Abstract
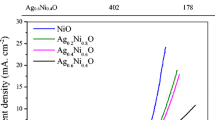
Silver nanoparticles(Ag NPs) were prepared by dealloying Mg-Ag alloy precursor. The obtained Ag NPs have an average ligament size of (50±10) nm. Electrocatalytic activity of Ag NPs towards oxygen reduction reaction( ORR) in 0.1 mol/L NaOH solution was assessed via cyclic voltammetry(CV), rotating ring disk electrode(RRDE) techniques, and electrochemical impedance spectroscopy(EIS). The electrochemical active area for the ORR was evaluated by means of the charge of the underpotential deposition(UPD) of lead(Pb) on Ag NPs. The CV results indicate that Ag NPs have a higher current density and more positive onset potential than the bulk Ag electrode. RRDE was employed to determine kinetic parameters for O2 reduction. Ag NPs exhibit a higher kinetic current density of 25.84 mA/cm2 and a rate constant of 5.45×10–2 cm/s at–0.35 V vs. Hg/HgO. The number of electrons(n) involved in ORR is close to 4. Further, EIS data show significantly low charge transfer resistances on the Ag NPs electrode. The results indicate that the prepared Ag NPs have a high activity and are promising catalyst for ORR in alkaline solution.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Borup R., Meyer J., Pivovar B., Kim Y. S., Mukundan R., Garland N., Myers D., Wilson M., Garzon F., Wood D., Zelenary P., More K., Stroh K., Zawodzinski T., Macgrath J. E., Inaba M., Miyatake K., Hori M., Ota K., Ogumi Z., Miyata S., Nishikata A., Siroma Z., Uchimoto Y., Yasuda K., Kimijima K., Iwashita N., Chem. Rev., 2007, 107(10), 3904
Liang Y., Li Y., Wang H., Zhou J., Wang J., Regier T., Dai H., Nat. Mater., 2011, 10(10), 780
Liu M., Zhang R., Chen W., Chem. Rev., 2014, 114(10), 5117
Voiry D., Yamaguchi H., Li J., Silva R., Alves D. C. B., Fujita T., Chen M., Asefa T., Shenoy V. B., Eda G., Chhowalla M., Nat. Mater., 2013, 12(9), 850
Suntivich J., Gasteiger H. A., Yabuuchi N., Nakanishi H., Goodenough J. B., Yang S. H., Nat. Chem., 2011, 3(7), 546
Cui C. H., Li H. H., Yu J. W., Gao M. R., Yu S. H., Angew. Chem. Int. Ed., 2010, 49(48), 9149
Lu Y. C., Xu Z., Gasteiger H. A., Chen S., Hamad-Schifferli K., Yang S. H., J. Am. Chem. Soc., 2010, 132(35), 12170
Wang Y. J., Wilkinson D. P., Zhang J., Chem. Rev., 2011, 111(12), 7625
Guo S. J., Zhang S., Sun S. H., Angew. Chem. Int. Ed., 2013, 52(33), 8526
Gewirth A. A., Thorum M. S., Inorg. Chem., 2010, 49(8), 3557
Choe J. E., You J. M., Yun M., Lee K., Ahmed M. S., Üstündag Z., Jeon S., J. Nanosci. Nanotechnol., 2015, 15(8), 5684
Lee K., Ahmed M. S., Jeon S., J. Electrochem. Soc., 2015, 162(1), F1
Lu Y., Chen W., J. Power Sources, 2012, 197, 107
Zhao D. J., Ma S. Y., Chem. Res. Chinese Universities, 2015, 31(3), 447
Jiang H. J., Wang F. B., Gao Y., Xing W., Shi B. C., Feng Y. Y., Yang H., Lu T. H., Chem. Res. Chinese Universities, 2003, 19(1), 54
Jeong E., Ahmed M. S., Jeong H., Lee E., Jeon S., Bull. Korean Chem. Soc., 2011, 32(3), 800
Wiley B., Sun Y., Xia Y., Acc. Chem. Res., 2007, 40(10), 1067
Demarconnay L., Coutanceau C., Leger J. M., Electrochim. Acta, 2004, 49(25), 4513
Blizanac B. B., Ross P. N., Markovic N. M., J. Phys. Chem. B, 2006, 110(10), 4735
Blizanac B. B., Ross P. N., Markovic N. M., Electrochim. Acta, 2007, 52(6), 2264
Ahern A. J., Nagle L. C., Burke L. D., J. Solid State Electrochem., 2002, 6(7), 451
Chatenet M., Genies-Bultel L., Aurousseau M., Durand R., Andolfatto F., J. Appl. Electrochem., 2002, 32(10), 1131
Kou T. Y., Li D. W., Zhang C., Zhang Z. H., Yang H., J. Mol. Catal. A: Chem., 2014, 382, 55
Singh P., Buttry D. A., J. Phys. Chem. C, 2012, 116(19), 10656
Guo S., Zhang X., Zhu W., He K., Su D., Mendoza-Garcia A., Ho S. F., Lu G., Sun S., J. Am. Chem. Soc., 2014, 136(42), 15026
Herrero E., Buller L. J., Abruna H. D., Chem. Rev., 2001, 101(7), 1897
Kirowa-Eisner E., Bonfil Y., Tzur D., Gileadi E., J. Electroanal. Chem., 2003, 552, 171
Stevenson K. J., Hatchett D. W., White H. S., Langmuir, 1996, 12(2), 494
Damjanovic A., Genshaw M. A., Bockris J. O. M., J. Electrochem. Soc., 1967, 114, 466
Reyes-Rodriguez J. L., Godinez-Salomon F., Leyva M. A., Solorza-Feria O., Int. J. Hydrog. Energy, 2013, 38(28), 12634
Bard A. J., Faulkner L. R., Electrochemical Methods, Wiley, New York, 1980
Chatenet M., Aurousseau M., Durand R., Andolfatto F., J. Electrochem. Soc., 2003, 150(3), 47
Lima F. H. B., Ticianelli E. A., Electrochim. Acta, 2004, 49(24), 4091
Sepa D. B., Vojnovic M. V., Vracar L. J. M., Damjanovic A., Electrochim. Acta, 1981, 26(6), 781
Tammeveski K., Tenno T., Claret J., Ferrater C., Electrochim. Acta, 1997, 42(19), 893
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the Strategic Priority Research Program of the Chinese Academy of Sciences(No.XDA09010401).
Rights and permissions
About this article
Cite this article
Cui, Q., Zhang, Y. & Peng, Z. Dealloyed silver nanoparticles as efficient catalyst towards oxygen reduction in alkaline solution. Chem. Res. Chin. Univ. 32, 106–111 (2016). https://doi.org/10.1007/s40242-016-5277-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-016-5277-5