Abstract
The current study evaluated the prospective toxicity of hydroalcoholic extract of Piper longum L. (HEPL) dried fruits with acute and sub-acute oral administration in Wistar rats. During acute toxicity study, female rats were orally administered with HEPL at a single dose of 300 mg/kg and repeated dose of 2000 mg/kg (OECD guidelines-423). Subacute toxicity of HEPL (250, 500, and 1000 mg/kg p.o.) was studied with the control group (1% CMC) by daily dosing in Wistar rats of both sexes for 28 days. To assess reversibility, other satellite groups were kept for another two weeks post-treatment. The acute toxicity study showed no lethal effects, and toxicity signs and LD50 transpire more significantly than 2000 mg/kg. In subacute toxicity study, oral administration of extract for 28 days; caused no significant changes in body weights; food and water consumption of rats were observed. In addition, no toxic effects of the extract on rats for hematological and biochemical parameters were observed. Histopathological analysis revealed no signs of degeneration for kidneys, testis, and ovaries; however liver showed mild multifocal hepatocellular necrosis with infiltration of inflammatory cells in rats treated with 1000 mg/kg of HEPL. These results showed that 250 mg/kg and 500 mg/kg were found safe, in addition to some liver toxicity was found for 1000 mg/kg. It is concluded that the oral LD50 of HEPL was more significant than 2000 mg/kg and is considered safe. Hence, Piper longum is safe in the short and long term for an oral dose.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The purpose of acute toxicity studies is to identify the dosage that produces death or severe toxicological consequences once or for a few administrations. They are also used to give information on dosages for further research. These investigations offer a further chance in morphology, clinical chemistry, or other assessments to identify compound-induced consequences (Haschek et al. 2013; King 2002). The assessment of possible harmful effects of a new medication after a treatment term of 2–4 weeks is performed in subacute toxicity studies. In order to determine dose ranges for future subchronic and chronic toxicity studies, subacute toxicity studies are performed as range-finding studies. Subacute toxicity studies may also assist early clinical trials for up to 4 weeks of treatment duration(“A Compr. Guid. to Toxicol. Nonclinical Drug Dev.,” 2017). In recent years, different medicinal plants have been used for good health and the management of diseases. About 8% of the world’s population depends upon traditional medicine as primary health care because it is part of their culture and a trusted healing remedy (Ekor 2014). The use of traditional remedies improves due to their effectiveness, accessibility, and social acceptability by patients (Benzie et al. 2011; Patil et al. 2010). These traditional remedies can tackle diseases such as arthritis, diabetes, obesity, cardiovascular diseases, renal and liver diseases (Kunle 2012). Traditional remedies are usually considered safe and low toxic based on humans’ long-term history of use (Ugwah-Oguejiofor et al. 2019). The recent surveys showed that many traditional remedies revealed adverse effects (Koduru et al. 2006). Since safety profile is determined by conducting toxicity studies on them, it is necessary to evaluate and standardize different traditional or herbal remedies to be used in animals and humans to assess potentially toxic or adverse effects.
Piper longum L., commonly known as Pipali, is a small aromatic plant with erect and thin branches. It belongs to the family Piperaceae. In India, the species is distributed across the Western Ghats, Maharashtra, Central Himalayas, to Assam Khasi and Milker Hills of Bengal (Kapoor 2017). The fruits of Piper longum Linn contain piperine and starch, carbohydrates, proteins, alkaloids, amygdalin, volatile oil, and saponins (Dasgupta et al. 1980; Zaveri et al. 2010). In traditional medicines, parts or sometimes whole plant is used to treat diabetes (Nabi et al. 2013), cardiovascular diseases (Shoji et al. 1986), snake bite (Shenoy et al. 2013), arthritis (Bang et al. 2009), fungal infections (Lee et al. 2001), depression (Li et al. 2007), and asthma (Bui et al. 2017). Also antiulcer (Bajad et al. 2001), analgesic (Vedhanayaki et al. 2003), anti-inflammatory (Stöhr et al. 2001), and hepatoprotective (Koul et al. 1993) activities have been reported.
