Abstract
Background
The concept of pharmaceutical care is operationalized through pharmaceutical professional services, which are patient-oriented to optimize their pharmacotherapy and to improve clinical outcomes.
Objective
The objective of this study was to estimate the incremental cost-effectiveness ratio (ICER) of a medication review with follow-up (MRF) service for older adults with polypharmacy in Spanish community pharmacies against the alternative of having their medication dispensed normally.
Methods
The study was designed as a cluster randomized controlled trial, and was carried out over a time horizon of 6 months. The target population was older adults with polypharmacy, defined as individuals taking five or more medicines per day. The study was conducted in 178 community pharmacies in Spain. Cost-utility analysis adopted a health service perspective. Costs were in euros at 2014 prices and the effectiveness of the intervention was estimated as quality-adjusted life-years (QALYs). In order to analyze the uncertainty of ICER results, we performed a non-parametric bootstrapping with 5000 replications.
Results
A total of 1403 older adults, aged between 65 and 94 years, were enrolled in the study: 688 in the intervention group (IG) and 715 in the control group (CG). By the end of the follow-up, both groups had reduced the mean number of prescribed medications they took, although this reduction was greater in the IG (0.28 ± 1.25 drugs; p < 0.001) than in the CG (0.07 ± 0.95 drugs; p = 0.063). Older adults in the IG saw their quality of life improved by 0.0528 ± 0.20 (p < 0.001). In contrast, the CG experienced a slight reduction in their quality of life: 0.0022 ± 0.24 (p = 0.815). The mean total cost was €977.57 ± 1455.88 for the IG and €1173.44 ± 3671.65 for the CG. In order to estimate the ICER, we used the costs adjusted for baseline medications and QALYs adjusted for baseline utility score, resulting in a mean incremental total cost of −€250.51 ± 148.61 (95 % CI −541.79 to 40.76) and a mean incremental QALY of 0.0156 ± 0.004 (95 % CI 0.008–0.023). Regarding the results from the cost-utility analysis, the MRF service emerged as the dominant strategy.
Conclusion
The MRF service is an effective intervention for optimizing prescribed medication and improving quality of life in older adults with polypharmacy in community pharmacies. The results from the cost-utility analysis suggest that the MRF service is cost effective.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Polypharmacy is a particular concern in older adult populations, and is associated with negative health outcomes. |
Medication review with follow-up (MRF) is a service that attempts to optimize pharmacotherapy, not just by focusing on the process of the use of medication, but also by improving clinical outcomes for older adults. |
The MRF service is an effective intervention for optimizing prescribed medication and improving the quality of life of older adults with polypharmacy in community pharmacies. Compared with usual dispensing, this service is cost effective. |
1 Introduction
In 2013, 18 % of the Spanish population was made up of older adults, conventionally defined as aged 65 or over. The percentage was higher among the female (20 %) than the male population (16 %) [1]. As a result of population aging, this percentage will represent 22 % of the Spanish population in the next 10 years (20 % of males and 24 % of females) [2]. Older adults usually present a wider variety of health problems and therefore make more intensive use of medication than the rest of the population. This often leads to drug-related problems (DRPs) [3] and negative outcomes associated with medication (NOMs) [4].
Polypharmacy is a common phenomenon that increases with age. This increase in medication use is in turn associated with increased morbidity. Although there are a wide range of definitions and different situations associated with this phenomenon [5], the most widespread strategy is to target patients with co-morbidities who are using five drugs or more [6].
Medication is the most widely used healthcare technology for dealing with health problems. In Spain, public expenditure through official prescriptions in the National Health Service (NHS) amounted to €9183 million in 2013 [7]. Although this figure represents a 6 % drop from previous years, such a high level of expenditure requires methods to ensure the rational use of medication, to optimize the results obtained from their use, and to ensure that these medications actually do control health problems. Failures in effectiveness and safety have a cost for the health of patients and also in terms of hospital admissions and accident and emergency (A&E) department visits, appointments with general practitioners, and increased pharmacological treatments.
The concept of pharmaceutical care promotes different pharmaceutical professional services, which are patient-oriented in an attempt to optimize their pharmacotherapy [8, 9]. Medication review with follow-up (MRF) is a service in which the pharmacist evaluates patients’ pharmacotherapies and intervenes in collaboration with the general practitioner and the patients themselves, in order to ensure that therapeutic goals are achieved. One of the main points of the MRF service is that it attempts to optimize pharmacotherapy, not just by focusing on the process of the use of medication, but also by improving clinical results for the patients. When pharmacists play a proactive role in performing medication reviews and in the active education of other healthcare professionals, pharmacotherapy for older patients is improved [10]. However, the evidence of the impact of pharmacists’ interventions on health outcomes, quality of life, or cost effectiveness of care is mixed [11]. Few studies of pharmaceutical care programs for older adults have undertaken a rigorous economic evaluation, and a more standardized approach to data collection is required [12].
