Abstract
Angiopoietin-2 (Ang-2) has been reported to be involved in the development of type 2 diabetes (T2D) and accelerates micro and macro vascular complication. The current study aimed to investigate serum Ang-2 levels in Saudi patients with type 2 diabetes and its complications. In this observational cross-sectional study, serum was collected from 329 T2D subjects aged 34–79 years. All subjects were evaluated for fasting blood glucose (FBG), HbA1c, lipid profile, and kidney profile. Serum concentration of Ang-2 was assayed by ELISA-based method. Mean serum Ang-2 level was significantly higher in female T2D subjects (2881 ± 1550 pg/ml) compared with male T2D subjects (2450 ± 1477 pg/ml). The mean serum Ang-2 levels in female T2D subjects significantly increased with increase in their age and BMI, while mean serum Ang-2 levels did not significantly increase among the male with their age and BMI. A significantly higher level of Ang-2 was found in patients with diabetic complications like neuropathy (3293 ± 1728 pg/ml), retinopathy (2986 ± 1645 pg/ml), and vascular complications (3152 ± 1571 pg/ml). The study has established comprehensive Ang-2 levels in various subsets of adults with diabetes. While interpreting Ang-2 levels, the differences in age and sex need to be taken into account. Diabetic patients with higher Ang-2 levels should be monitored closely and level of serum Ang-2 should be served as a marker to identify patients at high risk of diabetic complications.
Similar content being viewed by others

Avoid common mistakes on your manuscript.
Introduction
Diabetes mellitus (DM) is a growing health concern globally and International Diabetes Federation (IDF) estimated that more than 415 million people were affected with diabetes in 2015. It is also estimated that the number of diabetes cases will increase to 642 million by 2040, putting a huge burden on health costs globally [1]. People with diabetes have an increased risk of developing microvascular complications that lead to blindness, renal failure, nerve damage, and limb amputation [2].
In our body, growth, development, and repair of the tissues are controlled by the process called angiogenesis. For the formation of vascular network in the body, the roles of vascular endothelial growth factor (VEGF) and angiopoietins are well characterized [3]. Any dysfunction in angiogenesis will cause serious health disorders [4]. To stimulate or maintain a healthy vasculature, performance of both VEGF and angiopoietins must be synergistic [5]. The angiopoietin family comprises growth factors—Ang 1–4—that act as ligands for the Tie-2 receptor which is a tyrosine kinase receptor [6]. Among them, Angiopoietin-1 (Ang-1) and Ang-2 have distinctive role in developing and maintaining circulatory vasculature. The action of VEGF is more important during initial stages and Angiopoietin-tie 2 signaling is important towards the later stages. VEGF helps in the initiation of vasculogenesis while Angiopoietin-tie 2 signaling works towards vessel remodeling and maturity processes. The action of Ang-2 is always an antagonist to Tie2/Ang-1 signaling and depends on the tissue conditions and presence of VEGF; it assists vessel growth as well as vessel regression [7].
Several studies reported the role of different markers in diabetes and its complications. Recently, studies on biomarkers (cystatin C, NGAL, transferrin, metalloproteases etc.) in Saudi diabetic subjects have shown good diagnostic accuracy in diabetic complications [8,9,10]. Even though, these studies did not consider sexual dimorphism in the levels of biomarkers and its application in clinical practices is not yet proven because of the study design. A recent study reported the sexual dimorphism in diabetic vascular complications as well as microvascular complications [11]. In view of growing importance for Ang-2 as prospective marker of vascular health status and diabetic complications, we evaluated the gender differences in the level of serum Ang-2 in Saudi diabetic adult and its relevance with the diabetic complications. The study will establish the levels of Ang-2 in Saudi type 2 diabetic adults with different complications.
Materials and methods
This is an observational cross-sectional study conducted in the University Diabetes Center at King Saud University. The study protocol was approved by the Institutional Review Board (IRB) and was conducted in accordance with the guidelines set by Ethics Committee of the College of Medicine, King Saud University.
