Abstract
Ceftazidime–avibactam (CZA), meropenem–vaborbactam (MVB) and imipenem–relebactam (I–R) are combinations of old ß-lactams with novel non-ß-lactam ß-lactamase inhibitors (BLBLIs) able to inhibit some carbapenemases, such as the KPC-type, thus are becoming the standard for difficult-to-treat carbapenemase-producing Enterobacterales (CPE); a practical question is whether these novel BLBLIs should be used as monotherapy or as part of a combination regimen with other antibiotics, and if so, with which ones, to reduce the emergence of resistant strains and to optimize their efficacy. In this short review, we assessed clinical outcomes in patients with CPE-infections treated with the novel BLBLIs as mono- or combo-regimens, and laboratory studies on the synergistic effects with other antimicrobials. Available evidence on combination therapy is scarce and mainly limited to retrospective studies involving 630 patients treated with CZA: aminoglycosides were used in 39.6% of 336 patients treated with combo-regimens, followed by polymyxin B/colistin (24.4%), tigecycline (24.1%), carbapenems (13.4%) and fosfomycin (5.4%). Aminoglycosides could be useful in case of bloodstream and severe urinary infections. Pneumonia is a risk factor for CZA-resistance emergence: fosfomycin, due to favorable lung pharmacokinetics/pharmacodynamics, could represent an interesting partner; fosfomycin could be added also for osteomyelitis. Tigecycline could be preferred for intrabdominal and skin-soft tissue infections. Due to nephrotoxicity and lack of in vitro synergy, the association CZA/colistin seems not optimal. MVB and I–R were mostly used as monotherapies. Currently, there is no definitive evidence whether combinations are more effective than monotherapies; further studies are warranted, and to date only personal opinions can be provided.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ceftazidime–avibactam (CZA, CAZ–AVI), meropenem–vaborbactam (MVB, MEM-VAB) and imipenem/cilastatin–relebactam (I–R, IPM–REL) represent recent or upcoming combinations of old ß-lactams with novel synthetic non-ß-lactam ß-lactamase inhibitors (BLBLIs) able to inhibit extended-spectrum β-lactamases (ESBLs), AmpCs and some carbapenemases, such as the Klebsiella pneumoniae carbapenemase (KPC). AVI displays activity also towards some class D β-lactamases (such as OXA-48); VAB also restored the activity of meropenem against class A and class C ß-lactamase-producing K. pneumoniae strains with reduced permeability due to porin mutations [1,2,3]. Therefore, these novel BLBLIs represent an interesting option for managing severe infections due to carbapenem-resistant (CR) carbapenemase-producing Enterobacterales (CPE), although they are not active on strains producing class B metallo-ß-lactamases (MBLs), such as the New-Delhi metallo-ß-lactamase (NDM). Zidebactam and nacubactam are ongoing enhancer derivatives of the diazabicyclooctane scaffold with promising in vitro activity against MBL-producing Enterobacterales; taniborbactam is a boronic-acid-containing ß-lactamase inhibitor able to inhibit ß-lactamases belonging to class A, B, C and D [3].
KPC-producing Klebsiella pneumoniae (KPC-Kp) is nowadays endemic in several countries, is mainly driven by nosocomial spread [4], and is associated with a mortality of about 40% [5], but even over 50% [6] for bloodstream infections (BSIs) when treated with the best available therapy (BAT) prior these new options. The optimal treatment of infections due to KPC-Kp is not known, nor whether combination therapy would be superior to monotherapy; lacking the results of well-conducted randomized clinical trials (RCTs), the therapeutic choice is mainly based on clinical experience and expert opinions. For CPE other than KPC-Kp, such as NDM-producers that recently resulted in sustained transmission in the North-West area of Tuscany, Italy [7], data on the most appropriate treatment is even more scarce.
BAT against KPC-producers classically consisted of various combinations of antibiotics displaying in vitro activity against the isolate (i.e., aminoglycosides, colistin, fosfomycin and tigecycline), often in associations with high-dose meropenem (especially for MIC ≤ 8 mg/L) [8]; in the more severely ill patients the combination therapy has been shown to be associated with a lower mortality compared to monotherapy [5]. Other agents, such as cefiderocol, plazomicin and eravacycline, represent novel interesting not-BLBLI options for CPE treatment [9, 10].
Tumbarello et al. [6] showed that in patients with BSIs due to KPC-Kp, a salvage therapy with CZA was associated with a 30-day mortality significantly lower than that of a matched cohort treated with second-line regimens containing other antibiotics (36.5% vs 55.8%, p = 0.005), suggesting a survival benefit relatively to the commonly used regimens.
