Abstract
Objectives
Optimal care of rheumatoid arthritis (RA) patients entails regular assessment of disease activity and appropriate adjustment of disease-modifying antirheumatic drugs (DMARDs) until a predefined treatment goal is achieved. This raises questions about the approach to treatment decision making among RA patients and their preference for associated treatment changes. We aimed to systematically identify and synthesize the available evidence of RA patients’ preferences regarding DMARD modification with an emphasis on escalating, tapering, stopping, or switching of DMARDs.
Methods
A scoping review was undertaken to gauge the breadth of evidence from the range of studies relating to RA patients’ preferences for DMARD modification. Pertinent databases were searched for relevant studies published between 1988 and 2019. Conventional content analysis was applied to generate themes about how patients perceive changes to their RA treatment.
Results
Of the 1730 distinct articles identified, 32 were included for review. Eight studies investigated RA patients’ perceptions of switching to other DMARDs, 18 studies reported RA patients’ preferences for escalating treatment, and six studies explored the possibility of tapering or stopping of biologic DMARDs. Four overarching themes relating to RA patients’ preferences for treatment modification were identified: (i) patient satisfaction, (ii) patients’ beliefs, (iii) information needs, and (iv) patient–clinician relationships.
Conclusion
Uptake of treatment changes in clinical practice can be improved by understanding how RA patients approach the decision to modify their treatment and how this relates to their satisfaction, beliefs, information needs, and relationships with clinicians. Future work is needed to systematically determine the significance of these factors in RA patients’ decision-making processes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Rheumatoid arthritis patients’ approach to treatment decision making and their perspectives towards disease-related outcomes differs to those of clinicians. Decisions that are shared between RA patients and clinicians lead to better disease response and higher patient satisfaction. |
Patients draw upon self-reported satisfaction, past experiences, self-belief, and possible future impact when considering treatment modification. A strong patient–clinician relationship and meeting patients’ needs for information influences the social context of decision making among RA patients. |
Understanding the underlying reasons for RA patients’ preference for treatment modification and addressing their concerns may improve the uptake of treatment changes in clinical practice. |
1 Introduction
Rheumatoid arthritis (RA) is a chronic debilitating disease characterized by pain, inflammation, and potential erosion of the joints and resultant reduced quality of life [1]. It is one of the most prevalent chronic inflammatory diseases, affecting 0.5–1.0% of the population [2]. The primary therapeutic goal in RA is to attain a state of clinical remission or low disease activity (LDA), which typically can only be achieved through the use of disease-modifying antirheumatic drugs (DMARDs). At present, a wide array of DMARDs are available for the treatment of RA, including conventional synthetic DMARDs (csDMARDs), biologic DMARDs (biologics), and targeted synthetic DMARDs (tsDMARDs) [3]. Biologics now include the originator biologic and ‘generic’ biologics known as biosimilar agents [4].
Optimal care of people with RA entails regular assessment of disease activity and appropriate adjustment of medications until the predefined treatment goal is achieved [5]. Clinical trials have demonstrated that tight disease control significantly improves clinical and radiographic outcomes of RA [6, 7] and is associated with higher rates of remission in clinical practice [8]. Tight disease control requires systematic escalation of DMARD treatment either by increasing DMARD dose, combination use with prednisone and/or other DMARDs, or switching DMARDs when RA is not well controlled. For patients who achieve sustained remission for at least 6 months, a reduction in DMARD dose/frequency can be considered to reduce medication burden [9]. Considering the high cost of biologics, tapering or switching to less expensive biosimilars may be an attractive option to reduce individual, medical and societal health care costs if they are equally effective [10].
Providing patient-focused care that is responsive to patient preferences is paramount to achieving therapeutic success [11]. Previous studies have shown the approach to treatment decision making, as well as perspectives on disease-related outcomes among RA patients, differ substantially between patients and clinicians [12, 13]. Therefore, the decision to modify treatment to achieve better disease control and improve RA patients’ quality of life should ideally be aligned with both clinicians’ recommendations and patients’ preferences [14]. Decisions that are shared between people with RA and their clinicians are associated with good disease response [15], overcoming patients’ resistance towards treatment changes [16], and can lead to better health outcomes with regards to treatment adherence and satisfaction [17]. Patients’ values and preferences are advocated as essential elements of decision making and are acknowledged in international guidelines for treating RA patients [18, 19].
Previous literature reviews have focused on describing patients’ experiences of DMARD treatment, and preferences for DMARD, health states, and DMARD-related treatment outcomes [20, 21]. Based on our literature search, no systematic or scoping reviews have examined patients’ values and preferences surrounding treatment modification in RA. Therefore, the aim of this review was to systematically identify and synthesize the available evidence of RA patients’ preferences towards DMARD modification, with an emphasis on escalating, tapering, stopping, or switching of DMARDs. A secondary aim of this review was to determine factors influencing RA patients’ decision making when considering treatment modification. The notion of preference varies between disciplines and can be regarded as a quantitative valuation of outcomes [22] or construction of a utility function based on selection among a set of alternatives [23]. For this review, preference was defined in a broad sense and encompasses not only patients’ perspectives, but also their attitude towards health and health care [24].
2 Methods
A scoping review was undertaken to map the existing body of literature [25] to provide the breadth of evidence from a range of studies but not necessarily the depth that is consistent with a systematic review [26]. A scoping review is appropriate for our research questions because the scope of our review covers a wide range of factors associated with treatment modification. Guided by Arkey and O’Malley’s framework for conducting scoping reviews [27] and further refined by the Joanna Briggs Institute [28], this review followed five iterative stages: (1) identifying the research question, (2) identifying relevant studies, (3) selecting suitable studies, (4) extracting the data, and (5) collating and summarizing the results.
2.1 Research Questions
The research questions applied in this review were intentionally broad to capture a wide breadth of literature relating to RA patients’ preferences for treatment modification encompassing initiation of a new medication, switching to an alternative medication, altering the dose, or stopping a current medication. The term ‘medication’ refers to any DMARD used to treat rheumatoid arthritis, including csDMARDs, biologics, tsDMARDs, or biosimilars. The Patient, Intervention, Comparison, and Outcome (PICO) strategy was used to formulate our research questions and to identify the keywords that were used in the next stage of the review:
-
a.
