Abstract
Plants in various republics of the world face many dangers, including diseases that threaten crop productivity. The development and increase of novel species of infectious pathogens have made plant growth threatened. Fusarium wilt is one of the fiercest diseases affecting vegetables, which causes a great loss in the quality and quantity of pepper plants all over the world. In this study, stimulation of physiological immune responses in pepper plant using ecofriendly inducers (Aspergillus alabamensis, Aspergillus oryzae, and Aspergillus tubingensis) against Fusarium wilt had been studied. Endophytic fungi were assayed for their capability to synthesize hydrocyanic acid, phosphate solubilization, siderophores, and indole acetic acid synthesis, and the antifungal potential of ecofriendly inducers against F. oxysporum was also examined. A notable antifungal potential antifusarial with a supreme activity of A. tubingensis was found. More ultrastructure by TEM of Fusarium showed that sharp changes occurred in the cell wall, mycelium, and conidia as a result of treatment with A. tubingensis, A. oryzae, and A. alabamensis. The results demonstrated the high severity of F. oxysporum on pepper seedlings. Infected seedlings showed a high reduction in all vegetative parameters, photosynthesis, entire protein, and total carbohydrate. In the current study, the potential of endophytic fungi through foliar and soil application was applied to the Fusarium-infected pepper plants under pot conditions. Disease index, vegetative growth, photosynthetic pigments, osmolyte content, stress markers, and antioxidant isozymes were assessed. The achieved result indicates that tested endophytes through two modes (foliar and soil) lowered PDI and produced high protection, with the most protection influence represented by A. tubingensis (through the soil) by 83.33%. It was concluded that use of A. tubingensis, A. alabamensis, and A. oryzae could be commercially used as eco-friendly agents for the defense of pepper seedlings against Fusarium wilt disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Vegetables suffered from many pests, nematodes, and fungi that caused a severe loss in production pre- and postharvest [1]. The pepper plant is affected by different biotic diseases. Soil-borne plant pathogens mostly cause wilt and root rot diseases in pepper, thereby significantly affecting the growth and yield [2,3,4]. Fusarium wilt is one of the most destructive diseases of pepper in organic and conventional farming as it infects seedlings and kills them as soon as they germinate after their appearance on the surface of the soil, which leads to a small number of the resulting seedlings [5,6,7,8]. The infection of Fusarium can cause failure to capture light, reducing the efficiency of the photosynthesis process, and the difficulty of transporting water and salts, which causes burst condition inside cells in plants and causes an impact negative on plant growth and physiological signalization [9, 10]. The presence of these two free radicals can generate oxidative damage by encouraging the accumulation of superoxide (O2−), hydrogen peroxide (H2O2), and hydroxyl radicals (OH), and many compounds in plants are attacked by these species, including lipids, proteins, and nucleic acids [11]. Recently, in light of the economic crisis, there were voices calling for a stay away from environmental pollution to limit climate changes, which prompted plant pathologists to think of modern and effective methods that help in combating disease [12]. The use of biostimulants is the most important recent trend in the formation of a strong plant capable of resisting pathogens and creating an internal balance under stress conditions, which is known as induced resistance [13]. To protect themselves, plants developed the ability to scavenge those toxic species using a non-enzymatic pathway (accumulation of many secondary metabolites such as carotenoids, phenolics, soluble sugar, and proline content) and an enzymatic pathway (peroxidase and polyphenol oxidase) [14, 15]. The use of fungicides chemically synthesized has become less efficient in eliminating the disease in light of climatic changes, but plant resistance may be induced by biotic and abiotic elicitors [16, 17]. Plant physiological immunity can be stimulated by many means, perhaps the most important of which are growth-stimulating organisms against stress [3, 18, 19]. The harmful effect of Fusarium wilt can be minimized by the induction of natural exogenous inducers such as phytohormones and antioxidant molecules [20,21,22]. Furthermore, the molecules extracted from endophytes have been reported to be powerful biostimulants of growth, physiological immunity, and yield, as they enhanced tolerance to environmental stress by stimulating the antioxidant system and improving nutrient availability and nutrient uptake from the soil [23,24,25,26]. Application of endophytic Aspergillus resulted in a significant rise in the content of chlorophyll, protein contents, total sugar, and phenolic component of infected plants [18]. Endophytes are microorganisms that produce improved, growth-stimulating, and antimicrobial compounds that grow naturally within plants [27,28,29]. The application of Aspergillus as a biostimulant was documented to enhance chlorophyll contents and morphological growth attributes in different stressed crops via modifiable osmolytes and enzyme activities [30, 31]. Hence, the usage of endophytes in stimulating the synthetic immunity of plants was one of the most important biological factors in increasing crops yield [32,33,34,35]. Thus, the chief target of this study is to learn more about the mechanisms by which endophytic fungi Aspergillus spp. help plants resist wilt disease. Our study evaluates the effect of endophytic fungi Aspergillus spp. on F. oxysporum in vitro and then evaluates the effect of endophytic fungi on the induction of substances responsible for defense against Fusarium in pepper plants. Our study opens an effective way to control fungal phytopathogens in a way that is safe for the environment and has high effectiveness and efficiency instead of chemical fungicides that negatively affect the environment.
2 Materials and methods
2.1 Pepper seedlings
Three-week-old pepper seedlings were obtained from the Agricultural Research Center, Giza, Egypt (ARC).