Based on this, a national research project, the conSIGUE Program, has been implemented to assess the clinical and economic impact of the MRF service for older adults with polypharmacy in Spanish community pharmacies.
The objective of this study was to estimate the incremental cost-effectiveness ratio (ICER) of a MRF service for older adults with polypharmacy in community pharmacies against the alternative of usual dispensing.
2 Methods
2.1 Study Design
The study was designed as a cluster randomized controlled trial carried out over 6 months of follow-up. It was conducted in community pharmacies in Spain from November 2011 to January 2013. The target population comprised older adults, aged 65 years or over, with polypharmacy, defined as individuals taking five or more medicines per day. These five medicines are only prescription and over-the-counter medicines (officially registered medications).
Sample size was calculated according to the results of a pilot study, conducted in the Spanish province of Cádiz for 1 month. Taking into account the change in the number of medicines used that resulted from the pilot study, 530 patients were required in each group in order to obtain significant results. However, taking into account the cluster design and the confounding variables, 750 patients were required in each group.
The pharmacists of the participant provinces were informed about the conSIGUE Program through the Official College of Pharmacists, and voluntarily signed up to be included in the research study. Pharmacies were allocated into either the intervention group (IG) or the control group (CG). Each pharmacy selected ten patients who met the inclusion criteria: age of 65 years or over and taking five or more officially registered medicines.
Pharmacists allocated into the IG received a 3-day off-site training course and on-site visits by a facilitator during the 6-month follow-up. The functions of the facilitator were assisting pharmacists in the provision of the service and ensuring the quality and homogeneity of the interventions. Pharmacists and patients had follow-up visits every 1.2 months, and pharmacists were not compensated for providing the MRF service in the conSIGUE Program.
The variables recorded to measure the economic and clinical outcomes of the MRF service provided in community pharmacies to older adults with polypharmacy were the number of used medicines, number of uncontrolled health problems, health-related quality of life (HR-QOL), number of visits to A&E departments, number of hospital admissions, and the ICER of the MRF service. The secondary objectives were to describe the prevalence of DRPs and NOMs. Variables were recorded during the visits by the patient to the pharmacy where they interacted with a pharmacist.
The study was approved by the Ethics Committee of the Virgen de las Nieves Hospital in Granada in 2009. All patients signed an informed consent before their inclusion in the study.
The study was carried out taking into account the recommendations of the proposed guidelines for economic evaluation of health technologies in Spain [13], which have a high degree of consistency with CHEERS criteria [14], and using data from the NHS. It estimated all identifiable costs to the NHS and health outcomes in quality-adjusted life-years (QALYs). The alternatives compared were IG, older adults who received MRF service in community pharmacies, and CG, older adults who received usual dispensing in community pharmacies.
2.2 Medication Review with Follow-Up (MRF) Service
The Dader method for the MRF service was developed by the Pharmaceutical Care Research Group at the University of Granada, Granada, Spain [15]. The aim of the MRF service is to detect DRPs and to prevent and resolve NOMs [16]. In the context of this service it is important to establish the conceptual differences between the terms ‘medication-related problem’ and ‘NOM’ [4]. A NOM is the result affecting the health of the patient, which is or may be associated with the use of medications. Older adults in the IG received the MRF service as per the methodology agreed upon by a group of pharmaceutical care experts [3]. Table 1 shows the different phases of the MRF service. All sessions were conducted face to face.
2.3 Control Group
Older adults in the CG received usual dispensing in their community pharmacy.
2.4 Community Pharmacies
The study was performed in 178 community pharmacies of four Spanish provinces: 64 in Guipúzcoa (34 in IG vs. 30 in CG), 42 in Granada (24 in IG vs. 18 in CG), 39 in Santa Cruz de Tenerife (16 in IG vs. 23 in CG) and 33 in Las Palmas de Gran Canaria (14 in IG vs. 19 in CG).
The number of older adults from each province was: 525 in Guipúzcoa (278 in IG vs. 247 in CG), 324 in Granada (194 in IG vs. 130 in CG), 307 in Santa Cruz de Tenerife (115 in IG vs. 192 in CG) and 247 in Las Palmas de Gran Canaria (101 in IG vs. 146 in CG).
2.5 Primary Outcomes
2.5.1 Medication Prescribed
Information on medication prescribed was extracted six times between the beginning and the end of the study from community pharmacy databases, so the measurements were captured every 1.2 months.
2.5.2 Healthcare Resources
We analyzed A&E department visits and hospital admissions. For each older adult, data regarding healthcare resources used during the study were extracted from hospital information system databases for each province [18–20]. In order to analyze the impact of the MRF service, three experts independently evaluated the causes of hospital admissions. They identified hospital admissions related to NOMs that could have been avoided through a MRF service. Names and qualifications of three experts are included in the Acknowledgments section.
