Abstract
Background
Cognitive deficits are detectable in major depressive disorder (MDD). The cognitive impact of antidepressants remains unclear, as does the cognitive effects of aripiprazole in MDD, a commonly used adjunct with putative pro-cognitive properties.
Objectives
In this multi-centre, open-label study, cognitive changes associated with escitalopram monotherapy and adjunctive aripiprazole were examined.
Methods
Acutely depressed participants with MDD (n = 209) received 8 weeks of escitalopram. Non-responders received an additional 8 weeks of adjunctive aripiprazole (ESC-ARI, n = 88), while responders (ESC-CONT, n = 82) continued escitalopram monotherapy (n = 39 lost to attrition). ESC-ARI, ESC-CONT and matched healthy participants (n = 112) completed the Central Nervous System Vital Signs cognitive battery at baseline, 8 and 16 weeks. Linear mixed models compared participants with MDD cognitive trajectories with healthy participants.
Results
Participants with MDD displayed poorer baseline global cognition (assessed via the Neurocognitive Index), composite memory and psychomotor speed vs healthy participants. There were no statistically significant changes in participants with MDD receiving escitalopram monotherapy from baseline to week 8 in the neurocognitive index, reaction time, complex attention, cognitive flexibility, memory or psychomotor speed. Overall symptom severity changes were not associated with cognitive changes. The ESC-CONT group displayed no significant cognitive changes from weeks 8 to 16; reaction time worsened in the ESC-ARI group (p = 0.008) from weeks 8 to 16, independent of symptom change.
Conclusions
Escitalopram monotherapy in acute MDD did not result in significant cognitive improvements. We provide novel evidence that escitalopram continuation in responders does not adversely affect cognition, but adjunctive aripiprazole in escitalopram non-responders worsens reaction time. Treatments targeting cognitive dysfunction are needed in MDD.
ClinicalTrials.gov identifier
NCT01655706; 2 August, 2012.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Cognitive dysfunction commonly accompanies major depressive disorder (MDD); however, the potential cognitive effects of antidepressant treatments are unclear. |
In a multi-centre study, we found that 8 weeks of escitalopram monotherapy in currently symptomatic participants with MDD did not result in a statistically significant improvement in cognition. |
Continuing escitalopram for an additional 8 weeks in participants who experienced a significant improvement in their depressive symptoms did not further change cognition. |
The addition of aripiprazole (a commonly used add-on medication in MDD) to escitalopram in participants who did not respond to escitalopram monotherapy resulted in a worsening reaction time. |
These results suggest that targeted treatments for cognitive dysfunction in MDD are required. |
1 Introduction
Patients with major depressive disorder (MDD) display deficits in multiple cognitive domains including executive functioning, memory, attention and processing speed [1, 2]. These deficits contribute to functional impairment in MDD [3, 4], and can persist even after traditionally defined symptomatic remission from a major depressive episode is attained [5]. These findings indicate that cognitive dysfunction in MDD should be conceptualised as an important independent treatment target [4].
Accordingly, there is growing interest in understanding cognitive changes, positive or negative, associated with antidepressant treatment. Recent meta-analyses and systematic reviews have reported small positive effects on attention, executive function, memory, and psychomotor speed with antidepressant treatment [6,7,8,9]. However, these analyses noted significant methodological heterogeneity amongst studies in terms of cognitive assessment tools, sample size, duration of follow-up and study design. Although there have since been some larger studies assessing cognitive changes with antidepressant treatment [10], further comprehensive assessments in large samples are required to clarify the magnitude and nature of cognitive changes resulting from the pharmacological treatment of MDD.
There also remain further outstanding questions regarding the potential for antidepressant treatment to improve cognitive functioning in individuals with MDD. Antidepressants have been posited to exert a positive effect on cognition ‘directly’ by reversing stress-induced neurohistological changes, and improving neuroplasticity by stimulating postsynaptic monoamine receptors and modulating presynaptic glutamate release [11, 12]. Alternatively, any positive effect of antidepressants on cognition may be solely secondary to improvements in other depressive symptoms such as fatigue and lack of motivation [7]; however, the degree to which cognitive changes associated with antidepressant treatment are independent of improvements in symptom severity has been unclear in previous investigations [7]. Understanding this would guide investigation of specific mediators of cognitive improvement, and whether to target treatments toward cognition or other depressive symptoms. Furthermore, the long-term cognitive impacts of antidepressant treatment are not well understood. Certain antidepressant classes such as tricyclic antidepressants can negatively affect cognition, potentially through anticholinergic or anti-histaminergic mechanisms [13]. Selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors have generally not shown adverse effects on cognition; however, these studies have largely been limited to a short-term, 8-week treatment period [7]. It is unclear how antidepressants may impact cognition when used for a longer period of time following a symptomatic response, a particularly relevant consideration as an antidepressant is usually continued for several months following evidence of a treatment response. Understanding this would clarify the potential risks and benefits to cognition of continuing antidepressant therapy in participants whose other depressive symptoms have improved.
Finally, although effective treatment of MDD frequently requires the use of adjunctive medications [14], the impact on cognition of commonly used adjuncts is poorly understood [9]. Aripiprazole, an atypical antipsychotic and first-line recommended adjunctive agent in MDD, may be of particular interest in this regard [14]. Aripiprazole has been posited to have pro-cognitive effects via its unique profile of partial D2, 5-HT1A and 5-HT7 receptor agonist activity [15, 16], and has been shown to improve spatial memory in rodent models [15, 17]. Some authors have also reported improvements in verbal memory, fluency, processing and motor speed in participants with schizophrenia [18,19,20]. Objectively measured cognitive changes associated with aripiprazole treatment in MDD has received very little examination, with one open-label uncontrolled pilot study demonstrating that adjunctive aripiprazole resulted in improved executive functioning in treatment-refractory individuals with MDD [21]. Thus, although aripiprazole is commonly used in MDD and has biologically plausible mechanisms for improving cognition, its utility for this purpose remains unclear.