2.2 Endophytic fungi
Endophytic fungi A. alabamensis MW444552, A. oryzae MW444554, and A. tubingensis MW444553 were used in this study. The biochemical traits of endophytic fungi were completed as the following; the capability of the tested fungi to create hydrocyanic acid (HCN) was achieved according to the procedure described by Trivedi et al. [36]. The capacity of the fungi to solubilize phosphate was established according to Rezzonico et al. [37]. The assessment of siderophores’ creation was assayed according to Sujatha and Ammani [38]. The ability of fungi to create indole acetic acid (IAA) was established by the technique described by Leveau and Lindow [39].
2.3 Fungal pathogen
F. oxysporum was obtained from the Regional Center for Mycology et al.-Azhar University (RCMB) and then was established by pathogenicity test according to Hibar et al. [40]. The inoculum was ready according to Büttner et al. [41].
2.4 In vitro antifusarial activity of endophytic fungi
Well-diffusion method was used to determine the activity of ethyl acetate crude extract of endophytic against F. oxysporum. The extracts were inoculated on potato dextrose broth medium (PDB) and then incubated at 28 ± 2 °C for 15 days. Fungal inoculum of F. oxysporum was spread thoroughly on the sterilized potato dextrose broth medium (PDA). Wells (7 mm) were occupied with 100 µL of extract (10 mg/mL). The plates were incubated at 25 °C for 7 days, and the inhibition zones were measured. The cytological variations that occurred in F. oxysporum were examined with a JEOL-JEM 1010 transmission electron microscope employed by the Regional Center for Mycology and Biotechnology (RCMB), Al-Azhar University, to examine stained slices at a voltage of 70 kV. The samples were handled and post-fixed according to [42,43,44].
2.5 Pot experiment design
Three-week-old pepper seedlings were transplanted into 40 × 40-cm pots (each pot one seedlings and ten replicates). In the green plastic house, the pots contained 7 kg of 1:3 sand and clay (a temperature of 22 °C during the daylight hours and 18 °C at the nighttime, with a relative humidity of 70–85%). F. oxysporum (107 spores/mL) was inoculated into the soil after planting except for the healthy control. Endophytic-tested fungi were applied three times by soil treatment or foliar spraying. The pots were prepared as the following: T1-healthy control (pepper seedlings were sowing in sterilized soil), T2-infected control (pepper seedlings were sowing in infected soil with F. oxysporum), T3-infected plants soil treated with A. alabamensis, T4-infected plants soil treated with A. oryzae, T5-infected plants soil treated with A. tubingensis, T6-infected plants foliar treated with A. alabamensis, T7-infected plants foliar treated with A. oryzae, and T8-infected plants foliar treated with A. tubingensis. For plant resistance evaluation, disease development and severity and morphological and biochemical indicators for resistance analysis were recorded after the plant reaches the age of 60 days.
2.6 Disease index
Disease symptoms were daily observed until 40 days after inoculation, while disease index and protection were evaluated according to Farrag et al. [16], using five score classes: 0 (no symptoms), 1 (slight yellow of leaves), 2 (moderate yellow plant), 3 (wilted plant and browning of vascular bands), and 4 (plants completely destroyed). PDI was calculated by the equation PDI = (1n1 + 2n2 + 3n3 + 4n4) × 100/4nt, where n1–n4 are the number of plants in each class and Nt is the total number of plants. Percent protection was calculated by Protection % = A–B/A × 100%, where A = PDI in infected control plants and B = PDI in infected-treated plants.
2.7 Metabolic indicators for pepper resistance
The measurement of chlorophyll and carotenoids achieved by the procedure of Vernon and Seely [45]. Photosynthetic pigments were extracted from fresh leaves (1 g) using 100 mL of 80% acetone, and then the color was determined spectrophotometrically at 665, 649, and 470 nm after the extract was filtered. A method of Umbreit et al. [46] was used for assayed of total soluble carbohydrate in dried tissues. The dried shoots (0.5 g) from each treatments were diluted with 5 mL of 30% trichloroacetic acid (TCA) and 2.5 mL of 2% phenol and filtered through filter paper, and then 1 mL of the filtrate was treated with 2 mL of anthrone reagent (2 g anthrone/L of 95% H2SO4). 620 nm was used to determine the produced blue-green color.
Total soluble protein determined by the method [47]. One milliliter of plant extract was combined with 5 mL (50 mL of 2% Na2CO3 prepared in 0.1 N NaOH and 1 mL of 0.5% CuSO4) and 0.5 mL of Folin’s reagent (diluted by 1:3 v/v). After 30 min, a color change could be seen at a wavelength of 750 nm. Free proline and phenol in plants were altered in response to infection; thus, the content of free proline was established by the methods of Bates et al. [48] and Dai et al. [49] and was used to assessed the total phenolics. Adopted method of Srivastava [50] was applied to determine the peroxidase activity. The activity of polyphenol oxidase was measured by the method of Matta [51].
2.8 Statistical analyses
One-way variance analysis (ANOVA) was applied to the resulting data. Least significant difference (LSD test) by CoStat (CoHort, Monterey, CA, USA) was used to demonstrate statistically relevant differences between treatments at p < 0.05 [52].
3 Results
3.1 In vitro antifusarial activity of endophytic fungi
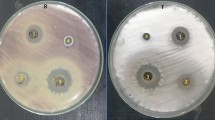
Results in Fig. 1 showed that tested endophytic fungi have great antifusarial activity, where A. tubingensis showed highly inhibition zone (25 mm diameter), A. oryzae (23 mm diameter), and then A. alabamensis (22 mm diameter).