2.5.3 Health-Related Quality of Life (HR-QOL)
HR-QOL was measured by the Spanish version of the EuroQol-5D-3L (EQ-5D-3L) questionnaire [21]. This questionnaire was administered in six personal interviews between the beginning and the end of the study, so the measurements were captured every 1.2 months. The EQ-5D-3L describes health status in terms of five dimensions (mobility, self-care, usual activities, pain/discomfort, and anxiety/depression), and each of these is defined with three levels of severity. The scores were used to estimate a utility score, a single index on HR-QOL, which ranges from 1 and 0, where 1 is the best possible state of health and 0 is death. In addition, a visual analog scale (VAS) was used to measure the subjects’ health status with scores at one end of the scale representing the ‘worst’ (0) and scores at the other end representing the ‘best’ (100) health state. QALYs were calculated by using an area-under-the-curve analysis, with linear interpolation of utility scores between six interviews (every 1.2 months, between baseline and 6 months of follow-up) [22].
2.5.4 Costs
We measured costs of the prescribed medication, of the time employed by the pharmacist, of A&E department visits, of hospital admissions, and of the investment in community pharmacy infrastructure and training. Costs were in euros at 2014 prices. Prices from previous years were updated according to the Spanish consumer price index [23]. The following variables were included in the analysis:
-
Time employed by the pharmacist in the training and in the MRF service was calculated for each patient. Pharmacists recorded the time dedicated in each stage of MRF service. In addition, pharmacists’ training was estimated, taking into account the total time of the training and the total number of customers a pharmacist could supply MRF service in 1 year (see Sect. 3.1). The unit cost of the community pharmacist was calculated taking into account the pharmacist’ salary in the Spanish community pharmacy agreement [24] and pharmacist’s annual working time.
-
Medication cost, excluding additional medication associated with hospitalizations, was calculated from the official list of drug prices [25].
-
A&E department visit cost was calculated using prices from the public system [26].
-
Hospital admission cost was calculated using costs from the public system for diagnosis-related groups [27].
-
The investment required to commence the MRF service in community pharmacies and its subsequent maintenance over time was estimated taking into account the following premises: a mean of 2500 customers per community pharmacy [28]; 16 % of customers being older adults with polypharmacy [29, 30]; and 60 % of older adults with polypharmacy accepting the MRF service, according to the results of a pilot study.
2.6 Cost-Utility Analysis
A health service perspective was adopted for the cost-utility analysis. The effectiveness of the intervention was estimated as QALYs. Results of the cost-utility analysis were expressed in terms of the ICER, calculated by dividing the difference in total costs between the IG and the CG by the difference in QALYs between both groups [31]. The total costs included the prescribed medication, A&E department visits, hospital admissions related to NOMs, the pharmacist’s intervention, and the required investment in the community pharmacy. Moreover, taking into account the differences in the number of medications and in basal utility scores, we used the costs adjusted for baseline medications and QALYs adjusted for baseline utility score.
Because the time horizon of the study did not extend beyond 12 months, discounting of costs and QALYs was not necessary. In order to analyze the uncertainty of the ICER results, we performed a non-parametric bootstrapping with 5000 replications [32]. The resulting 5000 ICER replicates were plotted on the cost-effectiveness plane and used to construct a cost-effectiveness acceptability curve.
The cost-effectiveness plane is a graphical way of presenting cost-effectiveness results, with the difference in costs on the vertical axis and the difference in health benefits on the horizontal axis [33]. The acceptability curve represents the proportion of simulations in which the intervention is considered cost effective over a range of values of the threshold cost per QALY [34]. Analyses were carried out with STATA® software, version 12 (StataCorp LP, College Station, TX, USA).
2.7 Statistical Analysis
Descriptive analyses of the primary variables included calculating the mean and standard deviation for the quantitative variables and absolute and relative frequencies for the qualitative variables. Bivariate analyses were performed to evaluate the differences between groups in terms of medication prescribed, healthcare resources, HR-QOL, and costs. Quantitative variables following a normal distribution were analyzed with the Student’s t-test or Mann-Whitney test for those variables non-normally distributed. The qualitative variables were analyzed with Pearson’s Chi-square (χ2) test or the Fisher test. Significance was set at p < 0.05. Prior to estimating the ICER, we adjusted the main variables of the outcome (cost and QALY) through simple linear regression (see the “Technical Appendix”).
3 Results
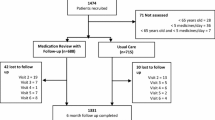
Community pharmacists recruited 1474 older adults, 71 of whom were excluded because they did not meet the inclusion criteria: 28 older adults were not old enough, 36 older adults did not take five or more drugs, and seven older adults were not old enough and did not take five or more drugs (Fig. 1).
A total of 1403 older adults were enrolled, 688 in the IG and 715 in the CG. Table 2 shows the socio-demographic characteristics of the participants.