What are the preferences of RA patients regarding the modification of their DMARD regimen?
-
b.
What are the key factors affecting RA patients’ decision making for DMARD modification?
2.2 Search Strategy
Following the finalization of keywords, a search strategy was developed in collaboration with an academic librarian. Medical Subject Heading (MeSH) terms and Boolean operators were used to narrow, widen, and combine the search (see Appendix 1 in the electronic supplementary material [ESM]). We used five diverse databases (PubMed, EMBASE, PsycINFO, CINAHL, and Web of Science) to identify potentially relevant studies. Complementing the database search were four grey literature databases: Google Scholar, Proquest dissertation and theses, Ethos, and OpenGrey using keywords as search filters. We also manually scanned the reference lists from included studies for potentially relevant studies.
2.3 Study Selection
Studies were included if they fulfilled the inclusion and exclusion criteria as outlined below.
Inclusion criteria:
-
Population (P): Patients age 18 years and above with RA. Studies involving a mixed population of patients with rheumatic diseases were accepted if patients with RA were included.
-
Intervention (I): Treatment modification surrounding DMARDs use in the treatment of RA. Treatment modification includes escalation, tapering, stopping, or switching of DMARDs.
-
Comparator (C): Continuing on current DMARD treatment.
-
Outcome (O): Patient values, preferences, or attitudes related to treatment modification.
-
Study type (S): Original, peer-reviewed studies of any design published in English only.
-
Full-text studies published between Jan 1, 1988 to Dec 31, 2019.
Exclusion criteria:
-
Studies describing patient-reported outcome measures of health-related quality of life only.
-
Studies assessing patients’ preferences about non-DMARD modifications including non-steroidal anti-inflammatory drugs (NSAIDs) or glucocorticoids.
-
Studies assessing patients’ perspectives towards non-pharmacological interventions in the management of RA which include, but are not limited to psychological therapies, physical activity/exercise, or complementary/alternative medicine.
-
Studies evaluating patients’ preferences for medication attributes as these were assessed in an existing systematic review [20].
-
Studies only published as abstracts or conference proceedings.
-
Studies describing treatment modification due to DMARD-induced adverse effects.
-
Studies describing patients’ self-adjustment of DMARD.
The references were first screened by title and abstract independently by two reviewers (JC and HY). If the eligibility of the study could not be determined from the title or abstract, the full-text article was retrieved and assessed. The final step before the full-text review involved a comparison of the screened results between the two reviewers. Any discrepancies were resolved through discussion until consensus was achieved.
2.4 Data Extraction and Synthesis
Data elements included were chosen through discussion between two authors (JC and HY), drafted, and revised iteratively throughout the stages of the review. The finalized data extraction form was designed to capture identifiers and variables including author(s) and year of publication, country of origin, sample size and characteristics (e.g., mean age and/or age range, proportion of males/females), study type, methodology, study objective, and key findings relevant to the research questions. All study data were extracted by JC, while HY validated the accuracy of the data by cross-checking against the included studies.
2.5 Collating and Summarizing the Results
Data collation and summarization was conducted in two steps. First, descriptive statistics were used to summarize studies by methodology and types of treatment changes. No inferential statistical testing or meta-analysis was performed due to the small numbers of studies using specific methods. Subsequently, a conventional content analysis was performed on all the data, and themes were developed to summarize study findings [29]. Conventional content analysis is appropriate when it is used to describe a phenomenon without using predetermined categories or theoretical perspectives, as is the case for this study [29]. All study data were read repeatedly to obtain a sense of the whole body of literature. Next, codes, which constituted a word or short phrases identifying features of data relevant to the research questions, were extracted and inductively sorted into categories based on how different codes were related and linked. Within each category were themes that represent how participants perceived changes to their RA treatment. This process was completed by JC and reviewed for appropriateness to the theme by two independent reviewers (HY and CM). Any differences were resolved through discussion among these three authors.
3 Results
3.1 Search Results
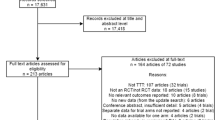
An initial electronic search of five databases yielded 1801 articles. An additional 289 studies were identified through searches of the grey literature for a total of 2090 articles. The screening for duplicates removed 360 articles, and another 1591 articles were excluded after title and/or abstract screening. Full texts of the remaining 139 articles were retrieved for further review, of which 107 articles were excluded leaving 32 studies included in the review. The main reasons for exclusion were studies eliciting patient preference for medication attributes only (n = 23), patient-reported outcomes (n = 19), and not involving treatment modification (n = 17). The study flow diagram is illustrated in Fig. 1.
3.2 Description of Included Studies
The 32 eligible studies are summarized in Table 1. A total of 20 quantitative studies were identified, together with 10 qualitative studies and two studies using mixed methods. Eight studies investigated RA patients’ perspectives on switching to DMARDs of a different route of administration [30,31,32,33,34,35,36,37], 18 studies reported RA patients’ perspectives for escalating RA treatment [38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55], and six studies explored the possibility of tapering or stopping biologics [56,57,58,59,60,61]. These studies were grouped into three broad categories that represent (i) switching medication, (ii) escalation of treatment, and (iii) tapering or stopping medication. A sub-analysis of findings that were unique to each category is presented separately in Table 2.
3.3 Themes
Four broad themes related to RA patients’ perspectives towards treatment modification were identified: (i) patient satisfaction, (ii) patients’ beliefs, (iii) information needs, and (iv) patient–clinician relationships. These themes and the constituent subthemes and identifying codes are summarized in Table 3.
Theme 1: Patient Satisfaction Satisfaction with current treatment or health state strongly influences RA patients’ preferences towards treatment changes in past research. Patients who perceive their disease to be stable [38,39,40,41, 48, 51, 52, 59] or were satisfied their current treatment was working [37, 41, 52, 56] preferred to maintain their current treatment. They did not perceive a need to add or change their treatment [42, 52], which may risk losing the currently acceptable health state [53, 61]. In particular, one study found that patients who were satisfied with RA control were almost seven times more unwilling to change therapy than those not satisfied with their disease control [52]. In contrast, patients who experienced persistent RA symptoms and declining functional capacity affecting their current quality of life were more willing to accept treatment modification [43, 46]. The more severe the RA symptoms were perceived by patients (pain and fatigue), the more likely patients were to change their treatment [42, 52, 54].
