Abstract
Purpose
The clinical course of COVID-19 has been complicated by secondary infections, including bacterial and fungal infections. The rapid rise in the incidence of invasive mucormycosis in these patients is very much concerning. COVID-19-associated mucormycosis was detected in huge numbers during the second wave of the COVID-19 pandemic in India, with several predisposing factors indicated in its pathogenesis. This study aimed to evaluate the epidemiology, predisposing factor, cumulative mortality and factors affecting outcomes among the coronavirus disease COVID-19-associated mucormycosis (CAM).
Methods
A multicenter retrospective study across three tertiary health care centers in Southern part of India was conducted during April-June 2021.
Results
Among the 217 cases of CAM, mucormycosis affecting the nasal sinuses was the commonest, affecting 95 (44%) of the patients, orbital extension seen in 84 (38%), pulmonary (n = 25, 12%), gastrointestinal (n = 6, 3%), isolated cerebral (n = 2) and disseminated mucormycosis (n = 2). Diabetes mellitus, high-dose systemic steroids were the most common underlying disease among CAM patients. The mucormycosis-associated case-fatality at 6 weeks was 14%, cerebral or GI or disseminated mucormycosis had 9 times higher risk of death compared to other locations. Extensive surgical debridement along with sequential antifungal drug treatment improved the survival in mucormycosis patients.
Conclusion
Judicious and appropriate management of the predisposing factor and factors affecting mortality associated with CAM with multi-disciplinary approach and timely surgical and medical management can be much helpful in achieving a successful outcome.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has taken the whole world under its clutches, forcing it into an unprecedented crisis. The virus with its high potential of a human to human spread has resulted in an expedited spread of the global pandemic. Secondary infections further worsen the course of the coronavirus disease (COVID-19). Although bacterial infections contribute to a majority of the secondary infections, the rising numbers of fungal infections being reported among COVID-19 patients are a major cause of concern [1]. Presence of pre-existing clinical conditions, diabetes, dysglycemia liberal use of glucocorticoids and higher antibiotics, and non-adherence to strict infection control practices have been ascribed to be the contributing factors leading to the rapid surge in the cases of fungal co-infection [2]. Fungal infections including candidemia, invasive aspergillosis and mucormycosis in COVID-19 patients has significantly increased [3,4,5]. This has led to the increased realization of the magnitude of the problem among clinicians and microbiologists. A literature search of the COVID-19-associated mucormycosis (CAM) cases reported 80 cases across 18 countries as of April 12, 2021 [5]. Another multicentric study from India reported 187 cases of CAM from September to December 2020 [6].
India contributes to the highest number of mucormycosis cases across the world due to its climatic conditions and the presence of large number patients with uncontrolled diabetes mellitus, the major predisposing factor for mucormycosis [7]. India has been hard-hit by the COVID-19 pandemic and thus was expected to have a large number of CAM cases. We conducted a multi-centre study to assess the clinical profile, identify the predisposing factor, cumulative mortality and factors affecting the outcome in the multisystemic cases of CAM.
Materials and methods
Study design
We conducted a retrospective, non-interventional, observational study of CAM patients, involving three tertiary health care centres in Hyderabad, India. We collected the details of the confirmed CAM cases reported during April 15-June 5, 2021 and followed up on the cases for 6 weeks. The ethics committee approval was obtained for the study protocol.
Study subjects and definitions
A case of mucormycosis was defined as one consistent with clinical, radiological findings and corroborating visualization of broad aseptate, ribbon-like fungal hyphae in the direct microscopy of tissue or sterile body fluids of a patient or histopathology specimen by fungal stains, or zygomycetes fungi isolated on culture [8]. COVID-19 diagnosis was confirmed in the patients positive for SARS-CoV-2 RNA by reverse transcriptase polymerase chain reaction (RT-PCR) on the respiratory samples or a positive rapid antigen test. CAM was defined as the detection of proven mucormycosis in COVID-19 patients. Mild COVID-19 infection was defined as those with upper respiratory tract symptoms (and/or fever) without shortness of breath or hypoxia. Moderate COVID-19 infection was defined as those with respiratory rate ≥ 24/min, breathlessness, and SpO2 ≤ 93% on room air. Severe COVID-19 infection was defined as those with SpO2 < 90% or with a respiratory rate > 30/min, requiring high flow oxygen or ventilator assistance at admission or at any time during the hospital stay. High-dose steroids have been defined as those receiving at least 1 mg/kg of methylprednisolone or equivalent doses of other steroids, for a minimum of 5 days.
