Abstract
Fungal diversity in the Hyrcanian forests can greatly vary due to diverse ecological conditions. The scope of the present research was to investigate the diversity of wood decay fungi at three sites in the northern forests of Iran. Fruiting bodies of fungi were collected in three plots dominated by Quercus castaneifolia C.A.M. (oak) and Carpinus betulus L. (hornbeam) in the Hyrcanian Forest. As many as 19 and 13 taxa were found on hornbeam and oak, respectively. The identification of these fungi revealed Fomes fomentarius (L.) Fr. and Ganoderma lucidum (Curtis) P. Karst. as highly abundant on hornbeam and oak, respectively. Highest fungal abundance was observed at an altitude range of 1150-1200 meters above sea level. Diversity of macro-fungi was determined and the mean Shannon diversity index was found to be 2.52 and 1.94 for hornbeam and oak, respectively, and mean equitability was calculated as 0.84 and 0.73 for hornbeam and oak, respectively. There were no significant differences in the Shannon Diversity Index or equitability. Overall, current work showed that most of the identified fungi were classified as white rot fungi.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Forest management is a branch of forestry that includes a variety of aspects: administrative, economic, technical, legal, social, and science [1]. Some forests are managed to only obtain products such as firewood, fiber for paper, and timber; however, with the progression of environmental awareness management of forests for additional uses is becoming increasingly common [2]. Wood decay fungi play an important role in forest ecosystems [3]. It is imperative to determine the role of wood decay fungi in relation to certain ecological processes [4, 5]. Aphyllophoroid fungi affect forest age, structure and gap dynamics [6]. Wood-inhabiting fungi are considered to be good indicators of dead wood communities and the overall naturalness of a particular forest area [7], to aid in the conservation of boreal forests [8], and contribute to species diversity of other dead-wood associated taxa [9]. In deciduous forests, there is also evidence that stumps and tree tops that remain after cutting develop different communities of decay fungi than unmanaged wood [10]. Old growth or virgin forests harbor more deadwood and associated fungal species than managed forests [11]. As a result of the reduced volume of dead wood in managed forests, there is a decline in the wood-inhabiting organisms [7, 12, 13] along with a reduction in fungal diversity [13,14,15,16]. Various forest activities affect the composition of fungal communities [17, 18], diversity [19, 20], species richness, abundance, [21] and fungal structure in forests [19, 22, 23]. Recently, it has been shown that fungal abundance is greater in un-managed forest [24, 25]. This text details the influence of forest management on fungal abundance and diversity of the Nowshahr forests, which are one of the main forests in northern Iran, with emphasis on management history.
Material and Methods
Test Plots
The study area, dominated by temperate broadleaved forests, extends throughout the southern coast of the Caspian Sea (Fig. 1). The studied areas was a section of a research and education forest, Kheyroud Forest, which is located 7 km East of Nowshahr in Mazandaran (36°27′ to 36°40′N, 51°32′ to 51°43′E). This area is limited to the Najardeh and Koliak villages from North to South, respectively. Due to the humid climate and fertile soil, the area under investigation is highly productive. The Hyrcanian forest typically contains oriental beech (Fagus orientalis Lipsky.), chestnut-leaved oak (Quercus castaneifolia C.A.M.), common hornbeam (Carpinus betulus L.), ironwood (Parrotia persica C.A.M.), false walnut (Pterocarya fraxinifolia Land. (Spach.) caucasian alder (Alnus subcordata C.A.M), Caspian honey locust (Gleditschia caspica Desf.), velvet maple (Acer velutinum Boiss.), and wild service (Sorbus torminalis (L.) Crantz.) as the dominant woody species. The climate in this region is sub-Mediterranean with a mean annual temperature of 15º to 18 °C and precipitation of 1000 mm [26]. The area of study contained a community of oak and hornbeam (Table 1).
Test Plot Conditions
Average annual rainfall and temperature in the Nowshahr forest station from 1994 to 2014 was recorded as 1273 mm and 19.6 °C, respectively. October was the wettest month (4/227 mm) and August was the driest month (1/32 mm) (Fig. 2).
Field Sampling
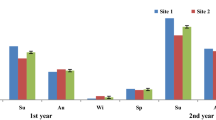
There were several sampling sessions during the summer through autumn months at the three Nowshahr forest plots: Pattom (P1), Namkhaneh (N2), and Gorazbon (G3). Of the three test locations selected, P1 is a research and managed forest with a long-term management history close to the Najardeh village. Test plot N2 is a moderately managed forest while test plot G3 is unmanaged. Details pertaining to the type of forest, tree health, and number of fungi at the test sites were also documented (Fig. 3), and tree health was also classified [27].
