Abstract
Wood bark is a source of extractives with potential for valorization based on their chemical functionalities. In this study, Acacia mangium bark was extracted with distilled water. Then bark extract solutions in distilled water at various concentrations (5%, 10%, 15%, and 20% w/w) were vacuum impregnated into rubberwood. The color of the impregnated rubberwood was observed. The retention of bark extract in rubberwood and weight percent gain (WPG) from it were calculated. The morphology, dimensional stability, resistance to fungal decay, and subterranean termite resistance were determined for impregnated rubberwood samples. It was found that a higher concentration of bark extract gave higher retention and WPG in vacuum-impregnated rubberwood. The color of impregnated wood got darker with the concentration of bark extract, from light brown to red brown and dark brown. The bark extract penetrated all the way to the center (in thickness direction) of the wood and remained in lumens of vessels, parenchyma, and ray cells. The bark extract can improve dimensional stability of rubberwood. Weight loss caused by the fungi Trametes versicolor and Gloeophyllum striatum decreased with retention of bark extract in the impregnated wood. The bark extract may be toxic to termites (Coptotermes gestroi) that consumed impregnated wood: termites died during the third and fourth weeks after they attacked wood impregnated with bark extract at 15% or 20% concentration.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Acacia mangium is an economic fast-growing tropical plantation tree that has been introduced to many countries, including Indonesia, Malaysia, China, Philippines, Thailand, Vietnam, Laos, India, and Sri Lanka [1]. Its desirable properties are good wood quality, rapid growth, and tolerance of a variety of soils and environments. Acacia mangium wood is useful for a variety of purposes and is used in furniture, cabinets, floors, particleboard, plywood, veneer, fence posts, firewood, and charcoal. Acacia mangium bark has not been utilized properly. Currently, the bark is incinerated or landfilled. Mainly, it is used as a low-cost resource of energy in saw mills. Acacia mangium bark is composed of holocellulose (36.2%), cellulose (22.4%), lignin (14.7%), and hot water-soluble extractives (17.4%) [2] and can serve as a renewable source of chemicals. Because of restrictions on the use of synthetic products, other environmentally friendly concepts, and growth of renewable material consumption [3], many studies have investigated potential value added products from bark extracts. Ecofriendly solvents have efficiently extracted antioxidant compounds from beech bark, which opens up opportunities for the utilization of bark extracts in wood technology in the future [4]. Acacia mangium bark extracts could possibly partially replace commercial phenol formaldehyde (PF) adhesives (by up to 90%) in the plywood-manufacturing industry [2]. Acacia confuse bark extracts can be used as antioxidant agents [5]. The bark of Eucalyptus sideroxylon contains extractives with potential for valorization based on chemical functionalities and bioactivities [6]. Mimosa and quebracho bark extracts can be used as wood preservatives against wood decaying fungi in indoor applications [7]. Tannin extracts isolated from Acacia crassicarpa and Acacia mearnsii barks affected termite feeding as deterrent [8]. Mimosa bark extracts improved thickness swelling of and formaldehyde emissions from particleboard [9].
Rubberwood from rubber trees is white to pale cream colored and sometimes has a pinkish tint. Its color changes to yellowish after seasoning [10]. It is mainly used in furniture. In addition, it also suitable for production of wood composite panels, for example particleboard and medium-density fiberboard [1, 11]. Rubberwood contains 36–43% alpha cellulose, 16–22% lignin, and 56–74% holocellulose [12, 13]. Fresh rubberwood also has relatively high contents of starch (8–10%) and free sugars (1–2%) [10, 14] and a low content of phenolic compounds (0.1%) [15]. As a result, the natural durability of rubberwood is low and it is prone to suffer attacks by fungi and insects [11, 16].