The primary outcome measure was cumulative mortality. The secondary outcome measures included the predisposing factors and the clinical characteristics of the patients with COVID-associated mucormycosis.
Statistical analysis
All continuous variables were presented as mean with standard deviation or median with interquartile range (IQR) and group differences were analyzed using the student’s t test or the Wilcoxon’s Rank sum test for non-parametric distributions. Comparisons across 3 or more groups were done using the analysis of variance (ANOVA). Categorical variables were presented as proportions (n, %) and group differences were analyzed using the chi-squared or Fisher’s exact test.
Death was used as the outcome variable and Kaplan–Meier (K–M) curves were plotted to show cumulative estimates of survival at various time points. Time to death was defined as the interval between the time of admission to the hospital for mucormycosis and death. Risk factors for death were assessed using the Cox Proportional Hazards Models and displayed using hazard ratios (HR) with 95% confidence intervals (CI). Potential covariates used for adjusting hazard ratios were those with a p < 0.05 in univariate models and those that have been shown to influence failure rates in previous studies.
All data were entered in Microsoft Excel and analyzed using STATA 12.1 I/c (Stata Corp, Fort Worth, Texas, USA) and all p values < 0.05 were considered statistically significant.
Results
Demographics
We included 217 consecutive patients with CAM during the study period. About half the cases were in the age group of 37–54 years (n = 109, 50%) while 22 (10%) were younger than 37 years and 86 (40%) were older than 55 years. Majority of patients were men (n = 177, 82%). Cases were managed by multiple consultants from different specialities across the three participating centres. Majority of the cases (n = 201, 93%) were referred to the participating centres for mucormycosis infection management after the primary COVID-19 infection was managed at the referring centre. The infecting organism was found to be Rhizopus in 144 cases, (66%) Mucor in 37 cases (17%), Syncephalastrum in 1 case. Mixed infection of Rhizopus with Aspergillus was detected in 10 (5%) cases while the species was not identified in 25 cases (12%).
Previous COVID-19 infection
The mean time between recovery from prior COVID-19 and onset of mucormycosis was 15.2 ± 9.1 days and ranged from 2 to 60 days. Recovery was as not as such defined and the date of discharge from the previous hospital, as obtained from the records was taken as presumed recovery. Records showed that nearly half the patients (n = 100, 46%) had severe COVID-19 in the past while another 83 (38%) had moderate COVID-19 and 21 (10%) had mild disease, while records were not available to classify COVID severity in 13 (6%) patients. During the study period, we treated nearly 1250 patients of severe COVID-19, of which 100 patients had mucormycosis. Most of the cases (95/100) of mucormycosis with severe COVID-19 were referred to the participating centres for mucormycosis tertiary care and management.
About half the patient’s required non-invasive ventilation (NIV) (n = 109, 50%), while a further 36 (17%) required mechanical ventilation and 37 (17%) needed supplemental oxygen alone while 35 (16%) did not need any oxygen. 14(7%) of the patients were on home-based care and not hospitalized for COVID-19 disease and yet developed CAM.
Predisposing factors for mucormycosis
Predisposing factors for mucormycosis seen in this cohort, along with prior COVID-19 is shown in Tables 1 and 2. Majority of patients were diabetic (n = 192, 88%) of which 27 (12%) were newly diagnosed. Overall, 83% received high-dose systemic steroids, 80% had uncontrolled diabetes and 12% had coexisting diabetic ketoacidosis. In addition to diabetes and high-dose steroids, patients had other comorbidities such as hypertension (n = 66, 30%), prior stroke (n = 4, 2%), chronic kidney disease (n = 4, 2%) and coronary artery disease (n = 9, 4%). Other immunosuppressive conditions included 3 post renal transplant recipients. Most patients classified as mild to moderate COVID-19 also received 1–2 mg/kg of methylprednisolone or its equivalent in the referring centres (Tables 1, 2). The mean blood sugar of patients admitted with mucormycosis was 273 ± 108 mg, the HbA1c was 9.4 ± 2.4 gm%, serum ferritin was 553 ± 400 ng/ml and C-Reactive protein was 53 ± 61 mg/dl. There were no patients without any known predisposing factor for mucormycosis.