Three studies found discrepancies between patient-reported satisfaction and assessment of disease activity in RA [46, 51, 52]. Takahashi et al. reported more than half of RA patients wanted to remain on current therapy and were satisfied with their current disease state despite having moderate to high disease activity [51]. Raczkiewicz et al. found a majority of RA patients (74.5%) declared satisfaction with therapy but only 44% of their sample had low disease activity or were in remission as defined by Disease Activity Score-28 (DAS28) [46]. Wolfe and Michaud discovered that of RA patients who reported being satisfied with their current treatment, 71.3% had moderate or high disease activity levels [52]. It therefore appears that patient-rated satisfaction does not depend on disease activity alone, but also on other factors such as perceived functional status and patients’ age [51, 52].
Theme 2: Patients’ Beliefs Patients’ health beliefs and their impact on decision making are critical in the understanding of patients’ receptiveness towards treatment changes in past research on RA. Three sub-themes were generated to illustrate the broad concept of patients’ beliefs, namely perception about medications, perception towards the consequences of change, and past experiences.
Perception about medications RA patients’ perceptions of medications and treatment modification have been found to range between two extremes. At one end, patients view DMARDs as a necessity as they provide symptom relief and/or prevention of joint damage [55]. At the other end, medications have been described as ‘poison’, unnatural, and toxic chemical substances that should be avoided [53, 54]. In addition, the action of taking medication is associated with the identity of being sick and of being a patient with a chronic, serious illness, influencing patients’ willingness to accept new treatments [53, 54]. DMARDs, in particular, are perceived as aggressive and harmful treatments [55]. However, patients often tolerate DMARDs as a ‘necessary evil’ to relieve RA symptoms [53] but often have strong desires to reduce DMARDs to alleviate medication burden [61].
For RA patients considering switching, escalating, or tapering treatment, information about the efficacy [48, 49], safety, and adverse effects profile [35, 38, 42, 48, 49, 51, 52, 57], and potential long-term use issues such as the risk of a weaker immune system and risk of developing cancer over time [48, 55] have been found to be equally important. For example, Funahashi and Matsubara found RA patients wanted to know about the types and frequency of adverse effects before starting a new treatment [49]. The route of drug administration was also mentioned as a potential barrier to the uptake of new treatment [35, 48]. Studies exploring RA patients’ perspectives towards biosimilars have revealed that patients generally prefer bio-originators over biosimilars for their perceived superior drug attributes (e.g., efficacy and safety) [30,31,32,33,34]. Frantzen et al. reported one of RA patients’ major concerns about biosimilars was the perception of inferior quality compared with the bio-originator [31], whereas van Overbeeke et al. reported patients are concerned about the suitability of biosimilars in treating RA [33].
Perception of the consequences of change RA patients were more likely to accept changes to their treatment if they perceived the change would significantly improve their current condition and future prognosis [40, 41, 43, 44, 47, 49, 53, 60]. The desire to return to normality has been found to be a recurrent theme motivating RA patients to accept treatment changes [35, 38, 42, 53, 55,56,57, 60, 61]. Some RA patients are even willing to accept potential adverse effects associated with treatment and did not mind taking large amounts of medication if this could improve their quality of life [38, 43, 53]. RA patients may be discouraged from accepting the proposed treatment changes when the risks of treatment are perceived to outweigh the benefits, and when there is uncertainty surrounding treatment outcomes [35, 36, 48, 52, 53, 55,56,57,58,59,60]. For instance, adding medication to control the disease was perceived as an additional health burden that some were unwilling to accept or it was perceived as a ‘threat’ that may jeopardize their perception that their disease is well controlled despite clinical indicators showing otherwise [56,57,58,59]. RA patients also considered the possible impact of treatment change on their family and employment situation when evaluating competing risks and benefits [42, 55, 61].
Past experiences RA patients tend to draw upon past disease or treatment experiences to help them make sense of potential treatment change decisions. Past experiences of poor disease control prior to biologics treatment and negative experiences of access to biologics were among the barriers that discouraged RA patients to taper or to stop their biologics if they were in remission [56, 61]. Equally, previous bad experiences with the symptoms of RA such as morning stiffness, joint pain, and fatigue compelled patients to accept intensive treatment in the hope of improving their disease outcomes [38, 43]. Unfamiliarity with subcutaneous injection was one of the main reasons RA patients rejected switching biologics from the intravenous to the subcutaneous route of administration despite the latter being a convenient route [35]. Past and current experiences with DMARD-related adverse effects and disease symptoms also affected RA patients’ perception of risks and beliefs, which in turn influenced their willingness to accept DMARDs [39, 52, 54]. RA patients who previously reported experiencing adverse effects of medications were more likely to be unwilling to change therapy (odds ratio [OR] 1.8, 95% confidence interval [CI] 1.6–2.1) [52].
Theme 3: Information Needs Fulfilling RA patients’ need for information has been found to facilitate the decision-making process about treatment modification. Pasma et al. suggested information shaped RA patients’ perception about medications, which in turn influenced adherence [54], whereas Frantzen et al. reported sufficient information was associated with reducing RA patients’ fear of treatment [31]. RA patients consistently wanted more information about the efficacy and safety [31,32,33,34, 38, 49, 56] and long-term effects [34] of the medications to make an informed choice. Information about other treatments to compare the potential benefits and risks [55], practical information on how to manage medication in daily activities [55], and information about the experiences of other RA patients [53] was beneficial. Also, RA patients wanted a clear rationale about why the treatment change was proposed. RA patients expected changes to their treatment to be clinically relevant and aligned with improving health as a priority and not for other purposes such as potential cost-savings [32, 56]. Not knowing enough about the proposed treatment was frequently mentioned across studies as a reason why RA patients were reluctant to modify their treatment [31, 34,35,36, 42, 54]. Despite a need for information, providing information that will increase relevant knowledge of treatment may not necessarily influence RA patients’ choice to escalate treatment [47]. Two studies further suggested that RA patients’ willingness to escalate treatment correlates with their ability to interpret and use medical information they have been provided with to make effective decisions related to health [39] and perceived ability to evaluate numerical information of both positive and negative outcomes associated with each option [45].
