Abstract
Phenological studies are fundamental for understanding the structure and dynamics of the plant community and assist in understanding the influence of climate on vegetation and for the analysis of plant-animal interactions. In this work, we present the phenology of 19 woody species of coastal dunes located in the eastern end of the Amazon coast. We evaluate the phenological aspects of the community and their relationship with climatic factors (photoperiod, precipitation, and temperature). For this evaluation, 19 woody species with a diameter at soil height (DBH) > 3 cm and whole crown visible were selected. Information on leaf abscission, new leaves, flowering, and fruiting was collected fortnightly. The activity and intensity of each phenophase were estimated according to the Fournier index. Circular statistic was employed to determine the mean dates for the start and peak of the phenophases, and the percentage of activity and intensity for each phenophase. Multiple linear regression analysis was applied to investigate the relationships between phenophases and climatic variables. Additionally, leaf habits and pollination and dispersion syndromes were defined for each species. The results reveal that the species evaluated did not show seasonally defined behavior, nor were they related to the climatic factors evaluated. Most species showed evergreen leaf habit (83.4%), melittophilly as the primary pollination syndrome (78.9%), and zoochoric dispersal (84.22%). Although it does not present a seasonally defined behavior, nor is it related to the climatic factors evaluated, the dry season presented the highest percentages for all phenophases, which may indicate that other factors associated with the dry period may influence the trigger off the phenophases. The information collected expands the knowledge about the functioning of this ecosystem and reinforces the need for more ecological studies and reduced impact management measures, aiming at coastal vegetation conservation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Dunes consist of sandy intertidal sediments that, once dry, are transported by wind action toward the continent (Cordazzo et al. 2006). They are characterized by specific environmental conditions, such as strong winds, high light and temperature intensity, and nutrient-poor soil (Britto et al. 1993; García-Mora et al. 2001; Ley Vega de Seoane et al. 2007). The variety of topographic features, and its relation with different environmental gradients, determines the coexistence of different types of plant communities in the space occupied by the dunes (Wilson and Sykes 1999; Frederiksen et al. 2006). Therefore, coastal dunes vegetation is not homogeneous, but arranged in patches, which leads to the formation of complex vegetation mosaics throughout this environment worldwide (Doing 1985; Cordazzo et al. 2006). In addition to their ecological importance, coastal dunes guarantee protection for coastal areas against sea waves, wind, floods, and erosion (Kiehl and Isermann 2007). However, coastal ecosystems, such as dunes, are increasingly threatened globally (Kutiel et al. 2000), mainly due to human actions led by the rapid progress of the urban occupation process (Rosa and Cordazzo 2007; Carboni et al. 2009).
Due to the sensitivity of coastal areas and the accelerated pressure and anthropogenic disturbance, studies on vegetation ecology are important for understanding and conserving these environments (Medeiros et al. 2007). In this respect, phenology stands out as an integrative environmental science and can be used in different approaches related to conservation practices (Morellato et al. 2016). Phenological studies are fundamental for understanding the influence of climate on vegetation and for the analysis of plant-animal interactions, such as pollination and seed dispersal (Williams-Linera and Meave 2002; Morellato 2003; Franco et al. 2014). These interactions are important for understanding the structure and dynamics of the plant community, essential in the elaboration of projects aimed at the conservation of biodiversity (Fournier 1976; Wunderlee 1997; Yamamoto et al. 2007; Dutra et al. 2009). However, studies on the phenological behavior of coastal dune vegetation are still scarce worldwide (Rodríguez-Gallego and Navarro 2015). Brazil follows the global trend, presenting few studies that specifically address the phenology of the coastal dune plant community. In studies carried out with dune vegetation at higher latitudes, temperature was identified as the main factor that influences reproductive phenophases, mainly flowering (Cordazzo e Seeliger 1988; Castellani et al. 1999; Morellato et al. 2013). In areas of coastal dunes in low latitudes and with little accentuated climatic seasonality, the regulation of phenophases may occur differently.