Sites of mucormycosis
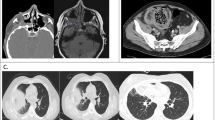
In our patient cohort, mucormycosis involved the nasal sinuses most commonly (n = 95, 44%) followed by rhino-orbital (ROM, n = 69, 32%), rhino-cerebral (RCM, n = 3), rhino-orbito-cerebral (ROCM, n = 15, 9%), pulmonary (n = 25, 12%), gastrointestinal (n = 6, 3%), isolated cerebral (n = 2) and disseminated mucormycosis (n = 2). In cases with rhino-orbital disease, the majority had bilateral involvement (n = 152, 70%). The maxillary sinus was the most commonly involved (n = 173, 80%) followed by the ethmoid (n = 165, 76%) and sphenoid sinuses (n = 162, 75%). One hundred and forty-seven patients (68%) showed involvement of all three sinuses. Intracranial extension was observed in 29 (13%) on imaging and an additional 8 patients (4%) had cranial nerve palsies. A comparison of predisposing factor and outcomes between mucormycosis at different sites is shown in Table 3. Those with involvement of the cerebral, GI or disseminated disease had significantly higher mortality, that occurred significantly sooner compared to mucormycosis involving the sinuses, orbit, or lungs. This subset was also marginally older, had a lower percentage of patients with high dose steroid administration and uncontrolled DM. The several sites of involvement and the operative findings have been shown in Fig. 1.
In those with sinus and orbital involvement (n = 179), the commonest symptoms were headache (n = 85, 47%), and pain in the facial region, jaw pain and retroorbital (n = 88, 49%) regions. Proptosis was the commonest symptom in those with orbital involvement (n = 68, 39%) while 53 (30%) presented with visual blurring or frank visual loss. In those with pulmonary involvement (n = 25), all had cough and dyspnea (100%) while 20 (80%) had chest pain and 11 (35%) had hemoptysis.
Treatment
The commonest medical treatment offered to patients was a combination of Liposomal amphotericin—B and Posaconazole, in a sequential manner (n = 141, 65%) followed by a combination of Liposomal amphotericin-B and Isavuconazole sequentially (n = 21, 10%) and lyophilized amphotericin B with Posaconazole sequentially (n = 22, 10%). Liposomal amphotericin B, the mainstay of medical management, was administered for a median of 17 days (IQR 11–22 days, range 1–36 days). Retrobulbar amphotericin-B was used in only 4 eyes. Similarly, functional endoscopic sinus surgery (FESS) was the mainstay of surgical management and was done in all cases of ROCM. Table 4 shows a summary of all surgical procedures done as per the anatomical site involved. Orbital exenteration was performed in 21% of cases with orbital involvement, while two-thirds of cases with pulmonary involvement underwent lobectomy and all patients with GI involvement underwent a hemicolectomy.
Outcomes and survival
A total of 31 (14%) patients died due to mucormycosis during the 6 weeks follow-up. Of these, 8 (26%) had sinus involvement, 9 (29%) had ROCM, 6 (19%) had pulmonary involvement, 5 (16%) had GI involvement, 1 (3%) had cerebral, 2 (7%) had disseminated mucormycosis. The cumulative probability of death (Fig. 2) increased from 9.7% (95% CI 6.4–14.5%) at day 10–14.4% (95% CI 10.2–20.2%) on day 20 and marginally more to 15.3% (95% CI 10.8–21.3%) on day 30. The cumulative probability of death was significantly higher in those with cerebral, GI and disseminated disease (Fig. 3) (log-rank p < 0.001). A univariate and multivariable Cox proportional hazards modeling showed that patients with cerebral or GI or disseminated mucormycosis had a 9 times higher risk of death (p < 0.001) compared to other locations. Pulmonary mucormycosis also had more than twice the higher risk of death compared to sinus involvement alone, though this was not statistically significant. Patients who required mechanical ventilation during their COVID-19 management were also at a 55% higher risk of mortality, but this was only marginally significant (p = 0.07). In pulmonary mucormycosis, of the 16 who underwent lobectomy, only 1 died (6%) while 5 out of 9 (55%) who did not undergo the procedure died (p < 0.001). However, 5/6 (83%) with GI involvement underwent hemicolectomy and still died (Fig. 4).
