Abstract
On-site handling of food wastes is an environmental alternative that will divert a significant portion of the wastes from landfills and reduce the collection costs subsequently. Regarding conventional composting systems such as windrows and aerated static piles that need large space and a long time to reach maturity; in-vessel bioreactors besides microbial inoculants can accelerate the process by occupying less space. This bioaugmentation was investigated during 40 days by using effective microorganisms, including fungi and bacteria such as Yeast, Lactic acid bacteria, and Actinomycetes, inside a 40-L composting vessel that was able to rotate and stimulate the temperature of the mass (30, 50, and 70 °C). The moisture content was maintained at over 40% with effective microorganism solutions. Food waste was prepared from the university restaurant kitchen then mixed with yard trimmings and wood chips as feedstock. In experiments with effective microorganisms, the thermophilic phase lasted longer (4 days), decomposition rate increased (16.1%), the compost reached maximum temperature faster (3 days), and the maturation period decreased (4 weeks). The most bioaugmentation occurred in the experiment with 50 °C thermal stimulation and 1.25-L effective microorganism solution which the carbon to nitrogen ratio of feedstock decreases 40.74% compared to 9.5% without effective microorganisms after two weeks. Proper carbon to nitrogen ratio (16 ± 0.5), negligible E.coli, Salmonella, and heavy metals of the humus, besides good germination index (> 100%), was accepted as a good quality compost. However, the maturation period would be more than 4 weeks without using effective microorganisms and thermal stimulation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Food waste (FW) is characterized by high organic matter and moisture content (Meng et al. 2015; Guo et al. 2018). FW includes uneaten food and food preparation from residences, institutional and industrial sources such as restaurants, school cafeterias, and factory lunchrooms. Most of them are kitchen waste produced from household and restaurant kitchens (Yang et al. 2013). Large amounts of FW generated by homes, restaurants, and food industries cause environmental problems, so the appropriate and efficient disposal of FW is a global environmental concern (Nourbakhshsamani et al. 2021; Charkhestani and Yousefi Kebria 2022).
Regarding FW characteristics, composting has proven to be a proper choice to deal with FW by less environmental pollutants and a more useful final product (Benito et al. 2006; Wei et al. 2017). According to the United States Environmental Protection Agency (EPA) and Alberta Agricultural, Food, and Rural Development (AAFRD) composting of organic municipal solid waste (MSW) can be done using three common methods including aerated windrow, aerated static pile, and in-vessel composting. Aerated or turned windrow composting is suited for large volumes such as that generated by entire communities and collected by local governments. This method includes the placement of the organic waste into long and narrow piles called “windrows” and aerating them periodically by either manually or mechanically turning the piles (EPA 2021). If the windrow is ventilated passively, there is a high risk of oxygen depletion, unpleasant odors, and greenhouse gases. The ideal pile height is between four and eight feet with a width of 14 to 16 feet. Windrow composting is a large-scale operation. Some advantages are quick drying with high temperatures, a drier product, the ability to handle high volumes of materials, good product stability, and low capital investment. Disadvantages are high operational costs, not space-efficient, vulnerability to climate changes, and emission of odors.
Alternatively, manure can be piled statically, and airflow can be introduced through the pile by installing ventilation pipes inside the system (Liu and Wang 2020). This method produces compost relatively quickly (within three to six months). The dimensions of the static pile are limited by the amount of aeration that can be supplied by the blowers and the characteristics of the waste (Dentel and Qi 2014). Since there is no physical turning, this method requires careful monitoring to ensure that the outside of the pile heats up as much as the core. Also, it may require significant cost and technical assistance to purchase, install, and maintain equipment such as blowers, pipes, sensors, and fans (EPA 2021). Compared to the windrow method shows that it requires less land and it is more vulnerable to climate impacts. Some other benefits of aerated static piles are high levels of pathogens destruction, good odor control, and good product stabilization (Abdoli et al. 2019).
The third common method which produces compost in just a few weeks is in-vessel composting. It can process large amounts of waste without taking up as much space as the windrow method. This method involves feeding organic materials into a drum, silo, or similar equipment which allows good control of temperature, moisture, and airflow. The material is mechanically turned or mixed to make sure the material is aerated. The size of the vessel can vary in size and capacity. This method is expensive and may require technical expertise to operate it properly. However, it uses much less land and labor than windrow composting besides producing very little odor or leachate (EPA 2021).
So, different factors such as capital and operating costs, land availability, operational complexity, odor emission, maturation, stabilization period, aeration system, and better processing control can be considered in choosing the best method (AAFRD 2005). By considering these tips, in-vessel composting is more controlled, requires less space, has high process efficiency, good odor control, and results in fewer operational problems than other methods (Cekmecelioglu et al. 2005; Kim et al. 2008). However, high capital cost and dependency on specialized mechanical and electrical equipment are some disadvantages of this method. The in-vessel composting technique allows the highest degree of temperature control (Thomas et al. 2020). Furthermore, it can be used to decrease the processing time of composting significantly (An et al. 2012).
In addition, if composting occurs on-site (e.g. at home), it will be diverting a significant portion of the waste stream from landfills and greenhouse gas emission will be reduced subsequently (Adhikari et al. 2013). Home-scale composting has not been studied broadly as compared to industrial-scale composting. Home composting, if conducted appropriately, can produce similar or even better levels of compost stability compared to industrial composting because of better control of the material treated, fewer impurities, higher moisture content, lower heavy metals concentration (Barrena et al. 2014). In addition, it reduces the produced leachate during waste transfer to landfill or composting plants (Nourbakhshsamani et al. 2021). Due to the undersized scale of home composters, the temperature is not sufficient to heat up to eliminate the pathogen effectively (Orthodoxou et al. 2015). Time is also an important factor in on-site composting; the less it takes the better.
The decomposition process is relatively lengthy. Human intervention is attempted to improve the efficiency of this natural process by utilization of microbial inoculants (MI) (Fan et al. 2017). Inoculants are products that have living microorganisms in their composition (Santos et al. 2019). It has been widely studied that the activity of microorganisms causes the decomposition of organic matter into simpler forms. For example, the widely used microbial Bacillus sp. is a cellulose and lignin degrader and Trichoderma sp. is known to degrade hemicelluloses (Wu et al. 2019). MI is sometimes referred to as activators and accelerators. Co-composting with MI seems to be a sustainable treatment option currently practiced, however, it is not possible to comment on its cost-effectiveness at industrial or small scale applications. A practice in India showed that the overall cost for co-composting of septage with mixed organic wastes was lower compared to other treatments because of higher available microorganisms in the organic waste (Thomas et al. 2020). However, a study in Thailand revealed that it might not be necessary to add commercial inoculants to facilitate composting of household organic waste due to the slight improvement of the finished compost (Karnchanawong and Nissaikla 2014). So, the efficiency of inoculants varies according to the composting conditions (Fan et al. 2018).