To clarify these questions, we examined cognitive changes in adults with MDD participating in a multi-site trial involving sequential escitalopram monotherapy and adjunctive aripiprazole treatment. Escitalopram was chosen for SSRI monotherapy because it is a first-line recommended antidepressant, is commonly used in clinical practice and has relative simplicity of dosing [22]. Participants received periodic assessments with a comprehensive cognitive battery. As ‘practice effects’ (improved performance resulting from increased familiarity and exposure to a test) are a common confound in serial neuropsychological assessments, a parallel healthy participant group acted as a test-retest control [23]. Our objectives were to determine: (1) whether an 8-week course of escitalopram monotherapy resulted in cognitive improvements in acutely depressed individuals with MDD; (2) whether the continuation of escitalopram in responders to acute monotherapy resulted in longer term cognitive changes; (3) whether the addition of adjunctive aripiprazole in escitalopram monotherapy non-responders led to additional cognitive improvements; and (4) the degree to which cognitive improvements were associated with changes in overall symptom severity in each treatment phase.
2 Materials and Methods
2.1 Participants
This was a post-hoc analysis of data from the Canadian Biomarker Integration Network in Depression Study-1 (CAN-BIND-1), a multi-centre discovery study designed to identify predictors of MDD treatment response (ClinicalTrials.gov identifier: NCT01655706) [22]. Full methods, clinical outcomes, and some cognitive data from baseline and week 16 time-points have been previously described [22, 24, 25]. Participants with MDD were recruited from six Canadian academic health science centres via community advertising, outpatient referrals and knowledge translation activities. Inclusion criteria were: (1) age 18–60 years; (2) Mini-International Neuropsychiatric Interview confirmed DSM-IV-TR-defined MDD, with current major depressive episodes [26]; (3) current major depressive episodes > 3 months; (4) free of psychotropic medications for five or more half-lives; (5) Montgomery-Åsberg Depression Rating Scale (MADRS) score ≥ 24 [27]; and (6) fluent in English. Exclusion criteria were: (1) bipolar disorder; (2) another primary psychiatric diagnosis; (3) high suicide risk; (4) substance abuse in the past 6 months; (5) neurological disorder/head trauma/unstable medical condition; (6) pregnant/breast feeding; (7) psychosis in the current episode; (8) high risk for hypomanic switch; (9) non-response to four or more adequate pharmacologic interventions; (10) previous failure/intolerance to escitalopram or aripiprazole; and (11) initiation of psychological treatment in the past 3 months.
Age- and sex-matched healthy comparison participants (HC) were recruited from the community, and had no lifetime or current psychiatric disorders as assessed by the Mini-International Neuropsychiatric Interview [22]. All participants provided written informed consent and received compensation for participation. The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1964, as revised in 2008. All procedures were approved by individual institutional research ethics boards.
2.2 Study Procedure
Following baseline assessment, participants with MDD were initiated on open-label escitalopram 10 mg daily. Escitalopram was increased to 20 mg daily at week 2 or 4 based on clinician judgement of effectiveness/tolerability. Participants who responded, defined as a ≥ 50% MADRS reduction from baseline, at week 8 continued the effective dose of escitalopram for another 8 weeks (ESC-CONT arm). Non-responders received 8 weeks of adjunctive aripiprazole therapy (ESC-ARI arm), flexibly dosed between 2 and 10 mg daily as needed and tolerated. The HC group received no medication.
2.3 Clinical Assessment
Prior to initiating treatment, participants with MDD and HC completed a baseline assessment consisting of a clinical interview, psychiatric history, medical history, Mini-International Neuropsychiatric Interview standardised interview and collection of demographic data. The MADRS scores were the primary measure of symptom severity at each time-point for all participants.
2.4 Cognitive Assessment
Cognitive functioning was evaluated in all participants with the Central Nervous System Vital Signs (CNS-VS) computerised battery at baseline, 8 and 16 weeks. The CNS-VS has shown robust and reliable psychometric properties [28], including concurrent and discriminant validity [29, 30]. It was also designed for repeated administrations, by altering the content of tests across sequential administrations [28]. It has been shown in multiple studies to be sensitive to cognitive deficits in mood disorders [31, 32], and has been previously used to detect clinically significant treatment-related improvements in MDD [33]. The CNS-VS contains 10 tests and generates 15 individual domain scores [28]. Raw domain scores were transformed into standard scores (mean 100, standard deviation 15) based on an age-matched normative sample [28]. The CNS-VS uses ‘validity indicators’ to flag low scores due to poor effort or misunderstanding instructions; invalid scores were discarded. Outlier scores ≥ 4 standard deviations were capped [34].
A measure of global cognition, the Neurocognitive Index (NCI), was calculated as an average of five cognitive domains [28]: composite memory (a composite of verbal and visual memory), psychomotor speed, reaction time, cognitive flexibility and complex attention. See Table 1 for individual test scores contributing to each domain.
2.5 Statistical Analysis
Statistical analyses were completed with SPSS 26.0 (Armonk, NY: IBM Corp); all tests were two-tailed. Demographic variables were compared amongst HC and participants with MDD using chi-square, Mann–Whitney U or t tests. As standard scores generated from the CNS-VS do not correct for education, analyses of covariance (ANCOVAs) covarying for years of education were used to compare NCI scores between MDD and HC at baseline, week 8 and week 16 time-points. Multivariate analyses of covariance (MANCOVA) covarying for education were conducted at each time-point to compare individual cognitive domain scores at each time-point amongst groups. Significant omnibus MANCOVA statistics (p < 0.05) were followed up with univariate ANCOVAs controlling for education, with Sidak correction for pairwise comparisons. Cohen’s d effect sizes [35] were calculated for the difference in cognitive scores between MDD and HC at each time-point.