3.2 Ultrastructure responses
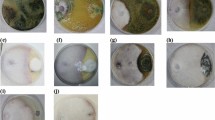
Results in Fig. 2 showed that the ultrastructure of F. oxysporum components was abnormal when applied to tested endophytes. There are detected abnormalities in fusarial cell wall and cytoplasmic substances compared with the control, where A. tubingensis caused distortion of conidia (macro- and microconidia). On the other hand, A. oryzae and A. alabamensis resulted in a moderately devastated Fusarium structure through the extension of macroconidia and microconidia, losing cytoplasmic components and proliferation wall thickness compared with the control.
3.3 Biochemical characteristics of endophytic fungi
Results in Table 1 revealed that the tested endophytic fungi have capability to produce HCN, IAA, and siderophores and can solubilize phosphorus, where A. alabamensis recorded the maximum activity of HCN and IAA production. Regarding to the ability of the tested endophytes to produce Siderophores, the results showed that A. oryzae recorded the highest siderophores amount, followed by A alabamensis and A. tubingensis. Also, A. alabamensis was best followed by A. tubingensis and then A. oryzae.
3.4 In vivo study
3.4.1 Disease severity (DS) and protection
The data in Table 2 revealed that F. oxysporum infection of pepper seedlings caused a high percent disease severity (PDI) 90.00%. Decreasing the severity of disease is the first evidence of the efficiency of the tested endophytes in inducing plant resistance. The results showed that both methods of treatment with the tested fungi, whether soil treatment or foliar spraying, reduced the severity of infection and increased the percentage of protection ranging (58.33: 83.33%), whereas during soil and foliar treatment, A. tubingensis recorded the lowest severity of infection by 15.00% and 25.00%, and the highest protection rate reached 83.33% and 72.22%.
3.4.2 Growth biomarkers
The severe decline in plant morphological characteristics (stem, root length, and leaves number) is the clear indications of the seriousness of the disease. It is clear from Table 3 that the infection with F. oxysporum caused a severe decrease in shoot length by 74.24%, root length by 59.48%, and number of leaves by 63.63%. On the other hand, application of all entophytic fungi, whether through soil or foliar led to the improvement morphological indicators of infected pepper seedlings, whereas treatment with A. tubingensis through soil and foliar recorded the highest shoot length (77.91 and 75.96 cm).
3.4.3 Effect of endophytic fungi on photosynthetic pigments
Photosynthesis is one of the most important vital processes in the development of plant growth stages, and at the same time, it is negatively affected by fusarial infection. Therefore, it was important to measure the photosynthesis pigments in this study. The data observed in Fig. 3 showed that F. oxysporum infection caused a significant deficiency of chlorophyll pigments a and b by 40.09% and 68.13%, respectively, and a significant increase in the carotenoid content by 87.78%. The results presented the improvement of photosynthetic pigments due to applying all tested endophytic fungi. These responses differed according to the method of application (soil or foliar). However, infected plants treated with A. tubingensis and A. alabamensis through the soil showed a significant improvement in chlorophyll a and b, followed by A. tubingensis through foliar, respectively. Also, the obtained results demonstrated that, the contents of carotenoids were decreased throughout the two-method application in response to the treatment with A. tubingensis, A. alabamensis, and A. oryzae).
Effect of endophytic fungi on photosynthetic pigments; T1-healthy control, T2-infected control, T3-infected plants soil treated with A. alabamensis, T4-infected plants soil treated with A. oryzae, T5-infected plants soil treated with A. tubingensis, T6-infected plants foliar treated with A. alabamensis, T7-infected plants foliar treated with A. oryzae, and T8-infected plants foliar treated with A. tubingensis
3.4.4 Effect of endophytic fungi on metabolic indicators
The results in Fig. 4 exhibited that the total sugars of fusarial-infected pepper seedlings declined significantly by 68.14%. The treatment of infected plants with endophytic fungi either through soil or foliar recovers the harmful effect of the F. oxysporum, by improving the sugar contents. Concerning the effect A. alabamensis, A. oryzae, and A. tubingensis through soil or foliar treatments on the infected plants with F. oxysporum, it was found that all fungi showed considerable increase in total carbohydrate, whereas the soil treatment of A. tubingensis and A. alabamensis were more efficient.
The results in Fig. 4 indicated that the protein content of infected pepper seedlings had a severe deficiency by 68.17 %. On the other hand, the application of endophytes recovers the damaging effect of the F. oxysporum infection through increasing the protein contents. Furthermore, the most effective treatments were A. alabamensis, A. oryzae, and A. tubingensis through foliar application. For more, the infected pepper plants showed an increase in the free proline and phenol contents by 22.13 % and 48.52 % compared to control healthy seedlings. Concerning the effect of tested entophytic fungi though soil or foliar treatment on the challenged plants with F. oxysporum, it was found that all tested entophytes causes an improvement of free proline and phenol content, whereas the treatment of A. alabamensis, A. oryzae, and A. tubingensis, respectively, through the foliar was more effective in increasing free proline. But the treatment of A. alabamensis and A. oryzae, respectively, through the foliar was more effective in increasing the phenol content.