The great majority of the early manufactured inoculants contained only one species of microorganism. However, over the past decade, the use of inoculants containing microorganisms of “different types” has increased (Santos et al. 2019). One of these MIs containing many kinds of naturally occurring beneficial microbes is Effective Microorganisms (EM) that have been used widely in nature and farming (Raja Namasivayam and Bharani 2012). The major microbiological components of compost are bacteria and fungi. Moreover, Actinomycetes, while a particular type of bacteria is considered as the third main component for their ability to degrade the more recalcitrant compounds (Karnchanawong and Nissaikla 2014). EM is a people-friendly and environmentally safe product of the EM Research Organization (EMRO) that achieves synergistic effects (bioaugmentation) by combining these beneficial microorganisms such as Lactic acid bacteria, Actinomycetes, Yeast, Fungi, and Phototrophic bacteria. It was developed by Professor Teruo Higa in 1982 (EMRO 2021). In some cases, native microorganisms cannot degrade a particular contaminant (Speight 2017). Therefore, bioaugmentation offers a way to supply enough specific microorganisms in sufficient numbers to improve the existing populations and complete the biodegradation (Maier and Gentry 2015).
Some studies found that the addition of certain EM microbial agents could increase the number of effective microorganisms, which could promote the decomposition of organic matter and accelerate the composting process to achieve rapid biological composting (Qu et al. 2019). Wang et al. (2014) researched cattle manure enhanced composting. Sharma et al. (2014) proved that the application of EM in compost resulted in hastening the composting of paddy straw. They showed high activity of hydrolytic enzymes and microbial activity (Sharma et al. 2014). Jusoh et al. (2013) stated that EM can reduce odor and increase the final product's decomposition rate and nutrient contents. The results showed that the composting period was shortened with the addition of the EM inoculum (Wang et al. 2014). However, Qian et al. (2014) showed that the cost of composting was also increased. They believed that the addition of the EM microbial agent was not worth considering (Qian et al. 2014).
There are different points of view about the bioaugmentation effect of EM in FW composting. Some researchers believe that the existing microbial community in the waste is enough for degradation (Abdullah et al. 2013), and EM does not have a remarkable effect in small-scale FW composting (Nair and Okamitsu 2010). Some others reported that it can enhance enzymatic activities and promote and accelerate biodegradation of organic matter and composting process (Sarkar et al. 2011; Patidar et al. 2013). So, investigation of the current status in on-site composting of FW with EM shows that the differences are in the type of reactors, the amount of feedstock used, and control of influencing factors which finally leads to a reduction in the maturation and stability time.
This study aims to investigate the bioaugmentation effects of EM on the on-site FW composting in an in-vessel bioreactor by different thermal stimulations and EM concentrations. To reach this goal, important physicochemical properties of the feedstock including temperature, pH, electric conductivity (EC), moisture content (MC), total organic carbon (TOC), total organic matter (TOM), total Kjeldahl nitrogen (TKN), and carbon to nitrogen ratio (C/N) were monitored along the composting process. In addition, germination index (GI), heavy metals, pathogens, nitrogen, potassium, and phosphate contents of the optimum experiment were determined to obtain the composting duration and quality. This study was conducted at Noshirvani University of Technology, Babol, Iran. The experiments were carried out from February 2020 to the end of January 2021.
Materials and methods
In-vessel bioreactor
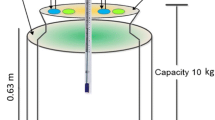
Figure 1 shows the schematic and general view of an aerobic in-vessel bioreactor used in this study. The average rate of Iran’s MSW generation in the last decade (2009–2019) was 0.745 kg/capita/day (Esmaeilizadeh et al. 2020). According to the literature, FW constitutes about 59% of MSW with about 0.5 kg/L wet bulk density (Adhikari et al. 2013; Charkhestani and Yousefi Kebria 2022). So, the vessel has been designed with 40-L usable volume which was proper for collecting FW during a week for a family with 5 persons. It has been designed suitably for on-site practice with an interior composting vessel of 35 cm * 30 cm * 45 cm (length*width*height) from stainless steel and an exterior size of 40 cm * 40 cm * 77.5 cm with a top cover from an Iron sheet with a thickness of 1 mm. Aeration occurred by gas outlet fan, which suctions the air through the air inlet window. Three attached bars to a shaft with 15 cm length were used for turning the mass. The feedstock was heated with a curve-shaped thermal element and a thermostat to set different temperatures from 0 to 100 °C as shown in Fig. 1. (II). The rate of turning was set at 2 rpm according to a study that works on the accelerated composting process of garbage for prolonged periods (Nishino et al. 2003). The bars turn the mass for 10 min, then rest for 20 min. During the turning heating element works simultaneously. It seems the mass has been turned and heated for 8 h during the day. It was assumed that if heating was not done simultaneously with the turning protocol, it would lead to a decrease in temperature of the mass and inhibit the activity of microorganisms consequently. On the other hand, heating for more than 8 h leads to a decrease in MC and also higher energy consumption.
Feedstock
For the experiment, the FW was collected from the University restaurant kitchen (Babol Noshirvani University of Technology, Iran), consisting of fruits scraps, vegetable scraps, tea wastes, steamed rice, and other organic discarded parts during a day. For example, it contained scraps of carrots, potatoes, onions, cucumbers, tomatoes, eggshells, etc. For more realistic results, simulated FW did not use. So, one plastic bin was placed in the kitchen on a specific day of the week and the shredded wastes were dumped into it. Then, the required volume of sample was taken from the bin then mixed with bulking agents (BA) and EM according to “Experimental design section”. BA like yard trimmings and wood chips can reduce odors and leachate generation besides C/N and MC regulation (Adhikari et al. 2009). Also, the proportion of starting materials used in the composting mixture influences the degradation of organic matter, nitrogen dynamics of the process, and its toxicity on germinating plants. The proportions with greater amounts of FW to a BA (70:30 v:v) had higher concentrations of mineral matter, higher peak temperature, and a better initial carbon-to-nitrogen ratio compared to 50:50 or 30:70 ratio (Guidoni et al. 2018). According to Adhikari et al. (2013), FW combined with yard trimmings gives higher MC and a lower C/N ratio than wood chips. So, in this study, both of them have been used equally as BA which was collected from the landscape in the university’s campus. By considering 50% of the bioreactor’s volume (20-L from 40-L), the 8 kg feedstock, including 6 kg restaurant kitchen waste, 1 kg yard trimmings, and 1 kg wood chips, were fed to the batch composting bioreactor (3:1 FW/BA ratio on a wet mass basis). The main characteristics of feedstock are presented in Table 1.