To address these problems, it is necessary to perform preservative treatment of rubberwood to prevent infestation by biodeteriorating agents. Several methods are used for protective treatment, such as dipping, dip diffusion, pressure treatment, vacuum pressure, and double vacuum processes. Protection of rubberwood is achieved mainly through use of chemical wood preservatives. An economical alternative in industrial-scale treatment of rubberwood is provided by boron compounds [11]. The esterification of rubberwood with hexanoylchloride, decanoyl chloride, and tetra-decanoyl chloride exhibited very good resistance to brown-rot (Polyporus meliae) and white-rot (Coriolus versicolor) fungi [16]. Phenylisothiocyanate-modified rubberwood showed good dimensional stability and was very resistant to decay fungi [17]. Rubberwood impregnated with zinc oxide and copper oxide dispersion in propylene glycol effectively resisted white-rot (Trametes hirsuta) and brown-rot (Polyporus meliae) decay and showed some reduction in moisture adsorption with lower equilibrium moisture content than untreated wood [18]. Rubberwood impregnated with micronized copper azole at minimum retention of 2.8 ± 0.3 kg/m3 by vacuum pressure full-cell process was found to be comparable to that subjected to chromated copper arsenate treatment at 15.1 ± 2.3 kg/m3 [19]. The health hazards and environmental pollution caused by chemical wood preservatives require alternative environmentally friendly approaches to wood protection. The application of tapioca starch coating incorporated with peppermint oil and lime oil at 1:3 ratio on rubberwood box surfaces suppressed mycelial growth and spore formation with minimum water absorption, stable wood composition, and structure after transportation [20]. Anise oil and garlic oil (10–100 μL mL−1) influenced the growth of Aspergillus niger on rubberwood surfaces [21]. Wettability of thermally modified rubberwood was enhanced from that of unmodified wood. It had higher contact angle, and the round-shaped water droplets on the surface of modified wood only slowly absorbed into wood [22].
According to the reviewed positive effects of bark extracts applied to wood, the aim of this study was the valorization of Acacia mangium bark extracts as wood preservatives. Water is generally recognized as a safe and low-cost solvent and was selected as solvent for the bark extraction. The chemical components in bark extracts were identified by gas chromatography–mass spectrometry analysis. Then various concentrations of the bark extracts were prepared in water. This may be the first report on vacuum impregnating rubberwood with Acacia mangium bark extracts. The morphology of vacuum-impregnated wood was studied. The effects of Acacia mangium bark extracts on dimensional stability, termite resistance, and fungal resistance of vacuum-impregnated wood were evaluated.
2 Materials and methods
2.1 Acacia mangium bark extraction
The bark of 12 years old Acacia mangium was obtained from Suratthani Silvicultural Research Station in Suratthani Province, Thailand. The bark was ground to particles passing through 40 mesh size and then extracted with distilled water (1:15 w/v) at 80 °C for 4 h. Approximately 100 g of ground bark and 1500 mL of distilled water were used for each extraction. At first, the solution was screened through Whatman filter paper no. 1 and further filtered through sintered glass (40–100 μm). The extract solution was then evaporated to retain its solids, at 50 °C in an oven until constant weight. The extraction yield was 14.49 (± 0.32)% of the initial dry weight of the bark. The solid concentrated extract from the bark of Acacia mangium was dissolved in distilled water at 4 concentration levels: 5%, 10%, 15%, and 20% by weight.
2.2 Preparation of wood specimens
Rubberwood logs were taken from a local saw mill in Suratthani Province, Thailand, and then cut into small-sized specimens. The specimens for dimensional stability test, termite resistance test, and fungal decay resistance test were of 20 (R) × 20 (T) × 20 (L) mm3 size, in which R, T, and L stands for radial, tangential, and longitudinal directions of the original log. All specimens were oven-dried at 105 ± 2 °C until constant weight. Then they were conditioned at 20 ± 2 °C and 65 ± 3% relative humidity until stable weight. The weights and dimensions of samples in both dry and equilibrium states were measured, before the subsequent vacuum impregnation with Acacia mangium bark extracts.
2.3 Acacia mangium bark extract impregnation
Rubberwood specimens were vacuum impregnated with the diluted bark extracts according to ASTM D1413-07e1 [23]. They were placed in desiccators and the air cooped up in the wood cells was removed with a 93.32-kPa relative vacuum for 30 min. Then a diluted bark extract solution was filled into the desiccator and the vacuum was held at 93.32 kPa for 1 h. After that, the solution was sucked from the desiccator and excess solution on the surfaces of impregnated wood was allowed to dry under 93.32 kPa vacuum level for 10 min. Finally, the impregnated wood samples were taken out from the desiccator. The weights of the samples were determined after impregnation, and they were oven-dried at 105 ± 2 °C until constant weight. Then the impregnated samples were conditioned at 20 ± 2 °C and 65 ± 3% relative humidity until stable weight. The weights and dimensions of samples in both dry and equilibrium conditions were measured. The retention of bark extract (kg/m3) was calculated as follows:
In equation, M0 is the stable weight of wood at 20 ± 2 °C and 65 ± 3% relative humidity before vacuum impregnation (g), Mf is the weight of impregnated wood before drying at 105 ± 2 °C (g), C is the concentration of Acacia mangium bark extract solution (%), and V is the volume of wood before vacuum impregnation (cm3).
