.org/10.1007/s11273-008-9119-1
Jindal R, Thakur RK, Singh UB, Ahluwalia AS (2015) Phytoplankton dynamics and water quality of Prashar lake, Himachal Pradesh, India. Sustain Water Qual Ecol 3–4:101–113. https://doi.org/10.1016/j.swaqe.2014.12.003
Joseph G, Francis MS (2019) First new record of two diatoms (Caloneis africana (Giffen) Stidolph and Luticola nivalis (Ehrenberg) D. G. Mann) from South West Coast of India (Cochin backwaters). Oceanologia 61:395–399
Karthick B, Hamilton PB, Kociolek JP (2013) An illustrated guide to common diatoms of Peninsular India, Gubbi Labs, 206
Karthick B (2010) Ecology of stream diatom community in Central Western Ghats (Ph.D. thesis). Department of studies in Botany, University of Mysore, Mysore
Kataria HC, Gupta MK, Kushwaha S, Kashyap S, Trivedi S, Bhadoriya R, Bandewar NK (2011) Study of physico-chemical parameters of drinking water of Bhopal city with reference to health impacts. Curr World Environ 6:95–99. https://doi.org/10.12944/CWE.6.1.13
Kennedy B, Buckley YM (2021) Use of seasonal epilithic diatom assemblages to evaluate ecological status in Irish lakes. Ecol Indic 129:107853. https://doi.org/10.1016/j.ecolind.2021.107853
Kivrak E, Uygun A (2012) The structure and diversity of the epipelic diatom community in a heavily polluted stream (the Akarcay, Turkey) and their relationship with environmental variables. J Freshw Ecol 27:443–457. https://doi.org/10.1080/02705060.2012.671147
Kock A, Taylor JC, Malherbe W (2019) Diatom community structure and relationship with water quality in Lake Sibaya, KwaZulu-Natal, South Africa. S Afr J Bot 123:161–169. https://doi.org/10.1016/j.sajb.2019.03.013
Komal KJIS, Singh DP, Singh Y (2021) New records of desmids from Ropar wetland (a Ramsar site) of Punjab, India. Plant Sci Today 8:1037–1048. https://doi.org/10.14719/pst.2021.8.4.1229
Kour G, Kothari R, Dhar S, Pathania D, Tyagi VV (2022) Impact assessment on water quality in the polluted stretch using a cluster analysis during pre- and COVID-19 lockdown of Tawi river basin, Jammu, North India: an environment resiliency. Energy Ecol Environ 7:461–472. https://doi.org/10.1007/s40974-021-00215-4
Kumar M, Prabhahar C (2012) Physico-chemical parameters of river water: a review. Int J Pharm Biol Arch 3:1304–1312
Kumar M, Singh PG (2017) Determination of the freshwater diatom flora of Hanumangarh district, Rajasthan, India. Indian J Pure Biol 32:227–238
Kumar G, Singh KK (2020) Mapping and monitoring the selected wetlands of Punjab, India, using geospatial techniques. J Indian Soc Remote Sens 48:615–625. https://doi.org/10.1007/s12524-020-01104-9
Lacerda SR, Koening ML, Neumann-Leitao S, Flores-Montes MJ (2004) Phytoplankton nyctemeral variation at a tropical river estuary (Itamaracá - Pernambuco - Brazil). Braz J Biol 64:81–94. https://doi.org/10.1590/s1519-69842004000100010
Ladhar SS (2002) Status of ecological health of wetlands in Punjab, India. Aquat Ecosyst Health Manag 5:457–465. https://doi.org/10.1016/j.sajb10.1080/14634980290002002
Ladhar SS (2005) Ropar wetland. Punjab State Council for Science and Technology, Chandigarh
Lawson EO (2011) Physico-chemical parameters and heavy metal content of water from mangrove swamps of Lagos Lagoon, Lagos, Nigeria. Adv Biol Res 5:08–21
Li L, Zheng B, Liu L (2010) Biomonitoring and bioindicators used for river ecosystems: definitions, approaches and trends. Procedia Environ Sci 2:1510–1524. https://doi.org/10.1016/j.proenv.2010.10.164
Mahananda MR, Mohanty BP, Behera NR (2010) Physico-chemical analysis of surface and ground water of Bargarh district, Orissa, India. IJRRAS 2:284–295
Maraşlıoğlu F, Bektaş S, Özen A (2020) Comparative performance of physicochemical and diatom-based metrics in assessing the water quality of Mert Stream, Turkey. J Ecol Eng 21:18–31. https://doi.org/10.12911/22998993/127392
Marbaniang DM, Paul RMP (2015) A study on diatom species in different water bodies in of Northern Eastern India (Meghalaya). Malays J Forn Sci 6:1–6