This paper presents the phenodynamic of the woody plant community on coastal dunes in eastern Amazon, as well as its leaf habits and pollination and seed dispersal syndromes, aiming to answer the question: is there any relation between climate factors (photoperiod, precipitation, and temperature) and the phenology of woody species from dunes in the eastern Amazon coast? In addition, in order to characterize the analyzed vegetation and to support discussions, pollination and dispersion syndromes and leaf habits of the species were determined.
2 Materials and methods
Field of study –
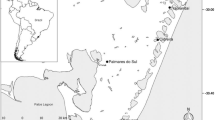
This study was carried out on a coastal dune area (02°29′7″ S 44°15′59″ W) located in Maranhão state, northeast Brazil (Fig. 1). This area is close to the urban perimeter and consists of primary and secondary dunes. The primary dunes found are narrow, with an almost completely flat area, probably as a result of disturbances such as the construction of the access road to the beach. On the other hand, the secondary dunes present a steep relief, reaching a height of 35 m (Silva et al. 2016).
The dune vegetation shows signs of anthropic impacts and is constantly affected by fire, indiscriminate cutting of vegetation, and tourists and beachgoers trampling (Silva et al. 2016). The vegetation extends over a total of about 4 km, with woody vegetation distributed throughout the whole area, whose species display, in general, smaller size and caliber compared to the same species occurring in forest areas (Araújo et al. 2016; Silva et al. 2016).
The area has a tropical savanna climate, type Aw, according to the Köppen climate classification (1948), with higher rainfall trends during the period from January to July, and the dry period between August and December (Alvares et al. 2013). According to historical data (1988 to 2018), temperatures ranged from 22 to 33 °C throughout the years, and the average annual rainfall was 1.977 mm (Fig. 2a) (INMET 2018). Data on total precipitation, average minimum and maximum temperatures (Fig. 2b) (Source: http://www.inmet.gov.br/portal/), and photoperiod (Fig. 2c) (Source: http://www.sci.fi/~benefon/sol.html) were collected monthly, from August 2017 to July 2018, to verify possible associations with phenological data.
a Historical climate data (1988—2018); b Climatic data of the study period (2017–2018): rain (filled area), maximum temperature (continuous line), average temperature (intermediate dashed line), and minimum temperature (dashed line underneath); c Photoperiod of the year of study. Information from an area of coastal dunes located in Maranhão state, northeastern Brazil
Phenological data –
To evaluate the phenological data, 19 woody species with a diameter at soil height (DSH) > 3 cm and whole crown visible were selected, including a species of cactus, based on the floristic list by Silva et al. (2016). The names of the species and their respective authors were consulted on the “Flora do Brasil 2020” website; and updates the names of species were carried out. We marked 2 to 15 individuals per species. During 12 consecutive months (August 2017 to July 2018), information on leaf abscission and production, flowering, and fruiting was collected fortnightly for all marked individuals.
The phenophases were estimated using the Fournier intensity index (1974), with values distributed on a semi-quantitative scale, and five categories grouped from 0 to 4, in 25% intervals, for vegetative and reproductive phenophases: leaf fall, new leaves, flowering, and fruiting. The phenophases were defined as (1) leaf abscission, as the proportion of the crown with no leaves, (2) new leaves, emerging or expanding, (3) flowering, the presence of flower buds and anthesis, and (4) fruiting, the presence of immature and ripe fruits. The activity percentage of each phenophase was determined, based on records of the presence or absence of the phenophases in each individual and the percentage of species that presented phenophases over the months observed.
The leaf habit of each species was defined, which were classified as evergreen, brevideciduous, and deciduous, according to Lenza and Kleink (2006), with some adjustments. Species with a crown coverage percentage always above 50% were recognized as evergreen. Species that remain without or with few senescent leaves for less than four weeks were classified as brevideciduous.
Pollination and seed dispersal syndromes –
Pollination and seed dispersal syndromes were determined by consulting specialized literature. For pollination, the following syndromes were considered: anemophily–wind, phalenophily–moths, melittophily–bees, and chiropterophily–bats. For seed dispersal analysis, species were categorized into the following groups: anemochoric–dispersed by the wind, autochoric–explosive or barocoric dispersal, and zoochoric–dispersed by animals.