Clasification of deciduous tree health in Northern forest of Iran [25]. 1 Living and healthy tree, standing, hard wood, hard skin, green top; 2 Living tree, dead top, standing, hardwood, dead crown; 3 Standing tree, soft wood, broken crown; 4 Snag, loosed wood, without crown; 5 Fallen dead wood, fine and coarse woody debris, log, loosed wood, soft wood
Identification of Fungi
The fungal fruiting bodies were collected from trees during the spring and autumn months (1 May to 31 October, 2010–2012) and immediately placed on ice. For identification of fungi, macro and microscopic characters were determined [28,29,30,31,32].
Statistical Analysis
The values of the Shannon index and equitability were computed using the PAST software. One- way ANOVA was performed and determined significant differences at the 95% confidence level. Grouping was made between factors using Duncan’s multiple range tests via SAS (Version 16) (Tables 2 and 3).
Results and Discussion
Fungal Taxa Encountered
As many as 25 fungal taxa were characterized from the study area (Fig. 4). The two tree species, hornbeam and oak, were colonized by 19 and 13 taxa, respectively. The results further reveal that wood decay fungi are more frequently reported in association with snags and fallen, dead wood (Table 4).
Fungal Diversity
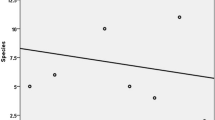
Fungal diversity was determined and the mean Shannon diversity index was found to be 2.52 for hornbeam and 1.94 for oak. The mean of equitability was 0.84 for hornbeam and 0.73 for oak (Table 2). Both the Shannon diversity index and equitability showed no significant differences between the two tree species. However, both these indices were statistically significant for the test sites (Table 3). Studies of fungal biodiversity in forest ecosystems can provide baseline information for determining interrelationships among organisms and indicate potential roles of fungi in forest ecosystem dynamics [24, 33, 34]. Understanding the role of fungi in forest ecosystems is key to characterizing stability and the succession of biological components (i.e. trees) [35], while information on fungal biodiversity can provide insight on sustaining fungi as beneficial resources (Fig. 5).
Presence of Wood Decay Fungi at Different Altitudes
The observations also demonstrate the abundance of wood decay fungi being significantly different (p = 0.05) at different altitudes. The trend of wood decay fungal abundance was observed to be ascendant then descendant. The species of Fomes, Ganoderma, Stereum, and Trametes were highly abundant in the three test sites. The increasing moisture content with increasing attitude along with management and logging practices at the P1 site also indicates the role of physiographic agents in the diversity of these fungi. Robledo and Renison [36] reported that attitude was conducive to the presence of fungal polypores and is highly affected by forest structure.
Relation of Wood Decay Fungi and Tree Health
The results showed that the relationship between wood decay fungi and tree health was significantly different (p = 0.05). Most of the fungi present were reported in association with sangs and fallen, dead wood (Fig. 3) as a result of the large substrate material for fungal colonization [22]. The main constituent of the carbon cycle in forest ecosystems is obtained by fungal decomposition of lignocellulosic material [24, 34]. Decay fungi play an important role in carbon and nitrogen cycling, while helping to convert organic debris into humus. Some fungi such as Fomes fomentarius, Ganoderma applanatum, and Pleurotus ostreatus attack living trees, while others especially Trametes versicolor invade dead or fallen timber and slash on the forest floor [37]. Wood-decay basidiomycetes colonize and degrade wood using enzymatic and mechanical processes [38, 39]. This group includes polypore or bracket fungi (a polyphyletic group with representatives in Hymenochaetales, Polyporales, Gloeophyllales and others), and corticioid fungi (another polyphyletic group represented in Hymenochaetales, Corticiales, Russulales and others) [34].
The species richness and equitability of fungi characterized in this study (Table 1) may seem small in comparison to other descriptions of macrofungi from this region [40, 41]. In the present research eight taxa (Armillaria mellea, Stereum sp., Pluteus cervinus, Ganoderma applanatum, Trichaptum sp., Fomes fomentarius, Pluteus sp. and Schizophyllum commune) on hornbeam and six taxa (Armillaria mellea, Hypholoma fasciculare, Crepidotus sp., Pluteus sp., Coprinus sp. and Ganoderma applanatum) on oak have been reported.
Conclusion
The study reported some effects of forest management on distribution of wood decay fungi in the forests of Northern Iran. It was determined that the highest fungal abundance found in the Kheyroud forests was characterized as Fomes, Ganoderma, Stereum, and Trametes. The current research demonstrates that fungal diversity and abundance becomes reduced when forest management practices occur, which is in accordance with the earlier reports. Assessing the ecology of wood decay fungi as well as their biodiversity has not yet been reported in Iran. In addition to sampling and identification of wood decay fungi at the three test sites, some wood decay fungi in hornbeam and oak are distinguished in Iran.