Study subjects
A total of 370 type 2 diabetic patients, who did not have any other illness, were recruited from general diabetes clinic. We excluded 6 female subjects who were pregnant at the time of recruitment. From the remaining 364 subjects, we excluded 35 subjects with > 30 years of diabetes duration. Finally, a total of 329 subjects were selected; 149 males (45.3%) and 180 females (54.7%), aged between 34 and 79 years.
Diagnosis of type 2 diabetic patients was based on the American Diabetes Association (ADA) criteria or reported to be taking treatment for diabetes [12]. The intact hepatic function was evaluated by aspartate aminotransferase (AST) or alanine aminotransferase (ALT). Patients with microvascular and macrovascular complications were included in this study. Diabetic nephropathy (DN) was evaluated by glomerular filtration rate < 90 mL/min/1.73m2. The diabetic neuropathy (DN) was evaluated by assessing upper and lower extremities of nerve conduction velocity. The presence of at least one definite microaneurysm in any field photographed was considered as the criterion for the diagnosis of diabetic retinopathy (DR) [13]. Macrovascular complications were confirmed by a history of previous myocardial infarction, angina, and coronary or peripheral revascularization. Selected subjects with different complication were controlled by antihypertensive medications such as Angiotensin II receptor antagonist, Thiazide diuretic, Angiotensin converting enzyme inhibitor (ACEI), Beta blocker, and Calcium Channel blockers (CCB). The diabetes was controlled either by insulin therapy or by oral antidiabetic therapy such as Metformin, Sitagliptin, or Sulfonylurea.
Clinical and demographic data
A research physician interviewed all subjects and clinical data were collected including age, gender, diabetes duration, systolic blood pressure (SBP), diastolic blood pressure (DBP), and smoking. Demographic data such as body mass index (BMI) were calculated as the quotient of weight (kg) divided by height squared (m2).
Laboratory measurements
After an overnight fast, 5 ml of venous blood sample were collected from each subject in a plain tube. Serum was separated and stored immediately at − 20 °C for further analysis. The biochemical assessments including FBG, HbA1c, lipid profile (triglyceride, high-density lipoprotein (HDL), low-density lipoprotein (LDL), and total cholesterol (TC)) were analyzed using routine laboratory procedures (RX Daytona clinical chemistry analyzer, Randox, UK). eGFR was estimated using CKD-EPI creatinine equation [14].
Serum Ang-2 was measured using the sandwich enzyme-linked immunosorbent assay (ELISA) (R&D Systems, UK). The Ang-2 values were expressed in pg/ml. The minimum detectable level of Ang-2 was 46.9 pg/ml, and the intra- and inter-assay coefficients of variation were 5.9 and 8.96%, respectively. The reference values of serum Ang-2 in normal healthy subjects were 800 ± 200 pg/ml and T2D subjects were 2200 ± 700 pg/ml [15].
Statistical analysis
Data entry and analysis were performed using SPSS version 21, IBM, Chicago, Illinois, USA. Results are represented as mean ± SD and percentage. Comparison of means between two groups was carried out using Student’s t test of normality and equality of variance. For all statistical tests a value of p < 0.05 was considered as statistically significant.
Results
The baseline demographic characteristics and biochemical parameters of Saudi type 2 diabetic subjects are shown in Table 1. There were no significant differences between male and female subjects with respect to age, duration of diabetes, SBP, and diabetic complications. Significantly higher BMI, TC, and HDL were observed among female diabetic subjects than males. The glycemic parameters such as mean FBG and HbA1c were also higher in female subjects. Kidney function tests such as serum creatinine were higher in males; while eGFR is higher in females. Diabetic neuropathy was present in 25.5% (n = 83) and diabetic retinopathy in 44.4% (n = 146) of subjects; while 18.8% (n = 62) and 14.6% (n = 48) of the total subjects had macrovascular complications and diabetic nephropathy respectively. Serum Ang-2 level was significantly higher in female subjects compared to male diabetic subjects (p = 0.011).
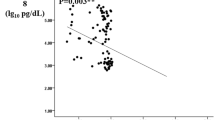
Table 2 presents level of Ang-2 based on different age groups. In type 2 diabetic subjects, the level of serum Ang-2 significantly increases with increase in their age (p = 0.002). Similar trend was found in female subjects (p = 0.018), while there is no significant increase in Ang-2 level in male subjects. Regardless of gender, when all subjects were considered, Ang-2 was significantly elevated in overweight and obese compared to lean (p = 0.017). The differences between the two groups were more marked in female subjects. When individual gender groups were considered, significant differences in Ang-2 levels were seen in female subjects (p = 0.05) (Fig. 1).