Effective microorganisms (EM)
The beneficial microorganisms such as Lactic acid bacteria, Actinomycetes, and Fungi are the major microbiological components of compost which have abilities to degrade the more recalcitrant compounds (Karnchanawong and Nissaikla 2014). These microorganisms are physiologically compatible with one another and can coexist in liquid culture. Lactic acid bacteria contributed to modify of environmental conditions allowing for the activity of important microorganisms in composting (Tran et al. 2015). They accelerated composting by solving the low pH problem, inhibiting the production of acetic acids, and increasing the proliferation of fungi having the ability to degrade organic matter (Tran et al. 2015; Nakasaki et al. 2019). Actinomycetes improved cellulase activities, increased the content of humic substances, and accelerated production of the key enzymes, including CMCase, Xylanase, lignin peroxidase, etc. (Zhao et al. 2017; Wei et al. 2019). They also alleviated CO2 emissions during composting. Fungi are the most abundant and fastest emerging microorganisms during composting which can produce extracellular enzymes accountable for cellulose and lignin degradation during composting (Awasthi et al. 2014). Yeast as a kind of fungi can degrade organic acids and thus increase the pH value beyond the neutral pH of composting material (Nakasaki et al. 2013). So, bioaugmentation with these EM offers a way to provide specific microorganisms in sufficient numbers to enhance the existing populations and complete the biodegradation (Maier and Gentry 2015). The commercial EM used in this study contains fungi like Yeast, and bacteria including Actinomycetes, Lactic acid, and Photosynthetic bacteria (Daly and Stewart 1999). EM includes both aerobic and anaerobic microorganisms which co-exist in an environment of around 3.5 pH (Mandalaywala et al. 2017). EM solution is a brownish liquid with a pleasant odor. The activated commercial EM (EM-1®) used in this study was supplied by Emkanpazir Pars Co., LTD., Iran. According to Fan et al. (2018), the activated EM contains ~ 107 Colony Forming Unit (CFU)/mL of fungi and ~ 108 CFU/mL of bacteria. The specified species are Streptomyces albus, Propionibacterium freudenreichii, Streptococcus lactis, Aspergillus oryzae, Mucor hiemalis, Saccharomyces cerevisiae, Candida utilis by 105 minimum viable organisms per mL and an unspecified number of Lactobacillus sp., Rhodopseudomonas sp., and Streptomyces griseus (Fan et al. 2018).
Experimental design
Nishino et al. (2003) worked on thermoacidophilic conditions for composting of garbage in prolonged periods with 85 °C constant temperature. It was ultrahigh temperature and consequently consumed high energy. The development of thermophilic composting with ultrahigh temperature (over 75 °C) would inactivate the potential in viral pathogens, but it can affect microbial activity if it lasts longer than 24 h (Chang et al. 2019). In addition, mesophilic microorganisms thrive in 20–45 °C, normal thermophilic microorganisms thrive in 45–65 °C, extreme thermophiles thrive in 65–80 °C and hyperthermophiles that grow optimally at 80 °C and above (Noll 2001; Zeldes et al. 2015; Schiraldi and De Rosa 2016). Therefore, to reduce energy consumption and highlight the bioaugmentation effect of EM, three intermittent heating regimes below 80 °C were applied in Run 4 (70 °C), Run 3 (50 °C), and Run 2 (30 °C). Also, Run 1 was conducted without heating stimulation to compare the results.
Experimental conditions are presented in Table 2. Four experiments (Run 1–4) were considered to investigate the bioaugmentation effect of EM during the composting process compared to four control groups that used distilled water instead of EM. In all Runs, 0.75-L EM was sprayed on the feedstock initially to regulate the MC around 75%. Many researchers reported that effective MC during composting is between 40 and 60% (Hubbe et al. 2010). So, the MC during the process was maintained above 40% using 0.5-L activated EM in main experiments or distilled water in control groups whenever MC reached 40 ± 2%. To reduce the number of study variables, EM was used as an MC regulator. Regarding different thermal regimes of the experiments, different concentrations of EM were used during the processes subsequently.
Monitoring parameters
Temperature measurements were done daily throughout the process by Testo 925 thermometric probe. MC was controlled by a portable REOTEMP long stem compost moisture meter during a day. This method is not very accurate like the hand-squeeze test but it gives good knowledge about the MC of the feedstock without sampling. So, some days, for more accuracy, the MC was also determined as weight loss through drying samples at 105 °C in an electronic oven (Memmert, UNE 400, Germany) for 24 h and reported as the exact MC of feedstock.
To determine pH and EC, each sample was mixed with distilled water (1:10 w/v ratio) to make a solution. The prepared solutions were stirred at 300 rpm for 30 min. Then, after 30 min rest, the electrodes (827 pH-lab Metrohm and WTW 9310) were dipped in each sample prepared solution and recorded when it was stabilized (Sánchez-Monedero et al. 2001; Fan et al. 2018). TOC and TKN were measured by Walkley–Black wet digesting and Kjeldahl (Vaposest 30 s, Gerhardt, Germany) methods (Walkley and Black 1934; Leege 1998). Subsequently, the C/N ratio was monitored during the process, too.
Statistical analysis
One-way analysis of variance (ANOVA) was applied using SPSS ver. 26.0 Software to find out whether there was a significant difference (p < 0.05) among the Runs and their controls. In Post Hoc Multiple Comparisons, LSD for normally distributed data sets and Games-Howell for not normally distributed data sets was used. Statistical analysis provides proper data to determine the optimum Run.
Maturation and quality of the final compost
The C/N is one of the essential parameters to determine the maturity of compost. When waste is composted, generally C/N ratio is stabilized in the range of 15–20 (Goyal et al. 2005). Maturity is not described by a single property, and therefore, it is best assessed by measuring two or more compost characteristics. So, to find the compost quality and minimum time for maturation, other parameters like germination index, heavy metals, nutrients, and pathogen contents were determined for the Run which its C/N ratio was placed faster in the range of 15–20.
According to Bernal et al. (2009), germination tests present an index for phytotoxicity of the final compost (Bernal et al. 2009). Zucconi et al. (1981) stated that a germination index below 50% is considered an immature compost. To determine germination index, the mixture for measurement of pH and EC was filtered through a 0.45 µm filter membrane, then 10 cress seeds (Lepidium sativum) were distributed on paper tissue in Petri dishes and moistened with 10 mL of the filtrate extraction (Yang et al. 2013). Three replicates for each sample were incubated at 25 °C for 3 days in Binder FD 23 drying and heating chambers, Germany, while distilled water was used as control. The germination index (GI %) was calculated using the following equation:
where Gsample and Gwater are the numbers of cress seeds germinated in the assay and water control.
According to Mao et al. (2021), pathogens are airborne and foodborne. Researchers are usually more concerned with foodborne pathogens like Escherichia coli (E. coli) and Salmonella because they use the compost for vegetable cultivation. (Mao et al. 2021). So, for the pathogen test, E. colii and Salmonella were determined according to the methods stated by Fan et al. (2018) based on the Food and Drug Administration’s (FDA) Bacteriological Analytical Manual. The nutrient contents (N, K2O, and P2O5) were determined according to the standard methods of compost maturity analysis (Leege 1998). To determine heavy metals (Zn, Cu, Cr, Ni, Co, As, Hg, Mo, Cd, and Pb) concentrations in the final compost, one gram of the sample was dried and ground then digested by 36 mL HNO3 and HClO4 (5:1 ratio, v/v) followed by atomic absorption spectrometry (Bazrafshan et al. 2016).
Results and discussion
Temperature
Temperature is one of the important parameters that reveal microorganisms’ activity (Sarkar et al. 2010; Kopčić et al. 2014). Figure 2 shows the temperature profile of interior mass during the experiments. As shown in Fig. 2, Runs with EM have a higher mass temperature, reach faster to maximum and the thermophilic phase (> 45 °C) lasts longer than the controls. However, statistical analysis shows that the temperature profile in each Run with EM is not significantly different (p < 0.05) from its control Run. Furthermore, ANOVA analysis between four main experiments with EM as well as four control groups indicates a significant difference in both of the groups that were acceptable regarding different thermal regimes.
