Abstract
In this paper, the authors consider a four-compartmental HIV epidemiological model, which describes the interaction between HIV virus and two target cells, CD4 T cells and macrophages in vivo. It is proved that the bilinear incidence can cause the backward bifurcation, where a locally asymptotically stable disease-free equilibrium co-exists with a locally asymptotically stable endemic equilibrium when the basic reproduction number \((R_{0})\) is less than unity. It is shown that a sequence of Hopf bifurcations occur at the endemic equilibrium by choosing one parameter of the model as the bifurcation parameter. Meanwhile, the global asymptotic stabilities of the equilibria are established by constructing suitable Lyapunov functions under some conditions. Furthermore, the authors develop an extended model by incorporating with the intracellular delays and derive global asymptotic stability of the delayed model by constructing Lyapunov functions. Some numerical simulations for justifying the theoretical analysis results are also given.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Human immunodeficiency virus (HIV), which mainly infects CD4 T cells and other target cells, threatens global human health and society development seriously (Perelson and Nelson 1999; Wodarz and Lloyd 1999). HIV, the cause of acquired immunodeficiency syndrome (AIDS), develops rapidly during the course of infection and the virus can evolve toward faster replication rates during late stages (Perelson and Nelson 1999; Regoes et al. 1998). There are approximately 6000 mm\(^-3\) white blood cells in a healthy body. It is estimated that between 1 and 6% of these are macrophages and approximately \(10\%\) are CD4 T cells Kirschner (1999). Clinical studies show that most viruses come from the infected macrophages during the late stages of the disease. Macrophages play an important role as a viral source and are considered as the second target cell of HIV (Perelson and Essunger 1997; Kirschner 1999). Moreover, macrophages are also the crucial immune responses and can clear certain HIV virus (Adams and Banks 2005; Kirschner 1999).
Mathematical models have made great contributions to getting insights into HIV infection dynamics in vivo. Great efforts were made to describe the interaction between HIV and CD4 T cells (Leenheer and Smith 2003; Hossein et al. 2014; Wang and Wang 2012; Hu and Liu 2010). Some other HIV models consider the interaction process of HIV not only with CD4 T cells but also with macrophages (Elaiw 2010; Adams and Banks 2005; Wodarz and Lloyd 1999; Shu et al. 2013). Generally speaking, the models with two target cells are more suitable than the models with only CD4 T cells (Elaiw 2010; Perelson and Nelson 1999; Kirschner 1999). Particularly, Elaiw proposed one model with two target cells and obtained the global asymptotical stabilities of the equilibria of the model by means of Lyapunov functions in Elaiw (2010). However, we should consider some other features including the proliferation of CD4 T cells stimulated by HIV virus and the loss term of the virus killed by macrophages (Ouattara 2005; Xia 2007). Based on above works, we introduce a four-dimensional model which is formulated by the following system of non-linear differential equations:
We briefly summarize the interpretation of different parameters in the model. \(T, T_m , T^{*},\) and v represent the uninfected CD4 T cells population, the uninfected macrophages population, the infected cells population, and the virus particles population in the blood, respectively. The terms \(\lambda _1\) and \(\lambda _2\) are the constant sources of new CD4 T cells and macrophages, respectively. \(\delta _1, \delta _2, a,\) and c denote the death rates of uninfected CD4 T cells, macrophages, infected cells, and virus particles, respectively. \(\beta _1 Tv\) represents the infection rate of uninfected CD4 T cells by virus. Because the number of CD4 T cells is large, it is reasonable to use the bilinear incidence rate (Elaiw 2010; Wang et al. 2014). \(\beta _{12} Tv\) is a proliferation term due to CD4 T cells immune response. \(\beta _2 T_mv\) and \(\beta _{22} T_mv\) could be explained in the same manner. \(bT^*\) is the source of HIV virus population and the constant b is the production rate of HIV virus by the infected CD4 T cells and infected macrophages. \(\beta _3T_m v\) is the loss term of HIV virus since macrophages can kill virus particles. c is the loss rate of HIV virus because of nature death or other immune response. Suppose all the parameters are nonnegative. Simultaneously, we give some brief definitions and reference values of the model parameters in Table 1. Denote \(\beta '_1=\beta _1-\beta _{12}>0\) and \(\beta '_2=\beta _2-\beta _{22}>0\). System (1–4) becomes the following system:
In this paper, we first discuss the positively invariant set, the equilibria and the backward bifurcation. By analyzing the characteristic equations, the local asymptotic stability of an endemic equilibrium of the model is established. Letting \(\beta _3\) be the bifurcation parameter, we show that system (5–8) can undergo Hopf bifurcation, that is, a family of periodic solutions bifurcates from the infected equilibrium when \(\beta _3\) passes through a critical value. To prove the global asymptotical stabilities of the equilibria, we construct Lyapunov functions, which are similar to those in Korobeinikov (2004), Elaiw (2010), Li et al. (2011), Tsuyoshi et al. (2015), Hossein et al. (2014), Roy and Roy (2016).