Theme 4: Patient–Clinician Relationships The patient–clinician relationship has been found to be central to the facilitation of effective treatment decision making in RA. Among various aspects of a patient–clinician relationship, trust was commonly mentioned by RA patients as a key influential factor related to treatment decisions [31,32,33, 37, 41, 43, 46, 54, 55, 58, 59]. Markusse et al. reported RA patients expressed a high level of trust in their rheumatologist to consider stopping biologics despite believing they needed life-long medication [58]. Nota et al. highlighted some RA patients did not hesitate to initiate DMARD treatment without considering other options but rather had complete trust in their clinician and the health care system [55].
Besides trust, the ability to communicate and interpersonal skills of physicians were also considered an integral part of the relationship by RA patients. Aladul et al. and Pasma et al. reported that RA patients valued having communication with their clinician when making a decision to accept treatment changes [32, 54]. RA patients described communication in the context of clinicians showing interest to know and address their needs, doubts, and fears [54]. Accordingly, empathy and clinicians’ experiences were held in high regard by patients when determining the next course of treatment action. RA patients felt their physician should be aware of their disease and medication history when prescribing treatment [42] and should have experience with the biologics [48] if initiating or changing.
Shaw et al. reported RA patients appreciate the role of clinicians as sources of information and opinion [53]. Patients described wanting guidance from clinicians to understand risk information associated with treatment. In other studies, clinicians were the most influential source of information [31], and their opinions were taken into consideration by RA patients when making treatment decisions [59]. RA patients recognized the value in taking a collaborative approach with clinicians in decision making [31, 32, 37, 56].
Continuity of care Six studies exploring continuity of care have been conducted with RA patients in Aotearoa New Zealand, France, the Netherlands, and the UK, all countries that have publicly funded universal health care [32, 35, 38, 57, 59, 60]. Having access to treatment [32, 57, 59, 60] and consultation with a clinician [38, 57, 60] when needed was valued by RA patients when considering changing their therapy. For RA patients considering whether they would accept biologics tapering, assurance of access to consultation with their clinician, and having the flexibility to increase the dose if disease symptoms worsened, were important determinants of the decision to taper [56, 57, 59, 60]. Similarly, RA patients emphasized the importance of convenient access to their rheumatologists if they were to escalate their treatment [38] and the ability to switch back to the bio-originator if they felt biosimilars were less effective [32]. In another study of intravenous infusions for RA, patients described the main reasons guiding the choice to refuse to switch over to subcutaneous injections were concerns about a lack of follow-up and medical assistance in the event of an adverse effect, given that subcutaneous injections were administered at home, unlike intravenous infusions [35].
4 Discussion
This scoping review found four overarching themes that influence patients’ preferences when considering changes to their treatment for RA. Patient-reported satisfaction is an important determinant of treatment decisions in RA. Willingness to accept treatment modifications is guided by RA patients’ past experiences, driven by their beliefs and the possible impact of the change on their disease and mental well-being. Accurate and complete information facilitates RA patients’ decision-making processes and the importance of patient–clinician relationships was crucial when considering changes to treatment.
RA patients are presumed to consider the available options and their possible outcomes when making treatment decisions [62]. However, past research shows that RA patients do not necessarily systematically approach these decisions but tend to base them on the current level of satisfaction with their disease state and treatments. RA patients who reported being satisfied with the treatment and their disease state were generally more resistant to the idea of changing treatments, even when clinical assessments indicated active disease. In this instance, RA patients may need to feel significantly worse before considering changing their treatment [63]. Patients’ beliefs about their future health are likely to modify the extent to which they are willing to accept treatment changes. Previous studies have shown RA patients’ beliefs are drawn from a wide range of external sources that may not be necessarily congruent with recommended biomedical concepts and can hamper effective disease management [64,65,66,67,68]. Nevertheless, these beliefs are dynamic and can be influenced using educational interventions [64], effective consultation [69], and biopsychosocial approaches [70, 71].
RA patients’ needs for information about medications have been found to be persistently high in past research [72,73,74] and informed patients tended to engage more in treatment decisions [75]. However, providing more information or increasing patients’ knowledge of disease treatment does not necessarily translate into higher uptake of treatment modification. This indicates that more emphasis should be given to ensure the information content is most salient to reduce patients’ cognitive load but still satisfy their need for information as well as meeting ethical requirements. Developing methods of effective dissemination of information that are easily accessible to patients can be improved by taking advantage of the widespread use of the internet and mobile-based technologies [76]. Online platforms have been used by health care organizations to provide news and information, patient education, and other various patient or stakeholder engagement activities [77]. It is equally important to recognize and acknowledge that health literacy varies across patients [78]. Therefore, clinicians and researchers should consider the ability of RA patients to process the information needed to make informed and appropriate health decisions.
Preferences for treatment have been shown to be motivated by what an individual thinks most people would choose in a given scenario (perceived social norm) [47] as well as their perception of the attending physician’s abilities and attributes [79]. For some patients, interactions with physicians are given substantially higher importance over information provided about treatment or disease-related factors in enabling treatment decisions. Thus, patients’ preference for treatment modification should also be understood in relation to the social context in which decisions are made, and not just focus on individuals making decisions in isolation.
The application of decision aids should be advocated to promote RA patient understanding and involvement in the decision-making process. Treatment decisions are often complex, and the process can be intimidating for RA patients to participate in and make a decision [80]. As discrepancy between patients and physicians regarding the management of RA is still an ongoing, significant concern [81], incorporating decision aids may help RA patients by eliciting, clarifying, and communicating their preferences to make decisions about a treatment modification that is aligned with their values [82]. Users of decision aids reported an increase in awareness and understanding of the choices made, as well as enhanced communication with the physician that led to improvement in the quality of the decision-making process [83]. In addition, past studies have demonstrated that decision aids can be integrated into practice to increase RA patients’ knowledge and reduce the uncertainty and conflict surrounding treatment decisions [84, 85].