Siharat Chunlaratthanaphorn et al. reported acute and subchronic toxicity study of the water extract from dried fruits of Piper nigrum L. in rats. The results suggest that the water extract from the dried fruits of P. nigrum does not cause acute or subchronic toxicities in either male or female rats (Jaijoy et al. 2010). Piperine (1-piperoyl piperidine) is a major alkaloid present in various piper species of Piperaceae. Pawinee Piyachaturawat et al. reported acute and subacute toxicity of piperine in mice, rats and hamsters. Most animals given a lethal dose died of respiratory paralysis within 3–17 min. In subacute toxicity studies, the rats died within l-3 days after treatment. Histopathologic changes included severe hemorrhagic necrosis and edema in gastrointestinal tract, urinary bladder and adrenal glands. Death of these animals may be attributable to multiple dysfunctions in their organs (Piyachaturawat et al. 1983).
The use of this plant obtained great potential and high popularity due to its toxicity profile. Therefore, the current study investigates the acute and sub-acute toxic effects of hydroalcoholic extract of dried fruits of Piper longum Linn in Wistar rats.
Materials and methods
Plant authentication and preparation of plant extract
Fruits of Piper longum L. were collected from the Pirangut area in Pune, Maharashtra, India, during October (2018) after the flowering season. Dr. C. R. Jadhav, Botanist, Botanical Survey of India, Ministry of Environment, Forests and Climate Change, Koregoan Road, Pune, carried out taxonomic identification and authentication. A voucher specimen (BSI/WRC/IDEN.CER./2029/H3) of the plant was deposited in our laboratory, Maratha Vidya Prasarak Samajs College of Pharmacy, Nashik.
The plant material was dried under normal conditions to maintain its active principles and reduced to a coarse powder using a mixer grinder. These powdered fruits were stored in a tight container. Powdered fruits of Piper longum L. were extracted separately with 1000ml of diluted alcohol (alcohol: water, 70:30) by heating under reflux on a water bath for 6 h at 55 °C. The mixture was then filtered, and marc was extracted twice again in the same manner. The filtrates from each extracted step were pooled and concentrated under a vacuum using a rotary vacuum evaporator. After the complete extraction process, the extract was stored in sterile bottles under refrigeration conditions for further use.
Ethics approval and experimental animals
Male and female Wistar rats weighing about 150–200 g were obtained from Crystal Biological Solutions, Handewadi, Pune, Maharashtra, India. Before the commencement of the study, all animals were acclimatized for one week. Standard commercial normal pellet diet (NPD) and water were provided ad libitum for the animals during an experiment. The animals were maintained at controlled temperature (22 °C ± 2 °C) and relative humidity (55% ±5%) with 12:12 h light and dark cycle. The study was approved by the Institutional Animal Ethics Committee (Protocol No: MVPCP/IAEC/2019/05) of Maratha Vidya Prasarak Samajs College of Pharmacy, Nashik constituted under Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Ministry of Environment and Forests, Government of India. According to established general guidelines in Guide for Care and Use of Laboratory Animals, ethical guidelines were strictly followed.
Preliminary phytochemical screening of extract
The preliminary phytochemical screening of the plant extract was carried out using different qualitative phytochemical tests. The presence of alkaloids, steroids, flavonoids, liganas, tannins, and saponins was tested (Khandelwal 2005).
Acute and subacute toxicity studies
Acute toxicity test
The oral acute toxicity study of the hydroalcoholic extract of Piper longum L. was carried out using a single dose of 2000 mg/kg following the Organization for Economic Co-operation and Development (OECD) Guidelines no. 423 (OECD 2008). The first set of three female rats were used for 300 mg/kg dose, and safety was observed. The second set of three female rats were used for 200 mg/kg dose level in the study, followed by the next three rats. An animal was weighed and dosed with an equivalent volume of extract dissolved in 1% carboxymethylcellulose. By using a gastric feeding tube, the plant extract was administered. All animals were observed after dosing for an initial 5 min for signs of regurgitation. Animals were observed individually at least once during the first 30, 60, 120, 180, and 240 min after dosing, with particular attention and once daily after that, for a total of 14 days for signs of toxicity (Changes in skin and fur, eyes and mucous membranes, respiratory, circulatory, autonomic and central nervous systems, somatomotor activity, behavior pattern, tremors, convulsions, salivation, lacrimation, diarrhea, lethargy, sleep, and coma) according to the specifications of OECD. All animals were monitored for 14 days for the long-term probable toxic outcome.