The weight percent gain (WPG) of wood was determined as follows:
where W1 and W2 are the oven-dried weights (g) of sample before and after the vacuum impregnation, respectively.
2.4 GC-MS analysis of Acacia mangium bark extracts
Analysis of chemical composition of the Acacia mangium bark extract was carried out using the GC-MS apparatus at the Scientific Equipment Center, Prince of Songkla University, Songkhla, Thailand. The Trace GC Ultra-ISQ Mass Spectrometer (Thermo Scientific, Austin, TX, USA) was connected with a direct capillary column AT-Wax MS (30 m × 0.25 mm, × 0.25 μm film thickness). Fine powder of Acacia mangium bark extract was dissolved in methanol and then filtered through a 25-mm polypropylene disposable syringe filter and a nylon filter before injecting in the GC-MS. The column temperature was primarily held at 50 °C for 10 min. After that, the temperature was increased at 5 °C/min rate to 200 °C, held for 2 min, and then increased to 250 °C at 10 °C/min and held for 1 min. The injector and detector temperatures were 250 °C. Helium was used as the carrier gas at a constant 1.5 mL/min flow rate. The solvent delay was 2 min and diluted samples of 1 mL were injected automatically in the splitless mode. EI mass spectra were collected at 70 eV ionization voltage over the 35–500 m/z range in full scan mode. The ion source and transfer line temperatures were set at 240 and 250 °C, respectively. The components were identified by comparison of their retention times and mass spectra with those in WIELY 09 database.
2.5 FTIR spectral analysis
Chemical analyses of surfaces of rubberwood and rubberwood vacuum impregnated with 10% Acacia mangium bark extract were performed using a Fourier transform infrared spectrometer (FTIR) (VERTEX 70, Bruker, Germany) in an ATR-FTIR mode. The spectra were recorded with a resolution of 4 cm−1 from 400 to 4000 cm−1 and were analyzed using the Spectrum software.
2.6 Dimensional stability test
The dimensional stability of impregnated wood was calculated from dimension changes along all edges from dry condition (105 ±2 °C) to equilibrium condition (20 ± 2 °C and 65 ± 3% relative humidity) by using the anti-swelling efficiency (ASE) method as reported previously [24]. ASE was calculated as follows.
Here, Su and Sm are the dimensional swelling of unmodified wood (%) and dimensional swelling of impregnated wood (%), respectively [25].
The moisture exclusion efficiency (MEE, %) is based on the equilibrium moisture content of impregnated wood (EMCm, %) compared with that of unmodified wood (EMCu, %). MEE was calculated as follows:
Ten replicates were tested for each treatment.
2.7 Fungal decay resistance test
Fungal decay resistances of rubberwood and rubberwood impregnated with Acacia mangium bark extract, against the white-rot fungus, Trametes versicolor, and the brown-rot fungus, Gloeophyllum striatum, were examined in accordance with ASTM D1413-07e1 standard [23] with some modifications. Soil block culture bottles were prepared using 90 g of sieved oven-dried soil filled in 8-oz culture bottles. Sterile water was added to maintain 130% of the water-holding capacity of soil in the bottle during the test. Rubberwood feeder strips (35 mm × 28 mm × 3 mm in tangential, radial, and longitudinal directions) were placed on the soil. The prepared bottles were then autoclaved at 121 °C for 30 min. After cooling down, the feeder strips placed in the sterilized bottles were inoculated with the fungal inoculum sections from freshly grown cultures. The bottle was incubated at room temperature for 2 weeks until mycelia of the decay fungus completely colonized the feeder strip. The dried wood specimens (dimension of 20 (R) × 20 (T) × 20 (L) mm3, in which R, T, and L stand for radial, tangential and longitudinal directions, respectively), steam-sterilized at 100 °C for 20 min, were placed on the feeder strips in contact with fungal mycelia. The bottles containing specimens were incubated at room temperature for 12 weeks. After incubation, the surface mycelia were removed and the damaged wood specimens were dried at 105 ± 2 °C until stable weight. Test results are expressed as mass loss percentages of wood specimens during the decay test. Ten replicates were used for each treatment group.