4.1 Strengths and Limitations of This Review
The main strength of this review is we systematically assessed and summarized past research on RA patients’ preferences for treatment modification, which provided insights that enhance the understanding of factors influencing RA patients’ acceptance or resistance towards changing their treatment, subsequently allowing the formulation of strategies that will improve patients’ willingness to modify therapy. In addition, we included a broad but systematic search across multiple databases including the grey literature that enabled us to comprehensively map the evidence related to RA patient preferences towards treatment modification.
The results of this review must be viewed with appropriate caution given a formal quality assessment of the studies under review was not able to be conducted given the wide variation in methodologies in the literature. Language bias is another potential limitation since studies published in English only were included and future reviews could include relevant studies published in other languages. Another limitation is that the search term ‘treatment modification’ is not consistently defined in the literature. While we acknowledged the term is subject to interpretation, we believe that our search was appropriately inclusive to ensure the major issues related to treatment modification were adequately covered.
5 Conclusion
Facilitation of uptake of recent advances in RA treatment, including introduction of more powerful therapeutic agents, affordable alternatives, and novel treatment approaches in clinical practice requires the understanding of factors that may influence how RA patients approach the decision to modify their treatment. By using a scoping review methodology, the breadth of literature providing insights into RA patients’ perspectives towards treatment modification were reliably captured, which adds to the depth of knowledge regarding RA patients’ preference for treatment modification. Future work is now needed to systematically determine the significance of these factors in the patient decision-making process in RA.
References
Aletaha D, Smolen JS. Diagnosis and management of rheumatoid arthritis: a review. JAMA. 2018;320(13):1360–72. https://doi.org/10.1001/jama.2018.13103.
Smolen JS, Aletaha D, Barton A, Burmester GR, Emery P, Firestein GS, et al. Rheumatoid arthritis. Nat Rev Dise Primers. 2018;4(1):18001. https://doi.org/10.1038/nrdp.2018.1.
Declerck P, Danesi R, Petersel D, Jacobs I. The Language of Biosimilars: Clarification, Definitions, and Regulatory Aspects. Drugs. 2017;77(6):671–7. https://doi.org/10.1007/s40265-017-0717-1.
Smolen JS, Goncalves J, Quinn M, Benedetti F, Lee JY. Era of biosimilars in rheumatology: reshaping the healthcare environment. RMD Open. 2019;5(1):e000900. https://doi.org/10.1136/rmdopen-2019-000900.
Burmester GR, Pope JE. Novel treatment strategies in rheumatoid arthritis. Lancet. 2017;389(10086):2338–48. https://doi.org/10.1016/S0140-6736(17)31491-5.
Schipper LG, van Hulst LT, Grol R, van Riel PL, Hulscher ME, Fransen J. Meta-analysis of tight control strategies in rheumatoid arthritis: protocolized treatment has additional value with respect to the clinical outcome. Rheumatology (Oxford, England). 2010;49(11):2154–64. https://doi.org/10.1093/rheumatology/keq195.
Vermeer M, Kuper HH, Hoekstra M, Haagsma CJ, Posthumus MD, Brus HL, et al. Implementation of a treat-to-target strategy in very early rheumatoid arthritis: results of the Dutch Rheumatoid Arthritis Monitoring remission induction cohort study. Arthritis Rheum. 2011;63(10):2865–72. https://doi.org/10.1002/art.30494.
Ramiro S, Landewé RB, Van Der Heijde D, Sepriano A, FitzGerald O, Ostergaard M, et al. Is treat-to-target really working in rheumatoid arthritis? a longitudinal analysis of a cohort of patients treated in daily practice (RA BIODAM). Ann Rheum Dis. 2020;79(4):453–9.
Schett G, Emery P, Tanaka Y, Burmester G, Pisetsky DS, Naredo E, et al. Tapering biologic and conventional DMARD therapy in rheumatoid arthritis: current evidence and future directions. Ann Rheum Dis. 2016;75(8):1428–37. https://doi.org/10.1136/annrheumdis-2016-209201.
Lenert A, Lenert P. Tapering biologics in rheumatoid arthritis: a pragmatic approach for clinical practice. Clin Rheumatol. 2017;36(1):1–8. https://doi.org/10.1007/s10067-016-3490-8.
Ibrahim SA. Patient preference as a barrier to needed care. Am J Public Health. 2015;105(4):613–4. https://doi.org/10.2105/AJPH.2015.302603.
Desthieux C, Hermet A, Granger B, Fautrel B, Gossec L. Patient-Physician Discordance in Global Assessment in Rheumatoid Arthritis: A Systematic Literature Review With Meta-Analysis. Arthritis Care Res (Hoboken). 2016;68(12):1767–73. https://doi.org/10.1002/acr.22902.
De Mits S, Lenaerts J, Vander Cruyssen B, Mielants H, Westhovens R, Durez P et al. A Nationwide survey on patient’s versus physician´ s evaluation of biological therapy in rheumatoid arthritis in relation to disease activity and route of administration: the Be-Raise Study. Plos one. 2016;11(11):e0166607. https://doi.org/10.1371/journal.pone.0166607.
O'Connor AM, Stacey D, Légaré F, Santesso N. Knowledge translation for patients: methods to support patients' participation in decision making about preference‐sensitive treatment options in rheumatology. Evid-Based Rheumatol. 2004; p. 41–62.
Sidiropoulos P, Bounas A, Athanassiou P, Koutsianas C, Petrikkou E, Kaltsonoudis E, et al. Correlation of patient preferences to treatment outcomes in patients with rheumatoid arthritis treated with tumour necrosis factor inhibitors in Greece. Clin Rheumatol. 2020. https://doi.org/10.1007/s10067-020-05171-8.
Zak A, Corrigan C, Yu Z, Bitton A, Fraenkel L, Harrold L, et al. Barriers to treatment adjustment within a treat to target strategy in rheumatoid arthritis: a secondary analysis of the TRACTION trial. Rheumatology. 2018;57(11):1933–7.
Smolen JS, Breedveld FC, Burmester GR, Bykerk V, Dougados M, Emery P, et al. Treating rheumatoid arthritis to target: 2014 update of the recommendations of an international task force. Ann Rheum Dis. 2016;75(1):3–15. https://doi.org/10.1136/annrheumdis-2015-207524.