Subacute toxicity test
Male and female Wistar rats of 150–180 gm were assigned randomly to six groups (n = 10/group: five males and five females). Group I was the control group, and group II was satellite control that received vehicle only (1% carboxymethylcellulose), group III was satellite test that received 1000 mg/kg dose while group IV–VI received 250, 500, and 1000 mg/kg of the extract respectively. All the rats were dosed using a curved and ball-tipped stainless steel feeding needle by oral gavage for 28 days as per OECD Guidelines-407. All animal weights were measured weekly while daily feed and water intake were recorded. The animals in the main groups were observed for 28 days, and those of the satellite groups were observed for 42 days after final dosing. On day 29, the rats from groups I, IV, V, and VI were weighed and sacrificed. Blood samples were collected by retro-orbital plexus for biochemical as well as hematological analyses. The Heart, Lungs, Ovary, Testis, Uterus, Kidney, Adrenal, Spleen, Pancreas, Liver were eradicated, weighed, trimmed, and then fixed in 10% formalin solution. Furthermore, histological analysis of organs like the heart, liver, testis, kidneys, and ovaries was done (OECD 2008).
Measurement of body and organ weights
Weights of all animals were measured weekly and weight gain was calculated using equation given below:
where, Wf = final weight, Wi = initial weight.
After blood collection, the heart, lungs, ovary, testis, uterus, kidney, adrenal, spleen, pancreas, and liver were eradicated and weighed. The relative organ weight of every collected organ was determined by following equation:
Assessment of haematological parameters
All the blood samples for hematology were kept in an Eppendorf tube along with anticoagulant K2 ethylene diamine tetra-acetic acid (EDTA). Hematological analysis was performed using a Triscan Duo Autohematology Analyzer (Operon). Parameters analyzed include erythrocyte (RBC), total and differential leukocyte (WBC), hemoglobin (HGB), hematocrit (HCT), platelet count (PLT), mean corpuscular hemoglobin (MCH), mean corpuscular volume (MCV), mean platelet volume (MPV), red distribution width (RDW), platelet distribution width (PDW), procalcitonin (PCT), platelet large ration count (LPCR) and some other parameters.
Assessment of serum biochemical parameters
For biochemical analysis, blood was quietly placed in an Eppendorf tube to avoid hemolysis of blood cells. Centrifugation of the blood sample was performed to obtain blood serum. Biochemical studies were carried out using standard methods for protein, albumin, glucose, urea nitrogen, creatinine, bilirubin, cholesterol, SGPT, and SGOT with the help of a smart 5 semi-auto biochemistry analyzer (Pathozyme).
Histopathological examination
On the 28th day, heart, ovaries, testis, kidney, and liver excised from the groups I and IV–VI were collected, weighed, and quickly set in 10% neutral buffered formalin at pH 7.4. It was also developed for histological studies. After the fixation procedure, tissues were cleansed in graded series of alcohol, washed in xylene, and inserted into paraffin. Tissues were segmented by a microtome (5 μm thin), stained with hematoxylin and eosin dye, dried, and mounted on a light microscope (40 X) for histological examinations. The same procedure was followed for Group II and III after 42 days, and the same organs were collected for histological examinations(Pieme et al. 2006).
Statistical analysis
All the results were expressed as the mean ± S.D. of five replicates. Using Graph Pad Prism Software, all the results were analyzed. All the groups were compared using a one-way analysis of variance (ANOVA) followed by Dunnett’s post hoc test. Values were considered statistically significant at p ≤ 0.05.
Results
Preliminary phytochemical tests
From the preliminary phytochemical analysis, it was observed that the HEPL exhibited the presence of alkaloids, steroids, flavonoids, terpenoids, and liganas.
Acute toxicity studies of HEPL in rats
The acute toxicity test at an oral dose of 2000 mg/kg of hydroalcoholic extract of Piper longum L. caused no death in the rats. No toxicity or lethal effects were observed in the animals throughout the 14 days study period. Hence, the extract may be safe and oral LD50 is more significant than 5000 mg/kg in Wistar rats.