Despite the unsatisfactory outcomes with the traditional BAT, and the obvious expectations placed on these novel carbapenemase-inhibitors, there is currently scarce evidence of clinical use of the novel BLBLIs for real-life management of the difficult-to-treat infections caused by CPE. Moreover, these drugs were often administered only as salvage therapy on a compassionate-use basis, because most phase 2 and 3 trials conducted to support their marketing authorization were conducted mainly in complicated intra-abdominal and urinary infections and compared with carbapenems so that the patients whose infections were caused by carbapenem-resistant strains were excluded from enrollment.
Many doubts are so nowadays unanswered by current literature. A practical question is whether these novel BLBLIs should be used, for the real-life treatment of CPE infections, as monotherapy or as part of a combination regimen with other antibiotics. And if so, with which ones? A combination therapy would have, in fact, the advantage of exploiting the bactericidal synergy between different drugs, as well as limiting the risk of selection of resistant mutant strains with a single antimicrobial, but these remain only hypotheses needing proof.
The aim of this short review is to try to address this knowledge gap, extracting from the available literature data regarding the clinical outcomes, mainly the mortality and clinical success (as defined in the different studies), for patients with infections due to carbapenem-resistant Enterobacterales (CRE) treated with the novel BLBLIs as mono- or combo-regimens. In addition, we searched in vitro studies investigating the effects of the mono- and combo-regimens.
Methods
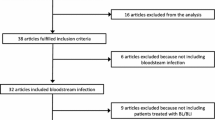
A comprehensive computerized search was performed using PubMed, through November 15, 2020, involving both Medical Subject Headings (MeSH) terminology and relevant keywords for search strings. The following terms were searched in combination: Enterobacteriaceae, Enterobacterales, carbapenemase, avibactam, relebactam, vaborbactam, combination, therapy, synergism/synergy, resistance, carbapenem-resistant. References of retrieved articles were manually searched to ensure the identification of studies not found in the initial literature search. The selection was limited to peer-reviewed publications written in English. After de-duplication, all authors independently screened titles and abstracts, and finally full texts, to identify all potentially relevant studies, resolving discrepancies through discussion and consultation between them. Moreover, a search was performed on https://clinicaltrial.gov for the studies on the new BLBLIs for CRE infections, in progress or waiting to begin or completed but not yet published. Authors of the included studies were contacted via e-mail if further study details were needed.
Results
We retrieved 11 observational retrospective [6, 11,12,13,14,15,16,17,18,19,20] and 2 prospective studies [21, 22] for a total of 630 patients treated with CZA (294 as monotherapy and 336 as combination therapy). Two of these studies specifically addressed the treatment of infections due to OXA-48 producing Enterobacterales, for a total of 81 patients treated with CZA as mono- or combo-therapy [18, 19]. We retrieved also a meta-analysis enrolling 11 retrospective studies comparing the clinical effects of CZA as monotherapy or combo against infections due to CRE and/or CR-Pseudomonas aeruginosa [23]: eight [6, 13,14,15,16,17,18,19] of the 11 studies included in the meta-analysis have been included in the present review.
As for MVB, we retrieved one RCT (TANGO II) with a total of 32 patients with CRE infections all treated with monotherapy [24] and a retrospective multicenter study enrolling 26 patients most of which (84.6%) treated with monotherapy [20].
We retrieved one RCT (RESTORE-IMI 1) [25] on imipenem non-susceptible bacterial infections, with a total of 21 patients treated with I–R as monotherapy compared with 10 patients treated with imipenem plus colistin: only 5 patients were treated with I–R for infections due to CRE, being the remaining CR-Pseudomonas aeruginosa infections, which are not the topic of this review.
Regarding the studies currently in progress (consultable on https://clinicaltrial.gov), we retrieved a phase 3 prospective, randomized, multicenter, open-label, parallel group trial (NCT03580044) designed to evaluate the association of aztreonam-avibactam as monotherapy for MBL-producing Gram-negative bacteria, but to date is not yet recruiting patients.
Most available evidence on the novel BLBLIs as mono or combo regimens for the treatment of infections due to CR-CPE are thus limited to CZA, and relies on relatively small sample size cohorts and on observational retrospective studies.
Table 1 shows the main characteristics of the clinical studies assessing the effectiveness of the novel BLBLIs as mono- or combo-regimens for CRE infections.
Regimens containing CZA were usually classified as combination therapy if they included at least one other antimicrobial displaying activity against the isolate, but also carbapenems were administered in several cases: carbapenems were associated in 45/336 cases (13.4%), but in the study of van Duin et al. [21] they were used in 46% of cases. Aminoglycosides were overall used in 133/336 patients (39.6%), representing the main class usually associated with CZA, followed by polymyxin B or colistin (82/336, 24.4%), tigecycline (81/336, 24.1%), and fosfomycin (18/336, 5.4%).