Singh JA, Saag KG, Bridges SL, Akl EA, Bannuru RR, Sullivan MC, et al. 2015 American College of Rheumatology guideline for the treatment of rheumatoid arthritis. Arthritis Rheumatol. 2016;68(1):1–26.
Smolen JS, Landewe R, Bijlsma J, Burmester G, Chatzidionysiou K, Dougados M, et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2016 update. Ann Rheum Dis. 2017;76(6):960–77. https://doi.org/10.1136/annrheumdis-2016-210715.
Durand C, Eldoma M, Marshall DA, Bansback N, Hazlewood GS. Patient preferences for disease-modifying antirheumatic drug treatment in rheumatoid arthritis: a systematic review. J Rheumatol. 2020;47(2):176–87. https://doi.org/10.3899/jrheum.181165.
Ayano N, David T, Kathleen T, Allison T, Chandima P. Patients’ attitudes and experiences of disease modifying anti-rheumatic drug use in rheumatoid arthritis and spondyloarthritis: Systematic review of qualitative studies. Intern Med J. 2016;46(Supplement 2):17.
Bridges JF, Hauber AB, Marshall D, Lloyd A, Prosser LA, Regier DA, et al. Conjoint analysis applications in health—a checklist: a report of the ISPOR Good Research Practices for Conjoint Analysis Task Force. Value Health. 2011;14(4):403–13. https://doi.org/10.1016/j.jval.2010.11.013.
Joy SM, Little E, Maruthur NM, Purnell TS, Bridges JF. Patient preferences for the treatment of type 2 diabetes: a scoping review. Pharmacoeconomics. 2013;31(10):877–92. https://doi.org/10.1007/s40273-013-0089-7.
Kim C, Armstrong MJ, Berta WB, Gagliardi ARJHE. How to identify, incorporate and report patient preferences in clinical guidelines: a scoping review. 2020.
Pham MT, Rajic A, Greig JD, Sargeant JM, Papadopoulos A, McEwen SA. A scoping review of scoping reviews: advancing the approach and enhancing the consistency. Res Synth Methods. 2014;5(4):371–85. https://doi.org/10.1002/jrsm.1123.
Brown T, Williams B. Evidence-based education in the health professions: promoting best practice in the learning and teaching of students. CRC Press; 2005.
Arksey H, O’Malley L. Scoping studies: towards a methodological framework. Int J Soc Res Methodol. 2005;8(1):19–32.
Peters MD, Godfrey CM, Khalil H, McInerney P, Parker D, Soares CB. Guidance for conducting systematic scoping reviews. Int J Evid Based Healthc. 2015;13(3):141–6. https://doi.org/10.1097/XEB.0000000000000050.
Hsieh HF, Shannon SE. Three approaches to qualitative content analysis. Qual Health Res. 2005;15(9):1277–88. https://doi.org/10.1177/1049732305276687.
Jacobs I, Singh E, Sewell KL, Sabbagh A, Shane LG. Patient attitudes and understanding about biosimilars: an international cross-sectional survey. Patient Prefer Adher. 2016;10:937.
Frantzen L, Cohen JD, Trope S, Beck M, Munos A, Sittler MA, et al. Patients’ information and perspectives on biosimilars in rheumatology: a French nation-wide survey. Jt Bone Spine. 2019;86(4):491–6. https://doi.org/10.1016/j.jbspin.2019.01.001.
Aladul MI, Fitzpatrick RW, Chapman SR. Patients’ Understanding and attitudes towards infliximab and etanercept biosimilars: result of a UK Web-Based Survey. BioDrugs. 2017;31(5):439–46. https://doi.org/10.1007/s40259-017-0238-1.
van Overbeeke E, De Beleyr B, de Hoon J, Westhovens R, Huys I. Perception of originator biologics and biosimilars: a survey among Belgian rheumatoid arthritis patients and rheumatologists. BioDrugs. 2017;31(5):447–59.
Waller J, Sullivan E, Piercy J, Black CM, Kachroo S. Assessing physician and patient acceptance of infliximab biosimilars in rheumatoid arthritis, ankylosing spondyloarthritis and psoriatic arthritis across Germany. Patient Prefer Adher. 2017;11:519–30. https://doi.org/10.2147/PPA.S129333.
Desplats M, Pascart T, Jelin G, Norberciak L, Philippe P, Houvenagel E, et al. Are abatacept and tocilizumab intravenous users willing to switch for the subcutaneous route of administration? A questionnaire-based study. Clin Rheumatol. 2017;36(6):1395–400. https://doi.org/10.1007/s10067-017-3587-8.
Chau J, Delate T, Ota T, Bhardwaja B. Patient Perspectives on Switching from infliximab to infliximab-dyyb in patients with rheumatologic diseases in the United States. ACR Open Rheumatol. 2019;1(1):52–7. https://doi.org/10.1002/acr2.1007.
Teeple A, Ginsburg S, Howard L, Huff L, Reynolds C, Walls D, et al. Patient attitudes about non-medical switching to biosimilars: results from an online patient survey in the United States. Curr Med Res Opin. 2019;35(4):603–9. https://doi.org/10.1080/03007995.2018.1560221.
Prothero L, Georgopoulou S, Galloway J, Williams R, Bosworth A, Lempp H. Patients’ and carers’ views and expectations about intensive management for moderate rheumatoid arthritis: a qualitative study. Psychol Health Med. 2016;21(8):918–25.
Martin RW, McCallops K, Head AJ, Eggebeen AT, Birmingham JD, Tellinghuisen DJ. Influence of patient characteristics on perceived risks and willingness to take a proposed anti-rheumatic drug. BMC Med Inform Decis Mak. 2013;13:89. https://doi.org/10.1186/1472-6947-13-89.
Hendrikx J, Kievit W, Fransen J, van Riel PL. The influence of patient perceptions of disease on medication intensification in daily practice. Rheumatology (Oxford, England). 2016;55(11):1938–45. https://doi.org/10.1093/rheumatology/kew041.
van Hulst LT, Kievit W, van Bommel R, van Riel PL, Fraenkel L. Rheumatoid arthritis patients and rheumatologists approach the decision to escalate care differently: results of a maximum difference scaling experiment. Arthritis Care Res (Hoboken). 2011;63(10):1407–14. https://doi.org/10.1002/acr.20551.