3.4.5 Effect of endophytic fungi on antioxidant enzyme activity
Results in Table 4 revealed that, generally, there were significant rises in the activities of peroxidase (POD) and polyphenol oxidase (PPO) in infected pepper seedlings. the activity of POD and PPO enzymes in pepper seedlings diver in response to soil or foliar treatment with endophytic fungi. Moreover, all treatments stimulated POD and PPO activities, and maximum values for PPO were observed due to the application of A. alabamensis and A. oryzae on the trough (soil), followed by followed by A. oryzae and A. alabamensis (foliar), respectively. Also, POD activity was significantly improved in response to soil or foliar treatment with endophytic fungi. Application of A. tubingensis and A. oryzae through foliar as well as trough (soil), respectively, were the best stimulators for POD antioxidant enzyme activity.
4 Discussion
The increasing severity of plant diseases in light of climatic aberration in all countries of the world resulted in to need of application of a safe and effective method to control plant diseases urgent and necessary. Scientific reports have proven the importance of endophytic microorganisms in terms of their ability to stimulate plant growth and their antimicrobial properties, antifungal, antioxidant, anticancer, antiviral, and antimalarial activities [18]. In the previous study, the three fungi tested in the current study confirmed that they are able to inhibit the fungus F. oxysporum f. sp. lycopersici RCMB008001 from tomato [18]. The endophytic fungi have the capability to supply vital compounds that recover the destructive impacts of fungal disease through enhancing plant health as well as inducing resistance [53]. HCN is bioactive compound produced as a biological control agent, based on its toxicity against fungal phytopathogens [54]. The current study presented the capacity of the tested fungi to supply HCN and IAA, where A. alabamensis documented the greatest content of HCN and IAA. HCN is a wide spectrum against fungal pathogens [54,55,56]. In this regard, Ramette et al. [57] mentioned that HCN act on fungal pathogen by causing a direct imbalance in the cytochrome of the fungus cells, which impedes the breathing process of the pathogenic fungus.
The current results confirm the capability of the tested fungi to supply IAA that shows an essential function in growth, which improves plant health [58]. Phosphorus in its organic formulae, which will not be taken up by plant cells, seems to have substantial qualities, but to be absorbed, organic phosphorus must first be changed into inorganic phosphorus through dissolving by microorganisms that result in enhanced plant health [59, 60]. By focusing on the efficiency of tested endophytes on phosphorus solubilization, the results recorded that A. alabamensis was the best isolate. One of the most vital features of growth-stimulating microorganisms is their ability on phosphorus solubilization, as they secrete acids that dissolve mineral elements in the soil, such as dissolving insoluble rock phosphate salts and transforming them into soluble phosphate salts [61, 62].
In this work, we studied the effect of endophytic fungi on infected pepper plants by two methods of uses (soil and foliar). The results indicated that both modes of treatment whether soil treatment or foliar recover the severity of infection and recover the protection against Fusarium wilt, whereas during soil treatment, A. tubingensis documented the lower severity of infection by 17.5% and the protection by 80%; these effects may be described by [63]; they reported that Aspergillus recorded the maximum protection against Fusarium wilt by 33% in tomato plants. The current results agree with Kriaa et al. [64]. A. tubingensis has antifungal activity and can be applied as a new biofungicide against fungal phytopathogens. It is interesting to apply A. tubingensis as a biofungicide against Fusarium wilt by breaking down and preventing the formation of toxic fusaric acid [65]. This antifusarial activity is evidenced by the development of phenols and flavonoids by endophytic A. oryzae [66].
Plants are affected by a clear effect that appears in the decreasing of growth indicators as a result of biotic stress with the fungal disease. Our current results showed a sharp decrease in all growth traits as a clear result of fusarial infection. These results are in agreement with several studies [18]; they proved that Fusarium infection leads to a significant decrease in all vegetative growth traits (stem length, root, and number of leaves). This harmful influence on the vegetative growth is due to the occurrence of disorders and severe imbalance in growth hormones and the generation of oxidative explosions within plant cells [67].
The improvement of pepper growth is a strong evidence of the plant’s recovery from disease and the increase in systemic resistance in the plant. The results of this study indicated the improvement of growth in response to the application of endophytic fungi through soil or foliar application. This improvement can be clarified by the statement that endophytic fungi contains stimulating compounds for plant growth, in addition to its antifungal ability that induces the growth of plants under unfavorable conditions [68, 69].
The process of photosynthesis is the most important indicators of plant health. The data observed in the current study showed that F. oxysporum infection caused a severe deficiency of chlorophyll pigments a and b by 40.09% and 68.13%, respectively, and a significant raised in the level of carotenoid by 87.78%. This marked decrease in chlorophyll pigments is the evidence of the interruption of chlorophyll and the failure of the photosynthesis process and at the same time a noticeable increase of carotene pigment which is a non-enzymatic antioxidant, as it confirms that the plant is under stress [70]. It is interesting that the management of infected pepper plants with A. tubingensis and A. alabamensis through soil showed a significant improvement in chlorophyll a and b, followed by A. alabamensis through foliar respectively, compared to control infected. Also, the obtained results demonstrated that, the contents of carotenoids were decreased throughout the two-method application in response to the treatment with tested endophytes. These results are in agreement with Aldinary et al. [18]; they indicated that the use of fungal endophytes increases and improves the efficiency of the photosynthesis process, due to many changes in the chloroplasts and the contents of carotene and chlorophyll.
Decreased total soluble carbohydrate in plants as a result of Fusarium wilt was observed in several studies [18, 71]. On the other hand, the application of tested endophytic fungi to infected plants either through soil or foliar significantly improve the carbohydrate contents of the infected pepper seedlings.
