Abstract
The aim of the study was to evaluate the performance of two newly developed plate-type biofilters in treating air contaminated with ammonia vapour at different inlet air temperatures (24, 28, and 32 °C). The biofilters had two different structures, straight lamellar plates (SLP) and wavy lamellar plates (WLP), with both having a built-in capillary system for humidifying the packing material made of synthetic hydrophilic fibres and wood fibre. In the packing material of the biofilters, different types of microorganisms were used, including bacteria, micromycetes and yeast. The efficiency of ammonia removal from the air using the biofilters with the two different plate types was investigated. The treatment efficiency of air-containing ammonia vapour reached 81.0–85.2% and 84.2–87.0% in biofilters with SLP and WLP, respectively. The highest ammonia treatment efficiency was obtained in the biofilter with WLP at 28 °C with 87.0% of ammonia being removed. The latter removal efficiency was obtained when a large population of the microorganisms was present with 1.0 ± 0.2 × 107, 1.0 ± 0.5 × 107 and 1.6 ± 0.1 × 109 CFU/g of micromycetes, yeast and bacteria, respectively. The results also demonstrated that at different temperatures of polluted air, different microorganisms predominated in the packing material of the biofilters.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Ammonia is a colourless gas with a strong odour that is generated from organic waste treatment plants and other industrial sources (Baquerizo et al. 2005). The process of air purification through biofiltration has attracted considerable attention with biofilters being used increasingly often to remove odours (Malhautier et al. 2005), toxic materials and volatile organic compounds (VOCs), such as NH3 (Chen et al. 2005), from air. Compared to traditional air treatment methods, biofiltration is an inexpensive and effective air purification technique (Baquerizo et al. 2005; Zigmontienė and Žarnauskas 2011).
Biofilters are biological systems accommodating a variety of microorganisms, such as bacteria and fungi (Martens et al. 2001). A biofilter is made of one or several beds consisting of activated biological or synthetic materials (Liang et al. 2000) functioning as a medium that provides nutrients for microorganisms that form a biofilm on the surface of the material used. The pollutant is absorbed into the biofilm where aerobic biodegradation of the pollutants occurs (Chen et al. 2005). Peat (Zilli et al. 2001), soil, wood chips or mixtures of these substances (Pagans et al. 2005) are the most common materials used in the biological removal of organic pollutants. Other materials, such as pine bark, sewage sludge, yard waste compost and other various types of waste, are used worldwide (Hort et al. 2009; Nicolai and Janni 2001). However, natural organic packing materials have a short period of usefulness and must be replaced within 3–5 years (Tymczyna et al. 2004). To avoid this issue, synthetic inert packing materials, such as plexiglass chips (Chan and Lai 2010), polyurethane foam cubes (Van Groenestijn and Liu 2002) or others that have high durability, have also been used for biofiltration. For the efficient elimination of high VOC concentrations, the constituents in the packing materials of biofilters should be made of several different materials (natural and synthetic) (Hernández et al. 2010). The use of packing materials consisting of two different components provides a larger reactive surface area and greater durability than packing materials produced from a single material (Chan and Lai 2010; Liang et al. 2000).
Bacteria, yeast and fungi are microorganisms that are most frequently applied in biofilters (Zigmontienė and Baltrėnas 2004). Microorganism biomass is inoculated into the packing material, which is then humidified with an activated nutrient-rich solution (Baltrenas and Zagorskis 2009). Biofiltration is based on the degradation of VOCs using specific cultures of microorganisms, such as Pseudomonas fluorescens (Kleinheinz et al. 1999) and Pseudomonas putida. To remove VOCs from polluted air, the bacteria Aureobasidium pullulans, Penicillium sp., Acremonium strictum, Gliocladium viride, Aspergillus versicolor and Cladosporium herbarum are suitable. Zhang and Pierce (2009) used Rhodococcus bacteria to decompose VOCs, resulting in a treatment efficiency of approximately 90%. Researchers from Italy and Tunisia have also used Rhodococcus bacteria to remove VOCs, arguing that these bacteria can purify VOCs with a treatment efficiency fluctuating from 81 to 100% (Borin et al. 2006). The number of bacteria in the packing material must vary from 108 to 1010 CFU/g (Malhautier et al. 2005). To promote the growth of microorganisms and their ability to remove VOCs from the polluted airflow, favourable conditions for the development of the microorganisms must be ensured (Baltrėnas and Zagorskis 2010).