Discussion
Our study is the first large study which looks into the multisystemic manifestations of CAM, its predisposing factor and outcome. Most of the CAM cases were detected within 15.2 + 9.1 days of COVID-19 diagnosis. Rhino-orbital mucormycosis was the commonest form of CAM, as has been detected in other studies, with relatively better outcome in comparison with the CAM of other sites. Uncontrolled diabetes mellitus, hypoxemia due to COVID-19 and inappropriate use of glucocorticoid drugs were independent predisposing factor for the development of CAM. The sequential use of antifungal drugs along with surgery was associated with improved survival at 6 weeks follow-up.
In our study, men were affected by CAM in 82% of the cases, as has been observed in previous studies [5, 6]. We identified diabetes mellitus to be the most common underlying disease in CAM patients, with 88% of them being diabetic, which corroborated that of the 2 large Indian studies on CAM, where 62.7% (n = 136) and in 78% (n = 2194) of the patients were diabetic [6, 9]. A meta-analysis of the cases CAM also showed that diabetes was the major risk factor for CAM. Diabetes mellitus in COVID-19 patients is a proinflammatory state and has been linked to the destruction of the pancreatic beta cells by the SARS CoV-2 virus [10, 11]. Newly detected diabetes mellitus was diagnosed in 27(12%) of our patients. The mean blood glucose was 273 + 108 mg, with mean glycated hemoglobin (HbA1c) of 9.4 + 2.4 gm% at admission, which was comparable to another study conducted in India [12]. Presence of diabetic ketoacidosis has been associated with CAM in 12% of the cases in our study, which is comparable to the diabetic ketoacidosis incidence of 14.9% in a systematic review of 101 CAM cases in India [13].
Use of high-dose glucocorticoids was present in about 80% of the CAM patients in our study, which was similar to the glucocorticoid use in the other large study from India, where 87% (n = 2073) received the glucocorticoids [9]. Even those with mild to moderate COVID-19 disease were given high-dose glucocorticoids and higher-order antibiotics, making such patients more predisposed to Mucor, especially in the setting of co-existent diabetes mellitus. Thus, there is a clear indication towards indiscriminate use of high-dose glucocorticoids. High-dose glucocorticoids and other immunomodulatory therapies have been associated with immune dysregulation [14, 15] and whether they predispose these patients to mucormycosis still needs to be proven. Lymphopenia is commonly seen in COVID-19 patients, and progressive lymphopenia has been associated directly with COVID-19 severity [16]. Lymphopenia was detected in 78% patients in our study, which could be associated with immune dysregulation and thus, increased risk of CAM in these patients. Immunomodulators such as tocilizumab have been shown to be associated with an increased risk of invasive candidiasis [17]. In our study, 11(5%) of the CAM patients had received immunomodulators. CAM has been associated with high mortality rates [18].
The appropriate management of mucormycosis involves effective control of hyperglycemia, and other predisposing factor, optimal surgical debridement and medical management with appropriate antifungal drugs for an adequate duration. Amphotericin B, the antifungal drug of choice was used in a sequential manner with the other antifungals including posaconazole and isavuconazole, in the consolidation phase [19], which was used in 184 (n = 85%)of the cases. The role of combined surgical debridement and medical approach with antifungals being associated with better survival has been supported in literature [20], and surgery was performed along with antifungals in 96% of our CAM cases (n = 208). Use of a combination of antifungals for the management of mucormycosis has not been well studied [21] and but was used in the management of our cases with extensive disease and multi-systemic involvement.