2.8 Termite resistance test
Termite resistance tests of wood and wood impregnated with Acacia mangium bark extract were carried out in the laboratory using the method described in ASTM D3345-17 [26] with slight modifications. Aseptic containers with 200 g of sand were filled with sufficient distilled water until the saturation point of sand. A total of 1 ± 0.05 g of Coptotermes gestroi including 90% workers and 10% soldiers from strong laboratory cultures were introduced into each test container. Then a wood specimen was placed at the center of a test container. The assembled containers were maintained at room temperature for 4 weeks in the dark. At the end of the first and fourth weeks, the containers were observed and the positions of the termites in the container, the existence of tunneling and termite mortality were recorded. After 4 weeks, the tested wood specimens were removed, cleaned, and visually rated using the rating system in ASTM D3345-17 [26]. The mass loss of each sample due to termite attack was calculated. Tests were performed in ten replicates and averages are reported
3 Results and discussion
3.1 Chemical components in Acacia mangium bark extract
In a GC-MS analysis of Acacia mangium bark extract, 70 chemical components were identified. The 30 chemical compounds (72.53%) specified by name are presented in Table 1, while 40 chemical compounds (27.47%) could not be identified by name based on their retention times and mass spectra with reference to the WIELY 09 database. The main constituents were 1,3,5-cycloheptatrienemethanol (14.93%), nitromethane (12.49%), 1,2,3-propanetrio (9.23%), hexadecanoic acid, ethyl ester (8.62%), acetol (2.61%), phenol, and 2-methoxy (2.05%) and methyl glycolate (1.78%). Gabr et al. [27] found that catechin is commonly present in the acacia bark. Several components of Acacia mangium bark extracts have been used as fungicides and insecticides. Nitromethane is a hazardous component in insecticides [28]. Li and Kubo [29] reported that benzaldehyde can be used as an insect (Sacrophaga neobelliaria) control agent.
3.2 The penetration of diluted Acacia mangium bark extracts into rubberwood
The penetration of diluted Acacia mangium barks extracts into rubberwood is reported in terms of solution uptake, retention, and WPG (Table 2). Acacia mangium extract solution uptake varied in the range from 132.34 to 137.17 kg/m3. Both retention and WPG of Acacia mangium bark extracts in wood depended on the concentration of diluted bark extract. Retention and WPG were both proportional to the concentration of extract.
A higher concentration bark extract gave higher retention in wood than a lower concentration. Visual inspection revealed that the bark extract solution changed the color of rubberwood from cream shade to red brown shade, depending on the concentration (Fig. 1).
Possibly a 10% concentration of bark extract was near optimal, because most of the bark extract penetrated into the wood instead of caking on the outer surfaces of wood specimens. Tondi et al. [30] found that the impregnation of tannin solution into the wood with a highly concentrated solution released a high amount of tannin content in the wood structure, but the high viscosity prevented deep penetration. When rubberwood was impregnated with Acacia mangium bark extracts at 15 and 20% concentrations, most of the extracts covered the outer surfaces of wood and were easily wiped off. A microscope investigation showed that the bark extract solution penetrated longitudinally along large and easily accessible vessels and across the radial direction trough ray cells. Then the red brown or dark brown bark extracts were deposited in the lumens. In the middle of the wood specimen (Fig. 2), not all vessels were entirely filled but only the larger vessels were impregnated. Possibly the smaller ones were plugged by the bark extract.
3.3 FTIR spectra
The FTIR spectra of the Acacia mangium bark extract, rubberwood, and rubberwood vacuum impregnated with 10% Acacia mangium bark extract are shown in Fig. 3. The spectrum of rubberwood is similar to the spectrum of rubberwood vacuum impregnated with 10% Acacia mangium bark extract, indicating similar functional groups. A broad band at 3200–3500 cm−1 was seen, which corresponds to −OH stretching vibrations in phenolic and aliphatic structures. Small peaks at 2920 and 2853 cm−1 were generated from –CH stretching vibrations in aromatic methoxyl groups and in methyl and methylene groups of side chains [31]. The peaks between 1400 and 2000 cm−1 show the aromatic nature of the structure [32]. Bands at 1505 cm−1 are attributed to aromatic squeal vibration, 1452 cm−1 corresponding to −CH deformation, and aromatic ring vibration [31]. Prior studies have reported that IR band around 1022 cm−1 represents cellulose [33]. Peaks at wavelengths 500–600 cm−1 are for aromatic –CH stretching vibrations that are intense in rubberwood. The lower intensities of IR bands at 1022 cm−1 and 500–600 cm−1 of Acacia mangium bark extract-impregnated rubberwood indicate that some water-soluble low-molecular weight carbohydrates were dissolved and removed by water during the vacuum impregnation. The higher intensity of IR band at 3200–3500 cm−1 is attributed to phenolic compounds in Acacia mangium bark extract-impregnated rubberwood. These may enhance the dimensional stability and fungal decay resistance of impregnated rubberwood. However, some bands are more intense on impregnated rubberwood, as indicated at the wave numbers 1600 and 841 cm−1, due to some bark extracts deposited in rubberwood. The results confirmed that the bark extracts were impregnated into the rubberwood by using vacuum impregnation method.