The intracellular delays, from entry into CD4 T cells or macrophages to the production of new viruses, have been incorporated into biological models in many papers (Wang et al. 2014; Regoes et al. 1998; Culshaw and Ruan 2000; Wang and Zhou 2009; Yuan et al. 2012). In this paper, we establish a delayed model based on system (5–8) to study the influence of the intracellular delays on the infection transmission. We construct Lyapunov functions to prove the global asymptotical stabilities for the delayed model (McCluskey 2010; Huang et al. 2010). It is theoretically shown that time delay has no effect on the asymptotic stability of the equilibria under some conditions.
The paper is ordered as follows: Equilibria and backward bifurcation are studied in Sect. 2. Hopf bifurcation of the model is discussed in Sect. 3. The global asymptotic stabilities of the two equilibria are considered in Sect. 4. Section 5 deals with the extended model with the intracellular delays. Finally, Sect. 6 presents the conclusions of the work.
2 Equilibria and backward bifurcation
In the absence of virus, it is easy to show that the number of CD4 T cells approaches \(T_0=\frac{\lambda _1}{\delta _1}\) and the number of macrophages approaches \(T_{m0}=\frac{\lambda _2}{\delta _2}\). It is straightforward to prove the positive invariance of the nonnegative orthant \(R_{+}^4\) because of biological sense by system (5–8). Furthermore, from (5) and (6), we obtain:
Therefore, we apply comparison lemma in Sharomi and Podder (2007) and obtain:
where \(T(0), T_m(0)\) are the initial conditions. It can be seen that the uninfected CD4 T cells and macrophages are always bounded. For simplicity, we may take
Therefore, \(T^*\) is bounded. From (8), we know that v is upper bounded too, say by \(M= \frac{b\varpi }{c\delta }.\) Define the region,
Then P is positively invariant with respect to system (5–8). Any solution of system (5–8) with initial point in P will stay in P. Furthermore, if \(T> T_0\) and \( T_m> T_{m0}\), either the solution of system (5-8) enters P in finite time, or T approaches \(T_0\) and \(T_m\) approaches \(T_{m0}\) asymptotically. Thus, P attracts all solutions in nonnegative orthant \(R_{+}^{4}\). This leads to the following result:
Proposition 1
The region P is positively invariant and attracting in nonnegative orthant \(R_{+}^{4}\) for system (5–8).
Next, we focus the dynamics behavior of system (5–8) in P. There always exists a disease-free equilibrium \(E_0(T_0, T_{m0},0,0)\), which represents the state with the absence of virus. The Jacobian matrix of system (5–8) at \(E_0\) is given as
with the characteristic equation
According to the fact \(| J(E_0)|=0\), it follows that \(\lambda _1 b\beta _1\delta _2+\lambda _2\delta _1 b\beta _2=ac\delta _1\delta _2+\lambda _2\delta _1a \beta _3\). So, we define the basic reproduction number as
which represents the average number of secondary cases that one infected case can generate. It can be easily verified that, \(E_0\) is locally asymptotically stable when \(R_0<1\) and is unstable when \(R_0>1\) from the characteristic equation.
When \(a\beta _3=b\beta _2\), system (5–8) has no endemic equilibrium if \(R_0\le 1\) and has only one endemic equilibrium \(\bar{E}_1(\bar{T}_1, \bar{T}_{m1},\bar{T^*}_1, \bar{v}_1)\) if \(R_0>1\), where
When \(a\beta _3\ne b\beta _2\), it can be computed that \(v\ne 0\) satisfies
which is equivalent to the quadratic equation
where
Therefore, we obtain the following result:
Proposition 2
When \(a\beta _3\ne b\beta _2\), system (5–8) has:
-
(1)
a unique endemic equilibrium if \(R_0>1\);
-
(2)
a unique endemic equilibrium if \(q<0\), and \(R_0=1\) or \(q^2-4pr=0\);
-
(3)
two endemic equilibria if \(R_0<1, q<0\) and \(q^2-4pr>0\);
-
(4)
no endemic equilibrium otherwise.
