Abstract
Purpose of review
To describe the imaging findings of tuberculosis with different imaging modalities, as well as the current usage and importance of imaging tuberculosis in resource-limited settings.
Recent findings
Primary pulmonary tuberculosis can manifest as lung parenchymal disease, lymphadenopathy, pleural effusion, and/or miliary disease. Differentiating features of post-primary tuberculosis include cavitation, bilateral tree-in-bud nodules, and an absence of lymphadenopathy. Extrapulmonary tuberculosis can involve nearly any organ system, including the heart, central nervous system, bones and abdominal organs. Many of these findings can be demonstrated with radiography and ultrasound in low-resource settings.
Summary
Tuberculosis remains prevalent in resource-limited settings. It is important for clinicians working in these areas to be familiar with both clinical manifestations and imaging findings of tuberculosis to adequately combat the disease. In order for imaging to help aid with diagnosis and treatment of tuberculosis, there needs to be an increasing focus on appropriate, low-cost imaging and imaging education.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Mycobacterium tuberculosis is the leading infectious cause of death globally, surpassing HIV/AIDS [1]. Tuberculosis (TB) is notable for its communicability, ability to form a latent disease state, and propensity to form drug-resistant strains. The World Health Organization (WHO) approximates that there were 10 million new cases and 1.6 million deaths related to TB in 2017 [1]. The WHO also estimates that 1.7 billion people (23% of the world’s population) have latent TB infections. These patients are at risk for reactivation (post-primary) TB, which has the potential to progress to localized destructive or disseminated disease. In order to combat this, the WHO implemented an “End TB” strategy that aims to decrease TB incidence rates by 4–5% per year [1]. While the global burden of TB has decreased, it has not done so at a rate that would achieve these goals [1].
Most cases of active and latent TB, as well as TB deaths, are concentrated in developing countries. In 2017, the incidence rate of new TB cases was 0.01% in most resource-rich countries, compared to a rate of 0.5% in resource-poor countries such as Mozambique, South Africa and the Philippines [1]. Discrepancies are also apparent in disease outcome between wealthy and developing nations. Case fatality rate, for example, is significantly higher in countries bearing the brunt of disease burden [1]. This reflects the vast inequalities in healthcare access for TB diagnosis and treatment. Additionally, the discrepancy is likely larger than reported, as many of the nations with highest disease burden underreport disease [1]. Widespread use of imaging to identify cases of TB is essential for diagnosis and management of tuberculosis in resource-limited settings.
Imaging Modalities Utilized in Resource-Limited Settings
In the developed world, evaluation of pulmonary tuberculosis is largely accomplished with Computed Tomography (CT). However, in the resource limited setting, there are significant barriers to accessing more advanced imaging techniques, and chest radiography, and increasingly chest ultrasound, are more frequently utilized.
Radiography
The WHO estimates that two-thirds of the world’s population does not currently have access to X-ray technology, and there are many barriers to implementation [2, 3]. Some of these barriers include that when new diagnostic imaging technology reaches low-resource countries, the technology is delivered to larger regional hospital centers first, leaving the rural health care facilities lacking. When equipment does reach these rural areas, staff is frequently not sufficiently trained to properly use and service the machine, rendering it unreliable [4, 5]. In order for a medical facility to offer chest radiography to aid in diagnosing tuberculosis, infrastructure specifications must be met. Standard stationary X-ray technology requires space for the X-ray generator and detector, as well as a reliable power source [6]. While these requirements may be trivial in hospitals in resource-rich countries, underdeveloped countries often have unreliable power grids and may need power generators to provide an adequate consistent power source [7].
Due to infrastructure limitations, many low-resource facilities have turned to mobile X-ray units to acquire chest radiographs. Companies like MinXray specialize in producing lightweight portable X-ray machines that can be set up in any room quickly and can easily be transported between sites (MinXray, Inc. 2015, Fig. 1). In addition, Delft imaging systems supplies portable X-ray units within a mobile van that combines X-ray technology and Xpert testing (Delft Imaging Systems, Fig. 2). These clinics help overcome barriers associated with medical facility infrastructure, as well as the difficulties of widespread distribution to rural sites.
Computed Tomography
Computed tomography (CT) is another valuable tool in diagnosis of pulmonary tuberculosis, and is complementary to conventional chest radiography and CAD, particularly when evaluating sequelae of pulmonary disease. The most significant limitation of CT use in resource-limited settings, however, is lack of access. A study conducted in South Africa in 2015 showed that general X-ray units were the most easily accessible resource throughout the country, with approximately 35 units available per million people [8]. CT was significantly less accessible, with 5 units available per million people, with significant variation in resource availability in different regions.