Subacute toxicity studies of HEPL in rats
It was observed that hydroalcoholic extract of Piper longum at the doses of 250, 500, and 1000 mg/kg were found to be safe throughout the 28 days of the treatment. Whereas at higher doses, it showed some toxicity. These were confirmed by less weight gain, biochemical parameters, and histopathology results. Satellite groups recovered these indications. In this investigation, all the extract-treated animals’ clinical signs, body weights, dietary intake, hematological and serum biochemical tests, organ weight, necropsy, and histological analysis did not display any significant toxicological signs. No specific toxicity signs were observed in the extract-treated rats compared with the control.
Effect of HEPL on feed and water intake
The effects of the HEPL on the food and water intakes in subacute treatment are depicted in Table 1, and the graphs of the same are illustrated in Fig. 1. The single oral daily administration of the extract HEPL at study doses (250, 500, and 1000 mg/kg) and satellite groups for 28 days caused no significant changes (p ≥ 0.05) in food and water intakes compared with the control group.
Effect of HEPL on the body weights of rats
The effects of the HEPL on the body weights of rats before, during, and after the subacute treatment are showed in Table 2, and the graphs are depicted in Fig. 2. The single oral daily administration of HEPL to rats at study doses (250, 500, and 1000 mg/kg) and satellite groups for 28 days did not produce any significant change (p > 0.05) in body weights compared with the control group. Hence, non-significant increases in body weights were observed in treatment and satellite groups compared to normal control.
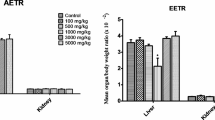
Effect of HEPL on relative organ weights
The effects of the HEPL on relative organ weights of rats after the subacute treatment are shown in Table 3. The single oral daily administration of HEPL to rats at study doses (250, 500, and 1000 mg/kg) and satellite groups for 28 days did not cause any significant alteration (p > 0.05) in relative organ weights compared with the control group.
The values are expressed as mean ± S.D., (n = 5).
Effect of HEPL on hematological parameters
The effects of the HEPL on hematological parameters of rats after the subacute treatment are tabulated in Table 4. All the tested different hematological parameters including Red blood cells (RBCs) count, differential leukocyte (WBC), hemoglobin (HGB), hematocrit (HCT), Platelet count (PLT), mean corpuscular hemoglobin (MCH), Mean corpuscular volume (MCV), Mean platelet volume (MPV), red distribution width (RDW), platelet distribution width (PDW), procalcitonin (PCT) and platelet large ration count (LPCR) were within the normal range compared to control group. No toxicologically significant changes (p > 0.05) in study doses (250, 500, and 1000 mg/kg), satellite groups and control group were found.
Effect of HEPL on biochemical parameters
The effects of the HEPL on biochemical parameters of rats after the subacute treatment for 28 days are depicted in Table 4. There was no significant difference (p > 0.05) in levels of protein, albumin, glucose, bilirubin, and cholesterol at the study doses (250, 500, and 1000 mg/kg) and satellite groups for 28 days when compared with normal. The creatinine kidney function test did not show any significant (p > 0.05) changes in study and satellite groups compared to normal. The significant changes (*p < 0.05) were observed for urea for 1000 mg/kg in male and female rats, which were recovered in satellite groups to normal levels. Furthermore, there was no significant difference (p > 0.05) in the levels of ALT (SGOT) of liver functions enzyme in study doses and satellite groups compared to normal while significant changes (*p < 0.05) were observed for urea for 1000 mg/kg dose in male and female which was recovered in satellite groups to normal levels (Table 5).
Effect of HEPL on histological parameters
Histological evaluations of heart, kidneys, ovaries, and testis in rats revealed no significant changes in study doses (250, 500, and 1000 mg/kg) and satellite groups compared with the control group, as shown in Figs. 3 and 4. However, liver of 1000 mg/kg HEPL treated test groups showed mild multifocal hepatocellular necrosis with infiltration of inflammatory cells, which was recovered in satellite groups to normal when observed under a light microscope.