Statistical analysis –
Directional statistics were applied to determine seasonal trends in the community. In order to do this, the first step was the conversion of the observation dates into angles, where a whole year corresponds to 360° (each day equals 0.986°). Then, the average start and peak dates of all individuals were determined to obtain averages by species and for the community, for each phenophases. The determination of the average data is the average of the degrees, subsequently converted into days, in which each individual manifested the phenophase for the first time (start) and the date when each individual had the highest intensity value for a given phenophase (peak).
The Rayleigh Z test was applied to test whether the circular distribution of the data collected presents seasonality. Significant probability values indicate that the concentration intensity around the average angle, represented by the vector “r,” is considered as a measure of seasonality degree, that ranges from zero (phenological activity evenly distributed throughout the year) to one (phenological activity concentrated around a single date or time of the year) (Morellato et al. 2000; 2010). All circular analyses were performed using the ORIANA 4.1 software (Kovach 2011).
Multiple regression analyses were performed to assess the possible relationships between the percentages of intensity of each phenophase (dependent variable) and the monthly climatic data of precipitation, average maximum and minimum temperatures and photoperiod (independent variables). These analyses were also carried out to verify whether there are relations between a phenological dynamics of the observed month and the climatic factors of the previous month. These analyses will be displayed only significant relationships and their beta values (the effect of the independent variable on the dependent variable) (Zar 2010).
3 Results
The research was conducted on 114 individuals, distributed in 19 species and 16 families (Table 1). Throughout the research, 10 individuals were lost as result of cutting and/or fire, which are some of the anthropic impacts considered as frequent in that area. These individuals were disregarded for statistical analysis, in view of the unfeasibility of observations throughout the study.
The community did not present seasonality for the phenophases, start and peak dates, with non-significant values on Rayleigh tests. It was not possible to determine the start dates for leaf fall and new leaves, because the species gained and lost leaves continuously, even though in small quantities (“Appendix 1 and 2”). Although the result was not significant, the angles of phenophases leaf fall and new leaves were concentrated in January. On the other hand, the flowering and fruiting started in April and May, respectively, and the peak for both phenophases was in September (Table 2). Two species did not produce fruit during the research period: Himatanthus obovatus (Müll. Arg.) Woods and Apeiba tibourbou Aubl.
The percentage of the number of species showing leaf fall was practically constant throughout the year (about 90%) with a decrease only in January. The species showed a high production of new leaves during the whole rainy season, extending until August, when this phenophase exhibited the highest percentage, reaching almost 100% of activity. After that, the production decreased (Fig. 3a).
Regarding the community activity percentage, there was a decrease in the monthly amount of abscission and new leaves production compared to the activity percentage of the species, although the variation on values was similar. Leaf abscission occurred throughout the year but decreased in January, whereas the production of leaves showed a growth pattern in dry/rainy and rainy/dry transition periods (Fig. 3b).
The intensity values determined using the Fournier index showed higher rates of leaf abscission between October and December, with percentages above 30%, followed by a decrease in January (16%). In the succeeding months, these values varied between 22 and 29%. The intensity percentage for new leaves exhibited the same pattern found for the community activity, with an increase during the dry/rainy transition periods (Fig. 3c). Regarding leaf habit, most species were classified as evergreen (83.4%), followed by brevideciduous (16.6%). No deciduous species were found (Table 1).
The species showed reproductive phenophases activity throughout the year. The highest percentages of flowering and fruiting occurred in September, during the dry period, and the lowest in May, during the rainy period. Despite following the same rate of growth and decrease, the values of fruiting dynamics were always lower than those of flowering (Fig. 4a).
The highest and lowest flowering activities percentages occurred largely during the dry season, from July to September, and from November to December, respectively. Regarding fruiting, the highest activity was registered during the dry season, from August to October, while the lowest occurred in the rainy season, in January (Fig. 4b). The Fournier intensity percentage for the reproductive phenophases exhibited the highest values during the dry season, both in September. The lowest flowering activity was also observed during the dry season, in November, and the lowest fruiting activity occurred during the rainy season, in April (Fig. 4c).