MRI
Due to its lack of ionizing radiation and improved contrast resolution compared with CT, there has been increased interest in utilization of magnetic resonance imaging (MRI) in evaluation of the chest, especially in the pediatric population. However, in the resource limited setting, cost, lack of availability and long procedure time often prove prohibitive. While likely not useful in the initial diagnosis of tuberculosis, if available MRI can be considered for assessing treatment response during prolonged antibiotic courses [9].
Ultrasound
Ultrasound (US) is more readily available than chest radiography in resource-limited settings. It is also cheaper, portable, and of course involves no radiation [10].
Imaging of Tuberculosis with Chest Radiography
Primary Tuberculosis
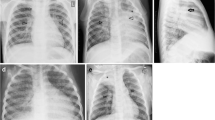
Primary tuberculosis develops with initial exposure to Mycobacterium tuberculosis and normally manifests as lung parenchymal disease, lymphadenopathy, pleural effusion and/or miliary disease. Parenchymal disease characteristically presents as dense homogeneous consolidation of any lobe, with lower and middle lobe predominance [11]. Multi-lobar consolidation is seen in approximately 25% of patients [12]. These findings are non-specific because lobar consolidation is much more likely to be caused by bacterial pneumonia. TB can sometimes be differentiated from bacterial pneumonia when bulky lymphadenopathy is present, or the patient has not responded to antibiotics [13]. Atypical infections, notably fungal, should also be considered in these clinical contexts. In addition to consolidation, in the pediatric population, lobar or segmental atelectasis is common [4]. In about two-thirds of patients, parenchymal disease resolves without permanent sequelae visible on chest radiography. However in about 15% of patients, a parenchymal scar that can calcify persists, called a Ghon focus [11, 14] (Fig. 3).
Lymphadenopathy is seen in up to 96% of pediatric patients and 43% of adults [5, 6]. In infants and young patients, lymphadenopathy in the hilar and para-tracheal region is one of the most important signs for diagnosing TB in suspect patients [15, 16]. Lymphadenopathy is often unilateral and may be the sole manifestation of primary tuberculosis (Fig. 4). The most common location for lymphadenopathy is right paratracheal and right hilar [14].
Disseminated miliary disease affects between 1 and 7% of patients, most often the elderly, infants, and the immunocompromised [11]. Chest radiography is often normal at the onset of symptoms, but within 6 months of exposure hyperinflation and diffuse, evenly distributed 2–3 mm nodules are seen with a slight lower lobe predominance [11] (Fig. 5).
Pleural effusion is also a common manifestation of primary tuberculosis and like lymphadenopathy is often unilateral and sometimes the sole manifestation of disease. Pleural thickening and calcification may ensue [11], sometimes resulting in significant contraction of the hemithorax (“fibrothorax”) [17].
Post-primary Tuberculosis
Post-primary tuberculosis refers to reinfection or reactivation of tuberculosis [11]. Findings of primary and post-primary tuberculosis may overlap. Differentiating features of post-primary tuberculosis include an upper lobe predominant or lower lobe superior segment cavitary mass, frequently with bilateral tree-in-bud nodules (representing dissemination of infection via the airways), in the absence of lymphadenopathy. The pulmonary parenchymal disease often displays patchy, poorly defined consolidation in apical and posterior segments of upper lobes [18], demonstrated in Figs. 6 and 7. Cavities with thick irregular walls are seen in approximately 50% of patients, eventually leading to scar or emphysematous change [11]. Pleural effusions are more common in primary tuberculosis but are seen in about 18% of post-primary patients [11].
Chest Radiograph Analysis in Resource-Limited Settings
In addition to mobile X-ray technology increasing accessibility of radiographic imaging to more resource-limited areas, there have been recent technological advances in diagnostic interpretation of chest radiographs with computer aided diagnostic systems (“CAD4TB”) [19]. Delft Imaging Systems has developed pattern recognition software, now in its sixth edition, that analyzes chest radiographs for findings associated with active pulmonary tuberculosis [20]. CAD4TB software combines multiple subsystems which analyze overall lung field shape (detecting large pleural-based abnormalities), inflammatory or fibrotic textural abnormalities of the lung parenchyma or pleura, and focal nodular abnormalities on chest radiographs in suspected cases of pulmonary tuberculosis [21]. As seen in Fig. 8, the software then generates heat maps that highlight radiographic findings associated with TB, generating a score from 0 to 100 on abnormalities consistent with TB. Computer aided detection software has been proven in multiple studies to have a high sensitivity and low-negative predictive value for pulmonary TB, allowing for increased detection rates and targeted follow up diagnostic laboratory studies [22, 23]. In its most recent edition, this technology is now available for the pediatric population and has been approved for children as young as 4 years of age (Fig. 9) [20].