Both groups of older adults had similar characteristics except for their level of education (27 % in the IG and 18.6 % in the CG had no formal education; p = 0.001) and the mean number of prescribed medications (7.74 drugs in the IG and 7.39 drugs in the CG; p = 0.009). Cost and QALY could not be obtained for 105 participants, who were excluded from the results.
3.1 Intervention Times for the MRF
The mean time employed by the pharmacist in the stages of the MRF service was 442.74 ± 652.24 min: 44.57 ± 29.77 min in the first interview; 40.26 ± 34.24 min for writing the situation assessment; 75.44 ± 87.26 min in the study phase; 39.05 ± 40.60 min in the evaluation phase; 29.93 ± 36.76 min in the therapeutic plan; 17.83 ± 21.67 min in the intervention phase; 162.47 ± 496.03 min for follow-up; and 33.19 ± 36.34 min for additional contacts.
Prior to the intervention, pharmacists were required to complete a 3-day off-site training course with a total duration of 900 min. When the provision of MRF service is on a per annum basis, the total time is calculated through doubling follow-up and additional contacts phases. As a result of this assumption, the mean time employed by the pharmacist in the stages of the MRF service would be 638 min/year. Dividing the annual working time by total minutes to provide MRF service during a year, a pharmacist could supply MRF service to 156 customers/year. This number of patients was used to estimate the training cost associated with each patient.
3.2 Prescribed Medication
Both groups reduced their mean number of prescribed medications (Table 3), although this reduction was greater in the IG (0.28 ± 1.25 drugs; p < 0.001) than in the CG (0.07 ± 0.95 drugs; p = 0.063). The difference in the observed reduction between the two groups was 0.21 ± 0.06 drugs (95 % CI 0.092–0.335).
3.3 Healthcare Resources
Both groups experienced a reduction in the percentage of older adults who at least visited the A&E department once during the 6 months before and during the 6 months of the study, although this reduction was greater in the IG (27.9 vs. 14.2 %; difference 13.7 %; p < 0.001) than in the CG (29.1 vs. 24.9 %; difference 4.2 %; p = 0.044).
The mean number of visits to the A&E department during the 6 months before and the 6 months of the study dropped in both groups, although this reduction was larger among older adults in the IG (0.43 ± 0.83 vs. 0.19 ± 0.51; difference 0.24; p < 0.001) than for older adults in the CG (0.55 ± 1.55 vs. 0.42 ± 1.21; difference 0.13; p < 0.001).
The percentage of participants with at least one hospital admission during the 6 months before and the 6 months of the study dropped among the IG (6.9 vs. 4.1 %; difference 2.8 %; p < 0.001), while this percentage increased among the CG (4.3 vs. 5.1 %; difference 0.8 %; p = 0.044).
This trend continued with the mean number of hospital admissions during the 6 months before and the 6 months of the study dropping among the IG (0.09 ± 0.35 vs. 0.05 ± 0.23; difference 0.04; p = 0.007) and increasing among the CG (0.05 ± 0.25 vs. 0.07 ± 0.36; difference 0.02; p = 0.106).
After the group of experts had reviewed the cause behind each hospital admission, the percentage of older adults with at least one hospital admission dropped to 3.2 % in the IG and 4.3 % in the CG (p = 0.285), with a mean of hospital admissions during the 6 months of the study of 0.03 ± 0.19 in the IG and 0.06 ± 0.31 in the CG. Table 4 shows the mean healthcare resources for the 6 months before and 6 months of the study.
3.4 HR-QOL
Table 4 shows the mean utility scores and VAS scores obtained from the EQ-5D-3L questionnaire at the six interviews. Participants in the IG reported an improvement in their quality of life of 0.0528 ± 0.20 in the utility score (p < 0.001) and 4.97 ± 15.29 in the VAS score (p < 0.001). In contrast, those in the CG saw a slight reduction in their quality of life of 0.0022 ± 0.24 in the utility score (p = 0.815) and 0.90 ± 15.19 in the VAS score (p = 0.127). The difference observed between both groups was 0.0550 ± 0.01 in the utility score (95 % CI 0.0306–0.0794) and 5.87 ± 0.85 in the VAS score (95 % CI 4.20–7.54).
The mean QALY scores, corresponding to the 6 months of the study, were 0.3721 ± 0.12 and 0.3488 ± 0.15 for the IG and CG, respectively, resulting in an incremental QALY score of 0.0233 (p = 0.002), largely due to the difference in utility scores at baseline (0.7148 vs. 0.6953 score for the IG and the CG, respectively).
3.5 Costs
A mean cost of €98.35 ± 143.03 was estimated for the time employed in the pharmacists’ interventions, including the training previous to the intervention. The mean cost of the prescribed medication for the 6 months was €655.91 ± 818.53 for the IG and €657.67 ± 600.09 for the CG. The IG saw a reduction in the mean daily cost of prescribed medication, while the CG experienced a slight increase (Table 5). Pharmacists’ interventions saved a mean medication cost of €0.17/day (p = 0.057), while in the CG there was an increase in the mean cost of €0.02/day (p = 0.774). The difference in the reduction observed between both groups was €0.19/day (p = 0.079).