Regarding pollination syndromes, 15 species (78.9%) were classified as mellitophilous, 2 (10.5%) as phalenophilous, 1 (5.3%) as chiropterophilous, and 1 (5.3%) as anemophilous. As for seed dispersal, 16 species exhibited zoochoric dispersal (84.3%), 2 (10.4%) autochoric, and 1 (5.3%) anemochoric.
According to the multiple regression results, the community has no relationship between the evaluated phenophases (leaf fall, new leaves, flowering, and fruiting) and the climatic data of the observed month, nor with the previous month (rain, temperature, photoperiod) (Table 3).
4 Discussion
The community did not show seasonality in its vegetative and reproductive phenophases. Besides, there was no relation to the climate elements evaluated (precipitation, temperature, and photoperiod), in the observed month or with the previous month of observation, indicating that other environment factors may be influencing the development of phenophases. The absence of relationships between leaf fall and new leaves, phenophases with climatic variables was also verified by Medeiros et al. (2007) in restinga vegetation in the state of Pernambuco, Brazil. Phenophase leaf fall (Silva et al. 2010) and new leaves (Cordazzo and Seeliger 1988; Bencke and Morellato 2002) have been related to rainfall, in restinga and dunes vegetation. In addition to rainfall, some coastal communities may still have relationships between vegetative phenophases (leaf fall and new leaves) with other climatic factors, such as temperature or photoperiod (Morellato et al. 2000; Marchioretto et al. 2007). This phenodynamic variation found in coastal areas may be related to the wide geographical location of the different communities.
The community evaluated in this study is located in low latitude, near the equator line, with so low variation of temperature and photoperiod. Probably, for this reason, the community did not show any relationship with temperature or photoperiod. Although it also does not show significant results in relation to rainfall, the dry season presented the highest percentages in almost all phenophases evaluated, which indicates that other factors associated with the dry period may be influencing the development of phenophases. In Brazil, coastal vegetation, such as those that develop in coastal dunes, has a wide geographical distribution, being subject to environmental conditions that vary according to latitude and that can influence the dynamic processes of communities (Marques and Oliveira 2004), exhibiting various phenological responses. In coastal areas located at low latitudes, where the annual photoperiod variation is small, the dry season generally tends to determine the phenological pattern (Medeiros et al. 2007; Silva et al. 2010). As the latitude increases, a greater photoperiod variation is noted; and since photoperiod and temperature are closely related, it exerts a greater influence on the phenophases (Marques et al. 2004; Marchioretto et al. 2007).
Regarding leaf habit, most species were classified as evergreen. The studied community is subjected to approximately four months with low or no rainfall, yet most species monitored maintained their leaves during the dry season, with constant leaf production and abscission. Evergreen species are not usually frequent in areas with water deficit, as the energy cost required for maintenance is higher (Holbrook et al. 1995). However, in coastal plains, which are normally environments of low soil fertility, an increase in leaf retention time can mean a nutrient-saving strategy, enabling the transfer of nutrients directly to new leaves (Jordan 1985).
In the flowering analysis, the phenodynamic observed in the community showed that flower production occurred throughout the year, showing an increase in the drier period, despite that, flowering did not show seasonality. These data converge with those obtained in other coastal areas, mainly with those found in lower latitudes (Medeiros et al. 2007; Silva et al. 2010). The occurrence of species in bloom throughout the year and many species with long blooms, as found in this study, is also reported for coastal areas located at higher latitudes (Castellani et al. 1999; Talora and Morellato 2000), where associations with climatic factors were verified, such as temperature and photoperiod (Marchioretto et al. 2007).
The long flowering periods of the species in the dune areas can represent an adaptive advantage to overcome the restriction that the strong action of the winds can induce in the pollinator activity in these areas (Albuquerque et al. 2007). Most of the species in the present study depended on pollinating agents for reproduction, with the majority presenting mellitophilous syndrome, information that converges with another study, previously carried out in the region's dunes (Gottsberger et al. 1988).