However, the mortality rate in our study was impressively low amounting to 31 (14%), which matches with the large series of 2826 cases of rhino-orbito-mucormycosis cases across India, published recently, which has a mortality rate of 14% (n = 305) [9]. The mortality rates in the said study was less than 10% in the group with nasal sinus involvement only and 11% in the cases with orbital involvement, with an overall mortality rate of 14%. The lower rates of mortality in our study could be attributable to the timely surgical debridement and FESS undertaken in most of the cases, which saves lives. The rates of death were significantly higher in those with cerebral, gastrointestinal and disseminated disease, having a nine times higher risk of death, as compared to other locations. Those with cerebral, GI and disseminated disease are at the highest risk of death and should be triaged and taken up for treatment on priority. In our study, the hemicolectomy could not salvage the life of the GI mucormycosis patients.
Our study has certain limitations. The first limitation is it included cases from a single state in India, and hence may not be representative of the appropriate incidence of the disease in India. We also did not include the non-COVID mucormycosis cases encountered during the study period, which could have helped us in finding the appropriate increase in the incidence of CAM. The follow-up of the patients is limited and many patients are still under active treatment, thus requiring the outcome to be analyzed with a bit of caution. There is also no large-scale data on the patients of COVID-19 who did not develop CAM, which could have served as a control group. Thus, prospective studies would be more helpful in ascertaining the predisposing factor. The strength of our study is a large number of cases which gives credibility to our findings and the inclusion of multisystemic cases of mucormycosis, which gives us an overview of the rare but not uncommon cases of CAM during the ongoing pandemic.
In conclusion, mucormycosis has emerged as an epidemic within the pandemic and it is very essential to curb its rising menace, with appropriate management by a multi-disciplinary approach including medical and surgical specialties. The predisposing factor identified included uncontrolled diabetes, indiscriminate usage of steroids and usage of higher antibiotics. The factors influencing mortality included the site of involvement of CAM, and the timing of administration of appropriate surgical and medical management. The incidence of mucormycosis has increased immensely during the second wave of COVID in India, and hence clinicians should be aware and have uptodate knowledge of the predisposing factor, clinical signs and symptoms, diagnostic modalities and treatment strategies of the various types of CAM. Also, the treating physicians should remember the predisposing factor involved in the development of CAM, the improper use of glucocorticoids, and should manage COVID cases accordingly with the appropriate drugs, thus preventing the huge increase in CAM cases.
References
Ripa M, Galli L, Poli A, Oltolini C, Spagnuolo V, Mastrangelo A, COVID-BioB study group, et al. Secondary infections in patients hospitalized with COVID-19: incidence and predictive factors. Clin Microbiol Infect. 2021;27:451–7. https://doi.org/10.1016/j.cmi.2020.10.021 (Epub 2020 Oct 24. PMID: 33223114; PMCID: PMC7584496).
Seaton RA, Gibbons CL, Cooper L, Malcolm W, McKinney R, Dundas S, et al. Survey of antibiotic and antifungal prescribing in patients with suspected and confirmed COVID-19 in Scottish hospitals. J Infect. 2020;81:952–60. https://doi.org/10.1016/j.jinf.2020.09.024 (Epub 2020 Sep 26. PMID: 32987097; PMCID: PMC7518971).
Nucci M, Barreiros G, Guimarães LF, Deriquehem VAS, Castiñeiras AC, Nouér SA. Increased incidence of candidemia in a tertiary care hospital with the COVID-19 pandemic. Mycoses. 2021;64:152–6. https://doi.org/10.1111/myc.13225 (Epub 2020 Dec 10. PMID: 33275821; PMCID: PMC7753494).
van Arkel ALE, Rijpstra TA, Belderbos HNA, van Wijngaarden P, Verweij PE, Bentvelsen RG. COVID-19-associated pulmonary aspergillosis. Am J Respir Crit Care Med. 2020;202:132–5. https://doi.org/10.1164/rccm.202004-1038LE (PMID:32396381;PMCID:PMC7328331).