Imaging of Tuberculosis with Computed Tomography
Although CT is less easily accessible in resource-limited settings, when available it can provide more detailed diagnostic information and better delineate the extent of TB infection. Similar to chest radiography, typical CT findings for primary TB are parenchymal consolidations, which can range from segmental to multi-lobar (Fig. 10) [14]. The manifestations of post-primary pulmonary TB on CT are upper lobe or lower lobe superior segment cavitary masses, with additional findings dependent on method of spread of the infection. Characteristically, centrilobular or tree-in-bud nodules are seen in endobronchial spread, randomly distributed nodules in miliary tuberculosis resulting from lymphohematogenous spread, and occasionally non-enhancing smoothly marginated masses with tuberculoma formation (Figs. 11, 12, 13) [24]. Hilar and mediastinal lymph nodes can be enlarged in up to 43% of adults and are classically peripherally enhancing and centrally hypoattenuating secondary to central caseating necrosis [14]. Pleural effusion is common particularly during primary infection and can often demonstrate the “split pleura sign” of enhancing pleura surrounding tuberculosis-infected empyema (Fig. 14).
Non-contrast coronal chest CT in a 79-year-old woman with history of hepatitis B and orthotopic liver transplant on immunosuppressive therapy, with recent history of travel to Bangladesh. The patient presented with complaints of back pain, fevers, and abdominal pain and was found to have diffuse, uniformly distributed 1–3 mm nodules representing miliary post-primary tuberculosis
The CT features of tuberculosis can also vary depending on the patient’s age and immune status. In immune deficient hosts, such as those with acquired human immune deficiency syndrome (HIV/AIDS), there is an increased incidence of miliary TB and pleural effusion compared to immunocompetent patients [24]. Similar to chest radiography, pediatric patients are more likely to present with lymphadenopathy as the primary manifestation of TB infection, and show parenchymal involvement less commonly than adults [14, 15, 25].
Overall, CT is more sensitive for detecting pulmonary tuberculosis than is chest X-ray, and it provides better characterization of both parenchymal lung and mediastinal lymph node involvement [14, 26]. While initial radiographs of patients with miliary tuberculosis may be interpreted as negative, the improved contrast resolution of CT can demonstrate subtle findings at an earlier stage of disease. CT can also provide more diagnostic information regarding extra-pulmonary manifestations of tuberculosis in comparison to radiography. CT is more sensitive for detecting mediastinal lymphadenopathy, skeletal involvement, and upper abdominal lymph node involvement in comparison with radiography [14]. If utilization of computed tomography for the diagnosis and characterization of tuberculosis infection is possible, its increased sensitivity and more detailed characterization of disease are valuable.
Imaging of Tuberculosis with Ultrasound
A recent systematic review evaluated studies that investigated the use of chest US for pulmonary TB and found five areas where chest US can be applied [27]. The first is for evaluation of pleural effusions, frequently at early stages, and which can often signify an underlying pulmonary lesion [28,29,30]. The second is for accurately detecting a septated sonographic pattern which can predict development of residual pleural thickening after TB treatment [31]. Third, an additional study described an US appearance including an interstitial pattern disseminated with B-lines in multiple lung areas and bilateral, diffuse sub-pleural granularity that is typically seen with miliary TB [32]. Fourthly, chest US can be utilized to guide aspirations or biopsies [33, 34]. Lastly, chest US can be used to assess mediastinal lymphadenopathy, discussed further below [10, 34,35,36].
Imaging of Multi-Drug-Resistant Tuberculosis
Multi-drug-resistant tuberculosis (MDR TB) refers to infection with mycobacterium tuberculosis strains that have evolved resistance to isoniazid and rifampin, two of the four antibiotics in the classic regimen used to treat tuberculosis [37, 38]. Although definitive diagnosis of MDR TB requires laboratory confirmation, imaging findings can suggest MDR TB infection. The most common imaging characteristics seen in MDR TB are similar to non-MDR TB and included centrilobular and tree-in-bud nodularity, consolidation, cavitation and bronchiectasis [37]. In MDR TB, there is classically more diffuse involvement of the bilateral lungs, and thick-walled cavities are more frequently seen in MDR TB than in non-MDR TB. One study suggested that the presence of > 3 cavities measuring > 3 cm in diameter in more than 2 lung zones was associated with pulmonary MDR-TB [39]. Although significant overlap between MDR-TB and non-MDR TB findings precludes definitive diagnosis based on imaging alone, radiologists can suggest the possibility of MDR-TB if extensive, thick-walled cavities are found in both lungs.