Levels of Ang-2 did not differ significantly between sexes whose age was < 50 years, while Ang-2 was significantly higher in diabetic females whose age is > 50 years (3046 ± 1574 vs 2583 ± 1561; p = 0.019). No statistically significant associations were observed between levels of Ang-2 between sexes whose BMI is < 30 while it was significantly higher among the obese female subjects. (3045 ± 1635 vs 2547 ± 1690; p = 0.043).
Table 3 shows the level of Ang-2 according to the diabetic complications. Ang-2 levels were significantly higher among subjects with diabetic neuropathy (p = 0.001) and diabetic retinopathy (p = 0.002). In addition, Ang-2 remained significantly higher among diabetic subjects with macrovascular complications when compared to those without (p = 0.008). However, levels of Ang-2 did not remain significantly different between participants with or without diabetic nephropathy. When compared to males Ang-2 remained higher among female subjects with microvascular complications but in macrovascular complications Ang-2 levels were higher in males.
Discussion
This is a comprehensive report evaluating the levels of Ang-2 in Saudi T2D adults. The diabetic females have comparatively higher Ang-2 level than diabetic males. Diabetic females have significantly increased Ang-2 level with increase in their age and BMI. A significantly higher level of Ang-2 was found in obese females with age group more than 50 years. The study also evaluates Ang-2 as a marker for the assessment of vascular health integrity and addresses DM patients at higher risk of diabetic complications. Since no previous data on reference levels of Ang-2 in Saudi adult are available, results of the present study cannot be compared with the other studies from the region.
Cardiovascular diseases are considered as one of the major complications of diabetes. There are only few studies reported on factors affecting cardiovascular diseases in Saudi population. In a previous study, diabetes, hypertension, obesity, dyslipidemia, smoking habits, and physical inactivity were mentioned as the major factors affecting the development of cardiovascular diseases in Saudi population. In addition, poor lifestyle habits such as lack of physical inactivity and high intake of total and saturated fat, and cholesterol are also related to the development of vascular disease [16]. Ang-2 is a glycoprotein exclusively expressed by the endothelial cells. It can act as an antagonist for Tie-2 and stimulate endothelial cell proliferation to induce vascular inflammation, accelerating the generation and development of vascular disease in patients with diabetes [17, 18].
In this study, we found that the Saudi diabetic females have comparatively higher Ang-2 level than diabetic males and it increases with increase in their BMI. To demonstrate the sexual dimorphism in serum concentrations of Ang-2 levels, a significantly higher level of serum concentrations of Ang-2 levels were observed in obese females compared to obese male subjects [19]. A previous study also demonstrated a gender dimorphism for Ang-2 levels, with women showing higher Ang-2 than men [20]. Differences in the gender on vascular functions and impact of feminine sex hormones on the expression of vascular growth factor have also been reported [21,22,23]. The exact mechanism behind the sexual dimorphism of Ang-2 has not been elucidated yet, so a detailed future study is suggested. In previous studies, it was reported that the Ang-2 expression may elevate by the inhibitory action of estrogen hormone on angiopoietin-1–Tie-2 receptor [24].
In the current study, we found that serum Ang-2 significantly increases with the increase in age for T2D and diabetic females but not in males and Ang-2 is significantly higher in diabetic females whose age is > 50 years. The angiogenic factors like VEGF, angiopoietins, fibroblast growth factor (bFGF) etc. are released by inflammatory cells having mitogenic and migratory effects on endothelium [25]. Increased transcription of angiogenic genes, such as VEGF and Ang-2, was triggered by a common stimulus for both chronic inflammation and angiogenesis [26]. Interleukin-6 (IL-6) is a proinflammatory cytokine, considered as mediator of chronic inflammation. The production of IL-6 is controlled by the macrophages produced by adipose tissue and adipocytes. In a previous study, it was found that postmenopausal women with the metabolic syndrome showed higher IL-6 as well as Ang-2 [27]. In our study, females above > 50 years were obese, and they were diabetic also. Hyperglycemia is considered as a major cause which exerts toxic effects on the endothelium and leads to inflammation which in turns increases the production of Ang-2 [28]. From the above possible reasons, we believe that females above 50 years are more likely to have chronic inflammation than males and the combined action of both hyperglycemia and inflammation leads to more circulating serum Ang-2 level.