The histological results (male rats) of heart, kidney, liver, and testis sections in control, satellite, and HEPL treated groups. The liver of the test groups of rats for 1000 mg/kg treated groups showed mild multifocal hepatocellular necrosis with infiltration of inflammatory cells, which was recovered in satellite groups to normal. The other organ sections were similar to the control
The histological results (female rats) of heart, kidney, liver, and testis sections in control, satellite, and HEPL treated groups. The liver of test groups of rats treated with 1000 mg/kg showed mild multifocal hepatocellular necrosis with infiltration of inflammatory cells, which was recovered in satellite groups to normal. The other organ sections were similar to the control
Discussion
Data from literature showed that piperine has a stimulating effect on the central nervous system and an increase in respiratory activity in all animal species studied. It is doubtful that piperine will have a poisonous effect on humans, given that the amount taken would be less than the toxic dose of 250 mg/kg body weight per day (Piyachaturawat et al. 1983). Piperine increases permeability of rat intestinal epithelial cells, possibly by interacting with the lipid environment (Johri et al. 1992). Furthermore, an aqueous extract (10% w/v) of black pepper increases gastric acid secretion in anesthetized albino rats (Vasudevan et al. 2000). This current study revealed the acute and subacute toxicity of Piper longum in Wistar rats. After the acute toxicity study, no toxicity or death-associated signs and symptoms were observed. A single dose of administration of HEPL (2000 mg/kg) did not display any significant changes in clinical signs and symptoms. Moreover, after the acute toxicity study period (14 days), no form of changes in any inner organs was evaluated compared to the control group. As per OECD criteria, category 5 with no animal death, the LD50 of the plant is more significant than 5000 mg/kg may be safe.
In the subacute toxicity study (28 days), after oral administration of HEPL (250, 500, and 1000 mg/kg) in study groups and satellite groups, no death of rats was observed, and no significant changes in food or water consumption compared to the control group. Hence, food and water intake are the symbols of usual health. The weights of the organs are the signs of the physiological and pathological health status of animals, while organ weights are indicators of toxicity in animals evaluated by toxicity tests (Cajuday et al. 2010; El Hilaly et al. 2004; Raina et al. 2015). Oral administration of herbal drugs sometimes shows toxic effects on essential organs like the heart, spleen, liver, and kidneys which play a vital role in normal physiological functioning (Ezeja et al. 2014). The liver and kidneys are the most important organs for xenobiotic actions like xenobiotic biotransformation and excretion of xenobiotics(Borawska et al. 2014). Our study showed no significant changes in organ weights (Table 3) of test and satellite groups compared with the control group, suggesting that the HEPL was not toxic to the animals. The nutritional status of animals is associated with changes in body weight(Kim et al. 2020). After a subacute toxicity study, all the animals revealed a steady rise in body weight. It showed that the daily intake of HEPL did not change the food and water consumption.
An assessment of hematological parameters plays an essential role in determining the health status of an individual (Rogić 2008). These parameters do not represent the toxic effects of compounds but also showed the changes in blood-related constituents. The hemopoietic system is an essential index of pathological and physiological status in both humans and animals. Our findings showed no noticeable hemolytic change in RBC, WBC, Hb, HCT, PLT, MCH, MCHC, MCV, and RDW. The extract evaluated no significant difference on the RBCs, which indicates that the HEPL does not affect morphology, hemorrhage, leukemia, erythropoiesis, or osmotic frugality of red blood cells (Porwal et al. 2017). WBCs play a vital role in defending the body’s response to tissue injury, infectious diseases, or any kind of inflammation. Furthermore, no significant differences were observed in the levels of WBCs or their subtypes, including lymphocytes, monocytes, and neutrophils, in study groups and satellite groups compared to the control (Table 4). This revealed that HEPL is non-toxic and safe. There was an increase in platelet count, but no significant changes were observed.
The functions of the liver were evaluated by the serum protein, albumin, and bilirubin. The increased serum levels depicted the cancerous conditions, while reduced serum levels indicate reduced synthetic functions (Ricchiuti 1999). Our findings showed no significant changes in the serum protein, albumin, and bilirubin were observed compared to the control group. Hence, it can be concluded that HEPL does not affect the normal functioning of the liver. Also, increased serum transaminase enzymes (AST and ALT) are an essential hallmark of liver impairment. The nonsignificant changes in AST and ALT activities were observed in the test groups. The retention of urea and creatinine are signs of kidney impairment[33]. The nonsignificant changes in urea and creatinine activities in study groups show no alteration in kidney functioning.