The biodegradation of VOCs using microorganisms depends on different parameters (i.e. temperature, humidity and pH in biofilters) (Yoon and Park 2002). The air temperature in biofilters has a significant impact on the efficiency of the biofiltration process as it plays an important role in the development and growth of different microorganisms. Thus, temperature is a crucial parameter for the effective performance of a biofilter. Asadi et al. (2009) examined the efficiency of a biofilter in the removal of acetone vapour from air using P. putida bacteria and found that an air treatment efficiency of 80.5% was reached when the inlet air temperature in the biofilter varied from 25 to 55 °C. An investigation of an airflow polluted with toluene vapour has suggested that the most effective biodegradation of this pollutant occurs at an air temperature of 30–35 °C (Yoon and Park 2002). To remove xylene from an airflow, Lee et al. (2002) reported that an optimal temperature of 30-35 °C is required. The effectiveness of biofiltration of air polluted by NH3 vapour from a composting process has been assessed using an air temperature of 35 °C in the biofilter (Pagans et al. 2005). A previous study on the treatment efficiency of a biofilter showed that a high biofilter purification efficiency can be obtained when the air humidity varies between 85 and 95% and when the air temperature in the biofilter fluctuates from 25 to 35 °C (Chang and Lu 2003). When a biofilter operates at higher temperatures, the population of microorganisms that adapts to higher temperatures increases in the medium. A reduction in the temperature of the biofilter system to ambient temperature may lead to a decrease in the population of these microorganisms and can promote the development of other microorganisms in the biofilter. Thus, to achieve optimal results, the air temperature in the biofilters should vary between 20 and 40 °C (Leson and Winer 1991).
Various biofilter designs have been used to remove VOCs from contaminated air (Farrokhzadeh et al. 2017; Lith et al. 1997). However, there is a lack of studies that apply lamellar plate-type biofilters for ammonia treatment. In addition, natural organic packing materials (Yoon and Park 2002) often used in biofilters have low durability and are more easily susceptible to degradation than synthetic materials (Chitwood and Devinny 2001). Therefore, two pilot plate-type biofilters with straight lamellar plates (SLP) or wavy lamellar plates (WLP) were constructed in this study for use with improved packing material and its associated humidification system for the air biofiltration of ammonia. The wood fibre used in the packing material was thermally treated to improve its durability and suitability for the growth of microorganisms. Because the developed biofilters use a capillary system to humidify the packing material, they do not require additional energy for the humidification of packing material. To reduce the resistance of airflow in biofilters, millimetre gaps between the plates of the packing material were made. The advantage of a lamellar plate structure is that the airflow resistance in the biofilters is reduced and no anaerobic zones remain where contaminants are not cleaned.
Using the newly developed biofilters with SLP and WLP, the aim of this study was to assess the efficiency of the biofiltration process of ammonia-contaminated air while maintaining different air temperatures and cultures of microorganisms in the biofilter. The second aim of this study was to assess the impact of the waviness of the plates in the biofilter on air treatment efficiency.
The effectiveness of the biofilters was studied at Vilnius Gediminas Technical University. Identification and quantification of the microorganisms were performed at Nature Research Centre (Vilnius, Lithuania). All experiments were performed between 2015 and 2019.
Materials and methods
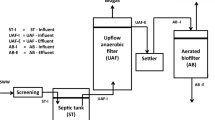
For this study, biofilters used as biological air treatment equipment were constructed (Fig. 1). The output of the biofilters with SLP (Fig. 1a) and WLP (Fig. 1b) reached 100 m3/h.
Schemes of the lamellar structure biofilters equipped with a capillary system for humidifying the packing material. a Biofiltration system made with straight lamellar plates. b Biofiltration system made with wavy lamellar plates. 1—The inlet duct of the contaminated air; 2—valve; 3—blower; 4—electric heater with the thermostat; 5—sampling sites (X1—before the biofilter, X2—after the biofilter); 6—air duct of the purified air; 7—temperature sensor; 8—plates; 9—perforated plate
Table 1 shows the technical characteristics of the biofilters. The packing material was made of hydrophilic synthetic texture [i.e. nonwoven caulking material (NWCM)] with a mass of 400–500 g/m2 and thermally treated (in a steam explosion reactor at 235 °C and under 32 bars of pressure) wood fibre (WF) attached to both sides of the NWCM. Both types of the biofilters had a structure of 44 plates arranged next to each other at a distance of 4 ± 0.2 mm. The capillary effect promotes the humidification of packing material when the water rises through the pores of the WF and NWCM with small distances between the plates.
The length, height and thickness of a single plate were 0.8 m, 1.3 m and 0.01 m, respectively, while the length, width and height of the equipment were 0.85 m, 0.7 m and 2.0 m, respectively.
Operating principle of biofilters with straight and wavy lamellar plates
Figure 1 shows the schemes of the biofilters with SLP and WLP. The ammonia-contaminated air was supplied to the biofilter through the inlet duct (1). The inner diameter of the inlet duct was 40 mm. A valve (2) installed in the inlet duct controlled the airflow rate. To measure the studied parameters, a sampling site was generated in the inlet duct. The contaminated airflow passed to the casing equipped with the biofiltration system. Through a perforated plate (9), the airflow was distributed throughout the entire packing material. The contaminated air moved between the plates of the packing material immersed in a solution and arranged at a distance of 4 ± 0.2 mm from each other towards the outlet. The treated airflow passed to the outlet duct (6), which had an inner diameter of 96 mm and was discharged. To measure the studied parameters, an opening (X2) in the outlet duct was made. The humidity of the packing material, the temperature of the liquid medium and airflow were regularly maintained. The temperature of the liquid medium was regulated with the aid of an electric heater (4), whereas the air temperature was controlled using a channel heater installed under the blower (3) of the supplied air.