Participants in the CG reported a greater consumption of healthcare resources, both in terms of visits to the A&E department and of hospital admissions. The mean A&E department visit cost was €33.05 ± 90.98 for the IG and €74.18 ± 213.93 for the CG (difference of €41.12; p < 0.001), and the mean hospitalization cost was €215.52 ± 1263.93 for the IG and €496.79 ± 3720.07 for the CG (difference of €281.27; p = 0.065). After excluding admissions for causes not related to NOMs, the mean hospitalization cost was €173.99 ± 1184.95 for the IG and €441.60 ± 3573.71 for the CG (difference of €267.61; p = 0.067). The mean healthcare cost was €248.58 ± 1285.76 for the IG and €570.97 ± 3765.75 for the CG (difference of €322.40; p = 0.037) and, after excluding admissions for causes not related to NOMs, €207.04 ± 1207.20 for the IG and €515.77 ± 3621.15 for the CG (difference of €308.73; p = 0.037). Finally, the mean cost estimated as the required investment was €4688.47 for a period of 5 years and a mean annual maintenance cost of €2967.02, resulting in a mean cost of €16.27 per person. Table 6 shows each category of total mean cost.
3.6 Cost-Utility Analysis
The total mean total cost was €977.57 ± 1455.88 for the IG and €1173.44 ± 3671.65 for the CG. To estimate the ICER, we used the costs adjusted for baseline medications and QALYs adjusted for baseline utility score, resulting in a mean incremental total cost of –€250.51 ± 156.82 (95 % CI −558.17 to 57.14) and a mean incremental QALY of 0.0156 ± 0.004 (95 % CI 0.008–0.023). Regarding the results from the cost-utility analysis, the MRF service emerged as the dominant strategy.
Based on 5000 bootstrap replications, most of the bootstrap simulations are located in the lower-right quadrant (96.8 %) and in the upper-right quadrant (3.2 %) of the cost-effectiveness plane (Fig. 2). The acceptability curve shows that if the willingness to pay is between €30,000/QALY and €45,000/QALY, the probability of the MRF service being cost effective, compared with usual dispensing, is 100 % (Fig. 3).
4 Discussion
Morbidity and mortality issues related to medication represent an important health and economic problem, which is particularly significant among older adults. This study suggests that an MRF service carried out in pharmacies by pharmacists is an efficient technique for dealing with a health problem that is increasingly prevalent in aging societies.
Pharmacists spent a mean of 443 min in the MRF service, which resulted in a cost of €98. If the MRF service was extended to 1 year, only the follow-up and additional contacts phases would have to be extrapolated to annual values. Pharmacists devoted an average of 199 min to the initial phases of the service: interview, situation assessment, study phase, and evaluation phase. These periods of time may indicate that the phases of the MRF service were conducted with rigor. The time employed in the implementation of the interventions was less than that registered in the previous phases, which seems to suggest an appropriate interaction with other health professionals. The aim of the MRF service is not only to reduce the consumption of medication used by the patient, but to improve the clinical results associated with the use of said medication. To achieve this objective, the pharmacist who performs the MRF service suggests interventions, which may or not be drug related. It is logical, therefore, that the number of drugs is reduced only in a few cases.
The effectiveness of services similar to the MRF service shows contradictory results in relation to their effect on the number of drugs [35]. In this study, we saw a drop in the number of drugs for both groups, although this tendency was greater among the older adults in the IG (0.28 drugs; p < 0.001) than for those in the CG (0.07 drugs; p = 0.063). In a recent study of an MRF service for nursing home residents in Spain [36], pharmacist interventions reduced the mean number of prescribed medications by 0.47 drugs (p < 0.001), whereas the mean number of prescribed medications increased by 0.94 drugs in the CG (p < 0.001). Participants were residents aged 65 years or over, but their health state was worse than in the case of the participants in the present study, and this may explain why that intervention was more effective in optimizing prescribed medication.
As for the impact of an MRF service on the consumption of healthcare resources, in the IG there was a reduction in the percentage of participants who visited A&E departments and who were admitted to hospital compared with the 6 months before the study. Among the CG there was also a reduction in the number of participants who visited A&E departments, although this was less pronounced than in the IG. There was an increase in the percentage of older adults who were admitted at least once to hospital. However, after reviewing the causes of the hospital admissions and ruling out those that could not have been avoided with this intervention, the percentage of older adults who were admitted at least once to hospital was the same before and during the study. Differences in A&E and hospitalizations between groups are very small but significant differences were found primarily due to the large sample size.