Imaging of Extrapulmonary Tuberculosis
It is important to recognize that TB can affect many organs other than the lungs. In fact, one in five cases of TB presents initially as extrapulmonary TB [40]. Extrapulmonary TB can involve nearly any organ system, including the heart, central nervous system, bones, and major abdominal organs, as well as the biliary tract, pancreas, adrenal glands and reproductive organs.
Cardiac
While cardiac involvement in TB is rare as a whole, pericardial involvement with or without pericarditis is relatively common, especially in low-resource areas [41]. Research suggests that while pericardial TB is rare in resource-rich nations, TB can be the most common cause of pericarditis and pericardial effusions in regions where TB is prevalent. This has been found to be at least in part due to increased prevalence of concurrent HIV/AIDS infection in these populations [42]. Pericardial effusions are easily diagnosed with echocardiography and/or point-of-care ultrasound [43]. In the chronic setting, pericardial calcifications can be visualized on chest radiography. Myocardial involvement is less common than pericardial involvement, and also difficult to diagnose with imaging [40].
Lymph Nodes
Lymph nodes are commonly affected in TB. In adults, cervical lymph nodes are most often involved, followed by mediastinal nodes. Usually, cervical lymphadenopathy is unilateral in cases of TB, but this does not allow reliable differentiation from alternative causes. In a resource-poor setting, point-of-care ultrasound plays a large role in reliably diagnosing TB. Research suggests that sonography with Power Doppler can demonstrate large lymph nodes with increased vascularity at the hilum, which differs from the peripheral vascularity seen in malignant lymph nodes [40, 44].
Within the abdomen, lymphadenopathy is the most common manifestation of tuberculosis [40]. On contrast-enhanced CT, this is visualized as large nodes with central hypoattenuation due to central necrosis (Fig. 15) [45]. Peri-portal and para-aortic lymphadenopathy is easily identified by ultrasound [30].
Abdomen
Gastrointestinal TB is not readily diagnosed by ultrasound and often requires CT or at the very least a contrast fluoroscopy study. While gastrointestinal involvement itself is rare, 90% of gastrointestinal TB cases involve the ileocecal region. This commonly manifests as nonspecific bowel wall thickening (Fig. 16) [46, 47].
In contrast to gastrointestinal involvement, hepatosplenic lesions are common [47]. Within the liver and spleen, involvement may be miliary or macronodular. Miliary involvement of the liver can appear as innumerable sub-2-mm nodules that may not be visualized on CT. By ultrasound, these miliary nodules often appear hypoechoic compared to parenchyma [48]. By contrast, the less commonly seen macronodular involvement demonstrates more obvious irregular, hypoattenuating lesions with peripheral enhancement on contrast-enhanced CT and as mass-like hypoechoic lesions on ultrasound [2, 40].
Approximately, 15–20% of extrapulmonary TB cases present as renal tuberculosis [40]. Renal TB commonly presents unilaterally as pyelonephritis or parenchymal nodules. The collecting system may also be involved by anterograde adjacent spread. On CT, this manifests as renal papillary necrosis with resultant irregular caliectasis. Further along in the disease process, imaging findings include a dilated collecting system with multifocal strictures and urothelial enhancement (Fig. 17). This can be seen anywhere from the renal calyces to the bladder. With chronic disease, calcifications occur along the course of the collecting system and may be seen on abdominal radiographs. With end-stage disease, radiographs, US, and CT may demonstrate a putty kidney (coarse, heterogeneous calcifications in the renal fossa) [40].
TB also commonly affects the peritoneum. There are three types of tuberculous peritonitis: wet, fibrotic and dry. Wet peritonitis is the most common and presents as large loculated or free ascites, which tends to be slightly hyperattenuating on contrast-enhanced CT. On ultrasound, loculated fluid pockets with internal debris are easily evident. Fibrotic peritonitis can be seen as omental caking with matted bowel. Dry peritonitis is seen as mesenteric thickening with adhesions and caseating nodules, similar to peritoneal carcinomatosis [2, 30].