Figure 2c reveals that mass temperature in Run 3 with 0.75 L initial EM reached thermophilic phase (> 45 °C) on the first day and reached its maximum temperature (76 ± 1 °C) after three days that is faster than other EM runs with the same concentration of EM. It indicates easy decomposing of organic matter and pathogens elimination (Strauch and Ballarini 1994; Awasthi et al. 2018). Fan et al. (2018) reached 45 °C after 5 days by using 1.2-L EM for 4 kg feedstock with the highest temperature (50 °C) on the 7 day. However, Awasthi et al. (2018) reached the thermophilic phase after 1 day by using a 2-L bacterial consortium for 25 kg feedstock with the highest temperature (68 °C) on the 5 day. One of the main differences between this study and the literature is thermal stimulation.
Thermal stimulation may help the mass temperature to be in the range of the thermophilic phase during the process. So, it is not clear the difference between real thermophilic phase duration with EM and control. However, Run 1 (with 0.75 L initial EM and without heating stimulation) highlighted this difference which showed a longer thermophilic phase for EM (7 days) compared to the control (3 days) that was due to the higher microbial activity. In addition, the thermophilic phase reached a faster rate rather than the control without EM (3 days faster).
Regarding Fig. 2, intermittent heating, while feedstock is turning, prevents the loss of mass temperature. In addition, at the early stages of the process when the activity of microorganisms is high, the temperature of the mass sometimes rises above the temperature of the element, and gradually, with the decrease of the activity of microorganisms, the temperature of the mass is almost constant and approaches the temperature of the element.
Moisture content
For all Runs, 0.75 L EM was sprayed on feedstock initially then obtained MC as presented in Fig. 3. In control Runs, distilled water was used. Regarding conducted pretests, except initial augmentation of EM, we decided to modify MC by spraying 0.5 L EM on feedstock in the bioreactor when MC reaches around 40 ± 2% which is clear in the Figures. As demonstrated, by increasing the bioreactor temperature, MC drastically decreases and control of the MC needs more EM. Apparently, in terms of additive consumption, 4 times addition of EM (2.75 L) in Run 4 is not cost-effective compared with the literature (Awasthi et al. 2018; Fan et al. 2018); however, discussing the proper augmentation of EM to obtain the best results needs the investigation of other maturation indices as follows.
pH and electrical conductivity
Microbial activity can affect pH during the initial stages of composting (Awasthi et al. 2018). Several studies suggested neutral pH for optimal composting (Zucconi et al. 1981; Kuok et al. 2012; Kopčić et al. 2014). Although in this study, pH was around 5.8 ± 0.15 and 5.08 ± 0.14 in the controls and Runs with EM, respectively, after initial moisture modifying. As shown in Fig. 4a, b, c the drop in pH occurred in the early stages of composting that is due to the formation of organic acids (Bazrafshan et al. 2016). According to Mandalaywala et al. (2017) acidic environment is favorable for the growth of EM. By decomposition of organic acids, releasing volatile ammonia, and mineralization, increasing pH was observed. Comparing the trends in Fig. 4 shows that pH increases 3.4, 2.7, 3.2, and 2.4 from the least to highest in 25, 20, 15, and 10 days in Runs 1 to 4, respectively. However, in control Runs, it increases 2.7, 2.9, 2.4, and 2. As demonstrated, in thermal stimulation above 30 °C (Run 3 and Run 4), pH changes have been stabled 2 weeks earlier due to the EM bioaugmentation effect that causes an increase in decomposition rate which is more significant when EM concentration and mass temperature have been increased. Statistical analysis showed that there is no significant difference (p > 0.05) between Run with EM and without EM in all 4 Runs.
Electrical conductivity is a factor of salinity and affects the quality of final compost. Figure 4 shows the variation of EC. As seen in Fig. 4, EC has been increased then decreased in Run 1 to 4. The increase occurs during the early thermophilic phase due to the release of mineral salts through the decomposition of organic substances (Gao et al. 2010). However, the reduction of water-soluble substances such as organic acids attributed to the reduction of EC in control groups (Tang et al. 2004). By thermal stimulation in different Runs, EC reduction has been accelerated and the largest reduction happened in Run 3 (6.4 to 2.9 ± 0.14 ds/m). Run 4 shows that more concentration of EM does not have any special bioaugmentation effect than Run 3 (6.1 to 3.5 ± 0.1 ds/m). In addition, statistical analysis revealed that a significant difference (p < 0.05) with its control has been observed only in Run 3. Although, ANOVA analysis between four main experiments with EM as well as four control groups showed no significant difference (p > 0.05).
Total Kjeldahl’s nitrogen
TKN depends on the rate of decomposition and initial nitrogen content of the waste (Kaviraj and Sharma 2003). TKN has a declining trend in the initial stage of composting due to the loss of nitrogen in the form of ammonia (Mengistu et al. 2017). Figures 5c, d show that the increasing rate of TKN begins about 5 days faster for EM Runs than the controls. It may be attributed to the higher loss of organic matter rather than the loss of NH3 during the active phase (Bernai et al. 1998). As demonstrated in Fig. 5c, d, the most TKN augmentation was 18.75 (Run 3) and 15.3% (Run 4) in the EM Runs verses 6.09 and 10% in the controls, respectively. So, compost with EM has more N due to the higher nitrogen-fixing bacteria activity (Seal et al. 2012; Jusoh et al. 2013). However, some studies state that EM did not have any special effect on composting of kitchen waste (Nair and Okamitsu 2010). Statistical analysis between the main Run and its control shows a significant difference between them in all Runs. Unlike Runs with EM, no significant difference was observed between the 4 control groups. LSD analysis showed that Run 1 (EM) has far from other EM Runs which highlights the effects of thermal stimulation and the role of EM.
Total organic carbon and C/N ratio
TOC contents in both EM and control Runs gradually decreased during composting. As shown in Fig. 6a–d, TOC has been reduced 25.7, 37.3, 42.1, and 42% in Runs 1 to 4, respectively, by the addition of EM and thermal stimulation. In all Runs, TOC reduction was higher in the EM inoculated mass rather than the controls (21.6, 29.9, 26, and 28.3%) which is similar to the results of Awasthi et al. (2018) that worked on in-vessel co-composting of FW with a bacterial consortium. Kim et al. (2008) worked on centralized in-vessel composting of FW. They reported TOC content decreases from 55 to 34% (38% reduction) during 35 days for 14,000 kg/day flow rate of mixing material. It is near to the best TOC reduction efficiency of this study (42.13% reduction in 40 days for Run 3) considering its different scale. By comparing Fig. 6c, d, it is obvious the temperature above 70 °C and subsequently higher EM consumption have no significant role in the TOC reduction trend. In addition, ANOVA analysis between the Runs by EM and without EM shows a significant difference (p < 0.05) only in Run 3.