Item (3) in Proposition 2 indicates the possibility of backward bifurcation when \(R_0<1\) (Sharomi and Podder 2007; Zhang and Liu 2008; Feng and Castillo-Chavez 2000). To verify the existence of backward bifurcation, we let the discriminant \(q^2-4pr\) be 0 and solve the equation in term of \(R_0\). We obtain
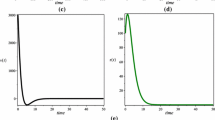
It can be shown that backward bifurcation occurs for the values of \(R_0\) if \(R^c_0<R_0<1\). We explore this phenomenon via numerical simulations and use the following parameter values: \(\lambda _1=8; \lambda _2=3; \delta _1=0.008; \delta _2=0.002; \beta '_1=0.000012; \beta '_2=0.0000211; \beta _1=0.0002; \beta _2=0.00025; a=0.48; b=50; c=4.3; \beta _3=0.05\) (Kirschner 1999; Lu and Huang 2014; Xia 2007). Then, we obtain \(R^c_0=0.2321\), \(R_0=0.7553\) and \(R^c_0<R_0<1\). It is clear that system (5–8) has a disease-free equilibrium \(E_0(1000, 1500, 0, 0)\) and two endemic equilibria, \(\hat{E}_1(278.5310,78.0510, 283.2904, 1.7268\times 10^{3})\) and \( \check{E}_1( 801.4765, 547.0186, 104.5313, 165.1315).\) The simulations depicted in Fig. 1 show that \(E_0\) and \(\hat{E}_1\) are locally asymptotically stable and \(\check{E}_1\) is unstable. As a result, a stable endemic equilibrium co-exists with a stable disease-free equilibrium for system (5–8) if \(R_0<1\). A bifurcation diagram is shown in Fig. 2. The above discussion is summarized below.
Theorem 1
System (5–8) exhibits backward bifurcation when \(q<0\), \(q^2-4pr>0\), and \(R^c_0<R_0<1\).
It should be noted that the term \(\beta _3T_mv\) plays an important role on the backward bifurcation. The phenomenon of backward bifurcation has been established in many papers (Sharomi and Podder 2007; Qesmi and Wu 2010; Feng and Castillo-Chavez 2000). It is worth stating that bilinear incidence in our model also can exhibit backward bifurcation (Hadeler and Driessche 1997; Wang and Wang 2012). This phenomenon has an important influence to control the disease. The existence of multiple endemic equilibria indicates that the asymptotical behavior of system (5–8) should depend on initial conditions. It is not enough for the eradication of the disease if \(R_0<1\). Therefore, it is equally important to identify possible backward bifurcation.
3 Existence of Hopf bifurcation at the endemic equilibrium
When \(R_0>1\) and \(a\beta _3\ne b\beta _2\), it is easy to derive that system (5–8) has a unique endemic equilibrium \(\tilde{E}_1(\tilde{T}_1, \tilde{T}_{m1},\tilde{T_1^*}, \tilde{v}_1)\), where
Therefore when \(R_0>1\), we always have one endemic equilibrium \(E_1(T_1, T_{m1},{T_1^*}, {v}_1)\), where \(E_1=\bar{E}_1\) if the condition \(a\beta _3= b\beta _2\) is satisfied and \(E_1=\tilde{E}_1\) if the condition \(a\beta _3\ne b\beta _2\) is satisfied. The characteristic equation corresponding to \(E_1\) is given by
where
By the Routh–Hurwitz criterion, it follows that all eigenvalues of Eq. (10) have negative real parts if and only if
By simply calculating, we derive that \(a_1>0, a_2>0, \) and \(a_1a_2>a_3\). Moreover, when \(a\beta _3= b\beta _2\), we can obtain \(a_4>0. \) So we derive the following proposition:
Proposition 3
When \(R_0>1\), the infected equilibrium \(E_1\) of system (5–8) is locally asymptotically stable if
In the following, we discuss the conditions for which \(E_1\) enters into Hopf bifurcation.