Histological studies are used to determine pathological changes in different tissues and organs. Histological examination of heart, liver, kidneys, testis, and ovaries of control group rats did not show any abnormalities. Microscopic examination of liver in test group rats at high dose showed mild multifocal hepatocellular necrosis with infiltration of inflammatory cells (Figs. 3 and 4). Similarly, when tested for the satellite control group, the first animal liver showed mild multifocal hepatocellular necrosis with infiltration of inflammatory cells, but the other two showed routine pathology. It indicates that the recovery takes place in animals after a given washout period. Hence, it suggests that the extract may not be toxic to the liver. Histological examination of the kidneys in male and female Wistar rats did not make any severe toxic alteration, confirming the safety of HEPL in the kidney.
It is concluded that the oral LD50 of hydroalcoholic extract of Piper longum L. is greater than 2000 mg/kg and is considered safe. The results obtained in 28 days oral subacute toxicity study indicates that the extract did not alter food intake, water consumption, body weight, or relative organ weights. Also, the extract did not have significant alteration in hematological and blood chemistry examination on both sexes. Furthermore, histological examinations after prolonged administration determined that it may not cause toxicity towards the heart, kidneys, ovaries, and testis, while mild toxicity to the liver was observed; this was recovered after the washout period. Hence, Piper longum L. is found to be safe in short and long-term use for an oral dose.
References
A Comprehensive Guide to Toxicology in Nonclinical Drug Development (2017) In A Comprehensive Guide to Toxicology in Nonclinical Drug Development. https://doi.org/10.1016/c2015-0-00147-2
Bajad S, Bedi KL, Singla AK, Johri RK (2001) Piperine inhibits gastric emptying and gastrointestinal transit in rats and mice. Planta Med 67(2):176–179. https://doi.org/10.1055/s-2001-11505
Bang JS, Oh DH, Choi HM, Sur BJ, Lim SJ, Yeon JY, Yang HI, Chul MC, Hahm DH, Kim KS (2009) Anti-inflammatory and antiarthritic effects of piperine in human interleukin 1β-stimulated fibroblast-like synoviocytes and in rat arthritis models. Arthritis Res Therapy. https://doi.org/10.1186/ar2662
Benzie IFF, Wachtel-Galor S (2011) Herbal medicine: biomolecular and clinical aspects: In: Herbal medicine: biomolecular and clinical aspects, 2nd edn
Borawska MH, Markiewicz-Zukowska R, Sawicka D, Naliwajko SK, Socha K, Omeljaniuk W, Car H (2014) Effects of chrysin on haematological parameters in rats. Farmacia 62(2):390–399
Bui TT, Piao CH, Song CH, Shin HS, Shon DH, Chai OH (2017) Piper nigrum extract ameliorated allergic inflammation through inhibiting Th2/Th17 responses and mast cells activation. Cell Immunol 322:64–73. https://doi.org/10.1016/j.cellimm.2017.10.005
Cajuday LA, Pocsidio GL (2010) Effects of Moringa oleifera Lam. (Moringaceae) on the reproduction of male mice (Mus musculus). J Med Plants Res 4(12):1115–1121. https://doi.org/10.5897/JMPR09.200
Dasgupta A, Datta PC (1980) Medicinal species of piper, pharmacognostic delimitation. Pharm Biol 18(1):17–25. https://doi.org/10.3109/13880208009065171
Ekor M (2014) The growing use of herbal medicines Issues relating to adverse reactions and challenges in monitoring safety. Front Neurol. https://doi.org/10.3389/fphar.2013.00177
El Hilaly J, Israili ZH, Lyoussi B (2004) Acute and chronic toxicological studies of Ajuga iva in experimental animals. J Ethnopharmacol 91(1):43–50. https://doi.org/10.1016/j.jep.2003.11.009