Mbao EO, Odinga ES, Nyika J, Ochieng B, Kitaka N (2022) A bibliometric study on the use of diatoms in water quality monitoring and bioassessment in Africa across 10-year (2012–2022) period. Aquat Sci 84:58. https://doi.org/10.1007/s00027-022-00891-2
Michelutti N, McCleary K, Douglas MSV, Smol JP (2013) Comparison of freshwater diatom assemblages from a high arctic oasis to nearby polar desert sites and their application to environmental inference models. J Phycol 49:41–53. https://doi.org/10.1111/jpy.12024
Morin S, Straub F, Weil F, Charlier P (2020) Diatoms on the hair of Holy Mary-Magdalene relics. Bot Lett. https://doi.org/10.1080/23818107.2020.1768892
Muñoz-López CL, Rivera-Rondón CA (2022) Diatom response to environmental gradients in the high mountain lakes of the Colombia’s Eastern Range. Aquat Sci 84:15. https://doi.org/10.1007/s00027-021-00838-z
Murulidhar VN, Murthy VNY (2014) Distribution and ecology of diatom communities in four lakes using Lange-Bertalot method. Int J Curr Microbiol Appl Sci 3:539–548
Nautiyal P, Verma J (2009) Taxonomic richness and diversity of the epilithic diatom flora of the two biogeographic regions of the Indian subcontinent. Bull NIE 19:1–4
Nautiyal P, Nautiyal R, Kala K, Verma J (2004) Taxonomic richness in the diatom flora of Himalayan streams (Garhwal, India). Diatom 20:123–132
Pande CB, Moharir K (2018) Spatial analysis of groundwater quality mapping in hard rock area in the Akola and Buldhana districts of Maharashtra. India Appl Water Sci 8:106. https://doi.org/10.1007/s13201-018-0754-2
Pandey LK, Bergey EA, Jie L, Park J, Choi S, Lee H, Depuydt S, Han T (2017) The use of diatoms in ecotoxicology and bioassessment: insights, advances and challenges. Water Res 118:39–58. https://doi.org/10.1016/j.watres.2017.01.062
Pareek RK, Khan AS, Srivastva P, Roy S, Grover S (2018) Ecological assessment of Ghaggar river with diatoms. Plant Arch 18:1074–1080
Paul SS, Malik B, Mandal M, Biswas B, Sekh S, Sarkar NS (2016) Epilithic diatoms as biological water quality indicators-a study in three geographically isolated hill streams in India. J Environ Biol 37:273–283
Pratiwi D, Sumiarsa D, Oktavia D, Sunardi S (2023) water quality influences self-purification in the Cihawuk and Majalaya segments upstream of the Citarum river, West Java. Indonesia Water 15:2998. https://doi.org/10.3390/w15162998
Qingmin Y, Yan L, Youfang W, Quanxi W (2008) Synedra ulna var. a repanda, a new variety of Synedra (Bacillariophyta) from Xinjiang. China Chin J Oceanol Limnol 26:419–420. https://doi.org/10.1007/s00343-008-0419-3
Reyes-Toscano CA, Alfaro-Cuevas-Villanueva R, Cortés-Martínez R, Morton-Bermea O, Hernández-Álvarez E, Buenrostro-Delgado O, Ávila-Olivera JA (2020) Hydrogeochemical characteristics and assessment of drinking water quality in the urban area of Zamora. Mexico Water 12:556. https://doi.org/10.3390/w12020556
Rusydi A (2018) Correlation between conductivity and total dissolved solid in various type of water: a review. IOP Conf Ser Earth Environ Sci 118:012019. https://doi.org/10.1088/1755-1315/118/1/012019
Sharma A, Sharma RC, Anthwal A (2007) Monitoring phytoplankton diversity in the hill stream Chandrabhaga of Garhwal Himalaya. Life Sci 4:80–84
Sharma C, Jindal R, Singh UB, Ahluwalia AS, Thakur RK (2013) Population dynamics and species diversity of plankton in relation to hydrobiological characteristics of river Sutlej, Punjab, India. Ecol Environ Conserv 19:717–724
Sharma C, Jindal R, Singh UB, Ahluwalia AS (2017) Assessment of water quality of river Sutlej, Punjab (India). Sustain Water Resour Manag 4:809–822. https://doi.org/10.1007/s40899-017-0173-9
Sharma P, Giri T, Kaur M, Gautam S (2019) Morphological identification of diatoms and their physical and chemical analysis from different water bodies of Punjab India. Think India J 22:679–690
Shibabaw T, Beyene A, Awoke A, Tirfie M, Azage M, Triest L (2021) Diatom community structure in relation to environmental factors in human influenced rivers and streams in tropical Africa. PLoS ONE 16:e0246043. https://doi.org/10.1371/journal.pone.0246043