The C/N ratio is an important factor to investigate compost maturity (Goyal et al. 2005). As seen in Fig. 6, by decomposition of organic matter through the microorganisms, the C/N ratio has been decreased. Run 3 and 4 demonstrated that enhancing bioreactor temperature and EM concentration make the C/N declining trend faster. In this study, the initial C/N ratio of the feedstock was 27 ± 1.2. To avoid nitrogen immobilization when matured compost is applied to soil, it should ideally have a value of about 15 (Erhart and Burian 1997). By considering this issue, in control Runs without EM during 40 days, C/N did not drop below 17.1 ± 0.56. In the experiments with EM, it dropped to 19.87 ( − 29.03%), 15.71 ( − 40.96%), 13.16 ( − 51.26%), and 13.01 ( − 49.73%) during 40 days in Run 1 to 4, respectively. According to Goyal et al. (2005), the C/N ratio stabilizes in the range of 15–20 when compost is matured. In Run 3 and 4, C/N reached 16 from an initial ratio of 27 and 25.9 after two weeks, respectively. Since the C/N ratio is one of the important indices of maturation, it is clear that most EM bioaugmentation effect occurs in Run 3 with a 40.7% reduction in C/N after 2 weeks which can decrease the maturation period to 14 days or less compared to 40 days or more without EM. In fact, in this stage, the compost is matured but not stabilized. Regarding the Temperature, OC, and C/N declining trend, it can be said that mature compost needs 1–2 weeks more to stabilize completely.
In comparing with literature, Awasthi et al. (2018) composted 25 kg FW mixed with sawdust using 2 L enriched bacterial consortium without thermal motivation, and the C/N ratio reached 16.18 (36.8% reduction) after 40 days. However, they could decrease only about 10% of C/N after 2 weeks. Fan et al. (2018) obtained a 61–66% final reduction of C/N after 8 weeks with 1.2 L EM for 4 kg simulated FW without daily turning, moisture-controlling, and thermal stimulation. However, they stated there was no significant difference in the C/N changes when using distilled water instead of EM. In addition, after 2 weeks, they did not obtain significant results in C/N reduction with EM compared to control. Abdullah et al. (2013) used a microbial solution consisting of seven types of bacteria and eight types of fungi isolated from soils as a starter culture for the kitchen-waste composting. They showed that the mixture with added starter culture and control have no significant difference in terms of TOC content, total nitrogen content, and C/N ratio and reached similar values at the end of composting. Nair and Okamitsu (2010) revealed that no significant difference was observed between the control and those inoculated with Trichoderma and EM in terms of the C/N ratio of the final product for on-site small scale household organic waste treatment during 28 days. However, it was observed that EM inoculation enhanced the reproductive rate of earthworms, and so probably created the best environment for vermicomposting. Patidar et al. (2013) observed significant decrement in MC, TOC, and C/N ratio and increment in temperature, and phosphate in thermophilic composting with microbial inoculation followed by vermicomposting. Sarkar et al. (2011) combined 35 different enzyme-producing bacterial strains by permutation combination to make different microbial consortia. They showed more than 50% degradation in 21 days for organic kitchen wastes while control with no inoculation showed only 36% degradation at the same time.
In this study, maximum C/N reduction occurred in Run 3 by 51.27% during 40 days compared to 30.28% in the control without EM. The significant results occurred after 2 weeks which C/N decreases 40.74% in Run 3 (with 1.25 L EM) compared to 9.5% in Run without EM. This trend was 38.17% compared to 14.4% for Run 4 (with 1.75 L EM). Lower energy consumption and less utilization of EM rather than Run 4, make Run 3 more cost-effective in C/N reduction.
In addition, statistical analysis of C/N changes during the experiments showed that there was a significant difference between all four Runs with EM (p < 0.05). However, this analysis did not present any differences between control groups (p > 0.05). So, Post Hoc Multiple Comparisons (LSD) were conducted between Runs with EM and results showed that Run 1 was far from the other Runs. It highlights the role of thermal stimulation that occurred in the other three Runs (2, 3, and 4). Comparing the C/N changes of each EM Run with its control demonstrated that there was a significant difference only between Run 3 and Run 4 with their control groups. Regarding higher consumption of EM and energy (higher temperature) in Run 4 besides other investigated conditions, Run 3 could be considered as the optimum Run of experiments. So, correlation analysis was conducted among parameters including Temperature, pH, EC, TOC, TKN, and C/N ratio for Run 3 and its control which has been presented in Table 3.
According to Table 3, in the optimum Run with EM, C/N is significantly correlated with parameters like pH and TKN compared to the control which is not. Regarding Fig. 4c pH of feedstock is almost acidic during the first 2 weeks which is favorable for the growth of EM (Mandalaywala et al. 2017). In addition, the greatest increase in pH (4.8–7.5) has occurred during this period that was coincided with the greatest decrease in C/N (40.74%) due to the decomposition of organic acids, releasing volatile ammonia and mineralization.
TKN is negatively correlated with TOC in a run with EM that is related to the activity and population of the presented humus. Higher consumption of organic carbon by microorganisms facilitates increasing the nitrogen content compared to the control Run without EM. In a control Run without EM, TOC is correlated with temperature which indicates that temperature stimulation can aid the composting process where the temperature is not high enough.
Quality of compost
During the first two weeks of the 40 days monitoring, Run 3 showed the best efficiency in terms of TOC and C/N reduction, temperature, and pH profile. Since it was assumed that EM shows their bioaugmentation effect in reducing compost production time, other parameters like heavy metals, nutrient contents, and germination index were examined to investigate the maturity and quality of the produced humus, after 2 weeks for Run 3. The results have been presented in Table 4.
The germination test presents the phytotoxicity and maturity of the compost. It directly indicates if the compost has an inhibitory effect on plant growth or not (Awasthi et al. 2018). Initially, the germination indices were very low due to the active decomposition of organic materials that generated a variety of toxic compounds and thus reduced the seed germination (Wang et al. 2018).
The germination index of the humus with EM reached over 100% after two weeks, while in the control Run, it did not reach over 80% after 40 days. It shows proper mineralization due to the bioaugmentation effect of EM. Comparing with literature shows that Fan et al. (2018) reached 306% in the compost with EM after 8 weeks, however, control Run without EM reached 401.6%. So, giving more time to the discharged compost until completely stabilized can improve the GI.
To control microbial parameters of the humus, E.Coli and Sallmonella were determined. They were negligible and met the standards for pathogen content according to Table 4. It is maybe due to the separated feedstock that is less likely to be contaminated by pathogens or thermophilic temperatures during the process (Fan et al. 2018). Temperature above 55 °C for at least three days inactivates pathogens (Wichuk and McCartney 2007). It was achieved by a curved-shaped element in the bioreactor besides the activity of EM.
In the case of nutrient contents of the composted raw mixture, initial Nitrogen, Potassium and Phosphorus of humus were 1.60 ± 0.07%, 0.79 ± 0.06% and 0.21 ± 0.05% that after composting were 1.85 ± 0.07%, 0.9 ± 0.05% and 0.6 ± 0.08%, respectively in Run 3 with EM compared to 1.56 ± 0.04%, 0.85 ± 0.05% and 0.54 ± 0.03% in control. So, no significant bioaugmentation effect of EM was seen except in the N content compared to the control. Fan et al. (2018) reported the same finding of the EM effect on the nutrient contents. Although Jusoh et al. (2013) revealed that composting of rice straw with EM had a significantly more N, P, and K content (P < 0.05) compared to compost without EM.