Theorem 2
Suppose \(a_3>0, a_4>0\) and \(R_0>1\). If there exists a critical value \(\beta _{30}>0\) such that \(\phi (\beta _{30})=0\) and \(\phi '(\beta _{30})\ne 0\), Hopf bifurcation occurs at \(E_1\) of system (5–8) when \(\beta _{3}\) passes through \(\beta _{30}\). That is, periodic solutions bifurcate from \(E_1\).
Proof
Suppose \(a_3>0\) and \(a_4>0\). Define the continuously differentiable function of \(\beta _3\):
If there exists \(\phi (\beta _{30})=0\), Eq. (10) becomes
It is easy to show that (12) has a pair of purely imaginary roots and either another pair of complex roots with negative real parts or two negative real roots. Since \(\phi (\beta _3)\) is one continuous function of all its roots in \(\beta _3\), we can derive that Eq. (10) has a pair of complex conjugate roots in a neighborhood of \(\beta _{30}\), denoted by \(\lambda _1\) and \(\lambda _2\). They are conjugated purely imaginary roots at \(\beta _3=\beta _{30} \). The transversality condition (Roy et al. 2017; Greenhalgh 1997; Liu 1994)
is equivalent to
This completes the proof.
We present some numerical results of system (5–8) for different values of \(\beta _3\) and use the parameter values as the same as in Sect. 2 except for \(\delta _2=0.008\) and \(\beta _3\). Let initial values be (500, 200, 300, 100). We obtain \(\beta _{30}\approx 0.057\) from \(\phi (\beta _3)=0\). Numerical simulations show that \(E_1\) is locally asymptotically stable if \(\beta _3=0.05< \beta _{30}\) (see Fig. 3). When \(\beta _3=\beta _{30}\), \(E_1\) loses its stability and Hopf bifurcation occurs. When \(\beta _3=0.058>\beta _{30}\), \(E_1\) becomes unstable and there are periodic solutions surrounding \(E_1\) (see Fig. 4).
4 Global stability of two equilibria
Theorem 3
If \(R_0\le 1\) and \(b\beta _2\ge a\beta _3\), the disease-free equilibrium \(E_0\) of system (5–8) is globally asymptotically stable in P.
Proof
Define a Lyapunov function
Calculating the time derivative of \(L_1\) along the solution of system (5–8), we obtain
Note that \(\lambda _1=\delta _1 T_0\) and \(\lambda _2=\delta _2 T_{m0} \). It follows from (13) that
Apparently, we can obtain \(2-\frac{T}{T_0}-\frac{T_0}{T}\le 0\) and \(2-\frac{T_m}{T_{m0}}-\frac{T_{m0}}{T_m}\le 0\). So we obtain \(\frac{\mathrm{d}L_1}{\mathrm{d}t}\le 0\) if \(R_0\le 1\). The largest compact invariant set in \(\{(T, T_m,T^*,v)\in P: \frac{\mathrm{d}L_1}{\mathrm{d}t}= 0 \}\) is the singleton \(\{E_0\}\). Using the Lasalle invariant principle, we derive that all solutions with initial conditions in P converge to \(E_0\). We complete the proof.
From Theorem 3, we obtain that the virus can be cleared under some conditions. In addition, we easily derive that system (5–8) does not exhibit backward bifurcation if \(b\beta _2\ge a\beta _3\).
Theorem 4
If \(R_0> 1\), \(b\beta _2> a\beta _3\) and \(b\lambda _1\beta _1\beta '_2\beta _3\le \beta '_1(b\beta _2- a\beta _3)(c\delta _2-\lambda _2\beta _3)\), the endemic equilibrium \(E_1\) of system (5–8) is globally asymptotically stable in P.
Proof
We consider a Lyapunov function
Calculating the time derivative of \(L_1\) along the solution of system (5–8), we obtain
From system (5–8), we obtain that
Then we get
If \(b\beta _2> a\beta _3\) and \(b\lambda _1\beta _1\beta '_2\beta _3\le \beta '_1(b\beta _2- a\beta _3)(c\delta _2-\lambda _2\beta _3)\), we can obtain
Then, we have:
Therefore, the endemic equilibrium \(E_1\) is globally asymptotically stable by the similar analysis in Theorem 3. Especially, we know \(E_1\) is globally asymptotically stable when \(\beta _3=0\). The proof is completed.