References
Shaanker RU, Aravind NA, Ganeshaiah KN (2004) Forest management for conservation. In: Burley J, Evans J, Youngquist J (eds) Encyclopedia of forest sciences, 4th edn. Elsevier Science, Amsterdam, pp 215–223
Smith DM, Young R (1982) Introduction to Forest Science. Wiley, Hoboken
Harmon ME, Franklin JF, Swanson FJ, Sollins P, Gregory SV, Lattin JD, Anderson NH, Cline SP, Aumen NG, Sedell JR, Lienkaemper GW, Jr Cromack, Cummins KW (1986) Ecology of coarse woody debris in temperate ecosystems. Adv Ecol Res 15:133–302. doi:10.1016/S0065-2504(08)60121-X
Aghajani H, Marvie Mohadjer MR, Asef MR, Shirvany A (2013) The relationship between abundance of wood macrofungi on chestnut-leave Oak (Quercus castaneifolia C.A.M.) and hornbeam (Carpinus betulus L.) and physiographic factors (Case study: Kheyroud forest, Noshahr). J Nat Environ Iran J Nat Res 66:1–12
Boddy L, Frankland JC, West PV (2008) Ecology of saprotrophic basidiomycetes. Elsevier, Amsterdam
Worrall JJ, Lee TD, Harrington TC (2005) Forest dynamics and agents that initiate and expand canopy gaps in Picea-Abies forests of Crawford Notch, New Hampshire, USA. J Ecol 93:178–190. doi:10.1111/j.1365-2745.2004.00937.x
Bader P, Jansson S, Jonsson BG (1995) Wood-inhabiting fungi and substratum decline in selectively logged boreal spruce forests. Biol Conserv 72:355–362
Kotiranta H, Niemelä T (1996) Threatened polypores in Finland. The Finnish Environment Institute, Helsinki (In Finnish with an English summary)
Similä M, Kouki J, Mönkkönen M, Sippola AL, Huhta E (2006) Co-variation and indicators of species diversity: can richness of forest-dwelling species be predicted in northern boreal forests? Ecol Indic 6:686–700
Heilmann-Clausen J, Boddy L (2008) Distribution patterns of wood-decay basidiomycetes at the landscape to global scale. In: Boddy L, Frankland JC, West PV (eds) Ecology of saprotrophic basidiomycetes. British Mycological Society Symposia Series Academic Press, pp 263–275
Blaser S, Prati D, Senn-Irlet B, Fischer M (2013) Effects of forest management on the diversity of deadwood-inhabiting fungi in Central European forests. For Ecol Manag 304:42–48. doi:10.1016/j.foreco.2013.04.043
Siitonen J (2001) Forest management, coarse woody debris and saproxylic organisms: fennoscandian boreal forests as an example. Ecol Bull 49:11–41. doi:10.2307/20113262
Penttila R, Siitonen J, Kuusinen M (2004) Polypore diversity in managed and oldgrowth boreal Picea abies forests in southern Finland. Biol Conserv 117:271–283. doi:10.1016/j.biocon.2003.12.007
Junninen K, Simila M, Kouki J, Kotiranta H (2006) Assemblages of woodinhabiting fungi along the gradients of succession and naturalness in boreal pine-dominated forests in Fennoscandia. Ecography 29:75–83. doi:10.1111/j.2005.0906-7590.04358.x
Muller J, Engel H, Blaschke M (2007) Assemblages of wood-inhabiting fungi related to silvicultural management intensity in beech forests in southern Germany. Eur J For Res 126:513–527. doi:10.1007/s10342-007-0173-7
Stokland JN, Larsson K (2011) Legacies from natural forest dynamics: different effects of forest management on wood-inhabiting fungi in pine and spruce forests. For Ecol Manag 261:1707–1721. doi:10.1016/j.foreco.2011.01.003
Kuffer N, Senn-Irlet B (2005) influence of forest management on the species richness and composition of wood-inhabiting basidiomycetes in Swiss forests. Biodivers Conserv 14:2419–2435. doi:10.1007/s10531-004-0151-z
Odor P, Heilmann-Clausen J, Christensen M, Aude E, van Dort KW, Piltaver A, Siller I, Veerkamp MT, Walleyn R, Standovar T, van Hees AFM, Kosec J, Matočec N, Kraigher H, Grebenc T (2006) Diversity of dead wood inhabiting fungi and bryophytes in semi-natural beech forests in Europe. Biol Conserv 131:58–71. doi:10.1016/j.biocon.2006.02.004
Abrego N, Salcedo L (2013) Variety of woody debris as the factor influencing wood-inhabiting fungal richness and assemblages: is it a question of quantity or quality? For Ecol Manag 291:377–385. doi:10.1016/j.foreco.2012.11.025
Halme P, Holec J, Heilmann-Clausen J (2017) The history and future of fungi as biodiversity surrogates in forests. Fungal Ecology. 27:193–201