Primary analyses were conducted with NCI scores. To evaluate NCI change following 8 weeks of escitalopram monotherapy, a repeated-measures linear mixed model (RM-LMM) was applied to all available data, using maximum likelihood estimation and a scaled identity covariance structure. Time-point (baseline vs 8 weeks), group (HC vs MDD), years of education and a time*group interaction term were first added as fixed effects. A randomly varying intercept significantly improved the model fit, as determined by Akaike’s information criterion and Bayesian information criterion values, and was retained in the model. Randomly varying slopes, site as a random or fixed effect, age or sex as fixed effects did not improve the model fit and were not retained. Similar RM-LMMs with 8- and 16-week NCI scores were separately constructed for the ESC-CONT and ESC-ARI arms. Time*group interaction terms in all RM-LMMs were examined to determine whether the MDD group displayed a significant (p < 0.05) cognitive change compared to HC over the same time period (i.e., whether the MDD group displayed a significant change beyond what would could be attributed to practice effects). A Cohen’s d effect size for repeated measures (drm), which accounts for correlations between the same measure pre and post-test, was calculated within subjects for each group [36].
The impact of overall symptom change on cognitive changes was assessed in two ways. First, similar RM-LMMs as above were constructed for the escitalopram monotherapy and ESC-ARI arms, with participants categorised as treatment responders or non-responders. Significant time*group (patient responders, non-responders and HC) interaction terms were followed up with post-hoc analyses between groups. This analysis was not done in the ESC-CONT arm, as only treatment responders at week 8 entered this arm. Second, in all the patient arms, percentage changes in MADRS scores between time-points of interest, age, sex and education were entered as predictors in linear regressions, with a change in NCI scores as the outcome variable.
Secondary analyses to determine changes in individual domains were completed by repeating RM-LMMs with each of the five cognitive domains making up the NCI score. For each set of analyses, a Bonferroni correction (0.05/5 = 0.01) was applied. Additional RM-LMMs dividing the patient group into responders vs non-responders, and linear regressions examining the relationship between a change in MADRS and a change in cognitive scores, were also conducted for individual domains. For these exploratory analyses, a significance level of p < 0.05 was used.
The NCI and cognitive domain scores except for psychomotor speed were negatively skewed; reflect and square root or logarithmic transformations of these scores normalised distribution. The ANCOVA results with transformed vs untransformed scores did not substantially change results, and for clarity results with untransformed scores are shown. Levene’s test for all ANCOVAs and Box’s test for all MANCOVAS were non-significant (all p > 0.05). Residuals of linear regression models and linear mixed models were normally distributed as determined by visual inspection of histograms [37], and variance inflation factor (VIF) values confirmed a lack of multicollinearity amongst predictor variables in linear regression models.
3 Results
3.1 Participant Characteristics
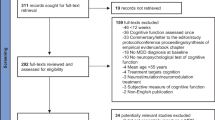
Two hundred and nine participants with MDD and 112 HC completed baseline clinical and cognitive assessment [24]. Healthy participants had significantly higher years of education compared with participants with MDD (Table 2).
One hundred and seventy-one participants with MDD completed week 8 testing (38/18% of the baseline sample not completing week 8 assessment) and 157 completed week 16 testing (14/4% who completed week 8 not completing week 16 assessment). There were no differences in age, education, baseline NCI/individual domain scores, sex proportion, or baseline MADRS scores (all p > 0.15) amongst the 157 patient participants who completed all three cognitive testing points and participants who did not.
Following 8 weeks of escitalopram monotherapy, 85 (49.7%) participants with MDD were classified as responders and 86 (50.3%) as non-responders. Because of protocol deviations in which two non-responders were placed in the ESC-CONT arm and four responders in the ESC-ARI arm, and one participant discontinuing the study after completing week 8 testing, 82 participants entered the ESC-CONT arm and 88 the ESC-ARI arm at week 8. Participants with MDD entering each arm did not differ in demographic variables, proportion with psychiatric comorbidities or prior illness course (Table 2). A proportion of participants with MDD completing week 16 testing did not significantly differ between the ESC-CONT (74/82, 90%) and ESC-ARI (83/88, 94%) arms (χ2 = 1.00, p = 0.32). Fifty-three (60%) participants in the ESC-ARI arm responded to treatment, while six participants (7%) in the ESC-CONT arm were reclassified as non-responders at week 16.
3.2 Cross-Sectional Comparison of Cognitive Scores at Baseline, Week 8 and Week 16
Participants with MDD had significantly lower baseline NCI scores compared with HC (p = 0.01, d = − 0.44) (Table 3, see Table 1 of the Electronic Supplementary Material [ESM] for detailed results). Multivariate analyses of covariance of individual baseline cognitive domains (p = 0.038) showed that participants with MDD scored lower in baseline psychomotor speed (p = 0.001, d = − 0.45) and memory (p = 0.043, d = − 0.36). At 8 weeks, NCI scores no longer differed amongst participants with MDD and HC as determined by ANCOVA (p = 0.40, d = − 0.25), and MANCOVA of week 8 individual domain scores (p = 0.014) showed that participants with MDD continued to display poorer psychomotor speed (p = 0.015, d = − 0.42).
Week 8 and 16 NCI scores did not significantly differ amongst HC, ESC-CONT or ESC-ARI arms (Table 2 of the ESM). Multivariate analyses of covariance of week 8 cognitive domains scores [F(10,500) = 1.86, p = 0.049] showed significant between-group differences in psychomotor speed (p = 0.03), with the ESC-CONT arm scoring lower than HC (p = 0.03). Multivariate analyses of covariance of week 16 cognitive domain scores showed no multivariate differences amongst groups [F(10,480) = 1.25, p = 0.26].