The results of our current study showed that carbohydrates decreased significantly due to Fusarium infection, which can be explained by the infection resulting in a minimized photosynthetic rate; thus, a high respiration rate causes the lower carbohydrate and protein content [18, 72,73,74].
Phenols play a vigorous role in building plant resistance against biotic stress. The results showed a proliferation in the content of infected plants from phenols in accordance with [75, 76]. The buildup of these compounds in the infected plants by fungi was reported in several studies [77]. These results suggest that each enhancement or accumulation in phenol content induces systemic resistance in the host to face the stress.
Under fungal infection, plants accumulate osmolytes such as proline that act as osmoregulator to scavenge reactive oxygen species [78]. The increase of proline contents in infected pepper plants was similarly to that in heavy studies [21, 79]. In addition, stressed plants treated with fungal endophytes (such as Piriformospora indica and Aspergillus ochraceus) have low levels of proline in comparison with non-treated plants [80]. The results of this study dealt with the estimation of the activity of antioxidant enzymes and indicated that infection with F. oxysporum caused a significant increase in enzymes (POD and PPO). The usage of fungal endophyte induced the enzymatic activity as enhanced agents of defense. Results exhibited that POD and PPO activity improved significantly in plants exposed to endophytic fungi to keep ROS at a lower level in the cell as POD helps in the conversion of H2O2 to H2O [81].
5 Conclusion
Endophytic fungi (A. alabamensis, A. oryzae, and A. tubingensis) isolated from healthy Moringa oleifera leaves can be used as a hopeful and safe alternative bio antifungal against F. oxysporum in vitro and in vivo. The results of this study include important recommendations for adding fungal endophytic in plant disease resistance, as it improves different growth characteristics and stimulates the formation of carbohydrates, proteins, proline, and antioxidants. However, the application of endophytic A. alabamensis, A. oryzae, and A. tubingensis through soil or foliar significantly offers the prospective to recovery F. oxysporum wilt disease in pepper plants through obstructing the F. oxysporum mycelium and conidia and improving the growth performance of the infected pepper plants.
Availability of data and materials
All data and materials are viable.
Abbreviations
- ARC:
-
Agricultural Research Center, Giza, Egypt
- RCMB:
-
Regional Center for Mycology et al.-Azhar University
- PDB medium:
-
Potato dextrose broth medium
- PDA:
-
Potato dextrose agar medium
- POD:
-
Peroxidase
- PPO:
-
Polyphenol oxidase
- TEM:
-
Transmission electron microscope
References
Kazerooni EA, Maharachchikumbura SS, Adhikari A, Al-Sadi AM, Kang SM, Kim LR, Lee IJ (2021) Rhizospheric Bacillus amyloliquefaciens protects Capsicum annuum cv. Geumsugangsan from multiple abiotic stresses via multifarious plant growth-promoting attributes. Front Plant Sci 12:669693
Graber ER, Frenkel O, Jaiswal AK, Elad Y (2014) How may biochar influence severity of diseases caused by soilborne pathogens? Carbon Manag 5(2):169–183
Attia MS, El-Sayyad GS, Abd Elkodous M, Khalil WF, Nofel MM, Abdelaziz AM, El Rouby WM (2021) Chitosan and EDTA conjugated graphene oxide antinematodes in Eggplant: Toward improving plant immune response. Int J Biol Macromol 179, 333–344
Coşkun F, Alptekin Y, Demir S (2022) Effects of arbuscular mycorrhizal fungi and salicylic acid on plant growth and the activity of antioxidative enzymes against wilt disease caused by Verticillium dahliae in pepper. Eur J Plant Pathol 1–15
Tsitsigiannis DI, Antoniou PP, Tjamos SE, Paplomatas EJ (2008) Major diseases of tomato, pepper and egg plant in green houses. Eur J Plant Sci Biotechnol 2(1):106–124
Khalil AM, Ahmed AF, Mahmoud EE, Abdelaziz AM (2015) Influence of organic farming system on microbial biomass and fungal communities of agricultural soil. Afr J Mycol Biotechnol 20(3):23–40
Worku M, Sahe S (2018) Review on disease management practice of tomato wilt caused Fusarium oxysporum in case of Ethiopia. J Plant Pathol Microbiol 9(11):2
Khan AR, El_Komy MH, Ibrahim YE, Hamad YK, Molan YY, Saleh A A (2020) Organic management of tomato Fusarium wilt using a native Bacillus subtilis strain and compost combination in Saudi Arabia Int J Agric Biol 23:1003–1012