The compost with hazardous components (i.e., heavy metals) limited its use in horticulture (Sánchez-Monedero et al. 2001). Heavy metals present in kitchen waste are extremely low which could be ignored in the humus that is following our results after two weeks (Manungufala et al. 2008). This low level of heavy metals or toxic compounds is a proper feature for the quality improvement of soil (Hogg et al. 2002).
The heavy metals, nutrient contents, microbial parameters, and germination index of the compost after two weeks were qualified as a first-grade compost according to the presented standards and it was suitable to use as fertilizer.
Conclusion
EM bioaugmentation in an on-site in-vessel FW composter was investigated. Thermal stimulation, turning the mass, and moisture controlling highlighted this bioaugmentation to reduce the time required for the maturation of compost. In experiments with EM, the thermophilic phase lasted longer, the decomposition rate of organic carbon increased, C/N reduced rapidly and the compost reached maximum temperature faster compared to controls without EM. The application of EM alone without an in-vessel bioreactor and suggested stimulations do not significantly affect the bioaugmentation of the process. Temperature stimulation above 50 °C not only had no significant effect but also increased energy consumption and moisture evaporation. The results indicate that compost inoculated with EM has rapid mineralization rates in the first 2 weeks. Proper C/N, negligible E.coli and Salmonella of the humus besides low-level content of heavy metals and good germination index (> 100%) after two weeks showed that maturation period decreases about four weeks compared to run without EM and thermal stimulation. This is an environmental alternative for on-site treatment of FW that will divert a significant portion of the waste stream from landfills, reduce collection costs and greenhouse gas emissions subsequently.
Of course, accurate discussion about the energy consumption and cost-effectiveness of the studied method compared to other alternatives, bedsides investigation of feedstock entry continuously instead of batch mode can be suggested as topics for future studies.
References
AAFRD (2005) Manure composting manual. Alberta agricultural, food, and rural development.https://www.agric.gov.ab.ca/$sdepartment/deptdocs.nsf/all/agdex 8875/$file/400_27-1.pdf?OpenElement. Accessed 30 Dec 2021
Abdoli MA, Omrani G, Safa M, Samavat S (2019) Comparison between aerated static piles and vermicomposting in producing co-compost from rural organic wastes and cow manure. Int J Environ Sci Technol 16:1551–1562. https://doi.org/10.1007/s13762-017-1607-5
Abdullah N, Chin NL, Mokhtar MN, Taip FS (2013) Effects of bulking agents, load size or starter cultures in kitchen-waste composting. Int J Recycl Org Waste Agric 2:3. https://doi.org/10.1186/2251-7715-2-3
Adhikari BK, Barrington S, Martinez J, King S (2009) Effectiveness of three bulking agents for food waste composting. Waste Manag 29:197–203. https://doi.org/10.1016/j.wasman.2008.04.001
Adhikari BK, Trémier A, Barrington S et al (2013) Gas emissions as influenced by home composting system configuration. J Environ Manag 116:163–171. https://doi.org/10.1016/j.jenvman.2012.12.008
An C-J, Huang G-H, Yao Y et al (2012) Performance of in-vessel composting of food waste in the presence of coal ash and uric acid. J Hazard Mater 203–204:38–45. https://doi.org/10.1016/j.jhazmat.2011.11.066
Awasthi MK, Pandey AK, Khan J et al (2014) Evaluation of thermophilic fungal consortium for organic municipal solid waste composting. Bioresour Technol 168:214–221
Awasthi MK, Wang Q, Wang M et al (2018) In-vessel co-composting of food waste employing enriched bacterial consortium. Food Technol Biotechnol 56:83–89
Barrena R, Font X, Gabarrell X, Sánchez A (2014) Home composting versus industrial composting: influence of composting system on compost quality with focus on compost stability. Waste Manag 34:1109–1116. https://doi.org/10.1016/j.wasman.2014.02.008
Bazrafshan E, Zarei A, Kord Mostafapour F et al (2016) Maturity and stability evaluation of composted municipal solid wastes. Health Scope 5:e33202
Benito M, Masaguer A, Moliner A, De Antonio R (2006) Chemical and physical properties of pruning waste compost and their seasonal variability. Bioresour Technol 97:2071–2076. https://doi.org/10.1016/j.biortech.2005.09.011
Bernai MP, Paredes C, Sánchez-Monedero MA, Cegarra J (1998) Maturity and stability parameters of composts prepared with a wide range of organic wastes. Bioresour Technol 63:91–99. https://doi.org/10.1016/S0960-8524(97)00084-9
Bernal MP, Alburquerque JA, Moral R (2009) Composting of animal manures and chemical criteria for compost maturity assessment. A Review Bioresour Technol 100:5444–5453. https://doi.org/10.1016/j.biortech.2008.11.027
Cekmecelioglu D, Demirci A, Graves RE, Davitt NH (2005) Applicability of optimised in-vessel food waste composting for windrow systems. Biosyst Eng 91:479–486. https://doi.org/10.1016/j.biosystemseng.2005.04.013
Chang R, Li Y, Li J et al (2019) Influences of the thermophilic period on biodegradation and nitrogen loss in stimulated vegetable waste composting. Glob Ecol Conserv 18:e00623. https://doi.org/10.1016/j.gecco.2019.e00623
Charkhestani A, Yousefi Kebria D (2022) Laboratory analysis to determine the accurate characteristics of urban food waste. Glob J Environ Sci Manag 8:225–236
Daly MJ, Stewart DPC (1999) Influence of “effective microorganisms” (EM) on vegetable production and carbon mineralization–a preliminary investigation. J Sustain Agric 14:15–25. https://doi.org/10.1300/J064v14n02_04
Dentel SK, Qi Y (2014) Management of sludges, biosolids, and residuals. In: Ahuja S (ed) Comprehensive water quality and purification. Elsevier, Waltham, pp 223–243
EMRO (2021) What is EM. https://www.emrojapan.com/what. Accessed 27 Nov 2021
EPA (2021) Composting basics. United States Environmental Protection Agency. https://www.epa.gov/sustainable-management-food/types-composting-and-understanding-process. Accessed 28 Jan 2022
Erhart E, Burian K (1997) Evaluating quality and suppressiveness of austrian biowaste composts. Compost Sci Util 5:15–24. https://doi.org/10.1080/1065657X.1997.10701881
Esmaeilizadeh S, Shaghaghi A, Taghipour H (2020) Key informants’ perspectives on the challenges of municipal solid waste management in Iran: a mixed method study. J Mater Cycles Waste Manag 22:1284–1298. https://doi.org/10.1007/s10163-020-01005-6
Fan YV, Lee CT, Ho CS et al (2017) Evaluation of microbial inoculation technology for composting. Chem Eng Trans 56:433–438. https://doi.org/10.3303/CET1756073