A non-significant trend of improvement of clinical success was reported for CZA combination therapy only in 3 studies [11, 12, 15], while in 6 studies this outcome was (albeit not significantly) better in the monotherapy group [13, 16,17,18,19,20]
Shields et al. [12] reported a higher mortality for CZA as monotherapy when compared with combination regimen with gentamicin (12.5% vs 0%); also Tumbarello et al. [6] and Guimaraes et al. [22] reported higher mortality rate for patients treated with CZA as monotherapy, while other authors [13, 16, 18,19,20] found higher mortality in combo group.
Tumbarello et al. [6] showed that, among patients with KPC-Kp BSIs, CZA was significantly more effective than alternative regimens in reducing 30-day mortality both as mono- or combination therapy. The mortality was 40.9% in the monotherapy group and 35.4% in the combination group: in the combo group, the mortality was 50% in association with amikacin, 38.5% with colistin, 37.5% with tigecycline, 36.9% with carbapenems, 32% with gentamicin, 28.6% with fosfomycin.
Guimaraes et al. [22], in case of severe infections caused by KPC-producing Enterobacterales co-resistant to carbapenems and polymyxins and treated with CZA as salvage treatment, reported a trend (although not significant) for a lower 14-day mortality for combo- than mono-therapy (21.4% vs 40%).
In the meta-analysis of Onorato et al. [23], among 377 patients with infections due to CRE, the mortality rate was 38.7% (74/191) for combo and 31.2% (58/186) for mono-therapy (RR = 1.20, 95% CI 0.89–1.61; p = 0.229). Similarly, no difference was found analyzing the rate of microbiological cure reported in 6 studies (63.3% for combo- vs. 61.9% for mono-therapy; RR = 1.05, 95% CI 0.84–1.31, p = 0.653). All studies except one reported data on the emergence of resistance to CZA, with a total of 8 patients (4.1%) in the mono and 6 patients (3.0%) in the combo group.
Combination therapy of MVB with other antibiotics has been evaluated by Ackley et al. [20], but data is limited to only 3 patients co-treated with polymyxins and 1 with tigecycline.
Table 2 shows some in vitro studies assessing the synergistic effects of the novel BLBLIs in combination with different antimicrobials on CRE. As clinical studies, most data refer to CZA.
Discussion
Currently, there is not enough high-level clinical evidence to make definitive conclusions whether the novel BLBLIs used as combo regimens are more effective than monotherapy for CPE infections; in some studies, the mortality rate among patients treated with combination therapy was even higher than in monotherapy group [13, 16, 18,19,20], but in retrospective studies an indication bias towards more severely ill patients could be the explanation for the lack of benefit of combo regimens observed.
Most of the studies we retrieved in the literature were, in fact, observational, a fact which does not allow assessment of efficacy, but only of effectiveness in real life. Definitive evidence from clinical trials of the adequate methodology is, therefore, crucial.
Despite the optimal combination regimen remains unknown, clinical and laboratory studies suggest some considerations; nevertheless, at the present time, only considerations reflecting personal opinion can be made about this specific topic.
Drug combinations are widely used in clinical practice to prevent the emergence of resistance; however, unfavorable effects on resistant selection cannot be excluded: Liu et al. [36] showed that the rapid evolution of tolerance is a major survival factor for bacteria, and tolerance can promote the evolution of resistance under antimicrobial combinations that were instead expected to prevent resistance.
CZA was used as combo regimen in over 50% of cases with CPE infections, in real-life. Aminoglycosides, such as gentamicin or amikacin (depending by in vitro susceptibility results), are rapidly and durably bactericidal and effective in the treatment of carbapenem-resistant K. pneumoniae BSIs, and are especially useful when the sources of infection are amenable to reliable pharmacokinetics, such as BSIs due to urinary sources; intraabdominal sources (especially in presence of biliary obstruction or hepatic damage) and respiratory tract and bone are instead characterized by a very poor penetration [37]. Shields et al. [12] effectively used combination therapy with short-course gentamicin followed by de-escalation to CZA alone as a strategy for maximizing treatment effectiveness while limiting toxicity, mainly represented by acute kidney injury. In the study of Tumbarello et al. [6], 8.7% of patients, mostly with BSIs, experienced KPC-Kp infection relapses after CZA alone treatment was discontinued: in all cases the isolates remained susceptible to CZA, and clinical and/or microbiological cures were achieved after retreatment with CZA plus gentamicin. Nevertheless, using a gradient synergy test, the combination of CZA with gentamicin (the same resulted also for ciprofloxacin) displayed no synergy against any of the KPC-Kp isolates tested in an in vitro study [26]. However, synergistic activities observed in vitro may not exactly correlate with clinical efficacy, for the peculiar in vivo pharmacokinetic properties displayed by the antibiotics.