3.3 Pre- to Post-Treatment Cognitive Change in Participants with MDD Receiving 8 Weeks of Escitalopram Monotherapy
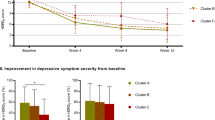
Participants with MDD did not show a statistically significant greater improvement in NCI scores from baseline to week 8 compared to HC (p = 0.066, drm = 0.59 in MDD vs 0.31 in HC, Table 4 and Fig. 1a). Similarly, participants with MDD did not show a statistically significant greater improvement in reaction time (p = 0.015, drm = 0.36 in MDD vs 0.01 in HC), cognitive flexibility (p = 0.024, drm = 0.33 in MDD vs 0.11 in HC), and complex attention (p = 0.048, drm = 0.51 in MDD vs 0.25 in HC), after correcting for multiple comparisons.
Change in cognitive performance in participants with major depressive disorder compared to healthy participants [HC] A following 8 weeks of escitalopram monotherapy and B following a further 8 weeks of either continuation of escitalopram monotherapy (in responders to 8 weeks of escitalopram monotherapy; ESC-CONT) or the addition of adjunctive aripiprazole (in non-responders to 8 weeks of escitalopram monotherapy; ESC-ARI). Y-axis values represent age-matched standardised cognitive scores as determined by Central Nervous System Vital Signs normative data (mean 100, standard deviation 15). NCI Neurocognitive Index
There were no significant time*group interactions for NCI, memory, psychomotor speed, complex attention or cognitive flexibility when participants with MDD were divided into responder and non-responder groups (Table 3 and Fig. 1a of the ESM). There was however a significant time*group interaction for reaction time [F(2,266.95) = 3.08, p = 0.048]. A post-hoc analysis showed no significant time*group interaction in reaction time between participants with MDD responders and non-responders (p = 0.78). Non-responders however showed significant improvement in reaction time compared with HC (parameter estimate = 3.66, p = 0.018), with responders showing a similar magnitude of improvement vs HC (parameter estimate = 3.22, p = 0.066).
Regression analyses in participants showed no significant associations between change in MADRS scores from baseline to week 8 and changes in NCI or any individual cognitive domain (see Table 4 of the ESM).
3.4 Pre- to Post-Treatment Cognitive Change in ESC-CONT from Weeks 8 to 16
Participants in the ESC-CONT arm did not display a significantly different trajectory in NCI (time*group interaction, p = 0.83), memory (p = 0.50), psychomotor speed (p = 0.26), reaction time (p = 0.29), complex attention (p = 0.70) or cognitive flexibility (p = 0.28) from weeks 8 to week 16 compared to HC (Table 5 and Fig. 1b). Regression analyses showed no associations between change in MADRS scores from weeks 8 to 16 and changes in NCI or any individual domain score (Table 4 of the ESM).
3.5 Pre- to Post-Treatment Cognitive Change in ESC-ARI from Weeks 8 to 16
Participants in the ESC-ARI arm also did not display significant differences in NCI change from weeks 8 to 16 compared to HC (time*group interaction p = 0.30, Table 5 and Fig. 1b). However, participants in this arm showed a significant decrease in reaction time from weeks 8 to 16 compared with HC, at the Bonferonni-corrected threshold (p = 0.008, drm = −0.35 in MDD vs 0.09 in HC).
Reaction time was also the only domain to show a significant time*group interaction when participants were divided into week 16 responders (n = 53) and non-responders (n = 30) [F((2,182.89) = 3.58, p = 0.030, see Table 3 and Fig. 1b of the ESM). Patient responders showed a greater decline over time compared to HC (estimate = − 4.06, p = 0.018), with non-responders showing a similar magnitude of decline vs HC (estimate = − 3.89, p = 0.057).
Regression analyses in participants in the ESC-ARI arm showed no associations between change in MADRS scores from week 8 to week 16 and change in NCI, memory, reaction time, complex attention or cognitive flexibility (Table 4 of the ESM). Improvement in psychomotor speed was significantly associated with improvement in MADRS scores (B = 0.30, p = 0.004; R2 change = 0.09, p = 0.004).
As a post-hoc exploratory analysis, Spearman’s correlation was calculated between the average dose of aripiprazole received by participants with MDD in the ESC-ARI group and the change in reaction time from weeks 8 to 16, which was non-significant (p = 0.077, p = 0.492). A partial correlation between average aripiprazole dose and reaction time change, controlling for change in the MADRS score from weeks 8 to 16, was similarly non-significant (r = 0.113, p = 0.314).
4 Discussion
Using data from a multi-centre study, we evaluated cognitive change in acutely symptomatic participants with MDD who received two 8-week phases of escitalopram monotherapy, and those who received 8 weeks of escitalopram followed by adjunctive aripiprazole during the second 8-week phase. Although showing trends towards improvement beyond what would be expected of practice effects, acutely depressed participants with MDD receiving 8 weeks of escitalopram monotherapy did not display statistically significant cognitive changes; furthermore, cognitive changes were not associated with treatment response or changes in overall symptom severity. Treatment responders who received an additional 8 weeks of escitalopram monotherapy did not show any further cognitive changes, negative or otherwise. Importantly, non-responders to 8 weeks of escitalopram monotherapy did not display cognitive improvements with 8 additional weeks of adjunctive aripiprazole; indeed, this group demonstrated a decline in reaction time independent of overall symptom change.