Fruiting recorded during all months of the year, and no significant relationship with climatic factors was found. The absence of relationships between this phenophase and the climate variables was also registered for restinga vegetation, in Pernambuco state (Medeiros et al. 2007). It was also verified the occurrence of the relationship between fruiting and precipitation in a restinga vegetation analyzed by Silva et al. (2010). Several ecosystems exhibit such a variable temporal pattern of fruiting (Machado et al. 1997; Justiniano and Fredericksen 2000; Morellato et al. 2013; Lacerda et al. 2017), which may indicate that, at the community level, there are different phenophase-related strategies associated with species seed dispersal, germination, and establishment (Morellato et al. 1989; Schaik et al. 1993; Griz and Machado 2001). These physiological mechanisms can be stronger drivers for fruiting than climatic variables.
As for fruits dispersion, more than 80% of the species in this study presented zoochoric syndrome. In Brazilian coastal areas, the proportion of zoochoric species are very similar to those observed in this study, always above 80% (Moraes and Mondin 2001; Almeida Jr. et al. 2008; Amaral et al. 2015), corroborating the assertion that most tropical species are animal-dispersed (Morellato and Leitão-Filho 1992; Talora and Morellato 2000).
Fruiting, which occurred throughout the year, with a subtle increase in the drier period, did not exceed the flowering percentages, differing from other coastal areas that, in at least some month, the fruiting values exceeded those of flowering (Castellani et al. 1999; Medeiros et al. 2007; Silva et al. 2010). This observation suggests that there may be little fruit production in the community. In addition, the species Himatanthus obovatus (Müll. Arg.) Woodson and Apeiba tibourbou Aubl did not fruiting throughout the study, despite having flowering, unlike studies carried out in different ecosystems in which it is reported that these species have an annual fruiting frequency (Pirani et al. 2009; Barbosa 2014). Such information of low fruit production and failure in the fruiting of some species calls attention to the health of the studied community, a fact that becomes even more worrying because this is a sensitive community due to the strong anthropic action. Anthropic interference is considered one of the causes that can influence the behavior of species, mainly in areas with a greater indication of degradation, there may be a difficulty in obtaining the necessary resources for the maintenance and growth of the plants, due to the scarcity or unavailability of resources in the area (Costa 2002). Therefore, it is important to emphasize that the anthropic interference can harm fruit production that is crucial for the balance of ecosystems, as this guarantees food for dispersers and availability of seed for maintaining the plant community.
Finally, the results of the present study reveal that the coastal dune community did not show a seasonally defined behavior, nor relationship to any of the climatic factors evaluated. Despite this, the dry season had the highest percentages in almost all of the evaluated phenophases, which may indicate that other factors associated with the dry period, such as wind speed, may be responsible for the intensification of these phenophases. The prolonged flowering behavior found in this study seems to be a strategy common to other coastal vegetation. Therefore, other studies that take these factors into account are strongly encouraged. The information collected expands the knowledge about the functioning of this ecosystem and reinforces the need for more attention to coastal lowland areas, through intensification of ecological studies and reduced impact management measures, aiming at coastal vegetation conservation.