3.4 Dimensional stability of rubberwood and Acacia mangium bark extract-treated rubberwood
Dimensional stabilities of rubberwood and Acacia mangium bark extract-treated rubberwood are reported in terms of moisture exclusion efficiency and anti-swelling efficiency in Table 2. The results show that Acacia mangium bark extract can improve the dimensional stability of rubberwood. It is possible that dry bark extract deposited in the lumens of wood cells slowed down the sorption of moisture into the wood. However, the highest concentration bark extract (20%) did not give the best dimensional stability of treated wood. The dimensional stabilities of rubberwood treated with Acacia mangium bark extract at 5%, 15%, and 20% concentrations were similar (P < 0.05). The rubberwood treated with Acacia mangium bark extract at 10% concentration showed the highest ASE and MEE. ASE in radial, tangential, and longitudinal directions were 89.02 ± 15.37%, 86.79 ± 15.32%, and 92.32 ± 7.53%, respectively. The MEE was 58.09 ± 1.76%.
This agrees with a previous report, which found that dimensional stability of wood was positively correlated with chemical (resin) concentration, but the largest effects on increasing stability per unit of chemical were at the lower concentration levels [34]. It was found that only lumen filling with rosin could not improve dimensional stability of wood, but the penetration of rosin in wood cell walls improved the ASE of wood [25]. In this study, ASE of treated woods was improved, so it is possible that Acacia mangium bark extracts penetrated wood cell walls to thereby enhance the dimensional stability of rubberwood. The bark extracts reduced hygroscopicity and enhanced dimensional stability of wood, because of reactions that mainly occurred between the fibers in amorphous regions of the cell wall [35]. However, chemical bonding between the bark extract and chemical components of wood cell walls, such as cellulose, hemicellulose, and lignin, is difficult to describe, so this is a potential topic for further studies. Prior research has found that condensed tannin from Pinus radiata bark improved the hydrophobicity of beech wood [9].
3.5 Fungal decay resistance
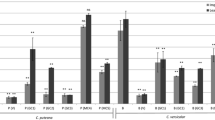
Mean mass losses of rubberwood specimens caused by T. versicolor and G. striatum are given in Fig. 4. The results indicate that rubberwood impregnated with Acacia mangium bark extracts had remarkably improved fungal decay resistance against the white-rot and brown-rot fungi, when compared to untreated rubberwood.
Rubberwood had an average mass loss of 28.26% or 26.36% after exposure to T. versicolor or G. striatum for 12 weeks. This indicates that damage from the white-rot fungus, T. versicolor, was more severe than that caused by the brown-rot fungus G. striatum. White-rot fungi can degrade lignin and cellulose components [36], while brown-rot fungi selectively attack cellulose and hemicelluloses, with little effect on lignin [37]. It was found that increased bark extract retention resulted in a significant reduction in mass loss. The highest concentration of Acacia mangium bark extract (20%) with 47.81 kg/m3 extract retention reduced mass loss of rubberwood from 28.26 to 9.23% and from 26.36 to 5.18% on exposure to T. versicolor and G. striatum for 12 weeks, respectively. The results of the present work agree with Rosdiana et al. [38], where Acacia mangium bark extracts were more effective against brown-rot fungi than white-rot fungi. The antifungal activity was attributed to phenolic compounds in the Acacia mangium bark extracts. Prior research has also found that Acacia mollissima bark extract-treated Scotch pine wood with 59.3 kg/m3 retention level had only 4.4% mean weight loss from exposure to T. versicolor for 16 weeks [7].