Fan YV, Lee CT, Klemeš JJ et al (2018) Evaluation of effective microorganisms on home scale organic waste composting. J Environ Manag 216:41–48. https://doi.org/10.1016/j.jenvman.2017.04.019
Gao M, Liang F, Yu A et al (2010) Evaluation of stability and maturity during forced-aeration composting of chicken manure and sawdust at different C/N ratios. Chemosphere 78:614–619. https://doi.org/10.1016/j.chemosphere.2009.10.056
Goyal S, Dhull SK, Kapoor KK (2005) Chemical and biological changes during composting of different organic wastes and assessment of compost maturity. Bioresour Technol 96:1584–1591. https://doi.org/10.1016/j.biortech.2004.12.012
Guidoni LLC, Marques RV, Moncks RB et al (2018) Home composting using different ratios of bulking agent to food waste. J Environ Manage 207:141–150. https://doi.org/10.1016/j.jenvman.2017.11.031
Guo W, Zhou Y, Zhu N et al (2018) On site composting of food waste: a pilot scale case study in China. Resour Conserv Recycl 132:130–138. https://doi.org/10.1016/j.resconrec.2018.01.033
Hogg D, Barth J, Favoino E et al. (2002) Comparison of compost standards within the eu, north america and australasia: main report. The Waste and Resources Action Programme, Banbury, UK
Hubbe MA, Nazhad M, Sánchez C (2010) Composting as a way to convert cellulosic biomass and organic waste into high-value soil amendments: a review. BioResources 5:2808–2854
ISIRI (2008) Compost: physical and chemical specifications, Standard No. 10716, Institute of Standards and Industrial Research of Iran (in Persian)
ISIRI (2011) Compost : microbial specification and test methods, Standard No. 13321–1, Institute of Standards and Industrial Research of Iran (in Persian)
Jusoh MLC, Manaf LA, Latiff PA (2013) Composting of rice straw with effective microorganisms (EM) and its influence on compost quality. Iran J Environ Health Sci Eng 10:17. https://doi.org/10.1186/1735-2746-10-17
Karnchanawong S, Nissaikla S (2014) Effects of microbial inoculation on composting of household organic waste using passive aeration bin. Int J Recycl Org Waste Agric 3:113–119. https://doi.org/10.1007/s40093-014-0072-0
Kaviraj SS (2003) Municipal solid waste management through vermicomposting employing exotic and local species of earthworms. Bioresour Technol 90:169–173. https://doi.org/10.1016/S0960-8524(03)00123-8
Kim J-D, Park J-S, In B-H et al (2008) Evaluation of pilot-scale in-vessel composting for food waste treatment. J Hazard Mater 154:272–277. https://doi.org/10.1016/j.jhazmat.2007.10.023
Kopčić N, Vuković Domanovac M, Kučić D, Briški F (2014) Evaluation of laboratory-scale in-vessel co-composting of tobacco and apple waste. Waste Manag 34:323–328. https://doi.org/10.1016/j.wasman.2013.11.001
Kuok F, Mimoto H, Nakasaki K (2012) Effects of turning on the microbial consortia and the in situ temperature preferences of microorganisms in a laboratory-scale swine manure composting. Bioresour Technol 116:421–427. https://doi.org/10.1016/j.biortech.2012.03.106
Leege PB (1998) Introduction of test methods for the examination of composting and compost. In: Brown S, Angle JS, Jacobs L (eds) Beneficial co-utilization of agricultural, municipal and industrial by-products. Springer, Dordrecht, pp 269–282
Liu Z, Wang X (2020) Manure treatment and utilization in production systems. In: Bazer FW, Lamb GC, Wu G (eds) Animal agriculture. Academic Press, pp 455–467
Maier RM, Gentry TJ (2015) Microorganisms and organic pollutants. In: Pepper IL, Gerba CP, Gentry TJ (eds) Environmental microbiology, 3rd edn. Academic Press, San Diego, pp 377–413
Mandalaywala HP, Patel PV, Ratna T (2017) Introduction and use of effective microorganisms for bioremediation processes- a review. Int J Sci Res Methodol 7:41–50
Manungufala TE, Chimuka L, Maswanganyi BX (2008) Evaluating the quality of communities made compost manure in South Africa: a case study of content and sources of metals in compost manure from Thulamela municipality, Limpopo province. Bioresour Technol 99:1491–1496. https://doi.org/10.1016/j.biortech.2007.02.006
Mao Y, Akdeniz N, Nguyen TH (2021) Quantification of pathogens and antibiotic resistance genes in backyard and commercial composts. Sci Total Environ 797:149197. https://doi.org/10.1016/j.scitotenv.2021.149197
Meng Y, Li S, Yuan H et al (2015) Effect of lipase addition on hydrolysis and biomethane production of Chinese food waste. Bioresour Technol 179:452–459. https://doi.org/10.1016/j.biortech.2014.12.015
Mengistu T, Gebrekidan H, Kibret K et al (2017) Comparative effectiveness of different composting methods on the stabilization, maturation and sanitization of municipal organic solid wastes and dried faecal sludge mixtures. Environ Syst Res 6:5. https://doi.org/10.1186/s40068-017-0079-4
Nair J, Okamitsu K (2010) Microbial inoculants for small scale composting of putrescible kitchen wastes. Waste Manag 30:977–982. https://doi.org/10.1016/j.wasman.2010.02.016
Nakasaki K, Araya S, Mimoto H (2013) Inoculation of Pichia kudriavzevii RB1 degrades the organic acids present in raw compost material and accelerates composting. Bioresour Technol 144:521–528. https://doi.org/10.1016/j.biortech.2013.07.005
Nakasaki K, Hirai H, Mimoto H et al (2019) Succession of microbial community during vigorous organic matter degradation in the primary fermentation stage of food waste composting. Sci Total Environ 671:1237–1244. https://doi.org/10.1016/j.scitotenv.2019.03.341
Nishino T, Nakayama T, Hemmi H et al (2003) Acidulocomposting, an accelerated composting process of garbage under thermoacidophilic conditions for prolonged periods. J Environ Biotechnol 3:33–36
Noll KM (2001) Thermophilic bacteria. In: Brenner S, Miller JH (eds) Encyclopedia of genetics. Academic Press, New York, pp 1961–1963
Nourbakhshsamani N, Fami HS, Amadeh H (2021) Quantitative and qualitative benefits of household efforts to dry food waste at source. Int J Environ Sci Technol. https://doi.org/10.1007/s13762-021-03614-6
Orthodoxou D, Pettitt TR, Fuller M et al (2015) An investigation of some critical physico-chemical parameters influencing the operational rotary in-vessel composting of food waste by a small-to-medium sized enterprise. Waste Biomass Valorization 6:293–302. https://doi.org/10.1007/s12649-014-9332-5
Patidar A, Gupta R, Tiwari A (2013) Potential of microbial inoculated water hyacinth amended thermophilic composting and vermicomposting in biodegradation of agro-industrial waste. J Bioremed Biodeg 4:119. https://doi.org/10.4172/2155-6199.1000191
Qian X, Shen G, Wang Z et al (2014) Co-composting of livestock manure with rice straw: characterization and establishment of maturity evaluation system. Waste Manag 34:530–535. https://doi.org/10.1016/j.wasman.2013.10.007