In Middle Eastern countries, prevalence of obesity is more likely to be among women than in men. Like Saudi Arabia, the high-income countries such as Kuwait, UAE, Bahrain, and Qatar also show > 25 kg/m2 of mean BMI for both males and females [29]. A pervious study in Saudi Arabia reported that prevalence of obesity was higher among Saudi women than men (33.5 vs 24.1%) [30]. In Saudi females, the average BMI over age 30 years was reported to be > 30 kg/m2; the prevalence of overweight and obesity increased with age [31]. In our study, we found that Ang-2 level significantly increases with BMI in all T2D and diabetic females. Different evidences suggest that angiogenesis may be enhanced in human obesity. The evidence from the experimental study with both human and mice revealed that increased obesity is associated with Ang-2 mRNA expression in visceral adipose tissue and insulin resistance is linked with adipose tissue inflammation [32]. The above findings speculate that increased levels of angiogenic factors lead to adiposity and are involved in the pathogenesis of obesity. It is also demonstrated that Ang-2 is elevated in overweight and obese compared to lean control subjects, although Ang-2 was significantly elevated only in female obese subjects compared to lean female subjects. The sexual dimorphism was observed in serum concentrations of Ang-2 levels with significantly higher levels in obese females compared to obese male subjects. Although several studies reported the association between Ang-2 in the regulation of angiogenesis, the exact mechanism of angiogenic factors in both serum and adipose tissues is not expounded yet [19].
Hyperglycemia promotes toxic effect on vascular endothelium by the production and activation of advanced glycation end products (AGE) and reactive oxygen species (ROS) which leads to the development of micro and macrovascular complications in diabetes mellitus [33, 34]. In our study, we found that serum Ang-2 was significantly higher in DR subjects than without retinopathy. Previous studies also confirmed that Ang-2 levels show a significant difference among retinopathy group and are more prominent in proliferative group [35].
Various growth factors and cytokines play crucial role in the development of proliferative diabetic retinopathy (PDR). The main process involved in the development of retinal angiogenesis in diabetic retinopathy is angiogenic switch and proteolytic processing of the extracellular matrix (ECM). In PDR, involvement of proteases in ECM remodeling and Ang–Tie system in vessel remodeling has been reported [36,37,38]. In earlier studies, it has been reported that the increased level of Ang-2 indicates the severity or progression of diabetic retinopathy especially in PDR [39]. A previous clinical study indicated that the increased level of Ang-2 in diabetic patients is not because of leakage of blood from ischemic retinal capillaries but due to excessive production of Ang-2 from endothelial cells. This confirms the above statement; upregulation of stored Ang-2 is found in Weibel-Palade bodies of retinal endothelial cells [40].
When compared to healthy controls, serum Ang-2 is significantly higher in non-diabetic patients with acute and chronic congestive heart failures, while the level of Ang-1 level remains same in both groups and comparison with chronic and acute cases reported that Ang-2 level was found to be elevated in non-diabetic patients with acute coronary syndrome [5, 41]. Without considering the presence and absence of CVD, Ang-2 level seems to be elevated in diabetic patients; while no differences were found in the level of Ang-2 in diabetic patients with and without CVD. A reduction of Ang-2 levels only in diabetics without CVD reveals the altered relationship of Ang-2 with glycemic control in the presence of CVD [42, 43]. In the present study, the Ang-2 levels were elevated in DM patients with vascular complications. These findings are in agreement with a previous cross-sectional study, among diabetic patients showing the elevated level of circulating Ang-2 and were also associated with various cardio-metabolic parameters [44].