Fraenkel L, Seng EK, Cunningham M, Mattocks K. Understanding how patients (vs physicians) approach the decision to escalate treatment: a proposed conceptual model. Rheumatology. 2015;54(2):278–85.
van Tuyl LH, Plass AM, Lems WF, Voskuyl AE, Kerstens PJ, Dijkmans BA, et al. Discordant perspectives of rheumatologists and patients on COBRA combination therapy in rheumatoid arthritis. Rheumatology (Oxford, England). 2008;47(10):1571–6. https://doi.org/10.1093/rheumatology/ken323.
Bolge SC, Goren A, Brown D, Ginsberg S, Allen I. Openness to and preference for attributes of biologic therapy prior to initiation among patients with rheumatoid arthritis: patient and rheumatologist perspectives and implications for decision making. Patient Prefer Adher. 2016;10:1079–90. https://doi.org/10.2147/PPA.S107790.
Fraenkel L, Cunningham M, Peters E. Subjective numeracy and preference to stay with the status quo. Med Decis Mak. 2015;35(1):6–11. https://doi.org/10.1177/0272989X14532531.
Raczkiewicz A, Juszkiewicz A, Tlustochowicz M, Tlustochowicz W. Polish rheumatologists and rheumatoid arthritis patients differ in their opinions about treatment inefficacy. Arch Rheumatol. 2015;30(3):238–43.
Martin RW, Enck RD, Tellinghuisen DJ, Eggebeen AT, Birmingham JD, Head AJ. Comparison of the effects of a pharmaceutical industry decision guide and decision aids on patient choice to intensify therapy in rheumatoid arthritis. Med Decis Mak. 2017;37(5):577–88. https://doi.org/10.1177/0272989X17696995.
Lisicki R, Chu L. What matters to patients and physicians when considering biologic therapy for rheumatoid arthritis. Postgrad Med. 2008;120(3):154–60.
Funahashi K, Matsubara T. What RA patients expect of their treatment—discussion over the result of our survey. Clin Rheumatol. 2012;31(11):1559–66.
Fraenkel L, Cunningham M. High disease activity may not be sufficient to escalate care. Arthritis Care Res. 2014;66(2):197–203.
Takahashi N, Sasaki K, Nishiyama T, Naniwa T. Satisfaction and attitudes toward therapy in patients with rheumatoid arthritis. Mod Rheumatol. 2012;22(3):376–81. https://doi.org/10.1007/s10165-011-0531-7.
Wolfe F, Michaud K. Resistance of rheumatoid arthritis patients to changing therapy: discordance between disease activity and patients’ treatment choices. Arthritis Rheumatol. 2007;56(7):2135–42.
Shaw Y, Metes ID, Michaud K, Donohue JM, Roberts MS, Levesque MC, et al. Rheumatoid arthritis patients’ motivations for accepting or resisting disease-modifying antirheumatic drug treatment regimens. Arthritis Care Res (Hoboken). 2018;70(4):533–41. https://doi.org/10.1002/acr.23301.
Pasma A, van’ t Spijker A, Luime JJ, Walter MJ, Busschbach JJ, Hazes JM. Facilitators and barriers to adherence in the initiation phase of disease-modifying antirheumatic drug (DMARD) use in patients with arthritis who recently started their first DMARD treatment. J Rheumatol. 2015;42(3):379–85.
Nota I, Drossaert CH, Taal E, van de Laar MA. Patients’ considerations in the decision-making process of initiating disease-modifying antirheumatic drugs. Arthritis Care Res (Hoboken). 2015;67(7):956–64. https://doi.org/10.1002/acr.22531.
Hewlett S, Haig-Ferguson A, Rose-Parfitt E, Halls S, Freke S, Creamer P. Dose reduction of biologic therapy in inflammatory arthritis: A qualitative study of patients’ perceptions and needs. Musculoskelet Care. 2019;17(1):63–71. https://doi.org/10.1002/msc.1367.
Wallis D, Holmes C, Holroyd C, Sonpal K, Zarroug J, Adams J, et al. Dose reduction of biological therapies for inflammatory rheumatic diseases: what do patients think? Philadelphia : Taylor & Francis Ltd; 2019. p. 251–2.
Markusse IM, Akdemir G, Huizinga TW, Allaart CF. Drug-free holiday in patients with rheumatoid arthritis: a qualitative study to explore patients’ opinion. Clin Rheumatol. 2014;33(8):1155–9. https://doi.org/10.1007/s10067-014-2500-y.
Verhoef LM, Selten EMH, Vriezekolk JE, de Jong AJL, van den Hoogen FHJ, den Broeder AA, et al. The patient perspective on biologic DMARD dose reduction in rheumatoid arthritis: a mixed methods study. Rheumatology (Oxford, England). 2018;57(11):1947–55. https://doi.org/10.1093/rheumatology/key205.
Chan SJ, Stamp LK, Liebergreen N, Ndukwe H, Marra C, Treharne GJ. Tapering biologic therapy for rheumatoid arthritis: a qualitative study of patient perspectives. Patient - Patient-Centered Outcomes Outcomes Res. 2020;13(2):225–34. https://doi.org/10.1007/s40271-019-00403-9.
Baker KF, Isaacs JD, Thompson B. “Living a normal life”: a qualitative study of patients’ views of medication withdrawal in rheumatoid arthritis. BMC Rheumatol. 2019;3:2. https://doi.org/10.1186/s41927-019-0070-y.
Mooney GH. Economics, medicine and health care. 3rd ed. Harlow, England: Financial Times Prentice Hall; 2003.
Dougados M, Nataf H, Steinberg G, Rouanet S, Falissard B. Relative importance of doctor-reported outcomes vs patient-reported outcomes in DMARD intensification for rheumatoid arthritis: the DUO study. Rheumatology (Oxford, England). 2013;52(2):391–9. https://doi.org/10.1093/rheumatology/kes285.
Wong PK. Medication adherence in patients with rheumatoid arthritis: why do patients not take what we prescribe? Rheumatol Int. 2016;36(11):1535–42.