Hoenigl M, Seidel D, Carvalho A, Rudramurthy SM, Arastehfar A, Gangneux JP, Nasir N, Bonifaz A, Araiza J, Klimko N, Serris A, et al. The emergence of COVID-19 associated mucormycosis: analysis of cases from 18 countries. Lancet Microbe. 2022. https://doi.org/10.1016/S2666-5247(21)00237-8.
Patel A, Agarwal R, Rudramurthy SM, Shevkani M, Xess I, Sharma R, MucoCovi Network, et al. Multicenter epidemiologic study of coronavirus disease-associated mucormycosis, India. Emerg Infect Dis. 2021. https://doi.org/10.3201/eid2709.210934 (Epub ahead of print. PMID: 34087089).
Patel A, Kaur H, Xess I, Michael JS, Savio J, Rudramurthy S, et al. A multicentre observational study on the epidemiology, predisposing factor, management and outcomes of mucormycosis in India. Clin Microbiol Infect. 2020;26:944.e9-944.e15. https://doi.org/10.1016/j.cmi.2019.11.021 (Epub 2019 Dec 4 PMID: 31811914).
Cornely OA, Alastruey-Izquierdo A, Arenz D, et al. Global guideline for the diagnosis and management of mucormycosis: an initiative of the European Confederation of Medical Mycology in cooperation with the Mycoses Study Group Education and Research Consortium. Lancet Infect Dis. 2019;19:e405–21. https://doi.org/10.1016/S1473-3099(19)30312-3.
Sen M, Honavar SG, Bansal R, Sengupta S, Rao R, Kim U, et al. Epidemiology, clinical profile, management, and outcome of COVID-19-associated rhino-orbital-cerebral mucormycosis in 2826 patients in India—Collaborative OPAI-IJO Study on Mucormycosis in COVID-19 (COSMIC), Report 1. Indian J Ophthalmol. 2021;69:1670–92. https://doi.org/10.4103/ijo.IJO_1565_21.
Müller JA, Groß R, Conzelmann C, Krüger J, Merle U, Steinhart J, et al. SARS-CoV-2 infects and replicates in cells of the human endocrine and exocrine pancreas. Nat Metab. 2021;3:149–65. https://doi.org/10.1038/s42255-021-00347-1 (Epub 2021 Feb 3 PMID: 33536639).
Accili D. Can COVID-19 cause diabetes? Nat Metab. 2021;3:123–5. https://doi.org/10.1038/s42255-020-00339-7 (PMID: 33432203).
Mishra Y, Prashar M, Sharma D, Akash KVP, Tilak TVSVGK. Diabetes, COVID 19 and mucormycosis: clinical spectrum and outcome in a tertiary care medical center in Western India. Diabetes Metab Syndr. 2021;15:102196. https://doi.org/10.1016/j.dsx.2021.102196 (Epub ahead of print. PMID: 34246939; PMCID: PMC8253699).
Singh AK, Singh R, Joshi SR, Misra A. Mucormycosis in COVID-19: a systematic review of cases reported worldwide and in India. Diabetes Metab Syndr. 2021;15:102146. https://doi.org/10.1016/j.dsx.2021.05.019 (Epub 2021 May 21. PMID:34192610; PMCID: PMC8137376).
Files JK, Boppana S, Perez MD, Sarkar S, Lowman KE, Qin K, et al. Sustained cellular immune dysregulation in individuals recovering from SARS-CoV-2 infection. J Clin Investig. 2021;131(1): e140491. https://doi.org/10.1172/JCI140491 (PMID:33119547;PMCID:PMC7773371).
Ghuman H, Voelz K. Innate and adaptive immunity to mucorales. J Fungi (Basel). 2017;3(3):48. https://doi.org/10.3390/jof3030048 (PMID:29371565;PMCID:PMC5715954).
Zhang X, Tan Y, Ling Y, Lu G, Liu F, Yi Z, et al. Viral and host factors related to the clinical outcome of COVID-19. Nature. 2020;583:437–40. https://doi.org/10.1038/s41586-020-2355-0 (Epub 2020 May 20 PMID: 32434211).