Mesophilic microorganisms were used in the biofilters developed in this study. Mesophiles are microorganisms that develop at temperatures from 20 to 45 °C (Schiraldi and De Rosa 2014). Because the optimum growth temperature of most of these organisms ranges from 25 to 37 °C (Abdel-Banat et al. 2010), the assays were conducted at temperatures suitable for the development of mesophilic organisms (24, 28 and 32 °C). The tests were performed in the laboratory premises. Ammonia was evaporated using an electric stove, and the released ammonia vapour was supplied through a duct into the biofilter.
In the biofilter, the air was heated to the desired temperature by heating elements. The same temperatures (24, 28 and 32 °C) were also maintained in the reservoir at the bottom of the biofilter biomedium, which humidifies the biofilter plates via the capillary effect.
Determining liquid medium pH and temperature
The porous plates of the packing material were immersed into the liquid medium (biomedium) saturated with nutrients. Table 2 shows the composition of used biomedium. The pH of the biomedium was maintained using buffer solutions. The pH value and temperature were measured daily.
The nutrients used in the medium were selected based on literature (Chang and Lu 2003; Den et al. 2004; Chen et al. 2005; Trejo-Aguilar et al. 2005; Liao et al. 2008; Ryu et al. 2010; Lebrero et al. 2013).
Determining air temperature and humidity in the biofilter
The measurements of air temperature and humidity in the biofilter were performed using a TESTO 400 instrument. The air temperature and humidity were measured at five sites evenly distributed over the packing material and were monitored at airflow inlets and outlets 500 mm from the opening of the airflow inlet.
Determining packing material humidity
The humidity of the packing material was measured using a M0290 moisture meter at five sites each day of the experiment. All the measurements were performed in triplicate.
Activation of packing material and determination of biofiltration efficiency
The ammonia-contaminated air was supplied through the packing material of the biofilter. The airflow rate between the plates reached 0.16 m/s on average and was measured and monitored on a daily basis using a Testo 400 airflow instrument with an accuracy of ± 0.01 m/s.
Both native and introduced microorganisms were used in this study. Cultures of microorganisms were selected by performing separate assays using microbial cultures capable of efficiently breaking down ammonia. Eight strains of micromycetes (A. strictum 1–40-L, A. versicolor BF-4, Aureobasidium pullulans BF-58, Cladosporium sp. L-7 pp, Penicillium sp. BF-2, G. viride BF-81, Stachybotrys sp. BF-90 and C. herbarum 7KA), two strains of yeast (Exophiala sp. and Aureobasidium pullulans BIA1.1.2) and two strains of bacteria (Rhodococcus sp. 30 and Bacillus subtilis 28) were selected. The microorganisms were inoculated on biopacking material by seeding, and 2 L of active solution containing the selected microorganisms was sprayed onto the packing material. The average concentration of each microorganism in the solution was 108 CFU/g.
The study was divided into three stages (at temperatures of 24, 28, and 32 °C). Before exploring the efficiency of the biofilter to purify the air from the abovementioned pollutant, the microorganisms were acclimated for 15 days. The concentration of the pollutant was 300 ± 25 mg/m3. Ammonia-contaminated air was supplied to the biofilters each day of the experiment. Following the acclimation period, investigation of the biofiltration efficiency and quantification of the microorganisms in the packing material using the ammonia vapour-polluted air were performed for additional 10 days.
The efficiency of ammonia removal was calculated by measuring the inlet and outlet ammonia concentration. The concentration of the pollutant was determined using a MiniRAE 2000 photoionization detector. The ammonia removal rate (the amount of ammonia removed per hour, R), space velocity (the relationship between volumetric flow and volume of packing material, SV) and air cleaning efficiency (the percentage of ammonia removed, RE) were determined using the following equations (Kim et al. 2000; Taghipour et al. 2006):
where R is removal rate, g/m3/h; Cin is inlet ammonia concentration, g/m3; Cout is outlet ammonia concentration, g/m3; SV is space velocity, 1/h; Qin is volumetric flow rate, m3; Vpacking material is volume of packing material, m3; and RE is air cleaning efficiency, %.
Before activating the packing material, the absorption of ammonia in water with biogenic elements was evaluated. Subsequently, experiments were repeated by inoculating the microorganism cultures into the biofilters.