Donovan J, Blake D, Fleming W. The patient is not a blank sheet: lay beliefs and their relevance to patient education. Br J Rheumatol. 1989;28(1):58–61.
Neame R, Hammond A. Beliefs about medications: a questionnaire survey of people with rheumatoid arthritis. Rheumatology (Oxford, England). 2005;44(6):762–7. https://doi.org/10.1093/rheumatology/keh587.
Van Den Bemt BJ, Van Lankveld WG. How can we improve adherence to therapy by patients with rheumatoid arthritis? Nat Clin Pract Rheumatol. 2007;3(12):681.
Wabe N, Lee A, Wechalekar M, McWilliams L, Proudman S, Wiese M. Factors associated with medication adherence in a longitudinal study of rheumatoid arthritis patients. Int J Clin Pract. 2019;73(7):e13375. https://doi.org/10.1111/ijcp.13375.
Main CJ, Buchbinder R, Porcheret M, Foster N. Addressing patient beliefs and expectations in the consultation. Best Pract Res Clin Rheumatol. 2010;24(2):219–25. https://doi.org/10.1016/j.berh.2009.12.013.
Vanhaudenhuyse A, Gillet A, Malaise N, Salamun I, Grosdent S, Maquet D, et al. Psychological interventions influence patients’ attitudes and beliefs about their chronic pain. J Tradit Complement Med. 2018;8(2):296–302. https://doi.org/10.1016/j.jtcme.2016.09.001.
van Middendorp H, Evers AW. The role of psychological factors in inflammatory rheumatic diseases: From burden to tailored treatment. Best Pract Res Clin Rheumatol. 2016;30(5):932–45. https://doi.org/10.1016/j.berh.2016.10.012.
Neame R, Hammond A, Deighton C. Need for information and for involvement in decision making among patients with rheumatoid arthritis: a questionnaire survey. Arthritis Rheum. 2005;53(2):249–55. https://doi.org/10.1002/art.21071.
Barber N, Parsons J, Clifford S, Darracott R, Horne R. Patients’ problems with new medication for chronic conditions. BMJ Quality & Safety. 2004;13(3):172–5.
Meesters J, de Boer I, van den Berg M, Fiocco M, Vlieland TV. Unmet information needs about the delivery of rheumatology health care services: a survey among patients with rheumatoid arthritis. Patient Educ Couns. 2011;85(2):299–303.
von Thiele SU. Co-care: Producing better health outcome through interactions between patients, care providers and information and communication technology. Health Serv Manag Res. 2016;29(1–2):10–5.
Kumar N, Abdullah K, Garg N, Pandey A. YouTube as a source of information on left ventricular assist devices. J Cardiac Fail. 2014;20(8):S84–5.
Courtney K. The use of social media in healthcare: organizational, clinical, and patient perspectives. Enabl Health Healthc ICT Avai Tail Closer. 2013;183:244.
Kim SP, Knight SJ, Tomori C, Colella KM, Schoor RA, Shih L, et al. Health literacy and shared decision making for prostate cancer patients with low socioeconomic status. Cancer Invest. 2001;19(7):684–91. https://doi.org/10.1081/cnv-100106143.
Martin RW, Head AJ, Rene J, Swartz TJ, Fiechtner JJ, McIntosh BA, et al. Patient decision-making related to antirheumatic drugs in rheumatoid arthritis: the importance of patient trust of physician. J Rheumatol. 2008;35(4):618–24.
Belcher VN, Fried TR, Agostini JV, Tinetti ME. Views of older adults on patient participation in medication-related decision making. J Gen Intern Med. 2006;21(4):298–303. https://doi.org/10.1111/j.1525-1497.2006.00329.x.
Sacristán JA, Dilla T, Díaz-Cerezo S, Gabás-Rivera C, Aceituno S. Patient-physician discrepancy in the perception of immune-mediated inflammatory diseases: rheumatoid arthritis, psoriatic arthritis and psoriasis. Qual Syst Rev Lit. 2020;15(6):e0234705.
Barry MJ, Edgman-Levitan S. Shared decision making—the pinnacle of patient-centered care. N Engl J Med. 2012;366(9):780–1.
Pablos JL, Jover JA, Roman-Ivorra JA, Inciarte-Mundo J, Dilla T, Sacristan JA et al. Patient decision aid (PDA) for patients with rheumatoid arthritis reduces decisional conflict and improves readiness for treatment decision making. Patient - Patient-Centered Outcomes Res. 2020;13(1):57–69. https://doi.org/10.1007/s40271-019-00381-y.
Barton JL, Trupin L, Schillinger D, Evans-Young G, Imboden J, Montori VM, et al. Use of low-literacy decision aid to enhance knowledge and reduce decisional conflict among a diverse population of adults with rheumatoid arthritis: results of a pilot study. Arthritis Care Res. 2016;68(7):889–98.
Li LC, Shaw CD, Lacaille D, Yacyshyn E, Jones CA, Koehn C, et al. Effects of a web-based patient decision aid on biologic and small-molecule agents for rheumatoid arthritis: results from a proof-of-concept study. Arthritis Care Res (Hoboken). 2018;70(3):343–52. https://doi.org/10.1002/acr.23287.
Acknowledgements
We thank Thelma Fisher, the subject librarian at Health Sciences Library, University of Otago for her assistance with the search strategy.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was supported by the Health Research Council of New Zealand (18/776) and the University of Otago Research Fund.
Conflict of interest
The authors report no conflicts of interest in this work.
Availability of data and material
The data supporting the findings of this scoping review was obtained from the literature. It is available within the article and its supplementary information files.
Code availability
Not applicable.
Author contributions
JC, HY, GT, LS, and CM contributed to the study conception and design. Data collection, synthesis, and analysis were performed by JC and HY with critical input from LS, GT, and CM. The first draft of the manuscript was written by JC and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chan, S.J., Yeo, H.Y., Stamp, L.K. et al. What Are the Preferences of Patients With Rheumatoid Arthritis for Treatment Modification? A Scoping Review. Patient 14, 505–532 (2021). https://doi.org/10.1007/s40271-020-00488-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40271-020-00488-7