References
Albuquerque PMC, Camargo JMF, Mendonça JAC (2007) Bee community of a beach dune ecosystem on Maranhão Island, Brazil. Braz Arch Biol Technol 50:1005–1018
Almeida EB Jr, Medeiros DPW, Vicente A, Lima LF, Lima PB (2007) Estudo comparativo entre síndromes de dispersão em quatro áreas de floresta Atlântica sensu lato, Nordeste-Brasil. Rev Bras De Biociênc 5:498–500
Alvares CA, Stape JL, Sentelhas PC, Gonçalves JD, Sparovek G (2013) Köppen’s climate classification map for Brazil. Meteorol Z 22:711–28
Amaral DD, Jardim MAG, Costa Neto SV, Carmo Bastos MDN (2015) Síndromes de dispersão de propágulos e a influência da floresta amazônica na composição de espécies lenhosas de uma restinga no litoral norte brasileiro. Biota Amazôn biote Amazon Biota Amazon Amazon Biota 5:28–37
Araujo ACM, Silva ANF, Almeida EB Jr (2016) Caracterização estrutural e status de conservação do estrato herbáceo de dunas da Praia de São Marcos, Maranhão. Bras Act Amazon 46:3
Barbosa JFM (2014) Ecofisiologia, fenologia e adaptação de espécies florestais arbóreas nas condições edafoclimáticas de Viana-ES. Dissertação de Mestrado (Mestrado em Biologia Vegetal) Universidade Federal do Espírito Santo
Bencke CS, Morellato LPC (2002) Estudo comparativo da fenologia de nove espécies arbóreas em três tipos de floresta atlântica no sudeste do Brasil. Rev Bras De Bot 25:237–248
Britto IC, Queiroz LP, Guedes MLS, Oliveira NC, Silva LB (1993) Flora fanerogâmica das dunas Lagoas de Abaeté, Salvador, Bahia. Sit Feira De Santana 11:31–46
Carboni M, Carranza ML, Acosta A (2009) Assessing conservation status on coastal dunes: a multiscale approach. Landsc Urban Plan 91:17–25
Castellani TT, Caus CA, Vieira S (1999) Fenologia de uma comunidade de duna frontal no sul do Brasil. Act Bot Bras 13:99–114
Cordazzo CV, Seeliger U (1988) Phenological and biogeographical aspects of coastal dune plant communities in southern Brazil. Vegetatio 75:169–173
Cordazzo CV, Paiva JB, Seeliger, U (2006) Plantas das dunas da costa sudoeste Atlântica: guia ilustrado. Useb
Costa FAPL (2002) Fenologia de árvores tropicais. La insignia. Disponível em: <http://www.lainsignia.org/2002/ diciembre/dial005.htm.>. Acesso em: 12 ago. 2008
Doing H (1985) Coastal fore-dune zonation and succession in various parts of the world. Ecology of coastal vegetation. Springer, Dordrecht, pp 55–75
Dutra VF, Vieira MF, Garcia FCP, Lima HCD (2009) Fenologia reprodutiva, síndromes de polinização e dispersão em espécies de Leguminosae dos campos rupestres do Parque Estadual do Itacolomi, Minas Gerais, Brasil. Rodriguésia 2:371–387
Fournier LA (1974) Un método cuantitativo para la medición de características fenológicas en arboles. Turrialba 24:422–423
Franco AC, Rossatto DR, Silva LDCR, Silva Ferreira C (2014) Cerrado vegetation and global change: the role of functional types, resource availability and disturbance in regulating plant community responses to rising CO 2 levels and climate warming. Theor Exp Plant Physiol 26:19–38
Frederiksen L, Kollmann J, Vestergaard P, Bruun HH (2006) A multivariate approach to plant community distribution in the coastal dune zonation of NW Denmark. Phytocoenologia 36:321–342
García-Mora MR, Gallego-Fernández JB, Williams AT, García-Novo F (2001) A coastal dune vulnerability classification: a case study of the SW Iberian Peninsula. J Coast Res 17:802–811
Gottsberger G, Camargo I, Silberbauer-Gottsberger JMF (1988) A bee-pollinated tropical community: the beach dune vegetation of Ilha de São Luis, Maranhão, Brasil. Bot Jahrb Für Syst 109:469–500
Griz LMS, Machado ICS (2001) Fruiting phenology and seed dispersal syndromes in caatinga, a tropical dry forest in the northeast of Brazil. J Trop Ecol 17:303–321
Holbrook NM, Whitbeck JL, Mooney HA (1995) Droughts responses of neotropical dry forest trees. In: Bullock SH, Mooney HA, Medina E (eds) Seasonally dry tropical forests. University Press, Cambridge