Identification and quantification of the microorganisms in the packing material
Packing material (1 g) was collected to identify and quantify microorganisms. Micromycetes were plated on an agar-solidified beer wort medium in Petri dishes that were incubated at 28 °C for 6–7 days. Subsequently, pure micromycetes cultures were identified using classical methods and descriptions for fungi (Ellis 1971; Pitt 1979; Pečiulytė and Bridžiuvienė 2008; Watanabe 2010). Yeast was plated on Rose Bengal CAF agar (Liofilchem, Italy) and Sabouraud agar with chloramphenicol (Liofilchem, Italy) in Petri dishes at 28 °C for 3–4 days. Yeast was identified using an Api 20 C AUX (bioMérieux, France) system. To culture bacteria from the tested samples, selective agar-solidified cetrimide (Pseudomonas (cetrimide) agar), nutrient agar and agar-solidified Bacillus cereus media were prepared. For seeding, bacterial suspensions at 1:10, 1:100, 1:1000, 1:10,000, 1:100,000, 1:1,000,000, 1:10,000,000, 1:100,000,000, 1:1,000,000,000 and 1:10,000,000,000 were prepared. Suspensions (0.1 ml) were poured onto the Petri dishes and rubbed with a spatula. Seeds were incubated at 28 °C for 2–4 days. The cultured bacteria were identified considering their morphological, physiological and biochemical properties and compared with those reported in the literature. Bergey’s Manual of Systematic Bacteriology was also used to identify bacteria (Palleroni 1984; Garrity 2005).
Statistical analysis
Microsoft Excel was used for statistical analysis. The measurements were repeated three times, and the mean and standard deviation of values were calculated. A one-way ANOVA test (p < 0.05) was used to differentiate between the means of different microorganisms present at different temperatures.
Results and discussion
Throughout the study, the air temperature in the biofilters was 24 ± 1, 28 ± 1 and 32 ± 1 °C. By adjusting the inlet air temperature, the effect of air temperature on the degree of ammonia vapour removal was monitored (Figs. 2, 3). The temperature of the treated air differed slightly from the air temperature in the biofilters. In some cases, the temperature of the inlet air was higher to maintain a stable air temperature in the biofilters because the inlet air stream flowed through the 25-cm layer of water and became cooler. A supplied air temperature greater than 40 °C may result in the death of the microorganisms unless thermophilic microorganisms are cultured in the biofilter (Leson and Winer, 1991).
Before inoculating the microorganisms, the ammonia absorption in water with nutrients reached 75.0 ± 2%. Figures 2 and 3 show the efficiency of the biofiltration process in the biofilters from 1 to 10 days after the acclimation period. Temperature is one of the most important factors determining the intensity of biochemical reactions and the growth of microorganisms (Leson and Winer 1991). At temperature of 24 °C, the purification of ammonia through the packing material made of NWCM and WF ranged from 81.0 to 84.0% and from 84.5 to 86.0% in the biofilters with SLP and WLP, respectively (Figs. 2, 3). Increasing the temperature to 28 °C resulted in a slight but significant increase in filtration efficiency, ranging from 82.5 to 85.2% (Fig. 2) and 85.0–87.0% (Fig. 3) in the biofilters with SLP and WLP, respectively. At 32 °C, the air purification efficiency of the biofilter with the SLP ranged from 82.0 to 83.3% (Fig. 2), while that of the biofilter with WLP ranged from 84.2 to 86.0% (Fig. 3). The effect of the temperature in the biofilters (between 24 and 32 °C) only slightly affected the ammonia removal efficiency. According to many studies, the optimum temperature for mesophilic microorganisms is between 20 and 35 °C (Delhoménie and Heitz 2005). Significantly higher ammonia purification efficiency was obtained in the biofilter with WLP. In this type of biofilter, the path of the treated air was increased. Compared to the biofilter with SLP, the treated air was retained longer in the biofilter with WLP, enabling longer contact with the packing material. Blázquez et al. (2017) studied the treatment of NH3 using a laboratory-scale biotrickling filter under different ammonia concentrations. When NH3 contact time with a packed bed was long (at inlet air rate of 38.4 m/h), nitrification rate was twofold higher than that observed at short NH3 contact time (at inlet air rate of 844 m/h) owing to a better distribution of ammonia through the packed bed (Blázquez et al. 2017).
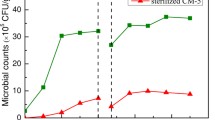
In the different types of biofilters, different tendencies of microorganisms’ growth in the packing material were observed. When filtering ammonia from air in the biofilter with SLP, 2.0–9.6 × 106, 4.2 × 106 to 5.6 × 107 and 3.1 × 107 to 2.5 × 108 CFU/g of micromycetes, yeast and bacteria were observed at 24 °C, respectively (Fig. 4). When the temperature was increased to 28 °C, a significant increase in the contents of all microorganisms was observed (for example, yeast levels increased to 8.0 × 108 CFU/g). However, increasing the temperature to 32 °C had no significant impact on the growth of microorganisms in the packing material. In the biofilter with SLP at 32 °C, the population of microorganisms varied from 1.0 ± 0.2 × 106 to 1.0 ± 0.3 × 108 CFU/g of micromycetes and from 4 ± 0.6 × 106 to 1.0 ± 0.2 × 1010 CFU/g of yeast, whereas the bacterial content was 2.3 × 109 CFU/g on average (Fig. 4). At 32 °C, a steady development of bacteria was observed, whereas the contents of micromycetes and yeast were unstable, due to variations in the humidity of the packing material.
