Abstract
A recent systematic review showed that hematological malignancy is often complicated by membranous nephropathy (MN). Histologically, the deposition of IgG subclasses other than IgG4 may imply secondary MN, such as malignancy-associated MN (M-MN). We describe a very rare case of concurrent isolated IgG2-positive MN and B-cell lymphoma. An 83-year-old woman was hospitalized at our institute for facial and lower extremity edema persisting for 2 months. Laboratory tests showed urinary protein level of 10.8 g/day, serum albumin level of 1.6 g/dl, and serum creatinine level of 2.34 mg/dl. Soon after diagnosis of nephrotic syndrome, treatment with corticosteroid was initiated, but it proved to be ineffective. Renal biopsy showed isolated IgG2-positive MN with highly infiltrated CD20-positive lymphoid cells in the kidney. Computed tomography revealed systemic lymphadenopathy, and aberrant B-cells with immunoglobulin light chain restriction were detected in peripheral blood and bone marrow, which led to the diagnosis of mature B-cell lymphoma. Although rituximab (375 mg/m2/week) was administered, the patient suddenly died from gastrointestinal bleeding on day 40 of hospitalization. It is, thus, necessary to consider hematological malignancy when a diagnosis of MN is made. Further studies are expected to elucidate the pathogenesis and to help establish the adequate treatment for this rare situation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The direct infiltration of a malignant lymphoma to the kidney is relatively common [1]. However, few reports have been published regarding an association between glomerular disease and lymphoma, particularly non-Hodgkin’s lymphoma [2]. Conventionally, membranous nephropathy (MN) is believed to complicate solid tumors more frequently than hematological malignancies. However, a recent meta-analysis showed that hematological malignancies are relatively common in malignancy-associated MN (M-MN) [3]. Several reports have demonstrated that a predominant glomerular deposition of IgG4 is characteristic of idiopathic MN [4], whereas significant deposition of other IgG subclasses may imply a secondary MN, such as M-MN.
Rituximab (RTX) has been originally used for CD20-positive B-cell lymphoma for more than a decade. In recent years, use of this novel agent has been extended to autoimmune diseases, such as systemic lupus erythematosus [5], anti-neutrophil cytoplasmic antibody-associated vasculitis [6], and minimal change nephrotic syndrome [7]. In addition, some smaller clinical studies have reported the effectiveness of RTX for treatment of other glomerular diseases, such as MN [8] and focal segmental glomerulosclerosis [9].
Here we describe a very rare case of concurrent isolated IgG2-positive MN and malignant B-cell lymphoma that was treated with RTX.
Case report
An 83-year-old woman was hospitalized at our institute for facial and lower extremity edema persisting for 2 months. The patient had suffered from mild hypertension and dyslipidemia for 20 years. On admission, physical exam revealed no abnormalities but prominent pitting edema was observed in her extremities. Laboratory tests showed urinary protein level of 10.8 g/day, serum albumin level of 1.5 g/dl, and serum creatinine level of 2.34 mg/dl. Serum soluble Interleukin-2 receptor level was 1327 U/ml. Serum free light chain kappa and lambda were 162.0 and 111.0 mg/l, respectively. Immunoglobulins and complements were within normal ranges.
Soon after diagnosis of nephrotic syndrome, prednisolone (PSL) was administered at a starting dose of 30 mg/day, followed by 250 mg/day of methylprednisolone pulses for 3 days and a dose increase of PSL to 40 mg/day. However, there were no changes in urinary protein and serum creatinine levels; furthermore, lymphocyte in the peripheral blood increased with emergence of atypical lymphocytes. Computed tomography revealed swelling of the cervical, axillary, and mesenteric lymph nodes. Flow cytometry analysis of peripheral blood and bone marrow detected aberrant B-cells which were CD5−, CD10−, CD19+, CD20+, CD23+ and HLA-DR+. They also revealed immunoglobulin light chain restriction with predominance of lambda chain. Considering above, we diagnosed the patient as advanced mature B-cell lymphoma with leukemic change.