Inmet, Instituto Nacional de Meteorologia. (2018) Banco de dados meteorológicos para ensino e pesquisa, BDMEP, http://www.inmet.gov.br/portal/. 06.Nov.2018
Jordan CF (1985) Nutrient cycling in tropical forest ecosystems principles and their application in management and conservation. Wiley, Hoboken
Justiniano MJ, Fredericksen TS (2000) Phenology of tree species in Bolivian dry forests. Biotropica 32:276–281
Kiehl K, Isermann M (2007) Restoration of coastal ecosystems an introduction. Coastline reports
Köppen W (1948) Climatologia. México. Fundo de Cultura Econômica
Kovach WL (2011) Oriana - Circular Statistics for Windows. Version 3. Pentraeth, Wales: Kovach Computing Services
Kutiel P, Peled Y, Geffen E (2000) The effect of removing scrub cover on annual plants and small mammals in a coastal sand dune ecosystem. Biol Conserv 94:235–242
Lacerda DMA, Araújo Barros JBH, Almeida EB Jr, Rossatto DR (2017) Do conspecific populations exhibit divergent phenological patterns? A study case of widespread savanna species. Flora 236:100–106
Lenza E, Klink CA (2006) Comportamento fenológico de espécies lenhosas em um cerrado sentido restrito de Brasília. DF Rev Bras Bot 29:627–638
Ley Vega de Seaone C, Gallego-Fernández JB, Vidal-Pascual C (2007) Manual de restauración de dunas costeras. Ministerio de Medio Ambiente, Dirección general de Costas
Machado IC, Barros LM, Sampaio EV (1997) Phenology of caatinga species at Serra Talhada, PE, northeastern Brazil. Biotropica 29:57–68
Marchioretto MS, Mauhs J, Budke JC (2007) Fenologia de espécies arbóreas zoocóricas em uma floresta psamófila no sul do Brasil. Act Bot Bras 21:193–201
Marques MCM, Oliveira PEAM (2004) Fenologia de espécies do dossel e do sub-bosque de duas Florestas de restinga na Ilha do mel, sul do Brasil. Rev Bras Bot 27:713–723
Marques MCM, Roper JJ, Salvalaggio APB (2004) Phenological patterns among plant life forms in a subtropical forest in Southern Brazil. Plant Ecol 173:203–213
Medeiros DPW, Lopes AV, Zickel CS (2007) Phenology of woody species in tropical coastal vegetation, northeastern Brazil. Flora Morphol Distrib Funct Ecol Plants 202:513–520
Moraes D, Mondin CA (2001) Florística e fitossociologia do estrato arbóreo em mata arenosa no balneário do quintão, palmares do sul, rio grande do sul. Pesqui Série Bot 51:87–100
Morellato LPC (2003) América do Sul. In: Schwartz M (ed) Phenology: uma ciência ambiental integrativa. Springer, Nova Iorque
Morellato LPC, Leitão-Filho HF (1992) Padrões de frutificação e dispersão na Serra do Japi. In: Morellato LPC (Ed.). História natural da Serra do Japi: ecologia e preservação de uma área florestal no Sudeste do Brasil. Campinas, Editora da Unicamp/Fapesp
Morellato LPC, Rodrigues RR, Joly L-F, CA, (1989) Estudo comparativo da fenologia de espécies arbóreas de floresta de altitude e floresta mesófila semidecídua na Serra do Japi, Jundiaí, São Paulo. Rev Bras Bot 12:85–98
Morellato LPC, Tarola DC, Takahasi A, Bencke CC, Romera EC, Zipparro VB (2000) Phenology of atlantic rain forest trees: a comparative study. Biotropica 32:811–823
Morellato LPC, Alberti LF, Hudson IL (2010) Applications of circular statistics in plant phenology: a case studies approach. In: Hudson LI, Keatley MR (eds) Phenological research. Springer, Netherlands, pp 339–359
Morellato LPC, Camargo MGG, Gressler E (2013) A review of plant phenology in South and Central America. Phenology: an integrative environmental science. Springer, Dordrecht, pp 91–113
Morellato LPC, Alberton B, Alvarado ST, Borges B, Buisson E, Camargo MGG, Cancian LF, Carstensen DW, Escobar DFE, Leite PTP, Mendonza I, Rocha NMWB, Soares NC, Silva TSF, Staggemeier VG, Streher AS, Vargas BC, Peres CA (2016) Linking plant phenology to conservation biology. Biol Cons 195:60–72
Pirani FR, Sanchez M, Pedroni F (2009) Fenologia de uma comunidade arbórea em cerrado sentido restrito, barra do garças, MT, Brasil. Act Bot Bras 23:1096–1109