Musculoskeletal
Worldwide, the incidence of musculoskeletal TB ranges from 10 to 35% [49]. This can present as tuberculous spondylitis, arthritis, osteomyelitis, or soft tissue disease [11]. The most important and common of these is tuberculous spondylitis, also known as Pott disease: TB of the vertebral bodies and intervertebral discs. Unlike with bacterial spondylitis, contiguous vertebral bodies may be involved without significant involvement of the intervertebral disc space [11]. Assessment of disease extent in the early stages generally requires MRI. In later stages, radiography can show reduction in vertebral body height, sclerotic vertebral bodies, and compression deformities. Untreated TB may lead to progressive kyphosis (Fig. 18). TB spondylitis may also be complicated by abscess formation in adjacent soft tissues. Radiographically evident calcifications within these abscesses are highly suggestive of TB [50].
Sagittal CT (a) and T2-weighted MRI (b) of the lumbar spine in a 61-year-old female with known TB spondylitis demonstrate destructive compression deformities of the T11 and T12 vertebral bodies with resultant focal kyphosis (a). MRI demonstrates severe spinal canal stenosis at these levels with involvement of the inferior T10 vertebral body. Images courtesy of Felipe Urdaneta, MD
Central Nervous System (CNS)
5–10% of patients with tuberculosis demonstrate involvement of the CNS [51]. Notably, the risk of CNS involvement is increased in immunocompromised individuals and is indeed five times greater in those with HIV/AIDS than in immunocompetent patients with TB [52]. In the CNS, TB presents most commonly as tuberculous meningitis or as an intracranial tuberculous granuloma. Typically, tuberculous meningitis presents on contrast-enhanced CT or MRI with thick leptomeningeal enhancement predominantly at the base of the brain, often leading to obstructive hydrocephalus (Fig. 19) [11].
Within the cerebral parenchyma, caseating tuberculous granulomas are seen as rim-enhancing lesions with marked surrounding edema [53]. Alternatively, non-caseating granulomas enhance homogeneously [40]. Radiography and US have no role in diagnosing CNS TB, and in resource-poor settings diagnosis usually rests on clinical findings.
Point-of-Care Ultrasound for Tuberculosis Diagnosis
Point-of-Care Ultrasound (POCUS) is especially useful in the evaluation of TB in the setting of HIV/AIDS, as TB often has extra-pulmonary manifestations in immunocompromised individuals. Extrapulmonary TB often poses a diagnostic challenge due to its non-specific presentation, pauci-bacillary nature and lack of pulmonary involvement (negative sputum smear) [54]. Additionally, TB is 20–30 times more common in HIV positive individuals and TB causes over 1/3 of HIV-related deaths, making early detection critical [55].
In 2017, Heller et al. modified the “focused assessment with sonography for trauma” (FAST) protocol to develop a technique specific for the diagnosis of TB in the setting of HIV, entitled “focused assessment with sonography for HIV-associated TB” (FASH) [54]. This ultrasound-based technique has become one of the most utilized point-of-care ultrasound techniques (POCUS) in resource-limited settings with high TB/HIV burdens, such as South Africa [56].
FASH aims to detect six key features of EPTB: pleural effusion, pericardial effusion, ascites, lymphadenopathy and splenic and hepatic abscesses (Fig. 20) [54, 56]. Unilateral pleural effusion presents on ultrasound as anechoic fluid accumulation in the costodiaphragmatic recess, and in the setting of HIV strongly suggests TB [55]. Bilateral pleural effusions, on the other hand, are more suggestive of congestive heart failure or Kaposi sarcoma [55]. Additionally, enlarged abdominal lymph nodes greater than 1.5 cm (especially para-aortic, epigastric, mesenteric, and splenic) warrant TB treatment in patients with HIV. Similarly, hypoechoic foci in the liver and spleen are suspicious for tuberculous abscesses, and if present should instigate treatment for disseminated TB [55]. Other findings such as pericardial effusion or ascites may represent sequelae of TB in high TB prevalence areas, although other etiologies (including metastatic disease, lymphoma or Kaposi’s sarcoma) should also be considered [55].
The Republic of Malawi Ministry of Health has adopted the FASH protocol and encourages its use in suspected TB cases. According to the Ministry of Health’s National Tuberculosis Control Program, a positive FASH exam in a resource-poor, high-prevalence setting is sufficient evidence to initiate empiric TB treatment [57].
POCUS can also be employed in the diagnosis of pediatric TB. As previously described, the hallmark of TB in the pediatric population is mediastinal lymphadenopathy [10, 25], and in children the mediastinum is often readily evaluated by US. However, mediastinal lymphadenopathy is not a specific finding for extrapulmonary TB and must be used in conjunction with clinical and laboratory findings to support the diagnosis [10].
