Abstract
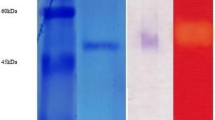
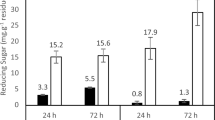
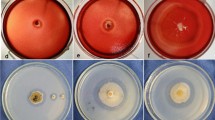
A strain of Trichoderma citrinoviride AUKAR04 was identified on the basis of morphological and 5.8S ribosomal RNA sequencing [GenBank: KF698728]. It produces cocktail of enzymes such as xylanase (55,000 IU gds−1), CMCase (375 IU gds−1) and β-1,3-glucanase (695 IU gds−1) after 72 h under solid-state fermentation. These enzymes were partially purified by a three-phase partitioning method, which recovered the maximum activities of xylanase (99.8 %) with 5.7-fold, CMCase (96.5 %) with 5.5-fold and β-1,3-glucanase (98.4 %) with 5.6-fold purification. The maximum activity of xylanase was observed at pH 5.0, CMCase at pH 5.0–6.0 and β-1,3-glucanase at pH 6.0. Optimum temperature of xylanase and β-1,3-glucanase was found to be at 50 °C, while for CMCase was at 60 °C. The activities of these enzymes were enhanced by Mg2+ and Mn2+ ions. Eucalyptus pulp fiber was incubated for 14 h with the enzyme cocktail. Xylanase hydrolyzed the pulp to yield arabinose (475 mg L−1) and xylose (1795 mg L−1), CMCase and β-1,3-glucanase released glucose (18763 mg L−1). The length of fiber was reduced from 0.881 to 0.056 mm. This is indicative of the potential application on bioconversion of lignocellulosic biomass into fermentable sugars by the enzyme cocktail produced from T. citrinoviride AUKAR04 for sustainable production of bioethanol.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Hahn-Hägerdal B., Galbe M., Gorwa-Grauslund M.F., Lidén G., Zacchi G.: Bio-ethanol-the fuel of tomorrow from the residues of today. Trends Biotechnol. 24, 549–556 (2006)
Binod P., Sindhu R., Singhania R.R., Vikram S., Devi L., Nagalakshmi S., Kurien N., Sukumaran R.K., Pandey A.: Bioethanol production from rice straw: an overview. Bioresour. Technol. 101, 4767–4774 (2010)
Kumar R., Singh S., Singh O.V.: Bioconversion of lignocellulosic biomass: biochemical and molecular perspectives. J. Ind. Microbiol. Biotechnol. 35, 377–391 (2008)
Rubin E.M.: Genomics of cellulosic biofuels. Nature 454, 841–845 (2008)
Kang S.: Production of cellulases and hemicellulases by Aspergillus niger KK2 from lignocellulosic biomass. Bioresour. Technol. 91, 153–156 (2004)
Guerriero G., Hausman J., Strauss J., Ertan H., Sohail K.: Destructuring plant biomass?: focus on fungal and extremophilic cell wall hydrolases. Plant Sci. 234, 180–193 (2015)
Menon V., Rao M.: Trends in bioconversion of lignocellulose: biofuels, platform chemicals & biorefinery concept. Prog. Energy Combust. Sci. 38, 522–550 (2012)
Geddes C.C., Nieves I.U., Ingram L.O.: Advances in ethanol production. Curr. Opin. Biotechnol. 22, 312–319 (2011)
Biswas R., Uellendahl H., Ahring B.: Conversion of C6 and C5 sugars in undetoxified wet exploded bagasse hydrolysates using Scheffersomyces (Pichia) stipitis CBS6054. AMB Express 3, 42 (2013)
Yin L., Lin H., Xiao Z.: Purification and characterization of a cellulase from Bacillus subtilis YJ1. J. Mar. Sci Technol. 18, 466–471 (2010)
Giese E.C., Dekker R.F.H., Barbosa A.M., De Lourdes M., Silva R.: Production of β-(1,3)-glucanases by Trichoderma harzianum Rifai: optimization and application to produce gluco-oligosaccharides from paramylon and pustulan. Ferment. Technol. 1, 1–5 (2012)
Sun H., Ge X., Hao Z., Peng M.: Cellulase production by Trichoderma sp. on apple pomace under solid state fermentation. Afr. J. Biotechnol. 9, 163–166 (2010)
Pandey A., Soccol C.R., Mitchell D.: New developments in solid state fermentation: I-bioprocesses and products. Process Biochem. 35, 1153–1169 (2000)
Sandrim V.C., Rizzatti A.C.S., Terenzi H.F., Jorge J.A., Milagres A.M.F., Polizeli M.L.T.M.: Purification and biochemical characterization of two xylanases produced by Aspergillus caespitosus and their potential for kraft pulp bleaching. Process Biochem. 40, 1823–1828 (2005)
Dogan N., Tari C.: Characterization of three-phase partitioned exo-polygalacturonase from Aspergillus sojae with unique properties. Biochem. Eng. J. 39, 43–50 (2008)
Choonia H.S., Lele S.S.: Three phase partitioning of β-galactosidase produced by an indigenous Lactobacillus acidophilus isolate. Sep. Purif. Technol. 110, 44–50 (2013)
Chaiwut P., Pintathong P., Rawdkuen S.: Extraction and three-phase partitioning behavior of proteases from papaya peels. Process Biochem. 45, 1172–1175 (2010)
Rajeeva S., Lele S.S.: Three-phase partitioning for concentration and purification of laccase produced by submerged cultures of Ganoderma sp. WR-1. Biochem. Eng. J. 54, 103–110 (2011)
Özer B., Akardere E., Çelem E.B., Önal S.: Three-phase partitioning as a rapid and efficient method for purification of invertase from tomato. Biochem. Eng. J. 50, 110–115 (2010)