Qu G, Cai Y, Lv P et al (2019) Effect of EM microbial agent on aerobic composting for dairy cattle manure. Int J Environ Sci Technol 16:6945–6958. https://doi.org/10.1007/s13762-018-2146-4
Raja Namasivayam SK, Bharani RSA (2012) Effect of compost derived from decomposed fruit wastes by effective microorganism (EM) technology on plant growth parameters of Vigna mungo. J Bioremed Biodeg 3:167. https://doi.org/10.4172/2155-6199.1000167
Sánchez-Monedero MA, Roig A, Paredes C, Bernal MP (2001) Nitrogen transformation during organic waste composting by the Rutgers system and its effects on pH, EC and maturity of the composting mixtures. Bioresour Technol 78:301–308. https://doi.org/10.1016/S0960-8524(01)00031-1
Santos MS, Nogueira MA, Hungria M (2019) Microbial inoculants: reviewing the past, discussing the present and previewing an outstanding future for the use of beneficial bacteria in agriculture. AMB Express 9:205. https://doi.org/10.1186/s13568-019-0932-0
Sarkar S, Banerjee R, Chanda S et al (2010) Effectiveness of inoculation with isolated Geobacillus strains in the thermophilic stage of vegetable waste composting. Bioresour Technol 101:2892–2895. https://doi.org/10.1016/j.biortech.2009.11.095
Sarkar P, Meghvanshi M, Singh R (2011) Microbial consortium: a new approach in effective degradation of organic kitchen wastes. Int J Environ Sci Dev 2:170–174
Saveyn H, Eder P (2014) End-of-waste criteria for biodegradable waste subjected to biological treatment (compost & digestate): technical proposals. Publications Office of the European Union, Luxembourg
Schiraldi C, De Rosa M (2016) Mesophilic organisms. In: Drioli E, Giorno L (eds) Encyclopedia of membranes. Springer, Berlin, Heidelberg, pp 1–2
Seal A, Bera R, Chatterjee AK, Dolui AK (2012) Evaluation of a new composting method in terms of its biodegradation pathway and assessment of compost quality, maturity and stability. Arch Agron Soil Sci 58:995–1012. https://doi.org/10.1080/03650340.2011.565410
Sharma A, Sharma R, Arora A et al (2014) Insights into rapid composting of paddy straw augmented with efficient microorganism consortium. Int J Recycl Org Waste Agric 3:54. https://doi.org/10.1007/s40093-014-0054-2
Speight JG (2017) Removal of organic compounds from the environment. In: Speight JG (ed) Environmental organic chemistry for engineers. Butterworth-Heinemann, pp 387–432
Strauch D, Ballarini G (1994) Hygienic aspects of the production and agricultural use of animal wastes1. J Vet Med Ser B 41:176–228. https://doi.org/10.1111/j.1439-0450.1994.tb00222.x
Tang J-C, Kanamori T, Inoue Y et al (2004) Changes in the microbial community structure during thermophilic composting of manure as detected by the quinone profile method. Process Biochem 39:1999–2006. https://doi.org/10.1016/j.procbio.2003.09.029
Thomas AR, Arulraj PR, Kranert M, Philip L (2020) Investigation on greenhouse gas emissions and compost dynamics during in-vessel co-composting of septage and mixed organic wastes. Int J Environ Sci Technol 17:1675–1690. https://doi.org/10.1007/s13762-019-02450-z
Tran QNM, Mimoto H, Nakasaki K (2015) Inoculation of lactic acid bacterium accelerates organic matter degradation during composting. Int Biodeterior Biodeg 104:377–383. https://doi.org/10.1016/j.ibiod.2015.07.007
WAC (2019) Washington composting rules about composting facilities, permit requirements and operating No. 17–350–220
Walkley A, Black IA (1934) An examination of the degtjareff method determining soil organic matter, and a proposed modification of the chromic acid titration method. Soil Sci 37:29–38
Wang S, Liu K, Li R et al (2014) Enhanced technology of cattle manure compost by microbial inoculum with high lignocellulose degradation ability. Trans Chin Soc Agric Mach 45:201–207. https://doi.org/10.6041/j.issn.1000-1298.2014.04.032
Wang X, Selvam A, Lau SSS, Wong JWC (2018) Influence of lime and struvite on microbial community succession and odour emission during food waste composting. Bioresour Technol 247:652–659. https://doi.org/10.1016/j.biortech.2017.07.091
Wei Y, Li J, Shi D et al (2017) Environmental challenges impeding the composting of biodegradable municipal solid waste: a critical review. Resour Conserv Recycl 122:51–65. https://doi.org/10.1016/j.resconrec.2017.01.024
Wei Y, Wu D, Wei D et al (2019) Improved lignocellulose-degrading performance during straw composting from diverse sources with actinomycetes inoculation by regulating the key enzyme activities. Bioresour Technol 271:66–74. https://doi.org/10.1016/j.biortech.2018.09.081
Wichuk KM, McCartney D (2007) A review of the effectiveness of current time–temperature regulations on pathogen inactivation during composting. J Environ Eng Sci 6:573–586. https://doi.org/10.1139/S07-011
Wu Y, Chen Y, Shaaban M et al (2019) Evaluation of microbial inoculants pretreatment in straw and manure co-composting process enhancement. J Clean Prod 239:118078. https://doi.org/10.1016/j.jclepro.2019.118078
Yang F, Li GX, Yang QY, Luo WH (2013) Effect of bulking agents on maturity and gaseous emissions during kitchen waste composting. Chemosphere 93:1393–1399. https://doi.org/10.1016/j.chemosphere.2013.07.002
Zeldes BM, Keller MW, Loder AJ et al (2015) Extremely thermophilic microorganisms as metabolic engineering platforms for production of fuels and industrial chemicals. Front Microbiol 6:1209. https://doi.org/10.3389/fmicb.2015.01209
Zhao Y, Zhao Y, Zhang Z et al (2017) Effect of thermo-tolerant actinomycetes inoculation on cellulose degradation and the formation of humic substances during composting. Waste Manag 68:64–73. https://doi.org/10.1016/j.wasman.2017.06.022
Zucconi F, Pera A, Forte M, De Bertoldi M (1981) Evaluating toxicity of immature compost. Biocycle 2:54–57
Acknowledgements
The authors wish to thank all who assisted in conducting this work.
Funding
No funding was received for conducting this study.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by AC and the guidance of DYK. The first draft of the manuscript was written by AC and all authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval and consent to participate
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Editorial responsibility: Samareh Mirkia.
Rights and permissions
About this article
Cite this article
Charkhestani, A., Kebria, D.Y. Investigation of effective microorganisms bioaugmentation in an on-site aerobic food waste composter. Int. J. Environ. Sci. Technol. 19, 8507–8522 (2022). https://doi.org/10.1007/s13762-022-04087-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-022-04087-x




 EM;
EM;  Control
Control

 EM (pH);
EM (pH);  Control (pH);
Control (pH);  EM (EC);
EM (EC);  Control (EC)
Control (EC)
 EM;
EM;  Control
Control
 EM (pH);
EM (pH);  Control (pH);
Control (pH);  EM (EC);
EM (EC);  Control (EC)
Control (EC)