The results of this study point to a positive effect of an MRF service on HR-QOL. Older adults who received the MRF service benefited from a significant increase in their quality of life as measured through the EQ-5D-3L questionnaire. One possible explanation for this could be linked to the high degree of personal contact between participants and pharmacists during the implementation of the MRF service. This was higher than the usual contact and may have had a great impact on the quality of life as perceived by the older adults in the study. Furthermore, it is only logical that better control of health problems is going to be reflected in an improvement in quality of life. In contrast, this improvement was not mirrored in the CG, where there was a decrease in their HR-QOL. Moreover, the CG had significantly more mobility problems than the IG and this could perhaps have contributed to the lack of quality-of-life improvements across the CG because mobility is a strong predictor of quality of life. The utility decrements in the CG were unexpected because we applied a random methodology for the selection of pharmacies. The difference in the utility scores between the two groups might be a consequence of the greater mobility problems in the CG and the improving quality of life in the IG. We believe that higher control of the IG due to the MRF service and greater personal inter-relationships between pharmacists and patients might have influenced this results. Likewise, this can lead to improvements in perceived health that are higher than those strictly derived from the MRF service. These emotional improvements for the MRF service should be analyzed in future research.
In Spain there is no official threshold of cost/QALY. With €30,000/QALY [37] or €30,000–45,000/QALY [38] being the threshold for determining whether a health technology is cost effective, we have determined that an MRF service for older adults with polypharmacy in community pharmacies is cost effective compared with usual dispensing. We estimated that an MRF service was a dominant intervention (less costly and more effective than usual dispensing) and the acceptability curve showed that there was little uncertainty, due to the fact that 100 % of the bootstrap simulations were below €30,000/QALY and €45,000/QALY.
Jódar-Sánchez et al. [36] evaluated the cost effectiveness of an MRF service for older adult residents in nursing homes in Spain, obtaining an adjusted ICER of €6574/QALY (in the more realistic scenario). The decreased results obtained in this study may be due to the fact that, rather than adopting the perspective of the NHS, it considered a more restrictive perspective, focusing on the variation in direct costs of medication and pharmacists’ time.
This study presents some limitations. First, although a cluster randomized trial is a recommended design [39], the main limitation of this study was the non-random selection of the sample of older adults. Randomizing the pharmacies instead of the participants was intended to ensure that the same pharmacist did not deliver the service to participants from both groups, which could have led to problems of selection bias, ethical conflicts, and potential problems of contamination between groups. Second, the cost of outpatient physician visits was not included, as this information was not available.
Despite these limitations, the study is important for several reasons. It strengthens the limited international evidence on the cost effectiveness of MRF service (or pharmaceutical care, in general) for improving the effectiveness and security of polypharmacy care for older adults in community pharmacies.
In Spain this type of service is not offered by the NHS or by any regional health service in the autonomous regions, yet the results of the study suggest high social benefits, particularly in relation to the number of potentially avoided A&E department visits and hospital admissions. The incorporation of this technique into the portfolio of services offered by the NHS, or by other health services committed to a universal system open to all citizens, provides options for policy decision makers. Adopting this strategy would be beneficial due to its potential to improve the efficiency of the treatment of older adults with polypharmacy, specifically because this population will inevitably grow in importance when population aging increases in most developed countries.
5 Conclusion
The MRF service is an effective intervention for optimizing prescribed medication and improving the quality of life of older adults with polypharmacy in community pharmacies. The results from the cost-utility analysis suggest that the MRF service is cost effective.
References
Spanish Statistical Office. Population figures. http://www.ine.es/en/inebaseDYN/cp30321/cp_inicio_en.htm. Accessed 7 Feb 2014.
Spanish Statistical Office. Short-term projections of the population. http://www.ine.es/jaxi/menu.do?type=pcaxis&path=%2Ft20%2Fp269&file=inebase&L=1. Accessed 7 Feb 2014.
Pharmaceutical Care Forum, expert panel. Consensus Document. January 2008. Ed. Consejo General de Colegios Oficiales de Farmacéuticos, Madrid. ISBN 978-84-691-1243-4.
Committee of Consensus. Third Granada’s consensus on drug-related problems (DRP) and negative outcomes associated with medication [in Spanish]. Ars Pharm. 2007;48(1):5–17.
Rollason V, Vogt N. Reduction of polypharmacy in the elderly: a systematic review of the role of the pharmacist. Drugs Aging. 2003;20(11):817–32.
Monane M, Monane S, Semla T. Optimal medication use in elders. Key to successful aging. West J Med. 1997;167(4):233–7.
Ministry of Health, Social Services and Equality. Medical prescription invoicing data. https://www.msssi.gob.es/profesionales/farmacia/datos/diciembre2013.htm. Accessed 7 Feb 2014.
Hepler CD, Strand LM. Opportunities and responsibilities in pharmaceutical care. Am J Hosp Pharm. 1990;47(3):533–43.
