Abstract
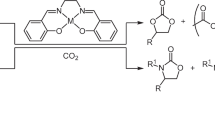
Metal acetylacetonates, a type of readily synthesized or commercially available metal complex, were demonstrated to be robust catalysts for the synthesis of oxazolidinone from CO2 and aziridine under condition of atmospheric pressure and mild temperature, exhibiting high chemo- and regio-selectivity. Tetrabutylammonium halides were employed as cocatalysts in the reaction, and it was believed that the reaction activity was influenced by a balance of anions’ two abilities, namely their nucleophilicity in ring-opening of aziridines and their leaving capacity in ring-closing of intermediate carbamate salts to form oxazolidinones. Notably, higher nucleophilicity of these anions resulted in increased formation of dimers, which served as by-products of the reaction. The study of mechanism suggested that there should be an alternative pathway involving CO2 derivative acting as a nucleophile during the ring-opening process, and this pathway could not be ignored when conventional nucleophiles, such as tetrabutylammonium halides are absent.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Lindsey R., Climate Change: Atmospheric Carbon Dioxide, https://www.climate.gov/news-features/understanding-climate/climate-change-atmospheric-carbon-dioxide (accessed 25/07/2023).
Global Monitoring Laboratory, Trends in Atmospheric Carbon Dioxide, https://www.esrl.noaa.gov/gmd/ccgg/trends (accessed 25/07/2023).
Zahasky C., Krevor S., Energ. Environ. Sci., 2020, 13, 1561
Burkart M., Hazari D. N., Tway C. L., Zeitler E. L., Acs Catal., 2019, 9, 7937.
Wang L., Qi C. R., Xiong W. F., Jiang H. F., Chin. J. Catal., 2022, 43, 1598.
Zhang Z., Deng Y., Zhang Q. F., Yu D. G., Chem. J. Chinese Universities, 2022, 43, 20220255.
Zhou C., Li M., Yu J. T., Sun S., Cheng J., Chin. J. Org. Chem., 2020, 40, 2221.
Ran C. K., Xiao H. Z., Liao L. L., Ju T., Zhang W., Yu D. G., Natl. Sci. Open, 2023, 2, 20220024.
Foti C., Piperno A., Scala A., Giuffre O., Molecules, 2021, 26, 4280.
Pons A., Delion L., Poisson T., Charette A. B., Jubault P., Acc. Chem. Res., 2021, 54, 2969.
Cannizzaro C. E., Ashley J. A., Janda K. D., Houk K. N., J. Am. Chem. Soc., 2003, 125, 2489.
Fournier A. H., Robillard G., Devillers C. H., Plasseraud L., Andrieu J., Eur. J. Org. Chem., 2016, 21, 3514.
Zhu H. T., Chen P. H., Liu G. S., J. Am. Chem. Soc., 2014, 136, 1766.
Liu X. F., Wang M. Y., He L. N., Curr. Org. Chem., 2017, 21, 698.
Ye J. H., Ju T., Huang H., Liao L. L., Yu D. G., Acc. Chem. Res., 2021, 54, 2518.
Ye J. H., Song L., Zhou W. J., Ju T., Yin Z. B., Yan S. S., Zhang Z., Li J., Yu D. G., Angew. Chem. Int. Ed., 2016, 55, 10022.
Zhang Z., Ye J. H., Wu D. S., Zhou Y. Q., Yu D. G., Chem. Asian J., 2018, 13, 2292.
Zhu X. Y., Ran C. K., Wen M., Guo G. L., Liu Y., Liao L. L., Li Y. Z., Li M. L., Yu D. G., Chin. J. Chem., 2021, 39, 3231.
Arshadi S., Banaei A., Ebrahimiasl S., Monfared A., Vessally E., RSC Adv., 2019, 9, 19465.
Chen Y. J., Ren Q. G., Zeng X. J., Tao L. M., Zhou X. T., Ji H. B., Chem. Eng. Sci., 2021, 232, 116380.
Chen Y. J., Luo R. C., Yang Z., Zhou X. T., Ji H. B., Sustain. Energ. Fuels, 2018, 2, 125.