Dehgahi R, Subramaniam S, Zakaria L, Joniyas A, Firouzjahi FB, Haghnama K, Razinataj M (2015) Review of research on fungal pathogen attack and plant defense mechanism against pathogen. Int J Sci Res Agric Sci 2(8):197–208
Ashraf M, Foolad MR (2007) Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ Exp Bot 59(2):206–216
Ercal N, Gurer-Orhan H, Aykin-Burns N (2001) Toxic metals and oxidative stress part I: mechanisms involved in metal-induced oxidative damage. Curr Top Med Chem 1(6):529–539
Gullino ML (2021) Spores tulips with fever, rusty coffee, rotten apples, sad oranges, crazy basil Plant Diseases that Changed the World as Well as My Life. Springer
Aamir M, Rai KK, Zehra A, Dubey MK, Kumar S, Shukla V, & Upadhyay RS (2020) Microbial bioformulation-based plant biostimulants: A plausible approach toward next generation of sustainable agriculture Microb Endophytes Woodhead Publishing pp. 195–225
Ejaz S, Fahad S, Anjum MA, Nawaz A, Naz S, Hussain S, Ahmad S (2020) Role of osmolytes in the mechanisms of antioxidant defense of plants. In Sustainable Agriculture Reviews 39. Springer, Cham pp. 95–117
Jogawat A, Yadav B, Lakra N, Singh AK, Narayan OP (2021) Crosstalk between phytohormones and secondary metabolites in the drought stress tolerance of crop plants: a review. Physiol Plant 172(2):1106–1132
Farrag AA, Attia MS, Younis A, Abd Elaziz AMA (2017) Potential impacts of elicitors to improve tomato plant disease resistance. Al Azhar Bull Sci 9:311–321
Omer AM, Osman MS, Badawy AA (2022) Inoculation with Azospirillum brasilense and/or Pseudomonas geniculata reinforces flax (Linum usitatissimum) growth by improving physiological activities under saline soil conditions. Bot Stud 63(1):1–15
Aldinary AM, Abdelaziz AM, Farrag AA, Attia MS (2021) Biocontrol of tomato Fusarium wilt disease by a new Moringa endophytic Aspergillus isolates. Materials Today: Proceedings
Hashem AH, Abdelaziz AM, Attia MS, Salem SS (2022) Selenium and nano-selenium-mediated biotic stress tolerance in plants. In Selenium and Nano-Selenium in Environmental Stress Management and Crop Quality Improvement. Springer, Cham pp. 209–226
Zehra A, Meena M, Dubey MK, Aamir M, Upadhyay RS (2017) Activation of defense response in tomato against Fusarium wilt disease triggered by Trichoderma harzianum supplemented with exogenous chemical inducers (SA and MeJA) Braz J Bot 40(3):651–664
Abdelaziz AM, Attia MS, Salem MS, Refaay DA, Alhoqail WA, Senousy HH (2022) Cyanobacteria-mediated immune responses in pepper plants against fusarium wilt. Plants 11(15):2049
Khattab AM, Abo-Taleb HA, Abdelaziz AM, El-Tabakh MA, El-Feky MM, Abu-Elghait M (2022) Daphnia magna and Gammarus pulex, novel promising agents for biomedical and agricultural applications Sci Rep 12(1):1–9
Oleńska E, Małek W, Wójcik M, Swiecicka I, Thijs S, Vangronsveld J (2020) Beneficial features of plant growth-promoting rhizobacteria for improving plant growth and health in challenging conditions: A methodical review. Sci Total Environ 743, 140682
Hamid B, Zaman M, Farooq S, Fatima S, Sayyed RZ, Baba ZA, Suriani NL (2021) Bacterial plant biostimulants: A sustainable way towards improving growth, productivity, and health of crops. Sustain 13(5):2856
Ali S, Moon YS, Hamayun M, Khan MA, Bibi K, Lee IJ (2022) Pragmatic role of microbial plant biostimulants in abiotic stress relief in crop plants J Plant Interact 17(1):705–718
Rakkammal K, Maharajan T, Ceasar SA, Ramesh M (2022) Biostimulants and their role in improving plant growth under drought and salinity. Cereal Res Commun 1–14
Iqbal MS, Ansari MI (2020) Microbial bioinoculants for salt stress tolerance in plants. Microbial Mitigation of Stress Response of Food Legumes. CRC Press, pp 155–163
Chaturvedi A, Saraswat P, Singh A, Tyagi P, Ranjan R (2022) 10 Biostimulants: An alternative to chemical pesticides for crop protection. Biostimulants for Crop Prod Sustain Agric 139
Sharaf MH, Abdelaziz AM, Kalaba MH, Radwan AA, Hashem AH (2022) Antimicrobial, antioxidant, cytotoxic activities and phytochemical analysis of fungal endophytes isolated from ocimum basilicum. Appl biochem Biotechnol 194(3):1271–1289
Asaf S, Hamayun M, Khan AL, Waqas M, Khan MA, Jan R, Hussain A (2018) Salt tolerance of Glycine max. L induced by endophytic fungus Aspergillus flavus CSH1, via regulating its endogenous hormones and antioxidative system. Plant Physiol Biochem 128:13–23
Arif Y, Bajguz A, Hayat S, (2022) Moringa oleifera extract as a natural plant biostimulant. J Plant Growth Regul p. 1–16.