Conclusion
Despite the low disease burden in resource-rich communities, tuberculosis remains prevalent in resource-poor communities, and accurate diagnosis is critical. To adequately combat TB, clinicians need to be aware of common and uncommon tuberculosis presentations as well as common and uncommon imaging findings. This is important, since TB can affect nearly every organ system in a variety of ways, often leading to diagnostic dilemmas. To appropriately combat the global burden that this disease entity presents, an increasing focus on appropriate, low-cost imaging and imaging education is essential. Although some manifestations of TB are only diagnosed with CT and MRI, radiography and US—the mainstays of imaging in resource-limited settings—allow diagnosis of many forms of chest, abdominal, and osseous disease.
References
Recently published papers of particular interest have been highlighted as: • Of importance •• Of major importance
••World Health Organization. Global tuberculosis report. Geneva: World Health Organization; 2018. p. 2018. This report provides an assessment of the TB epidemic at a global and regional level.
Shah N. Access to imaging technology in the developing world. In: Mollura DJ, Lungren MP, editors. Radiology in global health. New York: Springer; 2014. p. 13–7.
Waters H. Foundations-the first X-ray, 1895. Scientist. 2011;25(7):68.
Jiménez P, Medlen KP, Fleitas-Estévez I. Diagnostic imaging for global health: implementation and optimization of radiology in the developing world. In: Mollura DJ, Lungren MP, editors. Radiology in global health. New York: Springer; 2014. p. 127–37.
World Health Organization. Diagnostic Imaging: Global Collaboration. Geneva: World Health Organization; 2016.
Malkin R, Teninty B. Medical imaging in the global public health: donation, procurement, installation, and maintenance. In: Mollura DJ, Lungren MP, editors. Radiology in global health. New York: Springer; 2014. p. 33–9.
KNCV Tuberculosis Foundation. working document on chest X-ray equipment for use in TB prevalence surveys. Geneva: KNCV Tuberculosis Foundation; 2008.
Kabongo JM, Nel S, Pitcher RD. Analysis of licensed South African diagnostic imaging equipment. Pan Afr Med J. 2015;22:57.
Naranje P, Bhalla AS, Sherwani P. Chest tuberculosis in children. Indian J Pediatr. 2019. https://doi.org/10.1007/s12098-018-02847-7.
Pool K-L, Heuvelings CC, Bélard S, Grobusch MP, Zar HJ, Bulas D, Garra B, Andronikou S. Technical aspects of mediastinal ultrasound for pediatric pulmonary tuberculosis. Pediatr Radiol. 2017;47:1839–48.
Burrill J, Williams CJ, Bain G, Conder G, Hine AL, Misra RR. Tuberculosis: a radiologic review. Radiographics. 2007;27:1255–73.
Bomanji JB, Gupta N, Gulati P, Das CJ. Imaging in tuberculosis. Cold Spring Harb Perspect Med. 2015. https://doi.org/10.1101/cshperspect.a017814.
•Nachiappan AC, Rahbar K, Shi X, Guy ES, Mortani Barbosa EJ, Shroff GS, Ocazionez D, Schlesinger AE, Katz SI, Hammer MM. Pulmonary tuberculosis: role of radiology in diagnosis and management. Radiographics. 2017;37:52–72. This systematic review thoroughly summarizes the clinical and radiographic appearance of primary and postprimary tuberculosis. This review also discusses the role of imaging in the clinical management of TB infected patients.
Skoura E, Zumla A, Bomanji J. Imaging in tuberculosis. Int J Infect Dis. 2015;32:87–93.
Leung AN, Müller NL, Pineda PR, FitzGerald JM. Primary tuberculosis in childhood: radiographic manifestations. Radiology. 1992;182:87–91.
Mehrian P, Moghaddam AM, Tavakkol E, Amini A, Moghimi M, Kabir A, Velayati A. Determining the lymphadenopathy characteristics of the mediastinum in lung CT scan of children with tuberculosis. Int J Mycobacteriol. 2016;5:306–12.
Asri H, Zegmout A. Historic sequelae of lung tuberculosis. Pan Afr Med J. 2018;30:210.
Andreu J, Cáceres J, Pallisa E, Martinez-Rodriguez M. Radiological manifestations of pulmonary tuberculosis. Eur J Radiol. 2004;51:139–49.
Shen Rui, Cheng I, Basu A. A hybrid knowledge-guided detection technique for screening of infectious pulmonary tuberculosis from chest radiographs. IEEE Trans Biomed Eng. 2010;57:2646–56.