5 Analysis of the delayed model
In this section, we consider one differential equation model with a time delay, which denotes the time for the viruses from entry into CD4 T cells or macrophages to the production of new viruses. The model is given as follows:
The positive constant \(\tau \) represents the length of the delay. All the other parameters are the same as in system (5–8). The initial conditions are:
where \(\psi _3\) is a given constant, and \(\psi _1,\psi _2, \psi _4 \in C([-\tau , 0],R_{+})\) with \(R_{+}=[0, \infty )\).
Being similar to the analysis of system (5–8), we find system (14–17) always has one disease-free equilibrium \(E_0(T_0, T_{m0},0,0)\) and a unique positive equilibrium \(E_1(T_1, T_{m1}, T^*_1, v_1)\) if \(R_0>1\), where \( T_0, T_{m0}, T_1, T_{m1},T^*_1,\) and \(v_1\) are the same as in Sect. 2.
Let \(\bar{E}(\bar{T},\bar{T_m},\bar{T^*},\bar{v})\) be any equilibrium of system (14–17). Denote \( X =( T,T_m,T^*,v)^T. \) The linearized system in vector form is given as:
where \(A_1\) and \(A_2\) are \(4 \times 4\) matrices given by:
and
The characteristic equation for system (14–17) is given by:
Theorem 5
The disease-free equilibrium \(E_0\) of system (14–17) is locally asymptotically stable for all \(\tau \ge 0\) when \(R_0<1\), and is unstable when \(R_0>1\).
Proof
For the disease-free equilibrium \(E_0\), the characteristic equation reduces to,
When \(\tau =0\), we know that \(E_0\) is locally asymptotically stable if \(R_0<1\) and is unstable if \(R_0>1\) from former analysis. In the following, we discuss the case of \(\tau \ne 0\). Equation (18) has two negative solutions \(\lambda _1=-\delta _1\) and \(\lambda _2=-\delta _2\). The other eigenvalues of Eq. (18) satisfy the following transcendental equation:
Denote,
For the case of \(R_0>1\), we obtain \(F(0)<0\) and \(F(\lambda )\rightarrow +\infty \) (\(\lambda \rightarrow +\infty \)). Thus, Eq. (19) has at least one positive real root. So \(E_0\) is unstable. For the case of \(R_0<1\), we assume that \(\lambda =i\omega , ~~\omega >0.\) Substituting \(\lambda =i\omega \) into (19), we have:
We separate the real and imaginary parts and obtain,
Adding the squared Eqs. (20) and (21), it follows that,
If \(R_0<1\), Eq. (22) has no positive real roots and there is no \(i\omega (\omega \ne 0)\) satisfying Eq. (18). Equation (18) has roots with positive real parts if and only if it has purely imaginary roots by Rouch\(\acute{e}\)’s theorem (Culshaw and Ruan 2000). For all values of the delay \(\tau \ge 0\), all eigenvalues of Eq. (18) have negative real parts. This completes the proof.
Theorem 6
If \(R_0\le 1\) and \(b\beta _2\ge a\beta _3\), the disease-free equilibrium \(E_0\) of system (14–17) is globally asymptotically stable for any time delay \(\tau \ge 0\).
Proof
Define a Lyapunov function \(L_3\) as follows:
where
We calculate the time derivative of \( U_1(t)\),
Calculating the time derivative of \(L_1\) along the solution of system (14-17), we obtain
Note that \(\lambda _1=\delta _1 T_0\) and \(\lambda _2=\delta _2 T_{m0} \). Thus, we obtain
So, it follows that \(\frac{\mathrm{d}L_3}{\mathrm{d}t} \le 0\) if \(R_0\le 1\) and \(b\beta _2\ge a\beta _3\). It is clear that \(E_{0}\) is stable. Furthermore, the largest compact invariant set is the singleton \(\{E_0\}\). Accordingly, it follows from LaSalle invariance principle that \(E_{0}\) is globally asymptotically stable. We complete the proof.
The characteristic equation for the linearized system around the infected equilibrium \(E_1\) is given by
where
Substituting \(\lambda =i\omega \) with \(\omega >0\) into (23) and separating the real and imaginary parts, we yield
Adding up the squares of above both equations, we obtain
where
We put \(\omega ^2=\upsilon \) into Eq. (24) and obtain a fourth degree polynomial
By directly calculating, it is easy to show that \(p_1>0\) and \( p_4=(m_4-m_7)(m_4+m_7)>0\) if \(a_4>0\). Furthermore, we also obtain
If \( p_3\ge 0\), we know that Eq. (25) has no positive root. The real parts of all eigenvalues of Eq. (23) remain negative for all values of the delay \(\tau >0.\) Considering the special case of \(\beta _3=0\), we can obtain
Summarizing the above analysis, we have the following theorem.