When filtering ammonia in the biofilter with SLP, the composition of the types of micromycetes varied greatly depending on temperature. The fungus Chaetomium was dominant at 24 °C, whereas the fungus Geotrichum sp. was dominant at 28 °C. At the latter temperature, there were also many unidentified bacteria present that were resistant to the antibiotic chloramphenicol. At 32 °C, the inoculated micromycete C. herbarum predominated.
The yeast Exophiala jeanselmei grew best at 24 °C. During the removal of ammonia at 28–32 °C, the number of Ex. Jeanselmei decreased. In contrast, during the removal of ammonia in the biofilter with SLP, other species of yeast were not detected.
During the acclimation period, the bacteria B. subtilis, B. cereus, P. putida, P. fluorescens, Rhodococcus sp. and Staphylococcus aureus predominated in the packing material of the biofilter with SLP. In most cases, the bacteria B. subtilis, S. aureus, P. fluorescens, P. aeruginosa and Methylobacterium sp. grew at 24 °C and the bacteria B. subtilis, Rhodococcus sp., P. fluorescens, S. aureus and Methylobacterium sp. grew at 28 °C. Moreover, the bacteria B. subtilis, Pseudomonas aeruginosa, S. aureus, P. putida, Enterobacter sp. and Micrococcus sp. grew at 32 °C. Interestingly, the inoculated bacterium B. subtilis grew at all temperatures during the process of ammonia vapour removal. Borowski et al. (2017) used different types of bacteria in a microbial-mineral preparation for ammonia removal and observed that the most resistant strains of bacteria are B. subtilis and P. fluorescens during storage at 4 °C. Chung et al. (2001) studied the treatment of NH3 and H2S using biofilters containing the bacteria Arthrobacter oxydans and P. putida, and they reported that ammonia and hydrogen sulphide removal is greater than 95 and 90%, respectively.
The variation in temperature in the biofilter with WLP did not significantly influence the growth of microorganisms in the packing material. At 24 °C, the microorganism content in the packing material in the pilot-scale biofilter with WLP reached 106–108 CFU/g (Fig. 5). While filtering ammonia at 28 °C, the content of yeast and microscopic fungi fluctuated from 106 to 107 CFU/g. The content of bacteria did not exceed 109 CFU/g at 28 °C during the ammonia filtering process. At 32 °C, however, the content of microscopic fungi and yeast remained stable at 107 CFU/g, and the content of bacteria was 109 CFU/g.
During acclimation period, the fungus Trichoderma was dominant. After the microorganisms of the packing material were adapted to the ammonia, the fungus Trichoderma was replaced with micromycetes Chaetomium sp. and Stachybotrys sp. At the beginning of the ammonia vapour filtering process at 24 °C, however, Trichoderma sp. appeared again with Chaetomium sp., while it was close to the levels of Geotrichum sp at 28 °C. Furthermore, these micromycetes remained at the higher temperature of 32 °C where Stachybotrys sp. micromycetes could also be detected.
The adaptation of yeast in the packing material consisting of WLP to ammonia resulted in the number of yeast colonies E. jeanselmei being low. While removing ammonia vapour at 24–32 °C, only individual colonies of yeast E. jeanselmei were observed.
The removal of ammonia vapour showed that the number of bacterial species varied slightly at different temperatures. The bacteria B. subtilis, P. fluorescens, S. aureus, P. putida and Rhodococcus sp. were predominant at all investigated temperatures. Within the ammonia adaptation period, the bacterium Burkholderia convexa grew in addition to the abovementioned bacteria. At 24 and 28 °C, along with the previously mentioned bacteria, P. aeruginosa and B. cereus were predominant, and micromycetes began to develop. At 32 °C, the bacteria B. convexa, Enterobacter sp. and Erwinia sp. began to grow. At all investigated temperatures for ammonia removal, the inoculated bacterium B. subtilis grew.
Although the average number of different microorganisms in the biofilter with WLP was observed to be lower than that observed in the biofilter with SLP, the cleaning efficiency of this type of biofilter was higher. Thus, the biofilter design may have had a more significant contribution to NH3 degradation than the number of microorganisms.
Determining the changes in the number of various microorganisms is important to understand the ammonia filtration process. The results obtained in this study showed that the assayed microorganisms (micromycetes, yeasts and bacteria) adapted to a packing material consisting of WF and NWCM in developed biofilters with SLP and WLP. The microorganisms in the packing materials of biofilters effectively degraded ammonia and were stable under different temperatures for relatively long biofiltration conditions. The presence of a large number of bacteria, which had less influence on the physical properties of packing material, allowed for the effective elimination of NH3 from the air. In both biofilters, the number of yeast decreased from 1 to 10 days after the acclimation period at 28 °C. At this temperature, bacteria were predominant, suggesting that they significantly contributed to NH3 degradation.