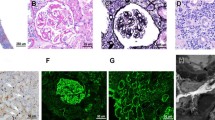
Renal biopsy was performed on day 19 of hospitalization. Periodic acid-Schiff staining showed highly infiltrated lymphoid cells in the kidney (Fig. 1a) with 40–50% of tubular atrophy and interstitial fibrosis. Only one out of 7 glomeruli exhibited global sclerosis. Remaining 6 glomeruli kept almost normal structure, without thickening of basement membrane or hypercellularity. On immunohistochemistry, granular IgG deposition along the glomerular basement membrane was observed, although bubbling and spike formation were not evident on the periodic acid-methenamine silver stain (Fig. 1b). Subclass analysis of IgG revealed isolated deposition of IgG2 (Fig. 1d), but not of IgG1, IgG3, or IgG4 (Fig. 1c, e, f). Infiltrating lymphoid cells did not express any subtype of IgG. Immunostaining for kappa and lambda light chains showed the same extent of deposition along the capillary wall (Fig. 1g, h). By electron microscopy, subepithelial electron dense deposits were sparsely located on glomerular basement membrane (Fig. 1i). Fibrils were absent. A histological diagnosis of MN (Ehrenreich and Churg stage 1) with infiltration of lymphoma cell was made.
a Highly infiltrated lymphoid cells in the kidney (Periodic acid-Schiff stain; magnification ×40). b Bubbling and spike formation are not evident (Periodic acid-methenamine silver stain; magnification ×400). c–f Isolated deposition of d IgG2, but not of c IgG1, e IgG3, or f IgG4 along the capillary wall, is found out (magnification ×100). Primary antibodies specific for: IgG1 (Mouse Anti-Human, clone 8c/6–39, Chemicon Europe), IgG2 (Mouse Anti-Human, clone HP6200, UK-Serotec Ltd.), IgG3 (Mouse Anti-Human, HP6050, Southern Biotech), IgG4 (Mouse Anti-Human, clone HP6023, Southern Biotech). g, h. Immunostaining for g kappa and h lambda light chains shows the same extent of deposition (magnification ×200). Primary antibodies specific for: kappa light chain (Rabbit Anti-Human, polyclonal, Dako), lambda light chain (Rabbit Anti-Human, polyclonal, Dako). i Subepithelial electron dense deposits (arrow) are observed by electron microscopy. Scale bar 1.0 µm
After the diagnosis of concurrent MN and B-cell lymphoma, administration of RTX (375 mg/m2/week) was started from day 29 of hospitalization, with informed consent from the patient and her family. RTX remarkably reduced lymphocyte number and serum creatinine level (Fig. 2), although massive proteinuria and hypoalbuminemia persisted. On the morning of day 40 of hospitalization, the patient died because of sudden cardiopulmonary arrest. An autopsy showed massive and diffuse hemorrhage from the erosive mucosa of the duodenum, ileum, and jejunum, where infiltration of CD20+ lymphoma cells were detected (Fig. 3a, b). These findings suggested that massive hemorrhage from gastrointestinal mucosa infiltrated with malignant lymphoma cells was the direct cause of death. Histopathological specimen of lymph nodes showed CD45+, CD20+, CD79a+, CD3−, CD5−, CD10−, BCL-2+, BCL-6− and c-Myc− lymphoma cells with nodular formation. Pathologically, it was diagnosed as follicular lymphoma, grade1. Infiltration of CD20+ lymphoma cells were also detected in bilateral kidneys (Fig. 3c).
Discussion
Conventionally, MN is believed to be a more frequent complication with solid tumors, such as those of the lungs, prostate, and colon, compared with hematological malignancies. However, a recent meta-analysis showed that hematological malignancies are relatively common in M-MN [3]. According to the meta-analysis, hematological malignancies are the third most common tumor, comprising 14.11% of M-MN cases. Therefore, it may be necessary to consider the coexistence of hematological malignancy when a diagnosis of MN is made.