Consistent with previous reports of cognitive dysfunction in MDD, acutely depressed participants in this study showed baseline decrements in global cognitive functioning compared to HC with a small-to-moderate effect size (d = − 0.44). Participants showed improvements in global cognitive functioning at a non-significant trend level (p = 0.066) with 8 weeks of escitalopram monotherapy, and no longer significantly differed in NCI scores vs HC at week 8 (d = − 0.25). The effect size of change in NCI scores from baseline to week 8 was 0.59, as opposed to 0.31 in HC. Participants with MDD also showed improvements in individual domains of reaction time, cognitive flexibility and complex attention at a non-significant trend level of p < 0.05. Similarly, the small magnitude numerical reductions in baseline reaction time (d = − 0.14), complex attention (d = − 0.27) and cognitive flexibility (d = − 0.33) seen in participants with MDD compared to HC were no longer present at week 8, with participants with MDD having equivalent or higher numerical scores vs HC at week 8. The changes in cross-sectional performance and the multiple individual cognitive domains showing trend-level time interactions suggest that these are not spurious findings, but rather a reflection of small magnitude cognitive improvements that may require larger samples to detect. This is consistent with previous reports that cognitive changes associated with SSRIs are small [6, 7], and a large randomised trial of escitalopram, sertraline and venlafaxine extended-release therapy reporting that cognitive improvements with these antidepressants were not significant after adjusting for multiple comparisons [10]. If so, this calls into question the clinical relevance of any cognitive improvements affected by escitalopram. Interestingly, there was negligible improvement in memory and psychomotor speed, the domains where participants with MDD showed the greatest magnitude baseline deficits compared with HC. Participants with MDD showed little change in week 8 cross-sectional memory (drm = 0.18 vs drm = 0.16 in HC) and psychomotor speed (drm = 0.38 vs drm = 0.30 in HC), and ongoing significant deficits in psychomotor speed were detectable at week 8. In sum, our results suggest that though short-term escitalopram monotherapy may improve cognitive functioning, the improvements are of small magnitude and may not address the domains where participants experience the greatest difficulties.
Although our results are consistent with previous studies finding minimal improvements in cognition with SSRI therapy in MDD cohorts [10], some other studies have reported improvements in memory and executive functioning [7, 38]. This raises the possibility that we may have been limited in our ability to detect cognitive changes owing to our participant cohort being relatively high functioning. Though moderate executive functioning deficits in acutely depressed individuals has been reported [1], we did not detect significant deficits in executive functioning-related measures such as cognitive flexibility. Indeed, even in the domains where participants with MDD displayed statistically significant differences compared with HC at baseline, standard scores based on age-matched population norms were still within an average range in the MDD group. Our patient group was relatively young, highly educated, had low rates of substance abuse and other comorbidities, were not treatment resistant and did not have psychotic features. All of these factors may have resulted in a higher cognitively performing cohort with higher cognitive reserve, thus limiting our ability to detect cognitive improvements compared to samples that include more treatment refractory or clinically severe participants. An emerging literature suggests that data-driven probabilistic segmentation techniques such as a cluster analysis or a latent latent class analysis can be used to delineate meaningful cognitive subgroups in mood disorder populations [39,40,41]. An extension to this would be to use group-based trajectory modelling to identify latent classes based on cognitive change, which may help identify discrete patient cohorts who display more pronounced cognitive improvements and who may be obscured by using group-level averages.
Any improvements in cognition following short-term escitalopram monotherapy appeared to be independent of changes in overall symptom response. Responders and non-responders experienced a similar trajectory of cognitive change, and a regression analysis did not identify any significant associations between changes in MADRS scores and cognitive performance in the first 8 weeks. This aligns with previous studies that did not find differences in cognitive changes based on remitter status [10, 38]. Vortioxetine is a novel multi-modal antidepressant agent that has shown consistent positive effects on cognition in MDD. As in our trial, studies suggest that the cognitive changes associated with vortioxetine are largely independent of reductions in other depressive symptoms [42]. We have previously shown in this MDD patient cohort that a history of childhood maltreatment was associated with cognitive deficits in acutely depressed and remitted participants, but did not impact cognitive change over the study term [25]. Future analyses may focus on changes in individual symptom domains such as sleep, energy and interest/motivation, on the basis that these may be more specific contributors to cognitive changes compared to overall symptom severity. Short-term antidepressant administration has also been shown to ameliorate negative affective biases prior to detectable changes in symptom severity [43]; it may be that these changes in negative affective bias (including the propensity to be distracted by negative internal stimuli and self-referential processing) or other social cognitive variables independently mediate improvements in objectively measured cognition. Antidepressant treatment is also associated with structural brain changes, such as increased hippocampal and striatal volumes, and functional changes in prefrontal regions [44, 45], which are plausible mediators of cognitive changes.
We did not find evidence that short-term treatment responders experienced further cognitive improvements with the continuation of escitalopram monotherapy. This may have been due to a ceiling effect in most cognitive domains; except for psychomotor speed, participants in the ESC-CONT arm performed comparably to HC at week 8, and thus likely had limited room for improvement. However, the ESC-CONT group did not experience any deterioration in cognitive functioning over an 8-week time span. Furthermore, the statistically significant psychomotor speed deficit in the ESC-CONT group at week 8 was not detected at week 16, with the ESC-CONT group demonstrating a larger effect size for improvement compared with HC (drm = 0.24 vs drm = 0.12 in HC). While this is suggestive of a small-magnitude improvement in psychomotor speed with continued escitalopram monotherapy, the time*group interaction term did not reach significance and the lack of a significant difference at week 16 may have been due to the smaller sample size at this time-point. Despite this uncertainty, our results do confirm that continuing escitalopram monotherapy in treatment responders for an additional 2 months is not associated with detrimental cognitive consequences. Further studies are required to fully assess the cognitive effects of continued antidepressant treatment over the long term in participants with mild or remitted symptoms.