In effect, a combination regimen could result in a synergistic activity, very useful in the most serious infections and in case of sepsis and septic shock, and it might suppress CZA resistance selection. Pneumonia has been recognized as a risk factor for CZA resistance among patients with CRE infections [17], and Krapp et al. [13] previously showed that patients with pneumonia due to CR-Kp had poorer outcomes (failure or relapse), suggesting potential limitations in the treatment of CR-Kp pneumonia with CZA and highlighting the need for adequate studies about the activity and bioavailability of CZA in the lungs, and therefore the need to assess the optimal dosing (such as increased or loading doses) of CZA, especially in septic patients. In effect, clinical data on optimal dosing are generally lacking for the novel BLBLIs thus, variations in extracellular volume and renal dysfunction often observed in critically ill patients may impact the disposition of both β-lactams and β-lactamase inhibitors [3]. Fosfomycin, which has excellent pharmacokinetic characteristics in the lung, could represent a very interesting option to guarantee a synergistic action and to intervene in cases where CZA resistance could more likely occur. The synergy of CZA in combination with fosfomycin against carbapenem-resistant K. pneumoniae has been shown in recent in vitro studies [33, 34].
Fosfomycin could be useful also for osteomyelitis, due to very high bone penetration of this drug. However, despite the manageability and the efficacy of this option, this association was rarely used in clinical practice, only in 5.4% of total patients we retrieved.
Polymyxin B and E (colistin, CST) have been used in combination with CZA in 24.4% of cases we retrieved, but despite the rationale that membrane permeabilization by colistin could facilitate increased access of CZA to its target sites, in an in vitro study [27] the combination of CZA and CST did not suppress CZA resistance and did not provide a benefit in potentiating the killing of most CRE isolates. Moreover, the study of van Duin et al. [21] showed that patients treated with CZA, compared to those treated with CST, were less likely to die and more likely to be discharged home at 30 days, and an analysis using the desirability of outcome ranking method showed that the IPTW(inverse probability of treatment weighting)-adjusted probability of a better outcome on CZA compared with colistin is 64%. No improvement of in vitro bactericidal activity and in vivo efficacy has been shown when polymyxin B is combined with CZA against KPC-Kp [35]. In addition, in consideration of the known nephrotoxicity of CST, if another option is available, the association of CZA with CST should be, in our opinion, avoided, reserving CST for cases in which it is the only active agent and the CZA monotherapy has the greatest risk of failure, as when the MIC of CZA is very close to the susceptibility breakpoint, or in case of pneumonia, as previously discussed.
In general, a combination therapy for CZA could be particularly useful when the MIC values for CZA are very close to the susceptibility clinical breakpoint (8 mg/L) [38, 39], as the in vitro study of Nath et al. [31] confirmed. However, it should also be noted that in the study of Shields et al. [17] a “monotherapy” did not represent a significant risk factor for CZA resistance among patients with microbiologic failure.
Tigecycline was often used in combination with CZA (24.1%), and could be useful when the infection source is the abdomen, especially the biliary tract, or skin and soft tissues, given its favorable pharmacokinetic in these districts, avoiding its use for pneumonia and BSIs. However, it is important to note that a recent in vitro study using the E-test MIC:MIC ratio synergy method [33] showed that the combination of CZA with tigecycline is synergistic only for 5% of carbapenemase-producing K. pneumoniae isolates tested (this percentage reached 12.5% for KPC-producing strains), being indifferent in the remaining cases.
Carbapenems were sometimes used in combination with CZA (13.4% of cases). The rationale goes through the reduction of MIC of carbapenems provided by avibactam, allowing to recover the favorable pharmacokinetics/pharmacodynamics of these antibiotics. Notably, CZA reduced MIC of meropenem and particularly for imipenem below the resistance breakpoints against KPC-Kp strains tested in an in vitro model using the gradient synergy test, displaying a synergistic activity both in CZA susceptible and resistant isolates, while for ertapenem a synergistic effect was observed only against CZA-susceptible strains [26]. Recently, CZA showed to be very active and synergistic with meropenem against multiresistant Serratia marcescens isolates carrying blaKPC-2 [40], and a synergy for CZA-ertapenem was found in all KPC-Kp isolates tested by Ojdana et al. [33]. Furthermore, in certain isolates of K. pneumoniae the development of resistance-conferring blaKPC-3 mutations during CZA exposure is associated with restoration of carbapenem susceptibility [41], thus the carbapenem/CZA combination might mitigate the emergence of blaKPC mutations or, alternatively, treat emerging resistant subpopulations. Notably, even 30% of 10 patients with microbiologic failure observed in the study of Shields et al. [11] developed CZA resistance (MIC > 8 mg/L) following a median of 15 days of treatment, but in 2 of these 3 patients the mutations so reduced the meropenem MIC to restore the full susceptibility and allowing an effective salvage treatment with the carbapenem therapy [41].