Sengoden M., North M., Whitwood A. C., Chemsuschem., 2019, 12, 3296.
Adhikari D., Miller A. W., Baik M.-H., Nguyen S. T., Chem. Sci., 2015, 6, 1293.
Xie Y., Lu C., Zhao B., Wang Q., Yao Y., J. Org. Chem., 2019, 84, 1951.
Bresciani G., Zacchini S., Marchetti F., Pampaloni G., Dalton Trans., 2021, 50, 5351.
Wang T., Mu Z., Ding X., Han B., Chem. Res. Chinese Universities, 2022, 38, 446.
Bresciani G., Bortoluzzi M., Pampaloni G., Marchetti F., Org. Bio. Chem., 2021, 19, 4152.
Zhao Y. N., Yang Z. Z., Luo S. H., He L. N., Catal. Today., 2013, 200, 2.
Yang Z. Z., He L. N., Peng S. Y., Liu A. H., Green Chem., 2010, 12, 1850.
Damiano C., Sonzini P., Manca G., Gallo E., Eur. J. Org. Chem., 2021, 19, 2807.
Sonzini P., Damiano C., Intrieri D., Manca G., Gallo E., Adv. Synth. Catal., 2020, 362, 2961.
Ueno A., Kayaki Y., Ikariya T., Green Chem., 2013, 15, 425.
Liu A. H., Dang Y. L., Zhou H., Zhang J. J., Lu X. B., Chemcatchem., 2018, 10, 2686.
Du Y., Wu Y., Liu A. H., He L. N., J. Org. Chem., 2008, 73, 4709.
Helal A., Fettouhi M., Arafat M. E., Khan M. Y., Sanhoob M. A., J. CO2 Util., 2021, 50, 101603.
Liu A. G., Chen Y., Liu P. D., Qi W., Li B., Inorg. Chem. Front., 2022, 9, 4425.
Wang X., Gao W. Y., Niu Z., Wojtas L., Perman J. A., Chen Y. S., Li Z., Aguila B., Ma S. Q., Chem. Commun., 2018, 54, 1170.
Shi Y., Zhao J., Xu H., Hou S.L., Zhao B., Sci. China Chem., 2021, 64, 1316.
Aresta M., Dibenedetto A., Angelini A., Chem. Rev., 2014, 114, 1709.
Vigato R. A., Peruzzo V., Tamburini S., Coord. Chem. Rev., 2009, 253, 1099.
Yang Z. Z., Li Y. N., Wei Y. Y., He L. N., Green Chem., 2011, 13, 2351.
Wang H. M., Zhang Z. L., Wang H. L., Guo L. P., Li L., Dalton Trans., 2019, 48, 15970.
Kumar S., Jain S. L., Sain B., Catal. Lett., 2012, 142, 615.
Guo L. P., Lamb K. J., North M., Green Chem., 2021, 23, 77.
Phung C., Ulrich R. M., Ibrahim M., Tighe N. T. G., Lieberman D. L., Pinhas A. R., Green Chem., 2011, 13, 3224.
Phung C., Tantillo D. J., Hein J. E., Pinhas A. R., J. Phys. Org. Chem., 2018, 31, e3735.
Dou X. Y., He L. N., Yang Z. Z., Wang J. L., Synlett., 2010, 14, 2159.
Acknowledgements
This work was supported by the Program for Public Technology of Jiaxing Province, China (No. 2023AD11041), the Natural Science Foundation of Zhejiang Province, China (No. LY22E020009), the National Undergraduate Training Programs for Innovation of China (No. 202110354038) and the Student Research Training Program of Jiaxing University, China (No. 8517231462).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare no conflicts of interest.
Rights and permissions
About this article
Cite this article
Li, B., Gong, Y., Lou, H. et al. Metal Acetylacetonates as Robust Catalysts for the Synthesis of Oxazolidinone from CO2 and Aziridine Under Atmospheric Pressure. Chem. Res. Chin. Univ. (2024). https://doi.org/10.1007/s40242-024-3253-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40242-024-3253-z