This analysis provides novel findings that adjunctive aripiprazole, though showing pro-cognitive effects in pre-clinical models, may detrimentally impact reaction time in individuals with MDD [15, 16]. The effect size of change in reaction time from week 8 to 16 in the ESC-ARI group was − 0.35, compared to 0.09 in HC and − 0.08 in ESC-CONT. Both responders and non-responders to adjunctive aripiprazole experienced a similar decline in reaction time, with no association between change in MADRS scores and change in reaction time. There has been very limited previous examination of the cognitive effects of aripiprazole in MDD, with one open-label study finding that adjunctive aripiprazole in non-responders to antidepressant monotherapy was associated with improvements in executive functioning [21]. However, as this was an open-label pilot study (n = 13) with no control group, practice effects may have contributed to the observed improvements. Although studies examining the cognitive effects of aripiprazole in schizophrenia have found improvements in verbal memory, fluency [18], working memory [19] and motor speed [20], there also exists evidence of adverse cognitive effects of aripiprazole in this patient population. One study found that though aripiprazole improved motor speed in participants with schizophrenia, it was also associated with a decline in verbal fluency and executive functioning [20]. Additionally, aripiprazole may interact differently with the distinct underlying pathophysiologies of schizophrenia vs MDD. Partial D2 receptor agonism may result in reduced dopamine output when dopamine levels are high, and increased dopamine output when dopamine levels are low [46]. Schizophrenia and MDD differ in basal striatal dopamine functioning, potentially resulting in differential modulation of striatal dopamine activity by aripiprazole and consequently different (and in MDD, potentially detrimental) effects on motor-based cognitive functions such as reaction time [47, 48]. Interestingly, improvement in depression symptom severity was significantly associated with improved psychomotor speed from weeks 8 to 16 in the ESC-ARI arm. Examining the trajectories of change in responders vs non-responders (Fig. 1b of the ESM) suggests that it was not that responders experienced improvements in psychomotor speed, but rather that non-responders experienced declining performance. Though by no means definitive, this suggests that participants with MDD who do not experience symptom improvement with adjunctive aripiprazole may be vulnerable to worse outcomes in motor-based cognitive domains other than reaction time.
These results are subject to certain limitations. Though our sample size was large compared to many other studies examining antidepressant-associated cognitive changes, it may still have been underpowered to detect, at the Bonferonni-corrected level of significance, small magnitude cognitive improvements. Because of study attrition, the sample sizes at weeks 8 and 16 may additionally have been underpowered to detect significant cross-sectional cognitive differences between groups. Furthermore, the study design did not include a placebo-treated patient control group. Although comparison to an HC group that underwent repeated testing provides some control for practice effects, we cannot attribute potential cognitive improvements in participants to active treatment vs placebo effects, nor can we determine whether improvements in the CAN-BIND patient cohort are greater in comparison to a matched untreated patient arm. Participants were also not randomised to the ESC-CONT or ESC-ARI arms, but assigned based on initial treatment response. Although these two groups did not differ on demographic or clinical variables such as comorbidities or prior illness course, they may have differed in other clinical or biological factors not captured here. This limits our certainty about what can be attributed to the effects of the treatment vs participant differences. However, this study was designed to mirror clinical practice, which enhances the clinical relevance of our results. In clinical practice, an individual with MDD is unlikely to receive aripiprazole unless they have demonstrated non-response to initial monotherapy; these results provide information on the cognitive effects of adjunctive aripiprazole in this patient group. Last, our patient cohort had an average of four previous episodes and approximate total illness duration of 15 years. As higher number of episodes have been associated with more severe cognitive deficits in remission [5], it is possible that greater treatment-related cognitive improvements may be detected in a group with a less chronic illness course.
5 Conclusions
This study provides further evidence that cognitive improvements associated with SSRI monotherapy are of small magnitude, may not address the most prominent cognitive deficits present at baseline and are not associated with changes in overall symptom severity. We also provide novel evidence that continuation of escitalopram in treatment responders is not associated with adverse cognitive consequences, but that adjunctive aripiprazole may have negative effects on reaction time. This supports the need to develop targeted treatments for cognitive dysfunction in MDD, and the need to identify specific mediators of cognitive changes other than changes in overall symptom severity.
References
Rock PL, Roiser JP, Riedel WJ, Blackwell AD. Cognitive impairment in depression: a systematic review and meta-analysis. Psychol Med. 2014;44(10):2029–40.
Bora E, Harrison BJ, Yucel M, Pantelis C. Cognitive impairment in euthymic major depressive disorder: a meta-analysis. Psychol Med. 2013;43(10):2017–26.
Evans VC, Iverson GL, Yatham LN, Lam RW. The relationship between neurocognitive and psychosocial functioning in major depressive disorder: a systematic review. J Clin Psychiatry. 2014;75(12):1359–70.
Bortolato B, Miskowiak KW, Kohler CA, Maes M, Fernandes BS, Berk M, et al. Cognitive remission: a novel objective for the treatment of major depression? BMC Med. 2016;14:9.
Semkovska M, Quinlivan L, O’Grady T, Johnson R, Collins A, O’Connor J, et al. Cognitive function following a major depressive episode: a systematic review and meta-analysis. Lancet Psychiatry. 2019;6(10):851–61.
Rosenblat JD, Kakar R, McIntyre RS. The cognitive effects of antidepressants in major depressive disorder: a systematic review and meta-analysis of randomized clinical trials. Int J Neuropsychopharmacol. 2015;19(2):pyv082.
Prado CE, Watt S, Crowe SF. A meta-analysis of the effects of antidepressants on cognitive functioning in depressed and non-depressed samples. Neuropsychol Rev. 2018;28(1):32–72.
Baune BT, Renger L. Pharmacological and non-pharmacological interventions to improve cognitive dysfunction and functional ability in clinical depression: a systematic review. Psychiatry Res. 2014;219(1):25–50.
Keefe RS, McClintock SM, Roth RM, Doraiswamy PM, Tiger S, Madhoo M. Cognitive effects of pharmacotherapy for major depressive disorder: a systematic review. J Clin Psychiatry. 2014;75(8):864–76.
Shilyansky C, Williams LM, Gyurak A, Harris A, Usherwood T, Etkin A. Effect of antidepressant treatment on cognitive impairments associated with depression: a randomised longitudinal study. Lancet Psychiatry. 2016;3(5):425–35.
Andrade C, Rao NS. How antidepressant drugs act: a primer on neuroplasticity as the eventual mediator of antidepressant efficacy. Indian J Psychiatry. 2010;52(4):378–86.
Liu B, Liu J, Wang M, Zhang Y, Li L. From serotonin to neuroplasticity: evolvement of theories for major depressive disorder. Front Cell Neurosci. 2017;11:305.