More recently, Gottig et al. [32], based on time-kill kinetics as well as an in vivo infection model, confirmed that the combination of CZA with IMP can represent an evaluable bactericidal strategy able to prevent the in vivo development of CZA resistance in KPC-producing Enterobacterales.
Unfortunately, the limit of combining CZA with a carbapenem is thwarting the carbapenem-sparing stewardship policies.
Awaiting the future marketing of aztreonam-avibactam association, the combination of the novel BLBLIs (CZA, MVB, I–R) with aztreonam (ATM) retains currently great importance in overcoming the resistance conferred by MBLs [42], taking advantage of the stability of aztreonam to hydrolytic activity of the MBLs and of the ability of the new inhibitors to block the activity of the ß-lactamases frequently co-expressed by the MBL-producing Enterobacterales, both of Ambler class A (such as the ESBLs or the KPCs), class C (AmpCs) and class D [1, 28, 30, 42]. For the combinations involving a double ß-lactam strategy (such as ATM plus ceftazidime or carbapenems), a possible benefit could arise by the simultaneous inhibition of multiple penicillin-binding proteins [42].
The combination with ATM seems particularly useful in geographical regions where MBL-producing Enterobacterales (e.g. NDM-producers) predominate, such as the Asian continent [43] and Tuscany, Italy [7]; if a molecular antibiogram directly detecting the resistance mechanisms at the molecular (mainly genic) level cannot be performed, and the only tool available to clinicians is represented by the conventional antibiogram based on phenotypic detection of bacterial growth, especially when the novel BLBLIs (such as CZA and MVB) are not tested or turn out in a resistance, it is very difficult to understand if the CRE-isolate is expressing class A (such as KPC) and/or class B (such as NDM) carbapenemases: in these cases, in areas endemic for MBL-producers, the use of a combination of ATM with a BLBLI could be the more effective and practical solution. ATM could be combined both with CZA and MVB and I–R; despite the novel BLBLIs could be realistically largely interchangeable for the treatment of infections due to CPE in real-life, the combination with AVI is preferable when OXA-48-producing Enterobacterales are present [29].
Only one study [20] directly compared clinical outcomes in patients who received CZA versus MVB as mono or combo regimens for CRE infections: KPC-Kp was the primary causative organism in both groups, but in the MVB group there were proportionally more patients with ampC-harboring Enterobacterales [44]. Patients in the CZA arm received combination therapy more often than patients in the MVB arm (61.0% versus 15.4%; p < 0.01), no difference of mortality was observed at 30-day (19.1% vs 11.5%, p = 0.57) and 90-day (28.6 vs 26.9%, p = 0.48) for CZA and MVB groups, respectively, and the rate of adverse events was similar [20]. Clinical success was observed in 61.9% of patients in the CZA group and 69.2% in the MVB group (p = 0.49). The median duration of combination therapy was 8.8 days with CZA and 3.1 days with MVB (p = 0.08). Emergence of MVB resistance was not observed within the study period, while among 15 patients who were treated with CZA and had recurrent infections three developed drug resistance within 90 days: interestingly, all three patients were found to have respiratory sources and received renal replacement therapy [20], confirming what has been previously discussed on pneumonia as a risk factor for CZA resistance and the need of adequate considerations about the pharmacokinetics/pharmacodynamics of CZA in particular high-risk populations [13, 17]. This study [20] supported the use of MVB as the preferred monotherapy agent for KPC-producing Enterobacterales.
In conclusion, the role of combination regimens with the novel BLBLIs for the treatment of infections due to CPE, in order to prevent bacterial resistance and optimize their overall efficacy, deserves further investigations. We hope that well-conducted RCTs will be performed to clarify these important aspects so that these agents can be effectively used for the longest possible period of time and minimizing the selection of resistance.
Conflict of interest
C.T. has received fees for speaking at symposia organized on behalf of Pfizer, Novartis, Merck, Angelini, Zambon, Thermofischer, Biotest, Gilead, Hikma, Biomerieux and Astellas. S.M. and B.V.: none to declare.
Change history
19 March 2021
A Correction to this paper has been published: https://doi.org/10.1007/s15010-021-01596-8
References
Stewart A, Harris P, Henderson A, Paterson D. Treatment of infections by OXA-48-producing Enterobacteriaceae. Antimicrob Agents Chemother. 2018;62:e01195-e1218.