Theorem 7
Suppose that the conditions in (11), \(R_0>1\) and \(p_3\ge 0\) hold, the infected equilibrium \(E_1\) of system (14–17) is locally asymptotically stable for all \(\tau \ge 0\).
Although we introduce the delay, \(E_1\) is also locally asymptotically stable under some conditions. Under the circumstances, system (14–17) does not undergo Hopf bifurcations.
Theorem 8
If \(R_0> 1\), \(b\beta _2> a\beta _3\) and \(b\lambda _1\beta _1\beta '_2\beta _3\le \beta '_1(b\beta _2- a\beta _3)(c\delta _2-\lambda _2\beta _3)\), the endemic equilibrium \(E_1\) of system (14–17) is globally asymptotically stable for all \(\tau \ge 0\).
Proof
We consider a Lyapunov function
where
Calculating the time derivative of \(L_4\) along the solution of system (14–17), we obtain
We calculate the time derivative of \(U_2(t)\) and \( U_3(t)\),
From system (14–17), we obtain that
Note that
Then we yield
It is known that the function \(f(x)=1-x+{\ln }x\) is always non-positive for \(x>0, \) and \(f(x)=0\) if and only if \(x=1\). Therefore, \(E_1\) is globally asymptotically stable by similar analysis as in Theorem 3. Especially, we know \(E_1\) is global asymptotically stable when \(\beta _3=0\). The proof is completed.
We use the parameter values in Sect. 2 except for \(\beta _3=0.01\). The condition (11), \( p_2>0\), and \( p_3>0\) are all satisfied. We also get \(R_0=3.1034>1\). It is shown that \(E_1 (164.3169, 40.7940, 319.2487, 1.3586\times 10^{3})\) is locally asymptotically stable for \(\tau =1 \) and \( \tau =10\) via simulating results (see Fig. 5). Time delay \(\tau \) does not change the stabilities of the equilibrium \(E_1\).
6 Discussions and conclusions
In the present paper, we propose a mathematical model, which describe the interactions of HIV virus and two target cells. The model can undergo the phenomenon of backward bifurcation if \(R_0<1\). Meanwhile, we find that Hopf bifurcation occurs under some conditions. By using Lyapunov functions, we obtain sufficient conditions for the global asymptotical stabilities of the equilibria. Especially, the two equilibria are globally asymptotically stable in case of \(\beta _3=0\). We cannot ignore the fact that macrophages really influence dynamics behavior of HIV virus from our analysis. What is more, we establish an extended model, and derive that the local asymptotical stabilities of the uninfected and infected equilibria are independent of the size of the delay if \(\beta _3=0\) by analyzing the transcendental characteristic equations. We also derive that the two equilibria are globally asymptotically stable for the delayed model under some conditions.
Finally, some interesting questions deserve further investigation about our model. One may consider the non-bilinear incidence rate for system (5–8). Moreover, we can study the influence of distributed delays not just discrete delays on system (14–17).