Moreover, in our study, we found a significant increase in Ang-2 level in diabetic patients with diabetic neuropathy. A previous study also reported that the polyneuropathy and insulin therapy were associated with higher Ang-2 levels. But even after controlling the presence of macrovascular complications, no differences were found between the levels of Ang-2 in subjects with and without diabetic polyneuropathy [44]. To our knowledge, the role of Ang-2 level in diabetic neuropathy is not yet reported anywhere. To clarify our results, future well-designed prospective studies on pathophysiological role of Ang-2 on diabetic neuropathy are needed. A well-defined role of Ang-2 in kidney diseases is already described in previous studies. The increased level of Ang-2 may lead to kidney damage [45]. A positive correlation with Ang-2 and proteinuria in kidney diseases was reported earlier [46]. In comparison with healthy controls, the diabetic patients with normoalbuminuria have significantly higher levels of both serum and urine Ang-2 and increased level of Ang-2 is related to the severity of diabetic nephropathy [47]. In our study, serum Ang-2 level was higher in DN group even though there is no significant difference among the patients with and without DN. This may be due to fact that majority of subjects in this study population were having eGFR > 90 ml/min/1.73 m2 and the sample size for DN group was very small. Another possible reason may be glycemic control which is considered as a primary determinant of reductions in serum Ang-2 [42].
In conclusion, present study establishes the levels of serum Ang-2 level in Saudi adult population with T2D in different subsets of age, gender, BMI, and diabetic complications. Our study shows that levels of Ang-2 are higher among female diabetic subjects. It also reveals that in female T2D subjects, Ang-2 levels rise with their age and BMI. A significantly higher level of Ang-2 was found in obese females with age group more than 50 years. Ang-2 levels are significantly higher in patients with diabetic neuropathy, retinopathy and diabetic vascular complications. Higher Ang-2 levels may serve as a marker to identify patients at high risk of vascular complications.
References
International Diabetes Federation. 7th Diabetes Atlas, 2015. http://www.diabetesatlas.org/resources/2015-atlas.html. Accessed 12 April 2017.
Fowler MJ. Microvascular and macrovascular complications of diabetes. Clinical diabetes. 2008;26:77–82.
Carmeliet P. Angiogenesis in health and disease. Nat Med. 2003;9:653–60.
Carmeliet P. Angiogenesis in life, disease and medicine. Nature. 2005;438:932–6.
Lee KW, Lip GY, Blann AD. Plasma angiopoietin-1, angiopoietin-2, angiopoietin receptor tie-2, and vascular endothelial growth factor levels in acute coronary syndromes. Circulation. 2004;110:2355–60.
Eklund L, Olsen BR. Tie receptors and their angiopoietin ligands are context-dependent regulators of vascular remodeling. Exp Cell Res. 2006;312:630–41.
Yancopoulos GD, Davis S, Gale NW, Rudge JS. Vascular-specific growth factors and blood vessel formation. Nature. 2000;407:242–8.
Al-Rubeaan K, Siddiqui K, Al-Ghonaim MA, Youssef AM, Al-Sharqawi AH, AlNaqeb D. Assessment of the diagnostic value of different biomarkers in relation to various stages of diabetic nephropathy in type 2 diabetic patients. Sci Rep. 2017;7(1):2684.
Abu El-Asrar AM, Mohammad G, Allegaert E, Ahmad A, Siddiquei MM, Alam K, et al. Opdenakker GMatrix metalloproteinase-14 is a biomarker of angiogenic activity in proliferative diabetic retinopathy. Mol Vis. 2018;24:394–406. eCollection 2018
Fawzy MS, Abu AlSel BT. Assessment of vitamin D-binding protein and early prediction of nephropathy in type 2 Saudi diabetic patients. J Diabetes Res. 2018;2018:8517929. https://doi.org/10.1155/2018/8517929. eCollection 2018
Maric-Bilkan C. Sex differences in micro-and macro-vascular complications of diabetes mellitus. Clin Sci. 2017;131:833–46.
American Diabetes Association. Classification and diagnosis of diabetes. Diabetes Care. 2015;38:S8–S16.
Early Treatment of Diabetic Retinopathy Study Research Group. Grading diabetic retinopathy from stereoscopic colour fundus photographs — an extension of the modified Airlie House classification. ETDRS report number 10. Ophthalmology. 1991;98:786–806.
Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF 3rd, Feldman HI, et al. CKD-EPI (Chronic Kidney Disease Epidemiology Collaboration). A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150:604–12.
Li L, ZHeng-Qing Y, Juan-Yu H, Jian-Yong X, Fan L, Guang-Chun Z, et al. Association between interleukin-19 and angiopoietin-2 with vascular complications in type 2 diabetes. J Diabetes Investig. 2016;7:895–900.
Alissa EM, Bahjri SM, Al-Ama N, Ahmed WH, Ferns GA. High cardiovascular risk in young Saudi males: cardiovascular risk factors, diet and inflammatory markers. Clin Chim Acta. 2006;365:288–96.
Thomas M, Augustin HG. The role of the Angiopoietins in vascular morphogenesis. Angiogenesis. 2009;12:125–37.
Maisonpierre PC, Suri C, Jones PF, Bartunkova S, Wiegand SJ, Radziejewski C, et al. Angiopoietin-2, a natural antagonist for Tie2 that disrupts in vivo angiogenesis. Science. 1997;277:55–60.
Silha JV, Krsek M, Sucharda P, Murphy LJ. Angiogenic factors are elevated in overweight and obese individuals. Int J Obes. 2005;29:1308–14.
Lieb W, Zachariah JP, Xanthakis V, Safa R, Chen MH, Sullivan LM, et al. Clinical and genetic correlates of circulating Angiopoietin-2 and soluble Tie-2 in the community. CircCardiovasc Genet. 2010;3:300–6.
Khalil RA. Sex hormones as potential modulators of vascular function in hypertension. Hypertension. 2005;46:249–54.
Ardelt AA, McCullough LD, Korach KS, Wang MM, Munzenmaier DH, Hurn PD. Estradiol regulates angiopoietin-1 mRNA expression through estrogen receptor-alpha in a rodent experimental stroke model. Stroke. 2005;36:337–41.
Albrecht ED, Babischkin JS, Lidor Y, Anderson LD, Udoff LC, Pepe GJ. Effect of estrogen on angiogenesis in co-cultures of human endometrial cells and microvascular endothelial cells. Hum Reprod. 2003;18:2039–47.
Ye F, Florian M, Magder SA, Sabah NA. Hussain. Regulation of angiopoietin and Tie-2 receptor expression in nonreproductive tissues by estrogen. Steroids. 2002;67:305–10.
Tahergorabi Z, Khazaei M. The relationship between inflammatory markers, angiogenesis, and obesity. ARYA Atheroscler. 2013;9:247–53.
Murdoch C, Muthana M, Lewis CE. Hypoxia regulates macrophage functions in inflammation. J Immunol. 2005;175:6257–63.
Eder K, Baffy N, Falus A, Fulop AK. The major inflammatory mediator interleukin-6 and obesity. Inflamm Res. 2009;58:727–36.
Tuo QH, Zeng H, Stinnett A, Yu H, Aschner JL, Liao DF, et al. Critical role of angiopoietins/Tie-2 in hyperglycemic exacerbation of myocardial infarction and impaired angiogenesis. Am J Physiol Heart Circ Physiol. 2008;294:H2547–57.
Musaiger AO. Overweight and obesity in eastern mediterranean region: prevalence and possible causes. J Obes. 2011;2011:1–17.
Memish ZA, Bcheraoui CE, Tuffaha M, Robinson M, Daoud F, Jaber S, et al. Obesity and associated factors — Kingdom of Saudi Arabia, 2013. Prev Chronic Dis. 2014;11:140236.
Al-Malki JS, Al-Jaser MH, WarsyA S. Overweight and obesity in Saudi females of childbearing age. Int J Obes. 2003;27:134–9.
Tabata M, Kadomatsu T, Fukuhara S, Miyata K, Ito Y, Endo M, et al. Angiopoietin-like protein 2 promotes chronic adipose tissue inflammation and obesity-related systemic insulin resistance. Cell Metab. 2009;10:178–88.
Pugliese G, Pricci F, Romeo G, Pugliese F, Mene P, Giannini S, et al. Upregulation of mesangial growth factor and extracellular matrix synthesis by advanced glycation end products via a receptor-mediated mechanism. Diabetes. 1997;46:1881–7.