Ezeja MI, Anaga AO, Asuzu IU (2014) Acute and sub-chronic toxicity profile of methanol leaf extract of Gouania longipetala in rats. J Ethnopharmacol 151(3):1155–1164. https://doi.org/10.1016/j.jep.2013.12.034
Haschek WM, Rousseaux CG, Wallig MA, Bolon B, Ochoa R (2013) Haschek and Rousseaux’s Handbook of Toxicologic Pathology. In: Haschek and Rousseaux’s handbook of toxicologic pathology. https://doi.org/10.1016/C2010-1-67850-9
Jaijoy K, Vannasiri S, Piyabhan P, Lerdvuthisopon N, Boonraeng S, Khonsung P, Lertprasertsuke N, Sireeratawong S (2010) Acute and subchronic toxicity study of the water extract from the fruits of Piper chaba hunter in rats. Int J Appl Res Nat Prod 3(4):29–35
Johri RK, Thusu N, Khajuria A, Zutshi U (1992) Piperine-mediated changes in the permeability of rat intestinal epithelial cells. The status of γ-glutamyl transpeptidase activity, uptake of amino acids and lipid peroxidation. Biochem Pharmacol 43(7):1401–1407. https://doi.org/10.1016/0006-2952(92)90195-O
Kapoor LD (2017) Handbook of ayurvedic medicinal plants: herbal reference library. In: Handbook of ayurvedic medicinal plants: herbal reference library. https://doi.org/10.1201/9780203719473
Khandelwal KR (2005) Practical pharmacognosy techniques and experiments. In: NiraliPrakashan, Pune, India (20th ed.). Nirali Prakashan Pune. Retrieved from http://www.bookganga.com/R/5FYVG
Kim HY, Zuo G, Lee SK, Lim SS (2020) Acute and subchronic toxicity study of nonpolar extract of licorice roots in mice. Food Sci Nutr 8(5):2242–2250. https://doi.org/10.1002/fsn3.1465
King GS (2002) Handbook of toxicologic pathology. In: Archives of Pathology & laboratory medicine, vol 126, No 9, pp 1138–1139. https://doi.org/10.5858/2002-126-1138b-hotp
Koduru S, Grierson DS, Afolayan AJ (2006) Antimicrobial activity of Solanum aculeastrum. Pharm Biol 44(4):283–286. https://doi.org/10.1080/13880200600714145
Koul IB, Kapil A (1993) Evaluation of the liver protective potential of piperine, an active principle of black and long peppers. Planta Med 59(5):413–417. https://doi.org/10.1055/s-2006-959721
Kunle, (2012) Standardization of herbal medicines - a review. Int J Biodivers Conserv. https://doi.org/10.5897/ijbc11.163
Lee SE, Park BS, Kim MK, Choi WS, Kim HT, Cho KY, Lee SG, Lee HS (2001) Fungicidal activity of pipernonaline, a piperidine alkaloid derived from long pepper, Piper longum L., against phytopathogenic fungi. Crop Prot 20(6):523–528. https://doi.org/10.1016/S0261-2194(00)00172-1
Li S, Wang C, Wang M, Li W, Matsumoto K, Tang Y (2007) Antidepressant like effects of piperine in chronic mild stress treated mice and its possible mechanisms. Life Sci 80(15):1373–1381. https://doi.org/10.1016/j.lfs.2006.12.027
Nabi SA, Kasetti RB, Sirasanagandla S, Tilak TK, Kumar MVJ, Rao CA (2013) Antidiabetic and antihyperlipidemic activity of Piper longum root aqueous extract in STZ induced diabetic rats. BMC Complement Altern Med. https://doi.org/10.1186/1472-6882-13-37
OECD (2008) OECD guidelines for the testing of chemicals: repeated dose 28-day oral toxicity study in rodents. Drug Chem Toxicol 34(1):13. Retrieved from http://www.oecd-ilibrary.org/environment/test-no-407-repeated-dose-28-day-oral-toxicity-study-in-rodents_9789264070684-en
Patil UH, Gaikwad DK (2010) Phytochemical profile and antibacterial activity of stem bark of Anogeissus latifolia. Pharmacognosy J 2(17):70–73. https://doi.org/10.1016/S0975-3575(10)80014-8
Pieme CA, Penlap VN, Nkegoum B, Taziebou CL, Tekwu EM, Etoa FX, Ngongang J (2006) Evaluation of acute and subacute toxicities of aqueous ethanolic extract of leaves of Senna alata (L.) Roxb (Ceasalpiniaceae). Afr J Biotechnol 5(3):283–289. https://doi.org/10.5897/AJB05.197