References
Adams BM, Banks HT et al (2005) HIV dynamics: modeling, data analysis, and optimal treatment protocols. J Comput Appl Math 184:10–49
Culshaw RV, Ruan S (2000) A delay-differential equation model of HIV infection of CD4+ T-cells. Math Biosci 165:27–39
Duffin RP, Tullis RH (2002) Mathematical models of the complete course of HIV infection and AIDS. J Theor Med 4:215–221
Elaiw AM (2010) Global properties of a class of HIV models. Nonlinear Anal Real World Appl 11:2253–2263
Feng Z, Castillo-Chavez C et al (2000) A model for tuberculosis with exogenous reinfection. Theor Popul Biol 57:235–247
Greenhalgh D (1997) Hopf bifurcation in epidemic models with a latent period and nonpermanent immunity. Math Comput Model 25:85–107
Hadeler KP, Driessche PV (1997) Backward bifurcation in epidemic control. Math Biosci 146:15–35
Hossein P, Sergei SP, Patrick DL (2014) Global analysis of within host virus models with cell-to-cell viral transmission. Discret Contin Dyn Syst Ser B 19:3341–3357
Hu Z, Liu X et al (2010) Analysis of the dynamics of a delayed HIV pathogenesis model. J Comput Appl Math 2:461–476
Huang G, Takeuchi Y, Ma W (2010) Lyapunov functionals for delay differential equations model of viral infection. SIAM J Appl Math 70:2693–2708
Kirschner D (1999) Dynamics of co-infection with M. tuberculosis and HIV-1. Theor Popul Biol 55:94–109
Korobeinikov A (2004) Global properties of basic virus dynamics models. Bull Math Biol 66:879–883
Leenheer PD, Smith HL (2003) Virus dynamics: a global analysis. SIAM J Appl Math 63:1313–1327
Li J, Yang Y, Zhou Y (2011) Global stability of an epidemic model with latent stage and vaccination. Nonlinear Anal Real World Appl 12:2163–2173
Liu W (1994) Criterion of Hopf bifurcations without using eigenvalues. J Math Anal Appl 182:250–256
Lu T, Huang Y et al (2014) A refined parameter estimating approach for HIV dynamic model. J Appl Stat 41:1645–1657
McCluskey CC (2010) Global stability fo ran SIR epidemic model with delay and nonlinear incidence. Nonlinear Anal Real World Appl 11:3106–3109
Ouattara DA (2005) Mathematical analysis of the HIV-1 infection: parameter estimation, therapies effectiveness, and therapeutical failures. In: 27th Annual international conference of the IEEE engineering in medicine and biology society, China, 9
Perelson AS, Essunger P et al (1997) Decay characteristics of HIV-1 infected compartments during combination therapy. Nature 387:188–191
Perelson AS, Nelson PW (1999) Mathematical analysis of HIV-1 dynamics in vivo. SIAM Rev 41:3–44
Qesmi R, Wu J et al (2010) Influence of backward bifurcation in a model of hepatitis B and C viruses. Math Biosci 224:118–125
Regoes RR, Wodarz D, Nowak MA (1998) Virus dynamics: the effect of target cell limitation and immune responses on virus evolution. J Theor Biol 191:340–351
Roy B, Roy SK (2015) Analysis of prey–predator three species models with vertebral and invertebral predators. Int J Dyn Control 3:306–312
Roy SK, Roy B (2016) Analysis of prey–predator three species fishery model with harvesting including prey refuge and migration. Int J Bifurc Chaos 26(1650022):1–19
Roy B, Roy SK, Gurung DB (2017) Holling–Tanner model with Beddington–DeAngelis functional response and time delay introducing harvesting. Math Comput Simul 142:1–14
Sharomi O, Podder CN et al (2007) Role of incidence function in vaccine-induced backward bifurcation in some HIV models. Math Biosci 210:436–463
Shu H, Wang L, Watmough J (2013) Global stability of a nonlinear viral infection model with infinitely distributed intracellular delays and CTL immune responses. SIAM J Appl Math 73:1280–1302
Tsuyoshi K, Toru S Yasuhiro (2015) Construction of Lyapunov functions for some models of infectious diseases in vivo: from simple models to complex models. Math Biosci Eng 12:117–133
Wang X, Wang W (2012) An HIV infection model based on a vectored immunoprophylaxis experiment. J Theor Biol 313:127–135
Wang Y, Zhou Y et al (2009) Oscillatory viral dynamics in a delayed HIV pathogenesis model. Math Biosci 219:104–112
Wang J, Pang J, Kuniya T (2014) Global threshold dynamics in a five-dimensional virus model with cell-mediated, humoral immune responses and distributed delays. Appl Math Comput 241:298–316
Wodarz D, Lloyd AL et al (1999) Dynamics of macrophage and T cell infection by HIV. J Theor Biol 199:101–113
Xia X (2007) Modeling of HIV infection: vaccine readiness, drug effectiveness and therapeutical failures. J Process Control 17:253–260
Yuan Z, Ma Z, Tang X (2012) Global stability of a delayed HIV infection model with nonlinear incidence rate. Nonlinear Dyn 68:207–214
Zhang X, Liu X (2008) Backward bifurcation of an epidemic model with saturated treatment function. J Math Anal Appl 348:433–443
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Florence Hubert.
This work is supported partially by Scientific Research Staring Foundation, Henan Normal University (qd13045).
Rights and permissions
About this article
Cite this article
Liu, Y., Liu, X. Global properties and bifurcation analysis of an HIV-1 infection model with two target cells. Comp. Appl. Math. 37, 3455–3472 (2018). https://doi.org/10.1007/s40314-017-0523-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40314-017-0523-0