Cipolle J, Strand LM, Morley PC. A reimbursement system for pharmaceutical care: pharmaceutical care practice. New York: McGraw-Hill; 1998.
Patterson SM, Hughes C, Kerse N, Cardwell CR, Bradley MC. Interventions to improve the appropriate use of polypharmacy for older people. Cochrane Database Syst Rev. 2012;5:CD008165.
Spinewine A, Fialova D, Byrne S. The role of the pharmacist in optimizing pharmacotherapy in older people. Drugs Aging. 2012;29(6):495–510.
Crealey GE, Sturgess IK, McElnay JC, Hughes CM. Pharmaceutical care programmes for the elderly: economic issues. Pharmacoeconomics. 2003;21(7):455–65.
Lopez-Bastida J, Oliva J, Antonanzas F, García-Altes A, Gisbert R, Mar J, et al. A proposed guideline for economic evaluation of health technologies. Gac Sanit. 2010;24(2):154–70.
Husereau D, Drummond M, Petrou S, Carswell C, Moher D, Greenberg D, et al. Consolidated health economic evaluation reporting standards (CHEERS) statement. Pharmacoeconomics. 2013;31(5):361–7.
Pharmaceutical Research Group of the University of Granada. Dader method to provide pharmacotherapy follow up. Ars Pharm. 2005;46(4):309–35.
Sabater Hernández D, Silva Castro MM, Faus MJ. Dáder method: pharmacotherapy follow-up guide. 3rd edn. Granada: Grupo de Investigación en Atención Farmacéutica. Universidad de Granada; 2007.
Spanish General Council of Official Colleges of Pharmacists. Bot PLUS web database. https://botplusweb.portalfarma.com/. Accessed 7 July 2013.
Andalusian Health Service. Regional Minister of Equality, Health and Social Policies. http://www.juntadeandalucia.es/servicioandaluzdesalud/principal/default.asp. Accessed 7 July 2013.
Basque Country Health Service (Osakidetza). http://www.osakidetza.euskadi.net/r85-ghhome00/es. Accessed 7 July 2013.
Islas Canarias Health Service. Regional Ministry of Health. http://www.gobiernodecanarias.org/sanidad/scs/. Accessed 7 July 2013.
Badia X, Roset M, Montserrat S, Herdman M, Segura A. The Spanish version of EuroQol: a description and its applications. Med Clin (Barc). 1999;112(Suppl 1):79–85.
Manca A, Hawkins N, Sculpher MJ. Estimating mean QALYs in trial-based cost-effectiveness analysis: the importance of controlling for baseline utility. Health Econ. 2005;14(5):487–96.
National Statistics Institute. Spanish Consumer Price Index. http://www.ine.es/en/inebmenu/mnu_nivel_vida_en.htm. Accessed 13 Nov 2013.
Spanish Official State Gazette. 2014; No. 112, Sec. III: p. 35242. http://www.boe.es/boe/dias/2014/05/08/pdfs/BOE-A-2014-4846.pdf. Accessed 12 Oct 2014.
Spanish General Council of Official Colleges of Pharmacists. Catalogue of medicines. 1st ed. Madrid: Spanish General Council of Official Colleges of Pharmacists; 2009.
Order of 14 October 2005, which are priced public health services provided by centers dependent of Andalusian Public Health System. Official Gazette of the Government of Andalusia. 2005;210.
Ministry of Health, Social Services and Equality. Analysis and development of the DRGs in the National Health System. Year 2010. http://www.msssi.gob.es/estadEstudios/estadisticas/inforRecopilaciones/anaDesarrolloGDR.htm. Accessed 7 July 2013.
Statistics Collegiate and Community Pharmacies. Spanish General Council of Official Colleges of Pharmacists. http://www.portalfarma.com/Profesionales/infoestadistica/Documents/Estadisticas2012.pdf. Accessed 7 Feb 2015.
Abellán A, Esparza C. A profile of the elderly in Spain. 2011. Basic statistical indicators. Madrid, Informes Portal Mayores no. 127. http://digital.csic.es/bitstream/10261/107721/1/pm-indicadoresbasicos11-1.pdf. Accessed 7 Feb 2015.
Cosby RH, Howard M, Kaczorowski J, Willan AR, Sellors JW. Randomizing patients by family practice: sample size estimation, intracluster correlation and data analysis. Fam Pract. 2003;20(1):77–82.
Drummond MF, O´Brien B, Stoddart GL, Torrance GW. Methods for the economic evaluation of health care programmes, vol. 3. Oxford University Press: Oxford; 2005.
Briggs AH, Wonderling DE, Mooney CZ. Pulling cost-effectiveness analysis up by its bootstraps: a non-parametric approach to confidence interval estimation. Health Econ. 1997;6(4):327–40.
Black WC. The CE plane: a graphic representation of cost-effectiveness. Med Decis Making. 1990;10(3):212–4.