NH3 may lower the durability of the biopacking material. Therefore, microscopic analysis of the structure of NWCM specimens of the tested biofilters was performed. NWCM specimens were assayed before and after the use of the biofilters for ammonia removal at different air temperatures with NH3 concentration of 300 mg/m3 and an airflow rate of 100 m3/h.
Figure 6 shows the images of the surfaces of both NWCM sides. The images were magnified 6 × and 25 × , and they were acquired using a Motic optical microscope. The NWCM comprised a woven base from a flat 1-mm-wide thread and smaller diameter fibre attached to the base. One side of the NWCM had a small amount of fibre and was named “base”, and the other side was named “fibre”. No extraneous formations were observed in the unprocessed specimens of the noncaulking material (Fig. 6a). After treatment with ammonia vapour, however, small quantities of a brown dispersible material were present on both sides of the SLP and WLP (Fig. 6b).
Electronic microscopy allowed determination of the structure of the materials used in this study (Fig. 7). The results demonstrated that small 15- to 25-µm-thick filaments comprised a major part of the NWCM. The spacing between the filaments was 5–10 times larger than their thickness, allowing for biofilm formation and avoiding anaerobic zones that can inhibit the growth of microorganisms. The disordered deployment of the filaments increased the specific surface area of the packing material and therefore the volume of the biomedium in it.
The results showed that an increase in the working time of the NWCM had no impact on its structure, which remained stable. However, an increase in operation time resulted in the increased amount of new brown-coloured formations on the side of the NWCM (Fig. 6). These formations were observed on the NWCM of the biofilter with both SLP and WLP.
Conclusion
The results obtained in this study showed that the cartridge of the biofilter consisting of wavy NWCM plates was superior to a biofilter consisting of straight plates. The results demonstrated that at an airflow rate of 0.16 m/s, the ammonia vapour removal efficiency reached 81.0–85.2% in the biofilter with SLP. When the biofilter with WLP was used, the efficiency of ammonia vapour removal reached 84.2–87.0%. In the biofilter with WLP, the contact time between the packing material and the air contaminated with ammonia was 1.5 times longer, leading to increased air treatment efficiency. The wavy lamellar plate-type biofilter was ~ 2–3% more efficient, and the structure was more rigid and stable, enabling it to retain more uniform spacing between the plates. Considering the reduction in ambient air pollution, a biofilter with WLP is beneficial because the manufacturing costs of straight and wavy NWCM plates are similar and the WF coating technology used is the same.
To achieve the air treatment efficiencies in the present study, an optimum mode for biofilter operation was maintained as follows: pH of 8.6, airflow humidity greater than 90%, aqueous medium temperature of 28–32 °C and airflow rate no greater than 0.16 m/s between the plates. Considering the growth of microorganisms in the packing materials of the pilot biofilters used in this study, 28 °C was the optimal temperature for the development of the microorganisms.
References
Abdel-Banat BM, Hoshida H, Ano A, Nonklang S, Akada R (2010) High-temperature fermentation: How can processes for ethanol production at high temperatures become superior to the traditional process using mesophilic yeast? Appl Microbiol Biotechnol 85:861–867. https://doi.org/10.1007/s00253-009-2248-5
Asadi M, Najafpour GD, Hashemiyeh BA, Mohammadi M (2009) Removal of acetone from contaminated air in biofilter using Pseudomonas putida. J Agric Environ Sci 5:712–719
Baltrenas P, Zagorskis A (2009) Investigation of cleaning efficiency of a biofilter with an aeration chamber. J Environ Eng Landsc Manag 17:12–19. https://doi.org/10.3846/1648-6897.2009.17.12-19
Baltrėnas P, Zagorskis A (2010) Investigation into the air treatment efficiency of biofilters of different structures. J Environ Eng Landsc Manag 18:23–31. https://doi.org/10.3846/jeelm.2010.03
Baquerizo G, Maestre JP, Sakuma T et al (2005) A detailed model of a biofilter for ammonia removal: model parameters analysis and model validation. Chem Eng J 113:205–214. https://doi.org/10.1016/j.cej.2005.03.003
Blázquez E, Bezerra T, Lafuente J, Gabriel D (2017) Performance, limitations and microbial diversity of a biotrickling filter for the treatment of high loads of ammonia. Chem Eng J 311:91–99. https://doi.org/10.1016/j.cej.2016.11.072