Delft Imaging Systems. Computer-aided detection of tuberculosis (CAD4TB). Veenendaal: Delft Imaging Systems; 2019.
Hogeweg L, Sánchez CI, Maduskar P, Philipsen R, Story A, Dawson R, Theron G, Dheda K, Peters-Bax L, van Ginneken B. Automatic detection of tuberculosis in chest radiographs using a combination of textural, focal, and shape abnormality analysis. IEEE Trans Med Imaging. 2015;34:2429–42.
Muyoyeta M, Maduskar P, Moyo M, Kasese N, Milimo D, Spooner R, Kapata N, Hogeweg L, van Ginneken B, Ayles H. The sensitivity and specificity of using a computer aided diagnosis program for automatically scoring chest X-rays of presumptive tb patients compared with Xpert MTB/RIF in Lusaka Zambia. PLoS ONE. 2014;9:e93757.
Zaidi SMA, Habib SS, Van Ginneken B, Ferrand RA, Creswell J, Khowaja S, Khan A. Evaluation of the diagnostic accuracy of computer-aided detection of tuberculosis on chest radiography among private sector patients in Pakistan. Sci Rep. 2018;8:12339.
Lee Kyung Soo, Jeong Yeon Joo. Muller’s imaging of the chest: pulmonary tuberculosis. 2nd ed. Amsterdam: Elsevier; 2018.
George A, Andronikou S, Pillay T, Goussard P, Zar HJ. Intrathoracic tuberculous lymphadenopathy in children: a guide to chest radiography. Pediatr Radiol. 2017;47:1277–82.
He W, Chen B-D, Lv Y, et al. Use of low-dose computed tomography to assess pulmonary tuberculosis among healthcare workers in a tuberculosis hospital. Infect Dis Poverty. 2017;6:68.
Di Gennaro F, Pisani L, Veronese N, et al. Potential diagnostic properties of chest ultrasound in thoracic tuberculosis—a systematic review. Int J Environ Res Public Health. 2018;1:1. https://doi.org/10.3390/ijerph15102235.
Carazo Martínez O, Vargas Serrano B, Rodríguez Romero R. Real-time ultrasound evaluation of tuberculous pleural effusions. J Clin Ultrasound. 1989;17:407–10.
Yuan A, Yang PC, Chang DB, Yu CJ, Lee LN, Wu HD, Kuo SH, Luh KT. Ultrasound guided aspiration biopsy for pulmonary tuberculosis with unusual radiographic appearances. Thorax. 1993;48:167–70.
Ahuja V, Gombar S, Kumar N, Goyal N, Gupta K. Pregnancy with bilateral tubercular pleural effusion: challenges. Trop Doct. 2014;44:116–8.
Lai Y-F, Su M-C, Weng H-H, Wu J-T, Chiu C-T. Sonographic septation: a predictor of sequelae of tuberculous pleurisy after treatment. Thorax. 2009;64:806–9.
Hunter L, Bélard S, Janssen S, van Hoving DJ, Heller T. Miliary tuberculosis: sonographic pattern in chest ultrasound. Infection. 2016;44:243–6.
Gulati M, Venkataramu NK, Gupta S, Sood BP, Sheena DM, Gupta SK, Suri S. Ultrasound guided fine needle aspiration biopsy in mediastinal tuberculosis. Int J Tuberc Lung Dis. 2000;4:1164–8.
Bosch-Marcet J, Serres-Créixams X, Zuasnabar-Cotro A, Codina-Puig X, Català-Puigbó M, Simon-Riazuelo JL. Comparison of ultrasound with plain radiography and CT for the detection of mediastinal lymphadenopathy in children with tuberculosis. Pediatr Radiol. 2004;34:895–900.
Bosch-Marcet J, Serres-Créixams X, Borrás-Pérez V, Coll-Sibina MT, Guitet-Juliá M, Coll-Rosell E. Value of sonography for follow-up of mediastinal lymphadenopathy in children with tuberculosis. J Clin Ultrasound. 2007;35:118–24.
Moseme T, Andronikou S. Through the eye of the suprasternal notch: point-of-care sonography for tuberculous mediastinal lymphadenopathy in children. Pediatr Radiol. 2014;44:681–4.
Wáng YXJ, Chung MJ, Skrahin A, Rosenthal A, Gabrielian A, Tartakovsky M. Radiological signs associated with pulmonary multi-drug resistant tuberculosis: an analysis of published evidences. Quant Imaging Med Surg. 2018;8:161–73.