Nagane A, Baba H, Nakano Y, Maeshima H, Hukatsu M, Ozawa K, et al. Comparative study of cognitive impairment between medicated and medication-free patients with remitted major depression: class-specific influence by tricyclic antidepressants and newer antidepressants. Psychiatry Res. 2014;218(1–2):101–5.
Kennedy SH, Lam RW, McIntyre RS, Tourjman SV, Bhat V, Blier P, et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) 2016 clinical guidelines for the management of adults with major depressive disorder: Section 3. Pharmacological treatments. Can J Psychiatry. 2016;61(9):540–60.
Burda K, Czubak A, Kus K, Nowakowska E, Ratajczak P, Zin J. Influence of aripiprazole on the antidepressant, anxiolytic and cognitive functions of rats. Pharmacol Rep. 2011;63(4):898–907.
Topolov MK, Getova DP. Cognitive impairment in schizophrenia, neurotransmitters and the new atypical antipsychotic aripiprazole. Folia Med (Plovdiv). 2016;58(1):12–8.
Russo E, Citraro R, Davoli A, Gallelli L, Di Paola ED, De Sarro G. Ameliorating effects of aripiprazole on cognitive functions and depressive-like behavior in a genetic rat model of absence epilepsy and mild-depression comorbidity. Neuropharmacology. 2013;64:371–9.
Bervoets C, Morrens M, Vansteelandt K, Kok F, de Patoul A, Halkin V, et al. Effect of aripiprazole on verbal memory and fluency in schizophrenic patients: results from the ESCAPE study. CNS Drugs. 2012;26(11):975–82.
Maat A, Cahn W, Gijsman HJ, Hovens JE, Kahn RS, Aleman A. Open, randomized trial of the effects of aripiprazole versus risperidone on social cognition in schizophrenia. Eur Neuropsychopharmacol. 2014;24(4):575–84.
Yasui-Furukori N, Kaneda A, Sugawara N, Tomita T, Kaneko S. Effect of adjunctive treatment with aripiprazole to atypical antipsychotics on cognitive function in schizophrenia patients. J Psychopharmacol. 2012;26(6):806–12.
Greer TLSP, Grannemann BD, Trivedi MH. Cognitive and psychosocial improvements following aripiprazole augmentation of SSRI antidepressant therapy in treatment-refractory depression: a pilot study. Open J Depression. 2013;2(4):45–53.
Lam RW, Milev R, Rotzinger S, Andreazza AC, Blier P, Brenner C, et al. Discovering biomarkers for antidepressant response: protocol from the Canadian Biomarker Integration Network in Depression (CAN-BIND) and clinical characteristics of the first patient cohort. BMC Psychiatry. 2016;16:105.
Woodward ND, Purdon SE, Meltzer HY, Zald DH. A meta-analysis of cognitive change with haloperidol in clinical trials of atypical antipsychotics: dose effects and comparison to practice effects. Schizophr Res. 2007;89(1–3):211–24.
Kennedy SH, Lam RW, Rotzinger S, Milev RV, Blier P, Downar J, et al. Symptomatic and functional outcomes and early prediction of response to escitalopram monotherapy and sequential adjunctive aripiprazole therapy in patients with major depressive disorder: a CAN-BIND-1 report. J Clin Psychiatry. 2019;80(2):18m12202.
Chakrabarty T, Harkness KL, McInerney SJ, Quilty LC, Milev RV, Kennedy SH, et al. Childhood maltreatment and cognitive functioning in patients with major depressive disorder: a CAN-BIND-1 report. Psychol Med. 2020;50(15):2536–47.
Sheehan DV, Lecrubier Y, Sheehan KH, Amorim P, Janavs J, Weiller E, et al. The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry. 1998;59(Suppl. 20):22–33 (quiz 4–57).
Montgomery SA, Asberg M. A new depression scale designed to be sensitive to change. Br J Psychiatry. 1979;134:382–9.
Gualtieri CT, Johnson LG. Reliability and validity of a computerized neurocognitive test battery, CNS vital signs. Arch Clin Neuropsychol. 2006;21(7):623–43.
Gualtieri CT, Johnson LG. Efficient allocation of attentional resources in patients with ADHD: maturational changes from age 10 to 29. J Atten Disord. 2006;9(3):534–42.
Gualtieri CT, Johnson LG. Antidepressant side effects in children and adolescents. J Child Adolesc Psychopharmacol. 2006;16(1–2):147–57.
Gualtieri CT, Johnson LG, Benedict KB. Neurocognition in depression: patients on and off medication versus healthy comparison subjects. J Neuropsychiatry Clin Neurosci. 2006;18(2):217–25.
Gualtieri CT, Morgan DW. The frequency of cognitive impairment in patients with anxiety, depression, and bipolar disorder: an unaccounted source of variance in clinical trials. J Clin Psychiatry. 2008;69(7):1122–30.
Lam RW, Iverson GL, Evans VC, Yatham LN, Stewart K, Tam EM, et al. The effects of desvenlafaxine on neurocognitive and work functioning in employed outpatients with major depressive disorder. J Affect Disord. 2016;203:55–61.
Field A. Discovering statistics using SPSS. 3rd ed. Thousand Oaks: SAGE Publications Ltd; 2009.
Cohen J. Statistical power analysis for the behavioral sciences. Abingdon: Routledge; 1988.
Morris SB, DeShon RP. Combining effect size estimates in meta-analysis with repeated measures and independent-groups designs. Psychol Methods. 2002;7(1):105–25.
Singer JDWJ. Applied longitudinal data analysis: modeling change and event occurrence. Oxford: Oxford University Press; 2003.
Herrera-Guzman I, Gudayol-Ferre E, Herrera-Guzman D, Guardia-Olmos J, Hinojosa-Calvo E, Herrera-Abarca JE. Effects of selective serotonin reuptake and dual serotonergic-noradrenergic reuptake treatments on memory and mental processing speed in patients with major depressive disorder. J Psychiatr Res. 2009;43(9):855–63.