Crass RL, Pai MP. Pharmacokinetics and Pharmacodynamics of β-Lactamase Inhibitors. Pharmacotherapy. 2019;39:182–95.
Yahav D, Giske CG, Grāmatniece A, Abodakpi H, Tam VH, Leibovici L. New β-Lactam-β-Lactamase inhibitor combinations. Clin Microbiol Rev. 2020;34:e00115-e120.
David 2019) David S, Reuter S, Harris SR, Glasner C, Feltwell T, Argimon S, et al. EuSCAPE Working Group; ESGEM Study Group, Feil EJ, Rossolini GM, Aanensen DM, Grundmann H. Epidemic of carbapenem-resistant Klebsiella pneumoniae in Europe is driven by nosocomial spread. Nat Microbiol. 2019;4:1919–1929
Gutiérrez-Gutiérrez B, Salamanca E, de Cueto M, et al; REIPI/ESGBIS/ INCREMENT Investigators. Effect of appropriate combination therapy on mortality of patients with bloodstream infections due to carbapenemase-producing Enterobacteriaceae (INCREMENT): a retrospective cohort study. Lancet Infect Dis 2017;17:726–34
Tumbarello M, Trecarichi EM, Corona A, De Rosa FG, Bassetti M, Mussini C, et al. Efficacy of Ceftazidime–avibactam Salvage Therapy in Patients With Infections Caused by Klebsiella pneumoniae Carbapenemase-producing K. pneumoniae. Clin Infect Dis. 2019;68:355–64.
Tavoschi L, Forni S, Porretta A, Righi L, Pieralli F, Menichetti F, et al. On Behalf Of The Tuscan Clinical Microbiology Laboratory Network. Prolonged outbreak of New Delhi metallo-beta-lactamase-producing carbapenem-resistant Enterobacterales (NDM-CRE), Tuscany, Italy, 2018 to 2019. Euro Surveill. 2020;25:2000085.
Bassetti M, Giacobbe DR, Giamarellou H, Viscoli C, Daikos GL, Dimopoulos G, et al.; Critically Ill Patients Study Group of the European Society of Clinical Microbiology and Infectious Disease (ESCMID); Hellenic Society of Chemotherapy (HSC) and Società Italiana di Terapia Antinfettiva (SITA). Management of KPC-producing Klebsiella pneumoniae infections. Clin Microbiol Infect. 2018;24:133–144
Rodríguez-Baño J, Gutiérrez-Gutiérrez B, Machuca I, Pascual A. Treatment of infections caused by extended-spectrum-beta-lactamase-, AmpC-, and Carbapenemase-producing Enterobacteriaceae. Clin Microbiol Rev. 2018;31:e00079-e117.
Doi Y. Treatment Options for Carbapenem-resistant Gram-negative Bacterial Infections. Clin Infect Dis. 2019;9:565-575
Shields RK, Potoski BA, Haidar G, Hao B, Doi Y, Chen L, et al. Clinical outcomes, drug toxicity, and emergence of ceftazidime–avibactam resistance among patients treated for carbapenem-resistant enterobacteriaceae infections. Clin Infect Dis. 2016;63:1615–8.
Shields RK, Nguyen MH, Chen L, Press EG, Potoski BA, Marini RV, et al. Ceftazidime–avibactam Is Superior to Other Treatment Regimens against Carbapenem-Resistant Klebsiella pneumoniae Bacteremia. Antimicrob Agents Chemother. 2017;61:e00883-e917.
Krapp F, Grant JL, Sutton SH, Ozer EA, Barr VO. Treating complicated carbapenem-resistant enterobacteriaceae infections with ceftazidime/avibactam: a retrospective study with molecular strain characterisation. Int J Antimicrob Agents. 2017;49:770–3.
Castón JJ, Lacort-Peralta I, Martín-Dávila P, Loeches B, Tabares S, Temkin L, et al. Clinical efficacy of ceftazidime/avibactam versus other active agents for the treatment of bacteremia due to carbapenemase-producing Enterobacteriaceae in hematologic patients. Int J Infect Dis. 2017;59:118–23.
