Abstract
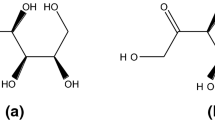
The heat capacities of D-galactose and galactitol were measured on a quantum design physical property measurement system(PPMS) over a temperature range of 1.9―300 K, and the experimental data were fitted to a function of T using a series of theoretical and empirical models in appropriate temperature ranges. The fit results were used to calculate thermodynamic function values, C θp,m , Δ T0 S θm , and Δ T0 H θm from 0 K to 300 K. The standard molar heat capacity, entropy and enthalpy values of D-galactose and galactitol at 298.15 K and 0.1 MPa were determined to be C θp,m =(227.96±2.28) and (239.50±2.40) J·K−1·mol−1, S θm = (211.22±2.11) and (230.82±2.30) J·K−1·mol−1 and H θm = (33.95±0.34) and (36.57± 0.37) kJ/mol, respectively.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Walker-Nasir E., Kaleem A., Hoessli D. C., Curr. Org. Chem., 2008, 12, 940
Fery P. A., Biochemistry, 1996, 35, 89
Topic E., Arambasin S., Zadro R., Annales De Biologie Clinique, 1998, 46, 546
Yeung H. W., NG T. B., Wong D. M., Int. J. Pept. Protein Res., 1986, 27, 208
Feng Y., Rainyeau D., Chachaty C., Yu Z. W., Wolf C., Quinn P. J., Biophys. J., 2004, 86(4), 2008
Wang S. R., Ru B., Lin H Z., Sun W X., Yu C. J., Luo Z. Y., Chem. Res. Chinese Universities, 2014, 30(5), 848
Ficicioglu C., Hussa C., Gallagher P. R., Clin. Chem., 2010, 56, 1177
Yager C. T., Chen J., Reynolds R., Molecular Genetics and Metabolism., 2003, 80, 283
Banipal P. K., Banipal T. S., Ahluwalia J. C., J. Chem. Thermodyn., 2002, 34, 1825
Banipal P. K., Banipal T. S., Lark B. S., J. Chem. Soc. Faraday Trans., 1997, 93, 81
Tewari Y. B., Goldberg R. N., Biophys. Chem., 1991, 40, 59
Daranas A. H., Shimizu H., Homans S. W., J. Am. Chem. Soc., 2004, 126, 11870
Wang J. J., Zhuo K. L., J. Chem. Soc. Faraday Trans., 1998, 94, 3359
Williams P. A. M., Etcheverry S. B., Barrio D. A., Carbohydr. Res., 2006, 341, 717
Shi Q., Snow C. L., Boerio-Goates J., Woodfield B. F., J. Chem. Thermodyn., 2010, 42, 1107
Shi Q., Boerio-Goates J., Woodfield B. F., J. Chem. Thermodyn., 2011, 43, 1263
Shi Q., Zhang L. Y., Schlesinger M. E., Boerio-Goates J., Woodfield B. F., J. Chem. Thermodyn., 2013, 61, 51
Gopal E. S. R., Specific Heats at Low Temperatures, Plenum Press, New York, 1966, 20
Phillips N. E., Rev C., Solid State Sci., 1971, 2, 467
Snow C. L. Shi Q., Boerio-Goates J., Woodfield B. F., J. Chem. Thermodyn., 2010, 42, 1136
Woodfield B. F., Boerio-Goates J., Shapiro L., Putnam R. L., Na v rotsky A., J. Chem. Thermodyn., 1999, 31, 245
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supported by the National Natural Science Foundation of China(No.21473198).
Rights and permissions
About this article
Cite this article
Cheng, Z., Xue, B., Tan, Z. et al. Low-temperature heat capacity and standard thermodynamic functions of D-galactose and galactitol. Chem. Res. Chin. Univ. 31, 987–991 (2015). https://doi.org/10.1007/s40242-015-5263-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-015-5263-3