Chiang C-Y, Centis R, Migliori GB. Drug-resistant tuberculosis: past, present, future. Respirology. 2010;15:413–32.
Chuchottaworn C, Thanachartwet V, Sangsayunh P, Than TZM, Sahassananda D, Surabotsophon M, Desakorn V. Risk factors for multidrug-resistant tuberculosis among patients with pulmonary tuberculosis at the central chest institute of Thailand. PLoS ONE. 2015;10:e0139986.
Gambhir S, Ravina M, Rangan K, Dixit M, Barai S, Bomanji J, International Atomic Energy Agency Extra-pulmonary TB Consortium. Imaging in extrapulmonary tuberculosis. Int J Infect Dis. 2017;56:237–47.
Rodríguez E, Soler R, Juffé A, Salgado L. CT and MR findings in a calcified myocardial tuberculoma of the left ventricle. J Comput Assist Tomogr. 2001;25:577–9.
Mayosi BM, Burgess LJ, Doubell AF. Tuberculous pericarditis. Circulation. 2005;112:3608–16.
Patel MN, Beningfield S, Burch V. Abdominal and pericardial ultrasound in suspected extrapulmonary or disseminated tuberculosis. S Afr Med J. 2011;101:39–42.
Ahuja A, Ying M, Yuen YH, Metreweli C. Power Doppler sonography to differentiate tuberculous cervical lymphadenopathy from nasopharyngeal carcinoma. AJNR Am J Neuroradiol. 2001;22:735–40.
Lee W-K, Van Tonder F, Tartaglia CJ, Dagia C, Cazzato RL, Duddalwar VA, Chang SD. CT appearances of abdominal tuberculosis. Clin Radiol. 2012;67:596–604.
Zissin R, Gayer G, Chowers M, Shapiro-Feinberg M, Kots E, Hertz M. Computerized tomography findings of abdominal tuberculosis: report of 19 cases. Isr Med Assoc J. 2001;3:414–8.
Leder RA, Low VH. Tuberculosis of the abdomen. Radiol Clin North Am. 1995;33:691–705.
Kakkar C, Polnaya AM, Koteshwara P, Smiti S, Rajagopal KV, Arora A. Hepatic tuberculosis: a multimodality imaging review. Insights Imaging. 2015;6:647–58.
Sexton C, McDonald M. Skeletal tuberculosis. UpToDate. Waltham, MA. UpToDate Inc. https://www.uptodate.com. Accessed 25 June 2019.
Resnick D. Tuberculosis infection. Diagnosis of bone and joint disorders. 3rd ed. London: Saunders; 2002.
Sanei Taheri M, Karimi MA, Haghighatkhah H, Pourghorban R, Samadian M, Delavar Kasmaei H. Central nervous system tuberculosis: an imaging-focused review of a reemerging disease. Radiol Res Pract. 2015;2015:202806.
Nelson CA, Zunt JR. Tuberculosis of the central nervous system in immunocompromised patients: HIV infection and solid organ transplant recipients. Clin Infect Dis. 2011;53:915–26.
Whiteman ML. Neuroimaging of central nervous system tuberculosis in HIV-infected patients. Neuroimaging Clin N Am. 1997;7:199–214.
Canan T, Hoffman RM, Schooley A. Training course in focused assessment with HIV prevalent medical centers in Malawi. J Global Radiol. 2018;4:2.
Heller T, Mtemang EA, Huson MAM, Heuvelings CC, Bélard S, Janssen S, Phiri S, Grobusch MP. Ultrasound for patients in a high HIV/tuberculosis prevalence setting: a needs assessment and review of focused applications for sub-Saharan Africa. Int J Infect Dis. 2017;56:229–36.
Kaminstein D, Heller T, Tamarozzi F. Sound around the world: ultrasound for tropical diseases. Infect Dis Clin North Am. 2019;33:169–95.
Ministry of Health. Republic of Malawi. National Tuberculosis Control Programme. Program Manual. Eighth Edition. 2018. p 38–39.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
Kelsey Pomykala, Ishita Desai, Meghan Jardon, Priyanka Naik, and Kara-Lee Pool each declare no potential conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical collection on Global Radiology.
Rights and permissions
About this article
Cite this article
Pomykala, K., Desai, I., Jardon, M. et al. Imaging of Tuberculosis in Resource-Limited Settings. Curr Radiol Rep 7, 23 (2019). https://doi.org/10.1007/s40134-019-0335-7
Published:
DOI: https://doi.org/10.1007/s40134-019-0335-7