Fenwick E, O’Brien BJ, Briggs A. Cost-effectiveness acceptability curves: facts, fallacies and frequently asked questions. Health Econ. 2004;13(5):405–15.
Sáez-Benito L, Fernández-Llimos F, Feletto E, Gastelurrutia MA, Martínez-Martínez F, Benrimoj SI. Evidence of the clinical effectiveness of cognitive pharmaceutical services for aged patients. Age Ageing. 2013;42(4):442–9.
Jódar-Sánchez F, Martín JJ, López Del Amo MP, García L, Araujo-Santos JM, Epstein D. Cost-utility analysis of a pharmacotherapy follow-up for elderly nursing home residents in Spain. J Am Geriatr Soc. 2014;62(7):1272–80.
Sacristán JA, Oliva J, Del LJ, Prieto L, Pinto JL. What is an efficient health technology in Spain? Gac Sanit. 2002;16(4):334–43.
De Cock E, Miravitlles M, González-Juanatey JR, Azanza-Perea JR. Valor umbral del coste por año de vida ganado para recomendar la adopción de tecnologías sanitarias en España: evidencias procedentes de una revisión de la literatura. Pharmacoecon Span Res Artic. 2007;4(3):97–107.
Campbell MK, Piaggio G, Elbourne DR, Altman DG. Consort 2010 statement: extension to cluster randomised trials. BMJ. 2012;345:e5661.
Acknowledgments
We thank all of the pharmacists who participated in the conSIGUE project. From Puerto Real University Hospital (Cádiz), we would like to thank Andrés Rabadán Asensio, Head of Public Health Department, and the three experts who evaluated the causes of hospital admissions: María José Pedrosa Martínez (specialist in Clinical Pharmacology), Julio González-Outón Velázquez (specialist in Preventive Medicine), and Emilio Jesús Alegre del Rey (specialist in Hospital Pharmacy).
The authors acknowledge health managers from the autonomous communities their collaboration.
Funding sources
We acknowledge and thank the financial support of the Spanish General Council of Official Colleges of Pharmacists and of CINFA Laboratory.
Disclosures
The authors have no conflicts of interest.
Author contributions
FM-M and SIB acquired funding and coordinated the study. AM-L, FM-M, MAG-G, VG-C, DS-H, LS-B, and SIB designed the study and database. AM-L registered the data. FJ-S, JJM, LG-M, and MPLA analyzed the data and interpreted the results. All authors participated in the preparation of the manuscript. JJM is the guarantor for the overall content of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Technical Appendix
Technical Appendix
1.1 Regression-Based Adjustment
-
1.
This approach allows the estimation of differential total cost, as well as the prediction of adjusted total cost, while controlling for baseline prescribed medications. The equation is:
$$ {\text{Cost}}_{i} = \beta_{0} + \beta_{1} \times T_{i} + \beta_{2} \times M_{i} $$where index i is the patient identifier (i = 1, 2, …, N); T i is the treatment arm dummy variable (0 = control; 1 = MRF service); and M i is the patient-specific baseline prescribed medication.
The coefficient β 1 represents the adjusted differential cost after controlling for imbalance in the mean prescribed medication at baseline.
Results:
$$ \begin{aligned} {\text{Cost}}_{i} = \, 255.18 \, {-} \, 250.51 \times T_{i} + \, 125.41 \times M_{i} \hfill \\ R{\text{-squared}} = 0.013 \hfill \\ \end{aligned} $$ -
2.
This approach allows the estimation of differential QALYs, as well as the prediction of adjusted QALYs, while controlling for baseline utility score. The equation is:
$$ {\text{QALY}}_{i} = \, \beta_{0} + \, \beta_{1} \times \, T_{i} + \, \beta_{2} \times \, U_{i} $$where index i is the patient identifier (i = 1, 2, …, N); T i is the treatment arm dummy variable (0 = control; 1 = MRF service); and U i is the patient-specific baseline utility score.
The coefficient β 1 represents the adjusted differential QALY after controlling for imbalance in the mean utility at baseline.
Results:
$$ \begin{aligned} {\text{QALY}}_{i} &= \, 0.077 \, + \, 0.016 \times T_{i} + \, 0.390 \times U_{i} \hfill \\ R{\text{-squared}}\;&= \;0.745 \hfill \\ \end{aligned} $$The regression-based approach not only generates an unbiased estimate of differential cost and QALYs between the arms of the study, but also increases the precision of the treatment effect estimate.
Rights and permissions
About this article
Cite this article
Jódar-Sánchez, F., Malet-Larrea, A., Martín, J.J. et al. Cost-Utility Analysis of a Medication Review with Follow-Up Service for Older Adults with Polypharmacy in Community Pharmacies in Spain: The conSIGUE Program. PharmacoEconomics 33, 599–610 (2015). https://doi.org/10.1007/s40273-015-0270-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40273-015-0270-2