Adeleke BS, Babalola OO (2021) The endosphere microbial communities, a great promise in agriculture. Int Microbiol 24(1):1–17
Sturz AV, Christie BR, Nowak J (2000) Bacterial endophytes: potential role in developing sustainable systems of crop production. Crit Rev Plant Sci 19(1):1–30
Collinge DB, Jørgensen HJ, Latz MA, Manzotti A, Ntana F, Rojas Tayo EC, Jensen B (2019) Searching for novel fungal biological control agents for plant disease control among endophytes. Endophytes Growing World 31:25
Attia MS, Hashem, AH, Badawy AA, Abdelaziz AM (2022) Biocontrol of early blight disease of eggplant using endophytic Aspergillus terreus: improving plant immunological, physiological and antifungal activities. Bot Stud 63(1):1–14
Trivedi P, Pandey A, Palni LMS (2008) In vitro evaluation of antagonistic properties of Pseudomonas corrugata. Microbiol Res 163(3):329–336
Rezzonico F, Zala M, Keel C, Duffy B, Moënne‐Loccoz Y, Défago G (2007) Is the ability of biocontrol fluorescent pseudomonads to produce the antifungal metabolite 2, 4‐diacetylphloroglucinol really synonymous with higher plant protection?. New Phytol 173(4):861–872
Sujatha N, Ammani K (2013) Siderophore production by the isolates of fluorescent Pseudomonads. Int J Curr Res Rev 5(20):1
Leveau JH, Lindow SE (2005) Utilization of the plant hormone indole-3-acetic acid for growth by Pseudomonas putida strain 1290. Appl Environ Microbiol 71(5):2365–2371
Hibar K, Edel‐Herman V, Steinberg C, Gautheron N, Daami‐Remadi M, Alabouvette C, El Mahjoub M (2007) Genetic diversity of Fusarium oxysporum populations isolated from tomato plants in Tunisia. J phytopathol 155(3):136–142
Büttner G, Pfähler B, Märländer B (2004) Greenhouse and field techniques for testing sugar beet for resistance to Rhizoctonia root and crown rot. Plant Breeding 123(2):158–166
Amin BH, Amer A, Azzam M, Abd El-Sattar NE, Mahmoud D, Al-Ashaal S, Hozzein WN (2022) Antimicrobial and anticancer activities of Periplaneta americana tissue lysate: An in vitro study. J King Saud Univ-Sci 34(5):102095
Hashem AH, Saied E, Amin BH, Alotibi FO, Al-Askar AA, Arishi AA, Elbahnasawy MA (2022) Antifungal activity of biosynthesized silver nanoparticles (AgNPs) against aspergilli causing aspergillosis: Ultrastructure Study. J Func biomater 13(4):242
Amin BH, Abou‐Dobara MI, Diab MA, Gomaa EA, El‐Mogazy MA, El‐Sonbati AZ, Salama HM (2020) Synthesis, characterization, and biological investigation of new mixed‐ligand complexes. Appl Organomet Chem 34(8):e5689
Vernon LP, Seely GR (2014) The chlorophylls. Academic press
Umbreit WW, Burris RH, and Stauffer JF (1964) Manometric techniques: a manual describing methods applicable to the study of tissue metabolism
Lowry OH, Rosebrough NJ, Farr AL, Rose J, Randall AJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Bates LS, Waldren RP, Teare I (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39(1):205–207
Dai GH, Andary C, Cosson-Mondolot L, Boubals D (1993) Polyphenols and resistance of grapevines to downy mildew. Int Symp Natur Phenols Plant Resist 381:763–766
Srivastava S (1987) Peroxidase and poly-phenol oxidase in Brassica juncea plants infected with Macrophomina phaseolina (Tassai) Goid. and their implication in disease resistance. J Phytopathol 120(3):249–254
Matta A (1969) Accumulation of phenols in tomato plants infected by different forms of Fusarium oxysporum. Phytopathology 59:512–513
Snedecor GW and WG Cochran, Statistical methods. 2nd printing. Iowa State Univ. press, Ame., USA, 1982. 507
Attia MS, Hashem AH, Badawy AA, Abdelaziz AM (2022) Biocontrol of early blight disease of eggplant using endophytic Aspergillus terreus: improving plant immunological, physiological and antifungal activities. Bot Stud 63(1):1–14
Ahmed AF, Attia MS, Faramawy F, Salaheldin MM Saudi J Pathol Microbiol (SJPM) ISSN 2518–3362 (Print)
Gorai PS, Ghosh R, Mandal S, Ghosh S, Chatterjee S, Gond SK, Mandal NC (2021) Bacillus siamensis CNE6-a multifaceted plant growth promoting endophyte of Cicer arietinum L. having broad spectrum antifungal activities and host colonizing potential. Microbiol Res 252, 126859
Ali S, Hameed S, Shahid M, Iqbal M, Lazarovits G, Imran A (2020) Functional characterization of potential PGPR exhibiting broad-spectrum antifungal activity. Microbiol Res 232, 126389
Ramette A, Frapolli M, Défago G, Moënne-Loccoz Y (2003) Phylogeny of HCN synthase-encoding hcnBC genes in biocontrol fluorescent pseudomonads and its relationship with host plant species and HCN synthesis ability. Mol Plant Microbe Interact 16(6):525–535
Chen B, Luo S, Wu Y, Ye J, Wang Q, Xu X, Yang X (2017) The effects of the endophytic bacterium Pseudomonas fluorescens Sasm05 and IAA on the plant growth and cadmium uptake of Sedum alfredii Hance. Front Microbiol 8:2538
Walpola BC, Yoon M-H (2012) Prospectus of phosphate solubilizing microorganisms and phosphorus availability in agricultural soils: a review. Afr J Microbiol Res 6(37):6600–6605
Igiehon NO, Babalola OO (2017) Biofertilizers and sustainable agriculture: exploring arbuscular mycorrhizal fungi. Appl Microbiol Biotechnol 101(12):4871–4881
Fatima F, Ahmad MM, Verma SR, Pathak N (2021) Relevance of phosphate solubilizing microbes in sustainable crop production: a review. Int J Environ Scie Technol 1–14