When focusing on lymphoma-associated MN, only a few reports have been published. Togashi et al. [10] described a case of co-occurrence of angioimmunoblastic T-cell lymphoma and MN. Larger retrospective observational study was described by Da’as et al. about renal manifestation in lymphoma patients [11]. Compared with these previous studies, the current case is unique in terms of the following two reasons. First, the current case was associated with B-cell, not T-cell, lymphoma. Second, deposition of specific subtype of IgG was proved. Renal biopsy showed isolated IgG2 deposition along the glomerular basement membrane. The absence of IgG4 deposition is considered to be characteristic of M-MN [12]. In another case series, the immunofluorescence intensities of IgG1 and IgG2 were significantly stronger in M-MN than in idiopathic MN (I-MN) [13]. These findings may support our speculation that MN in the present case was not just an accidental coincidence of I-MN and B-cell lymphoma but was M-MN. Still, the direct relationship between MN and lymphoma remains unclear, as infiltrating lymphoma cells did not express any subtype of IgG. Ohtani et al. previously described that malignancy related antigens stimulated Th-1 type immune response in M-MN [13]. This indirect mechanism might also cause MN in lymphoma.
Proliferative glomerulonephritis with monoclonal IgG deposits (PGNMID) is an important differential diagnosis in this setting. A previous study investigated 26 cases of glomerulopathy with monoclonal Ig deposits, of which some were IgG2 positive MN following hematological malignancy [14]. The current case had the same extent of deposition for both light chain subtypes (kappa and lambda). This finding did not fall in the category of PGNMID with MN features [15].
PSL alone did not decrease urinary protein levels in the present case. Therefore, we subsequently combined RTX with PSL, expecting effects on both MN and lymphoma. A prospective cohort study on I-MN [16] showed that RTX can lead to remission at 12 months. Renal function actually improved in the present case, leading us to believe that there was, at least, a short-term effect of RTX on the kidney infiltration of the lymphoma. Conversely, we could not evaluate the long-term effect of RTX on MN because the patient died shortly thereafter.
The autopsy showed multiple bleeding sites in the small intestine with infiltration of lymphoma cells. Though small intestinal hemorrhage caused by lymphoma is uncommon compared to gastro or rectal lymphoma, there are some reports [17, 18]. Therefore, we speculated that infiltration of lymphoma cells might probably cause gastrointestinal bleeding and this could be direct cause of death.
Conclusion
We report a very rare case of isolated IgG2-positive MN complicated by B-cell lymphoma. Further studies are expected to elucidate the pathogenesis and to help establish the adequate treatment for this rare situation.
References
Richmond J, Sherman RS, Diamond HD, Craver LF. Renal lesions associated with malignant lymphomas. Am J Med. 1962;32(2):184–207.
Lien Y-HH, Lai L-W. Pathogenesis, diagnosis and management of paraneoplastic glomerulonephritis. Nat Rev Nephrol. 2011;7(2):85–95.
Leeaphorn N, Kue-A-Pai P, Thamcharoen N, Ungprasert P, Stokes MB, Knight EL. Prevalence of cancer in membranous nephropathy: a systematic review and meta-analysis of observational studies. Am J Nephrol. 2014;40(1):29–35.
Doi T, Mayumi M, Kanatsu K, Suehiro F, Hamashima Y. Distribution of IgG subclasses in membranous nephropathy. Clin Exp Immunol. 1984;58(1):57–62.
Looney RJ, Anolik JH, Campbell D, Felgar RE, Young F, Arend LJ, Sloand JA, Rosenblatt J, Sanz I. B cell depletion as a novel treatment for systemic lupus erythematosus: a phase I/II dose-escalation trial of rituximab. Arthritis Rheum. 2004;50(8):2580–9.
Stasi R, Stipa E, Del Poeta G, Amadori S, Newland A, Provan D. Long-term observation of patients with anti-neutrophil cytoplasmic antibody-associated vasculitis treated with rituximab. Rheumatology. 2006;45(11):1432–6.
Gilbert RD, Hulse E, Rigden S. Rituximab therapy for steroid-dependent minimal change nephrotic syndrome. Pediatr Nephrol. 2006;21(11):1698–700.