3.6 Termite resistance
Table 3 summarizes mean mass loss, visual rating, and pattern of termite attack on a tangential section of bark extract-impregnated rubberwood. According to statistical analyses, rubberwood impregnated with bark extract at concentrations of 5%, 15%, and 20% did not have significantly reduced mass loss compared with untreated rubberwood. However, impregnation with bark extract at 10% concentration showed a significant reduction in mass loss, with the lowest mass loss of 20.50%. It was found that the visual rating depended on the retention of bark extract in rubberwood. The highest retention of bark extract in rubberwood was when impregnated with 20% concentration, and this case was only rated as 8.6 with a light attack by termites. Rubberwood samples impregnated with 10 and 15% concentrations were rated as 6.9 and 6.0, respectively, indicating moderate attacks by termites. The samples impregnated with 5% concentration were rated as 3.6 for heavy attack by termites. The visual rating scale of termite attack on rubberwood was 0.8, which means that these samples suffered from a strong attack. The bark extract may be toxic to termites (Coptotermes gestroi) that consumed impregnated wood. It was found that termites died during the third and fourth weeks after they attacked wood impregnated with bark extracts at 15% or 20% concentration. It might be concluded that bark extracts covering the outer surfaces of rubberwood annoyed the termites. Removing the bark extracts from the outer surfaces of rubberwood might make it easier for termite attacks to penetrate into wood. The microscopic investigation showed that bark extract at 10% concentration had deeply penetrated into the rubberwood specimens, while the bark extracts at 15 and 20% concentrations mostly covered outer surfaces of the rubberwood specimens. When termites entered into wood impregnated with bark extract at 10% concentration, the good penetration and distribution of the extract helped reduce mass loss. The bark extract may be toxic to termites (Coptotermes gestroi) that consumed impregnated wood: termites died during the third and fourth weeks after they attacked wood impregnated with bark extracts at 15% or 20% concentrations. Several studies have reported that bark extracts affect termite infestations on wood, such as extracts from Lawsania inermis bark [39], Acacia crassicarpa and Acacia mearnsii bark [8], mimosa (Acacia mollisima Willd.) bark, and quebracho (Shinopsis lorentzii Gresep.) bark [40].
4 Conclusions
This study evaluated the possibility of vacuum impregnating rubberwood with Acacia mangium bark extracts. Distilled water as an ecofriendly solvent was used in the extraction and in preparation of solutions of the bark extracts. The results show a positive correlation between the concentration of bark extract solution and the retention and WPG of bark extracts in rubberwood. With vacuum impregnation, aqueous solutions of Acacia mangium bark extracts penetrated deeply into the center of wood samples and remained in the lumens of vessels, parenchyma, and ray cells. In addition, the rubberwood components such as water-soluble low molecular weight carbohydrates were dissolved and removed by water during the vacuum impregnation. This may have improved the ASE and MEE of impregnated wood. The Acacia mangium bark extracts can enhance fungal resistance and termite resistance of rubberwood. The bark extract may be toxic to fungi and termites because of chemical components in the bark extracts that have been used as fungicides and insecticides, such as nitromethane and benzaldehyde. Further research is needed to determine whether they are effective alone or act synergistically with other bark components. However, the application of pressure impregnation process and bonding of the Acacia mangium bark extracts with wood are important aspects remaining for future studies.
References
Hegde M, Palanisamy K, Yi JS (2013) Acacia mangium Willd.-a fast growing tree for tropical plantation. J For Sci 29:1–14. https://doi.org/10.7747/JFS.2013.29.1.1
Hoong YB, Paridah MT, Jalaluddin H, Chuah LA (2011) A new source of natural adhesive: Acacia mangium bark extracts co-polymerized with phenol-formaldehyde (PF) for bonding Mempisang (Annonaceae spp.). Int J Adhes Adhes 31:164–167. https://doi.org/10.1016/j.ijadhadh.2010.12.002
Jansone Z, Muizniece I, Blumberga D (2017) Analysis of wood bark use opportunities. Energy Procedia 128:268–274. https://doi.org/10.1016/j.egypro.2017.09.070