Borin S, Marzorati M, Brusetti L et al (2006) Microbial succession in a compost-packed biofilter treating benzene-contaminated air. Biodegradation 17:79–89. https://doi.org/10.1007/s10532-005-7565-5
Borowski S, Matusiak K, Powałowski S et al (2017) A novel microbial-mineral preparation for the removal of offensive odors from poultry manure. Int Biodeterior Biodegrad 119:299–308. https://doi.org/10.1016/j.ibiod.2016.10.042
Chan WC, Lai TY (2010) Compounds interaction on the biodegradation of acetone and methyl ethyl ketone mixture in a composite bead biofilter. Bioresour Technol 101:126–130. https://doi.org/10.1016/j.biortech.2009.08.001
Chang K, Lu C (2003) Biofiltration of isopropyl alcohol and acetone mixtures by a trickle-bed air biofilter. Process Biochem 39:415–423. https://doi.org/10.1016/S0032-9592(03)00096-7
Chen YX, Yin J, Wang KX (2005) Long-term operation of biofilters for biological removal of ammonia. Chemosphere 58:1023–1030. https://doi.org/10.1016/j.chemosphere.2004.09.052
Chitwood DE, Devinny JS (2001) Treatment of mixed hydrogen sulfide and organic vapors in a rock medium biofilter. Water Environ Res 73:426–443. https://doi.org/10.2175/106143001X139470
Chung YC, Huang C, Tseng CP (2001) Biological elimination of H2S and NH3 from wastegases by biofilter packed with immobilized heterotrophic bacteria. Chemosphere 43:1043–1050. https://doi.org/10.1016/S0045-6535(00)00211-3
Delhoménie MC, Heitz M (2005) Biofiltration of air: a review. Crit Rev Biotechnol 25:53–72
Den W, Huang C, Li CH (2004) Effects of cross-substrate interaction on biotrickling filtration for the control of VOC emissions. Chemosphere 57:697–709. https://doi.org/10.1016/j.chemosphere.2004.06.018
Ellis MB (1971) Dematiaceous hyphomycetes. Commonwealth Mycological Institute, Kew
Farrokhzadeh H, Hettiaratchi JPA, Jayasinghe P, Kumar S (2017) Aerated biofilters with multiple-level air injection configurations to enhance biological treatment of methane emissions. Bioresour Technol 239:219–225. https://doi.org/10.1016/j.biortech.2017.05.009
Garrity GM (2005) The gammaproteobacteria. In: Brenner DJ, Krieg NR, Staley JR (eds) Bergey’s manual of systematic bacteriology. Springer, New York, pp 1–1108
Hernández J, Prado ÓJ, Almarcha M, Lafuente J, Gabriel D (2010) Development and application of a hybrid inert/organic packing material for the biofiltration of composting off-gases mimics. J Hazard Mater 178:665–672. https://doi.org/10.1016/j.jhazmat.2010.01.137
Hort C, Gracy S, Platel V, Moynault L (2009) Evaluation of sewage sludge and yard waste compost as a biofilter media for the removal of ammonia and volatile organic sulfur compounds (VOSCs). Chem Eng J 152:44–53. https://doi.org/10.1016/j.cej.2009.03.026
Kim NJ, Hirai M, Shoda M (2000) Comparison of organic and inorganic packing materials in the removal of ammonia gas in biofilters. J Hazard Mater 72:77–90. https://doi.org/10.1016/S0304-3894(99)00160-0
Kleinheinz GT, Bagley ST, John WP et al (1999) Characterization of alpha-pinene-degrading microorganisms and application to a bench-scale biofiltration system for VOC degradation. Arch Environ Contam Toxicol 37:151–157. https://doi.org/10.1007/s002449900500
Lebrero R, Volckaert D, Pérez R et al (2013) A membrane bioreactor for the simultaneous treatment of acetone, toluene, limonene and hexane at trace level concentrations. Water Res 47:2199–2212. https://doi.org/10.1016/j.watres.2013.01.041
Lee EY, Jun YS, Cho K-S, Ryu HW (2002) Degradation characteristics of toluene, benzene, ethylbenzene, and xylene by Stenotrophomonas maltophilia T3-c. J Air Waste Manag Assoc 52:400–406. https://doi.org/10.1080/10473289.2002.10470796
Leson G, Winer AM (1991) Biofiltration: an innovative air pollution control technology for VOC emissions. J Air Waste Manag Assoc 41:1045–1054. https://doi.org/10.1080/10473289.1991.10466898
Liang Y, Quan X, Chen J et al (2000) Long-term results of ammonia removal and transformation by biofiltration. J Hazard Mater 80:259–269. https://doi.org/10.1016/S0304-3894(00)00314-9
Liao Q, Tian X, Chen R, Zhu X (2008) Mathematical model for gas–liquid two-phase flow and biodegradation of a low concentration volatile organic compound (VOC) in a trickling biofilter. Int J Heat Mass Transf 51:1780–1792. https://doi.org/10.1016/j.ijheatmasstransfer.2007.07.007
Lith CV, Leson G, Michelsen R (1997) Evaluating design options for biofilters. J Air Waste Manag Assoc 47:37–48. https://doi.org/10.1080/10473289.1997.10464410