Temkin E, Torre-Cisneros J, Beovic B, Benito N, Giannella M, Gilarranz R, et al. Ceftazidime–avibactam as Salvage Therapy for Infections Caused by Carbapenem-Resistant Organisms. Antimicrob Agents Chemother. 2017;61
King M, Heil E, Kuriakose S, Bias T, Huang V, El-Beyrouty C, et al. Multicenter Study of Outcomes with Ceftazidime–avibactam in Patients with Carbapenem-Resistant Enterobacteriaceae Infections. Antimicrob Agents Chemother. 2017;61
Shields RK, Nguyen MH, Chen L, Press EG, Kreiswirth BN, Clancy CJ. Pneumonia and renal replacement therapy are risk factors for ceftazidime–avibactam treatment failures and resistance among patients with carbapenem-resistant Enterobacteriaceae infections. Antimicrob Agents Chemother 2018; 62. pii: e02497–17
Sousa A, Pérez-Rodríguez MT, Soto A, Rodríguez L, Pérez-Landeiro A, Martínez-Lamas L, et al. Effectiveness of ceftazidime/avibactam as salvage therapy for treatment of infections due to OXA-48 carbapenemase-producing Enterobacteriaceae. J Antimicrob Chemother. 2018;73:3170–5.
De la Calle C, Rodríguez O, Morata L, Marco F, Cardozo C, García-Vidal C, et al. Clinical characteristics and prognosis of infections caused by OXA-48 carbapenemase-producing Enterobacteriaceae in patients treated with ceftazidime–avibactam. Int J Antimicrob Agents. 2019;53:520–4.
Ackley R, Roshdy D, Meredith J, Minor S, Anderson WE, Capraro GA, Polk C. Meropenem–vaborbactam versus ceftazidime–avibactam for treatment of carbapenem-resistant enterobacteriaceae infections. Antimicrob Agents Chemother. 2020;64:e02313-e2319.
van Duin D, Lok JJ, Earley M, Cober E, Richter SS, Perez F, et al. Antibacterial Resistance Leadership Group. Colistin Versus Ceftazidime–avibactam in the Treatment of Infections Due to Carbapenem-Resistant Enterobacteriaceae. Clin Infect Dis. 2018; 66:163–171
Guimarães T, Nouér SA, Martins RCR, Perdigão Neto LV, Martins WMBS, et al. Ceftazidime/avibactam as salvage therapy for infections caused by Enterobacteriales co-resistant to carbapenems and polymyxins. Antimicrob Agents Chemother. 2019;63:e00528-e619.
Onorato L, Di Caprio G, Signoriello S, Coppola N. Efficacy of ceftazidime/avibactam in monotherapy or combination therapy against carbapenem-resistant Gram-negative bacteria: A meta-analysis. Int J Antimicrob Agents. 2019;54:735–40.
Wunderink RG, Giamarellos-Bourboulis EJ, Rahav G, Mathers AJ, Bassetti M, Vazquez J, et al. Effect and safety of meropenem–vaborbactam versus best-available therapy in patients with carbapenem-resistant Enterobacteriaceae infections: The TANGO II Randomized Clinical Trial. Infect Dis Ther. 2018;7:439–55.
Motsch J, Murta de Oliveira C, Stus V, Köksal I, Lyulko O, Boucher HW, et al. RESTORE-IMI 1: A multicenter, randomized, double-blind trial comparing efficacy and safety of imipenem/relebactam vs colistin plus imipenem in patients with imipenem-nonsusceptible bacterial infections. Clin Infect Dis. 2020;70:1799–1808.
Gaibani P, Lewis RE, Volpe SL, Giannella M, Campoli C, Landini MP, et al. In vitro interaction of ceftazidime–avibactam in combination with different antimicrobials against KPC-producing Klebsiella pneumoniae clinical isolates. Int J Infect Dis. 2017;65:1–3.
Shields RK, Nguyen MH, Hao B, Kline EG, Clancy CJ. Colistin Does Not Potentiate Ceftazidime–avibactam Killing of Carbapenem-Resistant Enterobacteriaceae In Vitro or Suppress Emergence of Ceftazidime–avibactam Resistance. Antimicrob Agents Chemother. 2018;62
Chew KL, Tay MKL, Cheng B, Lin RTP, Octavia S, Teo JWP. Aztreonam-Avibactam Combination Restores Susceptibility of Aztreonam in Dual-Carbapenemase-Producing Enterobacteriaceae. Antimicrob Agents Chemother. 2018; 27;62:18
Biagi M, Wu T, Lee M, Patel S, Butler D, Wenzler E. Searching for the Optimal Treatment for Metallo- and Serine-β-Lactamase Producing Enterobacteriaceae: Aztreonam in Combination with Ceftazidime–avibactam or Meropenem–vaborbactam. Antimicrob Agents Chemother. 2019;63:e01426-e1519.
Jayol A, Nordmann P, Poirel L, Dubois V. Ceftazidime/avibactam alone or in combination with aztreonam against colistin-resistant and carbapenemase-producing Klebsiella pneumoniae. J Antimicrob Chemother. 2018;73:542–4.