Giacco F, Brownlee M. Oxidative stress and diabetic complications. Circ Res. 2010;107:1058–70.
Loukovaara S, Robciuc A, Holopainen JH, Lehti K, Pessi T, Liinamaa J, et al. Ang-2 upregulation correlates with increased levels of MMP-9, VEGF, EPO and TGFb1 in diabetic eyes undergoing vitrectomy. ActaOphthalmol. 2013;91:531–9.
Sivak JM, Fini ME. MMPs in the eye: emerging roles for matrix metalloproteinases in ocular physiology. Prig Retin Eye Res. 2002;21:1–14.
Suri C, Jones PF, Patan S, Bartunkova S, Maisonpierre PC, Davis S, et al. Requisite role of angiopoietin-1, a ligand for the TIE2 receptor, during embryonic angiogenesis. Cell. 1996;87:1171–80.
Saharinen P, Bry M, Alitalo K. How do angiopoietins Tie in with vascular endothelial growth factors? CurrOpinHematol. 2010;17:198–205.
Abu El-Asrar AM, Missotten L, Geboes K. Expression of hypoxia-inducible factor-1alpha and the protein products of its target genes in diabetic fibrovascularepiretinal membranes. Br J Ophthalmol. 2007;91:822–6.
Fiedler U, Augustin HG. Angiopoietins: a link between angiogenesis and inflammation. Trends Immunol. 2006;27:552–8.
Chong AY, Caine GJ, Freestone B, Blann AD, Lip GY. Plasma angiopoietin-1, angiopoietin-2, and angiopoietin receptor tie-2 levels in congestive heart failure. J Am CollCardiol. 2004;43:423–8.
Lim HS, Blann AD, Chong AY, Freestone B, Lip GY. Plasma vascular endothelial growth factor, angiopoietin-1, and angiopoietin-2 in diabetes: implications for cardiovascular risk and effects of multifactorial intervention. Diabetes Care. 2004;27:2918–24.
Lim HS, Lip GY, Blann AD. Angiopoietin-1 and angiopoietin-2 in diabetes mellitus: relationship to VEGF, glycaemic control, endothelial damage/dysfunction and atherosclerosis. Atherosclerosis. 2005;180:113–8.
Rasul S, Reiter MH, Ilhan A, Lampichler K, Wagner L, Kautzky-Willer A. Circulating angiopoietin-2 and soluble Tie-2 in type 2 diabetes mellitus: a cross-sectional study. Cardiovasc Diabetol. 2011;10:55.
Woolf AS, Gnudi L, Long DA. Roles of angiopoietins in kidney development and disease. J Am Soc Nephrol. 2009;20:239–44.
Chang FC, Lai TS, Chiang CK, Chen YM, Wu MS, Chu TS, Wu KD, Lin SL. Angiopoietin-2 is associated with albuminuria and microinflammation in chronic kidney disease. PloS one. 2013;8(3):e54668.
Chen S, Li H, Zhang C, Li Z, Wang Q, Guo J, Luo C, Wang Y. Urinary angiopoietin-2 is associated with albuminuria in patients with type 2 diabetes mellitus. International journal of endocrinology. 2015;2015 https://doi.org/10.1155/2015/163120.
Acknowledgements
The authors would like to acknowledge the members of the research unit, at University Diabetes Center, for patient recruitment.
Author information
Authors and Affiliations
Contributions
KS conceived and designed the study. KS, SSJ, and SSN conducted the research, provided the research materials, and collected and organized the data. SSN analyzed and interpreted the data. KS wrote the initial and final draft of the article. All of the authors have critically reviewed and approved the final draft and are responsible for the content and similarity index of the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the College of Medicine, King Saud University and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Rights and permissions
About this article
Cite this article
Siddiqui, K., Joy, S.S. & Nawaz, S.S. Serum Angiopoietin-2 levels as a marker in type 2 diabetes mellitus complications. Int J Diabetes Dev Ctries 39, 387–393 (2019). https://doi.org/10.1007/s13410-018-0679-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13410-018-0679-1