Malhautier L, Khammar N, Bayle S, Fanlo J-L (2005) Biofiltration of volatile organic compounds. Appl Microbiol Biotechnol 68:16–22. https://doi.org/10.1007/s00253-005-1960-z
Martens W, Martinec M, Zapirain R et al (2001) Reduction potential of microbial, odour and ammonia emissions from a pig facility by biofilters. Int J Hyg Environ Health 203:335–345. https://doi.org/10.1078/1438-4639-00035
Nicolai RE, Janni KA (2001) Biofilter media mixture ratio of wood chips and compost treating swine odors. Water Sci Technol 44:261–267
Pagans EL, Font X, Sánchez A (2005) Biofiltration for ammonia removal from composting exhaust gases. Chem Eng J 113:105–110. https://doi.org/10.1016/j.cej.2005.03.004
Palleroni NJ (1984) Genus Pseudomonas migla. In: Noel R, Krieg J, Holf G (eds) Bergey’s manual of systematic bacteriology 1. William and Wilkins, Baltimore, pp 141–199
Pečiulytė D, Bridžiuvienė D (2008) Lietuvos grybai. II. Skurdeniečiai (Mortierellales) ir Pelėsiečiai (Mucorales) [Fungi of Lithuania. II Mortierellales and Mucorales]. Publishing office of the Institute of Botany, Vilnius
Pitt JI (1979) The genus Penicillium and its teleomorphic states Eupenicillium and Talaromyces. Academic Press, London
Ryu HW, Kim SJ, Cho KS (2010) Comparative studies on toluene removal and pressure drop in biofilters using different packing materials. J Environ Biol 31:315–318
Schiraldi C, De Rosa M (2014) Mesophilic organisms. In: Drioli E, Giorno L (eds) Encyclopedia of membranes. Springer, Heidelberg
Taghipour H, Shahmansoury MR, Bina B, Movahdian H (2006) Comparison of the biological NH3 removal characteristics of a three stage biofilter with a one stage biofilter. Int J Environ Sci Technol 3:417–424. https://doi.org/10.1007/BF03325951
Trejo-Aguilar G, Revah S, Lobo-Oehmichen R (2005) Hydrodynamic characterization of a trickle bed air biofilter. Chem Eng J 113:145–152. https://doi.org/10.1016/j.cej.2005.04.001
Tymczyna L, Chmielowiec-Korzeniowska A, Saba L (2004) Biological treatment of laying house air with open biofilter use. Polish J Environ Stud 13:425–428
Van Groenestijn JW, Liu JX (2002) Removal of alpha-pinene from gases using biofilters containing fungi. Atmos Environ 36:5501–5508. https://doi.org/10.1016/S1352-2310(02)00665-9
Watanabe T (2010) Pictorial atlas of soil and seed fungi: morphologies of cultured fungi and key to species. CRC Press, Boca Raton
Yoon IK, Park CH (2002) Effects of gas flow rate, inlet concentration and temperature on biofiltration of volatile organic compounds in a peat-packed biofilter. J Biosci Bioeng 93:165–169. https://doi.org/10.1016/S1389-1723(02)80009-3
Zhang J, Pierce GE (2009) Laboratory-scale biofiltration of acrylonitrile by Rhodococcus rhodochrous DAP 96622 in a trickling bed bioreactor. J Ind Microbiol Biotechnol 36:971–979. https://doi.org/10.1007/s10295-009-0576-1
Zigmontienė A, Baltrėnas P (2004) Biological purification of air polluted with volatile organic compounds by using active sludge recirculation. J Environ Eng Landsc Manag 12:45–52. https://doi.org/10.1080/16486897.2004.9636816
Zigmontienė A, Žarnauskas L (2011) Investigation and analysis of air-cleaning biofilter hybrid biocharge quantitative and qualitative parameters. J Environ Eng Landsc Manag 19:81–88. https://doi.org/10.3846/16486897.2011.557472
Zilli M, Palazzi E, Sene L et al (2001) Toluene and styrene removal from air in biofilters. Process Biochem 37:423–429. https://doi.org/10.1016/S0032-9592(01)00228-X
Acknowledgements
This study was performed in the framework of the project Applied Research and Technological Development of Plate Type Air Treatment Biofilter with a Capillary Humidification System for Packing Material “BIOFILTER” (Project No. VP1-3.1-ŠMM-10-V-02-015) under the Operational Programme for the Development of Human Resources 2007–2013, priority axis 3, “Strengthening Researcher Abilities”, measure VP1-3.1-ŠMM-10-V, “Promotion of High Level International Research”. This project was funded by the European Social Fund and was supported and co-funded by the European Union and the Republic of Lithuania.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Editorial responsibility: Samareh Mirkia.
Rights and permissions
About this article
Cite this article
Baltrėnas, P., Januševičius, T., Zagorskis, A. et al. Removal of ammonia by biofilters with straight and wavy lamellar plates. Int. J. Environ. Sci. Technol. 18, 1181–1190 (2021). https://doi.org/10.1007/s13762-020-02916-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-020-02916-5