Etesami H, Alikhani HA (2019) Halotolerant plant growth-promoting fungi and bacteria as an alternative strategy for improving nutrient availability to salinity-stressed crop plants. Saline soil-based agriculture by halotolerant microorganisms. Springer, pp 103–146
Rajeswari S, Umamaheswari S, Arvind Prasanth D, Rajamanikandan KCP (2016) Bioactive potential of endophytic fungi Aspergillus flavus (SS03) against clinical isolates. Int J Pharm Pharm Sci 8(9):37–40
Kriaa M, Hammami I, Sahnoun M, Azebou MC, Triki MA, Kammoun R (2015) Biocontrol of tomato plant diseases caused by Fusarium solani using a new isolated Aspergillus tubingensis CTM 507 glucose oxidase. CR Biol 338(10):666–677
Crutcher FK, Liu J, Puckhaber LS, Stipanovic RD, Duke SE, Bell AA, Nichols RL (2014) Conversion of fusaric acid to fusarinol by Aspergillus tubingensis: a detoxification reaction. J Chem Ecol 40(1):84–89
Qiu M, Xie RS, Shi Y, Zhang H, Chen HM (2010) Isolation and identification of two flavonoid-producing endophytic fungi from Ginkgo biloba L. Ann Microbiol 60(1):143–150
Bos L (1978) Symptoms of virus diseases in plants Third edition Centre for Agricultural Publishing and Documentation. Wageningen, the Netherlands
Ismail I, Hussain A, Mehmood A, Qadir M, Husna H, Iqbal A, Hamayun M, Khan N (2020) Thermal stress alleviating potential of endophytic fungus rhizopus oryzae inoculated to sunflower (Helianthus annuus L.) and soybean (Glycine max L.). Pak J Bot 52(5):1857–1865
Mousa W. (2016) Natural products and molecular genetics underlying the antifungal activity of endophytic microbes. 2016.
Patrzylas P, Wojcik M, and Skorzynska-Polit E (2015) Changes in the level of non-enzymatic antioxidants in leaves of Phaseolus coccineus (L.) plants under heavy metal stress. BioTechnologia. J Biotechnol Comput Biol Bionanotechnol 2015;96(1)
Wang M, Sun Y, Sun G, Liu X, Zhai L, Shen Q, Guo S (2015) Water balance altered in cucumber plants infected with Fusarium oxysporum f. sp. cucumerinum. Sci Rep 5(1):1–7
Ertunç F (2020) Physiology of virus-infected plants. Applied Plant Virology. Elsevier, pp 199–205
Bauriegel E, Herppich WB (2014) Hyperspectral and chlorophyll fluorescence imaging for early detection of plant diseases, with special reference to Fusarium spec. infections on wheat. Agriculture 4(1):32–57
Maust BE, Espadas F, Talavera C, Aguilar M, Santamaría JM, Oropeza C (2003) Changes in carbohydrate metabolism in coconut palms infected with the lethal yellowing phytoplasma. Phytopathol 93(8):976–981
Chrpová J, Orsák M, Martinek P, Lachman J, Trávníčková M (2021) Potential role and involvement of antioxidants and other secondary metabolites of wheat in the infection process and resistance to Fusarium spp. Agron 11(11):2235
Rao GS, Reddy NNR, Surekha C (2015) Induction of plant systemic resistance in Legumes cajanus cajan, Vigna radiata, Vigna mungo against plant pathogens Fusarium oxysporum and alternaria alternata–a Trichoderma viride mediated reprogramming of plant defense mechanism. Int J Recent Sci Res 6:4270–4280
Mikulic-Petkovsek M, Schmitzer V, Jakopic J, Cunja V, Veberic R, Munda A, Stampar F (2013) Phenolic compounds as defence response of pepper fruits to Colletotrichum coccodes. Physiol Mol Plant Pathol 84:138–145
Pandey S, Giri VP, Tripathi A, Kumari M, Narayan S, Bhattacharya A, Mishra A (2020) Early blight disease management by herbal nanoemulsion in Solanum lycopersicum with bio-protective manner. Ind Crops Prod 150:112421
Ashry NA, Mohamed HI (2011) Impact of secondary metabolites and related enzymes in flax resistance and or susceptibility to powdery mildew. World J Agric Sci 7(1):78–85
Jiang S, Han S, He D, Cao G, Fang K, Xiao X, Wan X (2019) The accumulation of phenolic compounds and increased activities of related enzymes contribute to early defense against walnut blight. Physiol Mol Plant Pathol 108:101433
Gill SS, Tuteja N (2010) Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem 48(12):909–930
Acknowledgements
The authors would like to thank the Botany and Microbiology Department, Faculty of Science, Al-Azhar University for promoting this research.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
Conceptualization: A. M. A. and M. S. A. Methodology: A. M. A., M.S.S, and M. S. A. Software: A. M. A and M. S. A. Formal analysis: A. M. A. and M. S. A. Investigation: A. M. A. and M. S. A. Resources: A.M.A. and M.S.A. Data curation: A.M.A. and M.S.A. Writing original draft preparation: A. M. A., M.S.S, and M. S. A. Writing review and editing: A. M. A., M.S.S, and M. S. A. Supervision: A. M. A. and M. S. A. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
All authors approved.
Consent for publication
All authors agree for publication.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Attia, M.S., Salem, M.S. & Abdelaziz, A.M. Endophytic fungi Aspergillus spp. reduce fusarial wilt disease severity, enhance growth, metabolism and stimulate the plant defense system in pepper plants. Biomass Conv. Bioref. 14, 16603–16613 (2024). https://doi.org/10.1007/s13399-022-03607-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-022-03607-6