Burdick KE, Russo M, Frangou S, Mahon K, Braga RJ, Shanahan M, et al. Empirical evidence for discrete neurocognitive subgroups in bipolar disorder: clinical implications. Psychol Med. 2014;44(14):3083–96.
Chakrabarty TTI, Su WW, Sawatzky R, Keramatian K, Yatham LN. Cognitive subgroups in first episode bipolar I disorder: relation to clinical and brain volumetric variables. Acta Psychiatr Scand. 2020;143:151–61.
Martin DM, Wollny-Huttarsch D, Nikolin S, McClintock SM, Alonzo A, Lisanby SH, et al. Neurocognitive subgroups in major depressive disorder. Neuropsychology. 2020;34(6):726–34.
Bennabi D, Haffen E, Van Waes V. Vortioxetine for cognitive enhancement in major depression: from animal models to clinical research. Front Psychiatry. 2019;10:771.
Harmer CJ. Emotional processing and antidepressant action. Curr Top Behav Neurosci. 2013;14:209–22.
Ma Y. Neuropsychological mechanism underlying antidepressant effect: a systematic meta-analysis. Mol Psychiatry. 2015;20(3):311–9.
Enneking V, Leehr EJ, Dannlowski U, Redlich R. Brain structural effects of treatments for depression and biomarkers of response: a systematic review of neuroimaging studies. Psychol Med. 2020;50(2):187–209.
Stahl SM. Stahl’s essential psychopharmacology: neuroscientific basis and practical applications. 4th ed. New York: Cambridge University Press; 2013.
Amalric M, Koob GF. Depletion of dopamine in the caudate nucleus but not in nucleus accumbens impairs reaction-time performance in rats. J Neurosci. 1987;7(7):2129–34.
Howes OD, Williams M, Ibrahim K, Leung G, Egerton A, McGuire PK, et al. Midbrain dopamine function in schizophrenia and depression: a post-mortem and positron emission tomographic imaging study. Brain. 2013;136(Pt 11):3242–51.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Funding
CAN-BIND is an Integrated Discovery Program carried out in partnership with, and financial support from, the Ontario Brain Institute [grant title “Canadian Biomarker Integration Network for Depression (CAN-BIND)”], an independent non-profit corporation, funded partially by the Ontario government. The opinions, results, and conclusions are those of the authors and no endorsement by the Ontario Brain Institute is intended or should be inferred. Additional funding was provided by CIHR, Lundbeck, Bristol-Myers Squibb, Pfizer and Servier. Funding and/or in kind support is also provided by the investigators’ universities and academic institutions. All study medications are independently purchased at wholesale market values.
Conflict of interest
Trisha Chakrabarty has received grant funding from the Michael Smith Foundation for Health Research and the National Research Council Canada. Shane J. McInerney has received advisory panel income from Janssen and research grant funding through the Healthy Minds Canada/Pfizer Canada Workplace Depression Awards. Ivan J. Torres has received consulting fees or sat on advisory boards for Lundbeck Canada, Sumitomo Dainippon and Community Living British Columbia. Benicio N. Frey has received research grants from Pfizer. Roumen V. Milev has received honoraria for ad hoc speaking or advising/consulting or received research funds from Lundbeck, Kye, Shire, Sunovion, Janssen, Lallemand, Allergan, Otsuka, Canadian Institutes of Health Research, Canadian Biomarker Integration Network for Depression, Ontario Brain Institute and Ontario Mental Health Foundation. Daniel J. Müller has received research funds from the Centre for Addiction and Mental Health Foundation, Canadian Institutes of Health Research and the National Institute of Health. Susan Rotzinger holds a patent “Teneurin C-Terminal Associated Peptides (TCAP) and methods and uses thereof. Inventors: David Lovejoy, R.B. Chewpoy, Dalia Barsyte, Susan Rotzinger.” Sidney H. Kennedy has received research funding or honoraria from the following sources: Abbott, Alkermes, Allergan, Boehringer lngelheim, Brain Canada, Canadian Institutes for Health Research, Janssen, Lundbeck, Lundbeck Institute, Ontario Brain Institute, Ontario Research Fund, Otsuka, Pfizer, Sanofi, Servier, Sunovion and Xian-Janssen. Sidney H. Kennedy holds stock in Field Trip Health. Raymond W. Lam has received honoraria for ad hoc speaking or advising/consulting, or received research funds from Akili, Allergan, Asia-Pacific Economic Cooperation, BC Leading Edge Foundation, Brain Canada, Canadian Institutes of Health Research, Canadian Network for Mood and Anxiety Treatments, Canadian Psychiatric Association, CME Institute, Hansoh, Janssen, Lundbeck, Lundbeck Institute, Medscape, Mind Mental Health Technologies, Otsuka, Pfizer, St. Jude Medical, University Health Network Foundation and Vancouver General Hospital Foundation.
Ethics approval
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1964, as revised in 2008. All procedures were approved by individual institutional research ethics boards.
Consent to participate
All participants provided written informed consent and received compensation for participation.
Consent for publication
All participants signed informed consent regarding publishing their data.
Data availability
The datasets generated during and/or analysed during the current study are not publicly available, but may be available on reasonable request upon permission by the CAN-BIND Investigator’s Team and Ontario Brain Institute. Syntax used for data analysis in SPSS are available upon reasonable request.
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by TC, SJM, IJT, BNF, RVM, DJM, SR, SHK and RWL. The first draft of the manuscript was written by TC and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chakrabarty, T., McInerney, S.J., Torres, I.J. et al. Cognitive Outcomes with Sequential Escitalopram Monotherapy and Adjunctive Aripiprazole Treatment in Major Depressive Disorder: A Canadian Biomarker Integration Network in Depression (CAN-BIND-1) Report. CNS Drugs 35, 291–304 (2021). https://doi.org/10.1007/s40263-021-00793-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40263-021-00793-1