Hofmann T, Nebehaj E, Bányai ÉS, Albert L (2015) Antioxidant capacity and total phenol content of beech (Fagus sylvatica L.) bark extracts. Ind Crop Prod 77:375–381. https://doi.org/10.1016/j.indcrop.2015.09.008
Tung YT, Wu JH, Huang CY, Kuo YH, Chang ST (2009) Antioxidant activities and phytochemical characteristics of extracts from Acacia confusa bark. Bioresour Technol 100:509–514. https://doi.org/10.1016/j.biortech.2008.01.001
Miranda I, Lima L, Quiho T, Knapic S, Pereira H (2016) The bark of Ecalyptus sideroxylon as a source of phenolic extracts with anti-oxidant properties. Ind Crop Prod 82:81–87. https://doi.org/10.1016/j.indcrop.2015.12.003
Tascioglu C, Yalcin M, Sen S, Akcay C (2013) Antifungal properties of some plant extracts used as wood preservatives. Int Biodeterior Biodegradation 85:23–28. https://doi.org/10.1016/j.ibiod.2013.06.004
Ismayati M, Nakagawa IA, Ohi H, Tarmadi D (2014) In: Forschler BT (ed) Efficacy of tannin extracts isolated from acacia bark waste against the subterranean termite Coptotermes gestroi Wasmann. Proceedings of the 10th Pacific-Termite Research Group Conference, Kuala Lumpur, Malaysia
Nemli G, Kırcı H, Temiz A (2004) Influence of impregnating wood particles with mimosa bark extract on some properties of particleboard. Ind Crop Prod 20:339–344. https://doi.org/10.1016/j.indcrop.2003.11.006
Killmann W, Hong LT (2000) Rubberwood-the success of an agricultural by-product. Unasylva 51(201):66–72
Teoh YP, Don MM, Ujang S (2011) Assessment of the properties, utilization, and preservation of rubberwood (Hevea brasiliensis): a case study in Malaysia. J Wood Sci 57:255–266. https://doi.org/10.1007/s10086-011-1173-2
Deraman M, Zakaria S, Husin M, Aziz AA, Ramli R, Mokhtar A, Yusof MNM, Sahri MH (1999) X-ray diffraction studies on fiber of oil palm empty fruit bunch and rubberwood for medium-density fiberboard. J Mater Sci Lett 18:249–253. https://doi.org/10.1023/A:1006642114056
Severo ETD, Oliveira EFJ, Sansıgolo CA, Rocha CD, Calonego FW (2003) Properties of juvenile and mature woods of Hevea brasiliensis untapped and with tapping panels. Eur J Wood Wood Prod 71:815–818. https://doi.org/10.1007/s00107-013-0731-2
Kadir AA, Sudin R (1989) Carbohydrates in rubberwood (Hevea brasiliensis Muell.). Holzforschung 43:173–178. https://doi.org/10.1515/hfsg.1989.43.3.173
Simatupang MH, Schmitt U, Kasim A (1994) Wood extractives of rubberwood (Hevea brasiliensis) and their influences on the setting of the inorganic binder in gypsum-bonded particle boards. J Trop For Sci 6(3):269–285
Jayashree PKK, Nagaveni HC, Mahadevan KM (2011) Fungal resistance of rubber wood modified by fatty acid chlorides. Int Biodeterior Biodegradation 65:890–895. https://doi.org/10.1016/j.ibiod.2011.05.001
Pandey KK, Jayashree, Nagaveni HC (2009) Study of dimensional stability, decay resistance, and light stability of phenylisothiocyanate modified rubberwood. Bioresources 4(1):257–267. https://doi.org/10.15376/BIORES.4.1.257-267
Nair S, Pandey KK, Giridhar BN, Vijayalakshmi G (2017) Decay resistance of rubberwood (Hevea brasiliensis) impregnated with ZnO and CuO nanoparticles dispersed in propylene glycol. Int Biodeterior Biodegradation 122:100–106. https://doi.org/10.1016/j.ibiod.2017.05.008
Shukla SR, Zhang J, Kamdemc DP (2019) Pressure treatment of rubberwood (Heavea brasiliensis) with waterborne micronized copper azole: effects on retention, copper leaching, decay resistance and mechanical properties. Constr Build Mater 216:576–587. https://doi.org/10.1016/j.conbuildmat.2019.05.013
Owolabi IO, Songsamoe S, Khunjan K, Matan N (2021) Effect of tapioca starch coated-rubberwood box incorporated with essential oils on the postharvest ripening and quality control of mangosteen during transportation. Food Control 126:108007. https://doi.org/10.1016/j.foodcont.2021.108007
Na M, Ni M, Ketsa S (2012) Effect of heat curing on antifungal activities of anise oil and garlic oil against Aspergillus niger on rubberwood. Int Biodeterior Biodegradation 75:150–157. https://doi.org/10.1016/j.ibiod.2012.03.012
Bakar BFA, Hiziroglu S, Tahir PM (2013) Properties of some thermally modified wood species. Mater Des 43:348–355. https://doi.org/10.1016/j.matdes.2012.06.054
ASTM D1413-07e1 (2017) Standard test method for wood preservatives by laboratory soil-block cultures (withdrawn 2016). ASTM International, West Conshohocken, PA