Junninen K, Komonen A (2011) Conservation ecology of boreal polypores: a review. Biolo Conserv 144(1):11–20
Heilmann-Clausen J, Christensen M (2004) Does size matter? On the importance of various dead wood fractions for fungal diversity in Danish beech forests. For Ecol Manag 201:105–117. doi:10.1016/j.foreco.2004.07.010
Debeljak M (2006) Coarse woody debris in virgin and managed forest. Ecolo Indica 6:733–742. doi:10.1016/j.ecolind.2005.08.031
Juutilainen K, Mnkknen M, Kotiranta H, Halme P (2014) The effects of forest management on wood-inhabiting fungi occupying dead wood of different diameter fractions. For Ecol Manag 313:283–291. doi:10.1016/j.foreco.2013.11.019
Runnel K, Lõhmus A (2016) Deadwood-rich managed forests provide insights into the old-forest association of wood-inhabiting fungi. Fungal Ecol 27:155–167
Marvie Mohadjer MR (2011) Silviculture. University of Tehran press, Tehran (in Persian)
Aghajani H (2012) Study on the oak (Quercus castaneifolia) and Hornbeam (Carpinus betulus) decaying macro fungi in mixed Oak-Hornbeam forest community in kheyroud Forest, North of Iran. M.Sc. thesis, Faculty of Natural resources. University of Tehran (in Persian)
Eriksson J, Ryvarden L (1975) The Corticiaceae of North Europe. Fungiflora, Norway, Oslo, pp 287–546
Gilbertson RL, Ryvarden L (1986) North American polypores, vol 1. Fungiflora, Oslo, p 433
Ryvarden L (1991) Genera of polypores, nomenclature and taxonomy. Syn Fung 5, Oslo, p 363
Ryvarden L, Gilbertson RL (1993) European polypores, vol 1. Fungiflora, Oslo, p 433
Kirk PM, Cannon PF, Minter DW, Spatafora JA (2008) Dictionary of the Fungi, 10th edn. CABI, The Netherlands, p 784
Heilmann-Clausen J, Christensen M (2003) Fungal diversity on decaying beech logs–implications for sustainable forestry. Biodivers Conserv 12:953–973. doi:10.1023/A:1022825809503
Stokland JN, Siitonen J, Jonsson BG (2012) Biodiversity in dead wood. Cambridge University Press, Cambridge
Baber K, Otto P, Kahl T, Gossner MM, Wirth C, Gminder A, Bässler C (2016) Disentangling the effects of forest-stand type and dead-wood origin of the early successional stage on the diversity of wood-inhabiting fungi. For Ecol Manag 377:161–169
Robledo GL, Renison D (2010) Wood-decaying polypores in the mountains of central Argentina in relation to Polylepis forest structure and altitude. Fungal Ecol 3:178–184. doi:10.1016/j.funeco.2009.10.003
Schmidt O (2006) Wood and tree Fungi: biology, damage, protection, and use. Springer- Verlag, Germany
Jasalavich CA, Ostrofsky A, Jellison J (2000) Detection and identification of decay fungi in spruce wood by restriction fragment length polymorphism analysis of amplified genes encoding rRNA. Appl Environ Microbiol 66(11):4725–4734
Bari E, Taghiyari HR, Naji HR, Schmidt O, Ohn MK, Clausen CA, Bakar ES (2016) Assessing the destructive behavior of two white-rot fungi on beech wood. Int Biodeterior Biodegr 114:129–140
Hallenberg N (1981) Synopsis of wood-inhabiting Aphyllophorales (Basidiomycetes) and Heterobasidiomycetes from Northern Iran [Wood-fungus]. Mycotaxon (USA)
Ershad D (2009) Fungi of Iran. Ministry of Jihad-e-Agriculture. Agricultural Research, Education and Extention Organization, Tehran, p 535
Acknowledgements
Thanks are due to managements of Mazandaran forestry for collaboration in collection of fungi.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Significance statement
The current work includes several first reports for the Iranian forest pathology and has not been reported before for the Iran. Therefore, the new host as well as fungal colonization behaviors were detected for fungal strategies on the hosts in Iran.
Rights and permissions
About this article
Cite this article
Aghajani, H., Marvie Mohadjer, M.R., Bari, E. et al. Assessing the Biodiversity of Wood Decay Fungi in Northern Forests of Iran. Proc. Natl. Acad. Sci., India, Sect. B Biol. Sci. 88, 1463–1469 (2018). https://doi.org/10.1007/s40011-017-0887-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40011-017-0887-3