Silva-Lehmkuhl AM, Tremarin PI, Vercellino IS, Ludwig TA (2019) Periphytic diatoms from an oligotrophic lentic system, Piraquara I reservoir, Paraná state. Brazil Biota Neotrop 19:e20180568. https://doi.org/10.1590/1676-0611-bn-2018-0568
Singh AL (2013) Nitrate and phosphate contamination in water and possible remedial measures. In: Dwivedi N (ed) Environmental problems and plants: current scenario and challenges. Lambert Academic Publishing, United Kingdom, pp 44–46
Singh UB, Ahluwalia AS (2013) Microalgae: a promising tool for carbon sequestration. Mitig Adapt Strateg Glob Chang 18:73–95. https://doi.org/10.1007/s11027-012-9393-3
Singh N, Kaur M, Katnoria JK (2017) Analysis on bioaccumulation of metals in aquatic environment of Beas River Basin: a case study from Kanjli wetland. Geo Health 1:93–105. https://doi.org/10.1002/2017GH000062
Singh D, Kumar K, Singh C, Ahmad T (2020) Assessment of water quality of Holy Kali Bein rivulet (Punjab) India, using multivariate statistical analysis. Int J Environ Anal Chem 102:6784–6800. https://doi.org/10.1080/03067319.2020.1817425
Singh Y, Singh G, Khattar JIS, Barinova S, Kaur J, Kumar S, Singh DP (2022) Assessment of water quality condition and spatiotemporal patterns in selected wetlands of Punjab, India. Environ Sci Pollut Res 29:2493–2509. https://doi.org/10.1007/s11356-021-15590-y
Siriwardhana KD, Jayaneththi DI, Herath RD, Makumbura RK, Jayasinghe H, Gunathilake MB, Azamathulla HM, Tota-Maharaj K, Rathnayake U (2023) A simplified equation for calculating the water quality index (WQI), Kalu River. Sri Lanka Sustain 15:12012. https://doi.org/10.3390/su151512012
Soeprobowati TR, Tandjung SD, Sutikno HS, Gell P, Hadiyanto SSWA (2016) The water quality parameters controlling diatoms assemblage in Rawapening Lake, Indonesia. Biodiversitas 17:657–664. https://doi.org/10.13057/biodiv/d170239
Thacker M, Karthick B (2022) Response of diatoms to changing water quality in the Myristica Swamps of the Western Ghats. India Divers 14:202. https://doi.org/10.3390/d14030202
Thakar MK, Singh R (2010) Diatomological mapping of water bodies for the drowning cases. J Foren Leg Med 17:18–25. https://doi.org/10.1016/j.jflm.2009.07.016
Verma J, Nautiyal P (2009) Longitudinal patterns of distribution of epilithic diatoms in a lesser Himalayan stream. J Hill Res 22:105–109
Verma J, Nautiyal P (2010) Floristic composition of the epilithic diatoms of central highland region of Indian subcontinent; Thalassiosiraceae, Fragilariaceae, Eunotiaceae and Achnanthaceae. J Indian Bot Soc 89:397–400
Verma J, Nautiyal P, Srivastava P (2016) Diversity of diatoms in the rivers of Bundelkhand plateau: a multivariate approach for floral patterns. Int J Geol Earth Environ Sci 6:66–77
Acknowledgements
Authors are thankful to the Vice-chancellor, Sri Guru Granth Sahib World University, Fatehgarh Sahib, Punjab for providing research facilities. Authors are also thankful to Dr. Balasubramanian Karthick (Scientist-E), Agharkar Research Institute, Pune, Maharashtra (India) for his expert suggestions and guidance. Yadvinder Singh thanks the SERB-DST, New Delhi for financial assistance in the form early career research grant (ECR/2017/001103).
Funding
No funding was received for conducting this study.
Author information
Authors and Affiliations
Contributions
Conceptualization YS, JISK and DPS; methodology YS, Komal and AS; software YS, GS and DPS; validation YS, JISK, and DPS; formal analysis YS, and Komal; investigation YS, Komal and AS; resources YS, JISK and DPS; writing–original draft preparation Komal; writing–review and editing YS, Komal and JISK; visualization Komal, GS and YS; supervision YS, JISK, and DPS; project administration YS; funding acquisition YS. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they not have any competing interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Singh, Y., Komal, Singh, G. et al. Spatial analysis of diatom diversity and water chemistry of ropar wetland (Ramsar site) of Punjab, India. Braz. J. Bot 46, 1095–1112 (2023). https://doi.org/10.1007/s40415-023-00951-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40415-023-00951-3