Thybring EE (2013) The decay resistance of modified wood influenced by moisture exclusion and swelling reduction. Int Biodeterior Biodegradation 82:87–95. https://doi.org/10.1016/j.ibiod.2013.02.004
Dong Y, Yan Y, Wang K, Li J, Zhang S, Xia C, Shi SQ, Cai L (2016) Improvement of water resistance, dimensional stability, and mechanical properties of poplar wood by rosin impregnation. Eur J Wood Wood Prod 74:177–184. https://doi.org/10.1007/s00107-015-0998-6
ASTM D3345-17 (2017) Standard test method for laboratory evaluation of solid wood for resistance to termites. ASTM International, West Conshohocken, PA
Gabr S, Nikles S, Wenzing EMP, Woelkart KA, Hathout RM, Ahmady SE, Motaal AA, Singab A, Bauer R (2018) Characterization and optimization of phenolics extracts from Acacia species in relevance to their anti-inflammatory activity. Biochem Syst Ecol 78:21–30. https://doi.org/10.1016/j.bse.2018.03.001
Yamamoto I, Casida JE (1999) Nicotinoid insecticides and the nicotinic acetylcholine receptor. Springer, Tokyo, New York
Li W, Kubo I (2004) QSAR and kinetics of the inhibition of benzaldehyde derivatives against Sarcophaga neobellaria phenoloxidase. Bioorg Med Chem 12:701–713. https://doi.org/10.1016/j.bmc.2003.11.014
Tondi G, Thevenon MF, Mies B, Standfest G, Petutschnigg A, Wieland S (2003) Impregnation of Scots pine and beech with tannin solution: effect of viscosity and wood anatomy in wood infiltration. Wood Sci Technol 47:615–626. https://doi.org/10.1007/s00226-012-0524-5
Chupin L, Motillon C, Bouhtoury FCE, Pizzi A, Charrier B (2013) Characterization of maritime (Pinus pinaster) bark tannins extracted under different conditions by spectroscopic methods, FTIR and HPLC. Ind Crop Prod 49:897–903. https://doi.org/10.1016/j.indcrop.2013.06.045
Soto R, Freer J, Baeza J (2005) Evidence of chemical reactions between di- and poly-glycidyl ether resins and tannins isolated from Pinus radiata D. Don bark. Bioresour Technol 96(1):95–101. https://doi.org/10.1016/j.biortech.2003.05.006
Can A, Huseyin S, Baki H (2018) Fungal inhibition and chemical characterization of wood treated with novel polystyrene-soybean oil copolymer containing silver nanoparticles. Int Biodeterior Biodegradation 133:210–215. https://doi.org/10.1016/j.ibiod.2018.06.022
Gabrielli CP, Frederick AK (2010) Phenol–formaldehyde impregnation of densified wood for improved dimensional stability. Wood Sci Technol 44:95–104. https://doi.org/10.1007/s00226-009-0253-6
Chen H, Miao X, Feng Z, Pu J (2014) In situ polymerization of phenolic methylolurea in cell wall and induction of pulse-pressure impregnation on green wood. Ind Eng Chem 53:9721–9727. https://doi.org/10.1021/ie5006349
Eaton RA, Hale MD (1993) Wood: decay, pests, and protection. Chapman and Hall, London
Carll CG, Highley TL (1999) Decay of wood and wood-based products above ground in buildings. J Test Eval 27(2):150–158. https://doi.org/10.1520/JTE12045J
Rosdiana NA, Dumarcay A, Gerardin C, Chapuis H, Medina FJS, Sari RK, Syafii W, Gelhaye E, Raharivelomanana P, Mohammed R, Gerardin P (2017) Characterization of bark extractives of different industrial Indonesian wood species for potential valorization. Ind Crop Prod 108:121–127. https://doi.org/10.1016/j.indcrop.2017.06.034
Adedeji GA, Olukayode YO, Taiwo OE (2017) Quantifications of phytochemicals and biocide actions of Lawsonia inermis linn. extracts against wood termites and fungi. Int Biodeterior Biodegradation 116:155–162. https://doi.org/10.1016/j.ibiod.2016.10.026
Tascioglu C, Yalcin M, Troya TD, Sivrikaya H (2012) Termiticidal properties of some wood and bark extracts used as wood preservatives. Bioresources 7(3):2960–2969. https://doi.org/10.15376/biores.7.3.2960-2969
Acknowledgements
The authors would like to thank Assoc. Prof. Dr. Seppo Karrila for the helpful comments and linguistic assistance.
Funding
This research was supported by the Office of the Higher Education Commission and the Thailand Research Fund. This work was supported by the Higher Education Research Promotion and National Research University Project of Thailand, Office of the Higher Education Commission.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yingprasert, W., Cherdchim, B. & Peaklin, S. Effects of Acacia mangium bark extracts on dimensional stability, termite resistance, and fungal decay resistance of rubberwood. Biomass Conv. Bioref. 13, 7623–7632 (2023). https://doi.org/10.1007/s13399-021-01484-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-021-01484-z