Remuzzi G, Chiurchiu C, Abbate M, Brusegan V, Bontempelli M, Ruggenenti P. Rituximab for idiopathic membranous nephropathy. Lancet. 2002;360(9337):923–4.
Fornoni A, Sageshima J, Wei C, Merscher-Gomez S, Aguillon-Prada R, Jauregui AN, Li J, Mattiazzi A, Ciancio G, Chen L, Zilleruelo G, Abitbol C, Chandar J, Seeherunvong W, Ricordi C, Ikehata M, Rastaldi MP, Reiser J, Burke 3rd GW. Rituximab targets podocytes in recurrent focal segmental glomerulosclerosis. Sci Transl Med. 2011;3(85):85ra46.
Togashi M, Wakui H, Kodama K, Kameoka Y, Komatsuda A, Nimura T, Ichinohasama R, Sawada K. Angioimmunoblastic T-cell lymphoma and membranous nephropathy: a still unreported association. Clin Exp Nephrol. 2010;14(3):288–93.
Da’as N, Polliack A, Cohen Y, Amir G, Darmon D, Kleinman Y, Goldfarb AW, Ben-Yehuda D. Kidney involvement and renal manifestations in non-Hodgkin’s lymphoma and lymphocytic leukemia: a retrospective study in 700 patients. Eur J Haematol. 2001;67(3):158–64.
Qu Z, Liu G, Li J, Wu L-h, Tan Y, Zheng X, Ao J, Zhao MH. Absence of glomerular IgG4 deposition in patients with membranous nephropathy may indicate malignancy. Nephrol Dial Transplant. 2012;27(5):1931–7.
Ohtani H, Wakui H, Komatsuda A, Okuyama S, Masai R, Maki N, Kigawa A, Sawada K, Imai H. Distribution of glomerular IgG subclass deposits in malignancy-associated membranous nephropathy. Nephrol Dial Transplant. 2004;19(3):574–9.
Guiard E, Karras A, Plaisier E, Van Huyen J-PD, Fakhouri F, Rougier J-P, Noel LH, Callard P, Delahousse M, Ronco P. Patterns of noncryoglobulinemic glomerulonephritis with monoclonal Ig deposits: correlation with IgG subclass and response to rituximab. Clin J Am Soc Nephrol. 2011;6(7):1609–16.
Ohashi R, Sakai Y, Otsuka T, Ohno D, Masuda Y, Murasawa T, Sato N, Shimizu A. Proliferative glomerulonephritis with monoclonal IgG2κ deposit successfully treated with steroids: a case report and review of the literature. CEN case reports. 2013;2(2):197–203.
Fervenza FC, Abraham RS, Erickson SB, Irazabal MV, Eirin A, Specks U, Nachman PH, Bergstralh EJ, Leung N, Cosio FG, Hogan MC, Dillon JJ, Hickson LJ, Li X, Cattran DC. Mayo Nephrology Collaborative Group. Rituximab therapy in idiopathic membranous nephropathy: a 2-year study. Clin J Am Soc Nephrol. 2010;5(12):2188–98.
Domizio P, Owen R, Shepherd N, Talbot I, Norton A. Primary Lymphoma of the Small Intestine A. Clinicopathological Study of 119 Cases. Am J Surg Pathol. 1993;17(5):429–42.
Howlett D, Farrugia M, Irvine A. Therapeutic transcatheter embolotherapy in the control of recurrent haemorrhage from lymphoma of the small bowel. Br J Radiol. 1995;68(808):431–4.
Acknowledgements
We appreciate Ryoko Saito for valuable advice on pathological findings and Kiyomi Kisu for her technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no conflict of interest exists.
Human and animal rights statement
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
About this article
Cite this article
Shimada, S., Nakamichi, T., Yamada, G. et al. Concurrent isolated IgG2-positive membranous nephropathy and malignant B-cell lymphoma. CEN Case Rep 7, 248–252 (2018). https://doi.org/10.1007/s13730-018-0336-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13730-018-0336-z