Roy I., Sharma A., Gupta M.N.: Three-phase partitioning for simultaneous renaturation and partial purification of Aspergillus niger xylanase. Biochim. Biophys. Acta 1698, 107–110 (2004)
Patagundi B.I., Shivasharan C.T., Kaliwal B.B.: Isolation and characterization of cellulase producing bacteria from soil. Int. J. Curr. Microbiol. Appl. Sci. 3, 601–614 (2014)
Deshpande S.K., Bhotmange M.G., Chakrabarti T., Shastri P.N.: Production of cellulase and xylanase by Trichoderma reesei (QM 9414 mutant), Aspergillus niger and mixed culture by solid state fermentation (SSF) of water hyacinth (Eichhornia crassipes). Indian J. Chem. Technol. 15, 449–456 (2008)
Azin M., Moravej R., Zareh D.: Production of xylanase by Trichoderma longibrachiatum on a mixture of wheat bran and wheat straw: optimization of culture condition by Taguchi method. Enzyme Microb. Technol. 40, 801–805 (2007)
Mekala N.K., Singhania R.R., Sukumaran R.K., Pandey A.: Cellulase production under solid-state fermentation by Trichoderma reesei RUT C30: statistical optimization of process parameters. Appl. Biochem. Biotechnol. 151, 122–131 (2008)
Sadhu S., Saha P., Sen S.K., Mayilraj S., Maiti T.K.: Production, purification and characterization of a novel thermotolerant endoglucanase (CMCase) from Bacillus strain isolated from cow dung. Springerplus 2, 10 (2013)
Jia H., Li Y., Liu Y., Yan Q., Yang S., Jiang Z.: Engineering a thermostable β-1,3-1,4-glucanase from Paecilomyces thermophila to improve catalytic efficiency at acidic pH. J. Biotechnol. 159, 50–55 (2012)
Miller G.L.: Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 31(3), 426–428 (1959)
Bayraktar H., S.: Concentration and purification of α-galactosidase from watermelon (Citrullus vulgaris) by three phase partitioning. Sep. Purif. Technol. 118, 835–841 (2013)
Iqbal H.M.N.: Purification and characterization of the kinetic parameters of cellulase produced from wheat straw by Trichoderma viride under SSF and its detergent compatibility. Adv. Biosci. Biotechnol. 02, 149–156 (2011)
Bajaj B.K., Abbass M.: Studies on an alkali-thermostable xylanase from Aspergillus fumigatus MA28. 3 Biotech 1, 161–171 (2011)
Ferreira, S.M.P.; Duarte, A.P.; Queiroz, J.A.; Domingues, F.C.: Influence of buffer systems on Trichoderma reesei Rut C-30 morphology and cellulase production. Electron. J. Biotechnol. 12 (2009)
Ketnawa S., Benjakul S., Martínez-alvarez O., Rawdkuen S.: Three-phase partitioning and proteins hydrolysis patterns of alkaline proteases derived from fish viscera. Sep. Purif. Technol. 132, 174–181 (2014)
Tseng M.-J., Yap M.-N., Ratanakhanokchai K., Kyu K.L., Chen S.-T.: Purification and characterization of two cellulase free xylanases from an alkaliphilic Bacillus firmus. Enzyme Microb. Technol. 30, 590–595 (2002)
Saha B.C.: Production, purification and properties of xylanase from a newly isolated Fusarium proliferatum. Process Biochem. 37, 1279–1284 (2002)
DaSilva Aires R., Steindorff A.S., Ramada M.H.S., de Siqueira S.J.L., Ulhoa C.J.: Biochemical characterization of a 27 kDa 1,3-β-d-glucanase from Trichoderma asperellum induced by cell wall of Rhizoctonia solani. Carbohydr. Polym. 87, 1219–1223 (2012)
Chen L.-L., Zhang M., Zhang D.-H., Chen X.-L., Sun C.-Y., Zhou B.-C., Zhang Y.-Z.: Purification and enzymatic characterization of two beta-endoxylanases from Trichoderma sp. K9301 and their actions in xylooligosaccharide production. Bioresour. Technol. 100, 5230–5236 (2009)
Fontaine T., Hartland R.P., Diaquin M., Simenel C., Latgé J.P.: Differential patterns of activity displayed by two exo-beta-1,3-glucanases associated with the Aspergillus fumigatus cell wall. J. Bacteriol. 179, 3154–3163 (1997)
Hauli I., Sarkar B., Mukherjee T., Mukhopadhyay S.K.: Purification and characterization of a thermoalkaline, cellulase free thermostable xylanase from a newly isolated Anoxybacillus sp. Ip-C from hot spring of Ladakh. Res. Biotechnol. 4, 30–43 (2013)
Leelasuphakul W., Sivanunsakul P., Phongpaichit S.: Purification, characterization and synergistic activity of β-1,3-glucanase and antibiotic extract from an antagonistic Bacillus subtilis NSRS 89-24 against rice blast and sheath blight. Enzyme Microb. Technol. 38, 990–997 (2006)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Periyasamy, K., Santhalembi, L., Mortha, G. et al. Production, Partial Purification and Characterization of Enzyme Cocktail from Trichoderma citrinoviride AUKAR04 Through Solid-State Fermentation. Arab J Sci Eng 42, 53–63 (2017). https://doi.org/10.1007/s13369-016-2110-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13369-016-2110-x