Antinori S, Bonazzetti C, Gubertini G, Capetti A, Pagani C, Morena V, et al. Tocilizumab for cytokine storm syndrome in COVID-19 pneumonia: an increased risk for candidemia? Autoimmun Rev. 2020;19:102564. https://doi.org/10.1016/j.autrev.2020.102564 (Epub 2020 May 5. PMID: 32376396; PMCID: PMC7200127).
Garg D, Muthu V, Sehgal IS, Ramachandran R, Kaur H, Bhalla A, et al. Coronavirus Disease (Covid-19) Associated Mucormycosis (CAM): case report and systematic review of literature. Mycopathologia. 2021;186:289–98. https://doi.org/10.1007/s11046-021-00528-2 (Epub 2021 Feb 5. PMID: 33544266; PMCID: PMC7862973).
Cornely OA, Alastruey-Izquierdo A, Arenz D, Chen SCA, Dannaoui E, Hochhegger B, et al. Global guideline for the diagnosis and management of mucormycosis: an initiative of the European Confederation of Medical Mycology in cooperation with the Mycoses Study Group Education and Research Consortium. Lancet Infect Dis. 2019;19:e405–21. https://doi.org/10.1016/S1473-3099(19)30312-3 (Epub 2019 Nov 5. PMID: 31699664).
Muthu V, Agarwal R, Dhooria S, Sehgal IS, Prasad KT, Aggarwal AN, et al. Has the mortality from pulmonary mucormycosis changed over time? A systematic review and meta-analysis. Clin Microbiol Infect. 2021;27:538–49. https://doi.org/10.1016/j.cmi.2020.12.035 (Epub 2021 Jan 5 PMID: 33418022).
Jeong W, Keighley C, Wolfe R, Lee WL, Slavin MA, Chen SC, et al. Contemporary management and clinical outcomes of mucormycosis: a systematic review and meta-analysis of case reports. Int J Antimicrob Agents. 2019;53:589–97. https://doi.org/10.1016/j.ijantimicag.2019.01.002 (Epub 2019 Jan 10 PMID: 30639526).
Acknowledgements
Members of MuCOVIDYH group include: Bharat Ashok Vaswani, Pradeep Kumar Mishra, Tushar Ramrao Nemmaniwar, Vighnesh Y. Naidu, Kavangal Dhananjay, Gurunath J.M., Bongu Karthik Rao, Narahari Yamjala, Bhavin Ram, Pranith Ram Mamidi, Prasad Babu TLVD, Vikas Agrawal, Kandaraju Sai Satish, Sapna Marda, Sashi K Srivastav, Shivaram Rao Komandla, Vallumkonda Suresh Babu, Urmila Anandh, Mahesh Gudelli, Radhika TK (Yashoda Hospital, Secunderabad unit), Anjani Kumar Darapureddy, Hemanth Chirumamilla, Naveen Reddy, Jaydip Ray Chaudhuri, Sastry VSSRK Kambhapati, K Seshikiran (Yashoda Hospital, Somajiguda unit), M Haritha, G Krishna Mohan Reddy, VenuGopal Reddy P, Viswesvaran Balasubramaniam, Abhijeet Ingle (Yashoda Hospitals, Malakpet unit); Tanya A Punjani (Emory College, Atlanta, Georgia, USA).
Funding
None to declare.
Author information
Authors and Affiliations
Consortia
Contributions
All listed authors meet the ICMJE criteria. We attest that all authors contributed significantly to the creation of this manuscript, each having fulfilled the criteria as established by the ICMJE.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
IRB approval
The study was approved by the Institute Ethics Committee, Yashoda Academy of Medical Education and Research, India. [IEC-YAMER DC/PP-02/2021].
Additional information
The members of MuCOVIDYH group are listed in acknowledgements.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sahu, M., Shah, M., Mallela, V.R. et al. COVID-19 associated multisystemic mucormycosis from India: a multicentric retrospective study on clinical profile, predisposing factors, cumulative mortality and factors affecting outcome. Infection 51, 407–416 (2023). https://doi.org/10.1007/s15010-022-01891-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s15010-022-01891-y