Piyachaturawat P, Glinsukon T, Toskulkao C (1983) Acute and subacute toxicity of piperine in mice, rats and hamsters. Toxicol Lett 16(3–4):351–359. https://doi.org/10.1016/0378-4274(83)90198-4
Porwal M, Khan NA, Maheshwari KK (2017) Evaluation of acute and subacute oral toxicity induced by ethanolic extract of Marsdenia tenacissima leaves in experimental rats. Sci Pharm. https://doi.org/10.3390/scipharm85030029
Raina P, Chandrasekaran CV, Deepak M, Agarwal A, Ruchika KG (2015) Evaluation of subacute toxicity of methanolic/aqueous preparation of aerial parts of O. sanctum in Wistar rats: clinical, haematological, biochemical and histopathological studies. J Ethnopharmacol 175:509–517. https://doi.org/10.1016/j.jep.2015.10.015
Ricchiuti V (1999) Tietz textbook of clinical chemistry. Am J Clin Pathol 112(1):120–121. https://doi.org/10.1093/ajcp/112.1.120
Rogić D (2008) Tietz fundamentals of clinical chemistry. In: Biochemia medica: Biochemia medica, vol 18, No 3
Shenoy PA, Nipate SS, Sonpetkar JM, Salvi NC, Waghmare AB, Chaudhari PD (2013) Anti-snake venom activities of ethanolic extract of fruits of Piper longum L. (Piperaceae) against Russell’s viper venom: characterization of piperine as active principle. J Ethnopharmacol 147(2):373–382. https://doi.org/10.1016/j.jep.2013.03.022
Shoji N, Umeyama A, Saito N, Takemoto T, Kajiwara A, Ohizumi Y (1986) Dehydropipernonaline, an amide possessing coronary vasodilating activity, isolated from Piper iongum L. J Pharm Sci 75(12):1188–1189. https://doi.org/10.1002/jps.2600751215
Stöhr JR, Xiao PG, Bauer R (2001) Constituents of Chinese Piper species and their inhibitory activity on prostaglandin and leukotriene biosynthesis in vitro. J Ethnopharmacol 75(2–3):133–139. https://doi.org/10.1016/S0378-8741(00)00397-4
Ugwah-Oguejiofor CJ, Okoli CO, Ugwah MO, Umaru ML, Ogbulie CS, Mshelia HE, Umar M, Njan AA (2019) Acute and sub-acute toxicity of aqueous extract of aerial parts of Caralluma dalzielii N. E. Brown in mice and rats. Heliyon. https://doi.org/10.1016/j.heliyon.2019.e01179
Vasudevan K, Vembar S, Veeraraghavan K, Haranath PSRK (2000) Influence of intragastric perfusion of aqueous spice extracts on acid secretion in anesthetized albino rats. Indian J Gastroenterol 19(2):53–56
Vedhanayaki G, Shastri GV, Kuruvilla A (2003) Analgesic activity of Piper longum Linn. root. Indian J Exp Biol 41(6):649–651
Zaveri M, Khandhar A, Patel S, Patel A (2010) Chemistry and pharmacology of Piper longum L. Int J Pharm Sci Rev Res 5(1):67–76
Acknowledgements
Authors are grateful to Maratha Vidya Prasarak Samaj’s College of Pharmacy, MVP Campus, Gangapur road, Shivaji Nagar, Nashik and Crystal Biological Solutions, Pune for providing all the facilities to complete this research work.
Funding
The authors declare that this study has no funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics statement
The study was approved by the Institutional Animal Ethics Committee (Protocol No: MVPCP/IAEC/2019/05) of Maratha Vidya Prasarak Samajs College of Pharmacy, Nashik constituted under Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Ministry of Environment and Forests, Government of India. According to established general guidelines in Guide for Care and Use of Laboratory Animals, ethical guidelines were strictly followed.
Conflict of interest
Swati Vinod Jogdand has no conflict of interest. Ghanshyam B. Jadhav has no conflict of interest. Yogesh P. Talekar has no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jogdand, S.V., Jadhav, G.B. & Talekar, Y.P. Acute and sub-acute toxicity studies of hydro-alcoholic extract of dried fruits of Piper longum Linn in Wistar rats. ADV TRADIT MED (ADTM) 24, 179–190 (2024). https://doi.org/10.1007/s13596-023-00680-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13596-023-00680-8