Nath S, Moussavi F, Abraham D, Landman D, Quale J. In vitro and in vivo activity of single and dual antimicrobial agents against KPC-producing Klebsiella pneumoniae. J Antimicrob Chemother. 2018;73:431–6.
Göttig S, Frank D, Mungo E, Nolte A, Hogardt M, Besier S, Wichelhaus TA. Emergence of ceftazidime/avibactam resistance in KPC-3-producing Klebsiella pneumoniae in vivo. J Antimicrob Chemother. 2019;74:3211–6.
Ojdana D, Gutowska A, Sacha P, Majewski P, Wieczorek P, Tryniszewska E. Activity of ceftazidime–avibactam alone and in combination with ertapenem, fosfomycin, and tigecycline against carbapenemase-producing Klebsiella pneumoniae. Microb Drug Resist. 2019;25:1357–64.
Mikhail S, Singh NB, Kebriaei R, Rice SA, Stamper KC, Castanheira M, Rybak MJ. Evaluation of the Synergy of Ceftazidime–avibactam in Combination with Meropenem, Amikacin, Aztreonam, Colistin, or Fosfomycin against Well-Characterized Multidrug-Resistant Klebsiella pneumoniae and Pseudomonas aeruginosa. Antimicrob Agents Chemother. 2019;63. pii: e00779–19
Borjan J, Meyer KA, Shields RK, Wenzler E. Activity of ceftazidime–avibactam alone and in combination with polymyxin B against carbapenem-resistant Klebsiella pneumoniae in a tandem in vitro time-kill/in vivo Galleria mellonella survival model analysis. Int J Antimicrob Agents. 2020;55:105852.
Liu J, Gefen O, Ronin I, Bar-Meir M, Balaban NQ. Effect of tolerance on the evolution of antibiotic resistance under drug combinations. Science. 2020;367:200–4.
Shields RK, Clancy CJ, Press EG, Nguyen MH. Aminoglycosides for Treatment of bacteremia due to carbapenem-resistant Klebsiella pneumoniae. Antimicrob Agents Chemother. 2016;60:3187–92.
Wang Y, Wang J, Wang R, Cai Y. Resistance to ceftazidime–avibactam and underlying mechanisms. J Glob Antimicrob Resist. 2020;22:18–27.
Nichols WW, Stone GG, Newell P, Broadhurst H, Wardman A, MacPherson M, et al. Ceftazidime- Avibactam Susceptibility Breakpoints against Enterobacteriaceae and Pseudomonas aeruginosa. Antimicrob Agents Chemother. 2018;62:e02590-e2617.
Gaudereto JJ, Perdigão Neto LV, Leite GC, Ruedas Martins R, Boas do Prado GV, Rossi F, et al. Synergistic Effect of Ceftazidime–avibactam with Meropenem against Panresistant, Carbapenemase-Harboring Acinetobacter baumannii and Serratia marcescens Investigated Using Time-Kill and Disk Approximation Assays. Antimicrob Agents Chemother. 2019;63. pii: e02367–18
Shields RK, Chen L, Cheng S, Chavda KD, Press EG, Snyder A, et al. Emergence of Ceftazidime–avibactam Resistance Due to Plasmid-Borne blaKPC-3 Mutations during Treatment of Carbapenem-Resistant Klebsiella pneumoniae Infections. Antimicrob Agents Chemother. 2017;61
Marshall S, Hujer AM, Rojas LJ, Papp-Wallace KM, Humphries RM, Spellberg B et al. Can Ceftazidime–avibactam and Aztreonam Overcome β-Lactam Resistance Conferred by Metallo-β-Lactamases in Enterobacteriaceae? Antimicrob Agents Chemother. 2017;61. pii: e02243–16.
Khan AU, Maryam L, Zarrilli R. Structure, Genetics and Worldwide Spread of New Delhi Metallo-β-lactamase (NDM): a threat to public health. BMC Microbiol. 2017;17:101.
Meini S, Tascini C, Cei M, Sozio E, Rossolini GM. AmpC β-lactamase-producing Enterobacterales: what a clinician should know. Infection. 2019;47:363–75.
Funding
No funding has been received for this work. This study was carried out as part of our routine work.
Author information
Authors and Affiliations
Corresponding author
Additional information
The original online version of this article was revised: The presentation of Table 1 was incorrect. The corrected table is given below.
Rights and permissions
About this article
Cite this article
Meini, S., Viaggi, B. & Tascini, C. Mono vs. combo regimens with novel beta-lactam/beta-lactamase inhibitor combinations for the treatment of infections due to carbapenemase-producing Enterobacterales: insights from the literature. Infection 49, 411–421 (2021). https://doi.org/10.1007/s15010-021-01577-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s15010-021-01577-x