Rodríguez-Gallego C, Navarro T (2015) Vegetative and reproductive phenological patterns in coastal dunes in S Spain. In Anales del Jardín Botánico de Madrid. 72:17
Rosa LSD, Cordazzo CV (2007) Perturbações antrópicas na vegetação das dunas da Praia do Cassino (RS)
Schaik CPV, Terborgh JW, Wright SJ (1993) The phenology of tropical forests: adaptive significance and consequences for primary consumers. Annu Rev Ecol Syst 24:353–377
Silva SSL, Medeiros DPW, Almeida Jr EB, Pessoa LM, Zickel CS (2010) Observações fenológicas em uma restinga da APA de Guadalupe, Tamandaré, Pernambuco. In: Albuquerque, U. P et al. (Org.). Biodiversidade, potencial econômico e processos eco-fisiológicos em ecossistemas nordestinos. Bauru, SP, Canal6/Recife, PE: NUPEEA
Silva, ANF, Araujo, ACM, Almeida Jr, EB (2016) Flora Fanerogâmica das Dunas da Praia de São Marcos, São Luís, Maranhão. In: Almeida Jr, E.B., Santos-Filho, F.S. (Orgs). Biodiversidade do Meio Norte do Brasil: Conhecimentos ecológicos e aplicações. Curitiba, CVR
Talora DC, Morellato LPC (2000) Fenologia das espécies arbóreas em floresta de planície litorânea do sudeste do Brasil. Rev Bras Bot 1:13–26
Williams-Linera G, Meave H (2002) Patrones fenológicos. In: Guariguata MR, Kattan GH (Eds.) Ecologia y Conservación de Bosques Neotropicales, Libro Universitário Regional, Cartago
Wilson JB, Sykes MT (1999) Is zonation on coastal sand dunes determined primarily by sand burial or salt spray? a test in New Zealand dunes. Ecol Lett 2:233–236
Wunderlee JM Jr (1997) The role of animal seed dispersal in accelerating native forest regeneration on degraded tropical lands. For Ecol Manag 99:223–235
Yamamoto LF, Kinoshita LS, Martins FR (2007) Síndromes de polinização e de dispersão em fragmentos da Floresta Estacional Semidecídua Montana, SP, Brasil. Acta Botanica Brasilica 3:553–573
Zar JH (2010) Bioestatistical analysis. Prentice Hall, New Jersey
Acknowledgements
The authors would like to thank the Coordination for the Improvement of Higher Education Personnel (CAPES), finance code 001, for the scholarship granted, the Programa de Pós-Graduação em Biodiversidade e Conservação (PPGBC). The Fundação de Amparo à Pesquisa e ao Desenvolvimento Científico e Tecnológico do Maranhão (FAPEMA). The Laboratório de Estudos Botânicos (LEB) of the Universidade Federal do Maranhão (UFMA) for the infrastructure provided for the execution of the study and the Luann Brendo da Silva Costa for his presence in all data collections. This research was supported by the Agency for Innovation, Entrepreneurship, Research, Postgraduate and Internationalization (AGEUFMA), Federal University of Maranhão, Brazil.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Aline Duarte Nascimento performed literature review, data collection, and the first draft of the manuscript. Aline Duarte Nascimento, Luann Brendo da Silva Costa, and Dinnie Michelle Assunção Lacerda performed statistical analysis and interpretation of data. Luann Brendo da Silva Costa, Dinnie Michelle Assunção Lacerda, and Eduardo Bezerra de Almeida Jr. performed critical analysis of paper and support in discussing the results of paper. All authors read and approved the final manuscript.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix 1
Phenophasic diagrams of leaf fall and new leaves of 19 species studied in coastal dunes in the state of Maranhão, northeastern Brazil.

Appendix 2
Phenophasic diagrams of flowering and fruiting of 19 species studied in coastal dune in the state of Maranhão, northeastern Brazil.

Rights and permissions
About this article
Cite this article
Nascimento, A.D., Costa, L.B.D., Lacerda, D.M.A. et al. Woody plants phenology of the coastal dunes in eastern Amazon, Brazil. Braz. J. Bot 44, 741–752 (2021). https://doi.org/10.1007/s40415-021-00716-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40415-021-00716-w