Abstract
A child with vertical transmission of human immunodeficiency virus refractory to therapy developed zoster-induced protein S deficiency and recurrent strokes. Extensive carotid arteritis was found postmortem. The carotid tissue was positive for herpes varicella zoster by polymerase chain reaction, as were immunofixation stains of the arterial wall.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In nonimmune children, varicella zoster virus (VZV) infection usually appears as chickenpox. Thereafter, the virus becomes latent by residing as proviral DNA in spinal ganglia. Immunocompromised states such as acquired immune deficiency syndrome (AIDS) frequently allow VZV reactivation. Such recrudescence usually manifests as a dermatomal vesicular rash called zoster or shingles (Nagel et al. 2010; Nagel and Gilden 2014).
During this recrudescence, concurrent VZV viremia rarely leads to inflammatory arteritis. Strokes due to vascular occlusion can follow (Newsome and Nath 2009; Teo et al. 2014). In AIDS cases, most VZV-related strokes occur during initial antiretroviral therapy (ART) owing to an inflammatory arteritis induced by immune reconstitution inflammatory syndrome (IRIS) (Newsome and Nath 2009; Teo et al. 2014).
Protein S (PS), a cofactor in the clotting pathway, prevents excessive coagulation. PS deficiency is highly associated with thromboembolisms, deep vein thrombosis, and pulmonary embolisms. Antibodies to PS have been shown to bind with VZV peptide chains, and PS deficiency has been reported in children with recent VZV infection (Manco-Johnson et al. 1996).
We describe the case of a child with AIDS refractory to ART who developed central nervous system (CNS) strokes as a consequence of bilateral intracranial vasculitis. Evaluation of her paraffin-preserved tissues revealed her vasculitis to be related to recrudescent VZV.
Case report
A 5-year-old girl was brought to our health system for treatment of AIDS. At age 2 months, she was found infected with human immunodeficiency virus (HIV) through vertical transmission. Her ART began with didanosine (DDI) and zidovudine (AZT), with subsequent multidrug combinations of indinavir, nelfinavir, saquinavir, ritonavir, D4T, and lamivudine/zidovudine over the next 4 years. Her CD4 count was never greater than 31 cells/μL, and her viral load remained uncontrolled, not due to viral resistance. At age 4, she developed chickenpox and was treated with acyclovir and two courses of zoster immunoglobulin. Between age 5 and her death, she tested negative by serology and/or polymerase chain reaction (PCR) for toxoplasmosis, cytomegalovirus (CMV), and herpes simplex virus, and by culture for fungus and mycobacteria. However, serological evidence for Epstein-Barr virus (EBV) and VZV was found.
At age 7, she had a vesicular lesion in a dermatomal distribution on the right nape of her neck. VZV was detected by direct fluorescent antibody, and she was started on valacyclovir and one dose of 125 units of VZV immune globulin. One month later, despite multiple ART attempts, her CD4 count was only 6 cells/μL.
Nine months post-zoster therapy, the patient had a stroke characterized by right hemiplegia and decreased ability to communicate. Seven weeks later, a second infarct presented with headache, drooling, and dysphagia. Infarcts were documented on computed tomography scan in the left and right basal ganglia. At that time, testing was again negative for syphilis, CMV, hepatitis B and C, cryptococcal antigen, and for growth in fungal and mycobacterial cultures. However, PS activity deficiency was detected (protein reactivity < 6%, free PS 10%, total PS antigen 90%), and Coumadin therapy was initiated. Following this repeat cerebrovascular accident (CVA), the patient suffered from recurring seizures until her death at age 9.
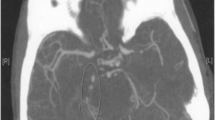
Postmortem evaluation reported AIDS as the cause of death, with evidence of cerebral atrophy, an incomplete circle of Willis, multiple thrombosed aneurysmal dilatations, and lymphocytic vasculitis of the external carotid artery (Fig. 1).
Sections of her preserved carotid arterial tissue were de-paraffinized and tested positive for VZV using standard PCR methods (Harbecke et al. 2009). The specimen was also strongly positive for VZV by immunohistochemistry polyantigen monoclonal staining (Nagel et al. 2015) (Fig. 2).
Discussion
HIV/AIDS-related strokes have been reported in children and young adults (Nagel et al. 2010; Gutierrez and Ortiz 2011; Benjamin et al. 2012). Possible etiologies of these strokes include immune reconstitution vasculitis following ART or IRIS (van der Ven et al. 2002; Newsome and Nath 2009; Teo et al. 2014), coagulopathy, and opportunistic infections such as Cryptococcus species, Mycobacterium species, syphilis, CMV or EBV, and as in this case, VZV (Benjamin et al. 2012; Teo et al. 2014; Pinninti et al. 2016).
When VZV CNS vasculitis occurs in immunocompetent individuals, it affects localized areas of small- to medium-sized arteries and can cause thrombosis in the internal carotid or basilar arteries (Gilden et al. 1996). In contrast, in this pediatric AIDS case, a medium- to large-vessel vasculitis with multiple thrombosed aneurysmal dilatations occurred. As others have hypothesized (Thomson et al. 2010; Nguyên et al. 1994; Josephson et al. 2001; Ferrara et al. 2013), we suggest that these findings stem from an acquired PS deficiency secondary to VZV viremia and a local, severe, persistent medium to large artery arteritis with ongoing VZV replication caused her strokes.
With zoster, VZV usually produces a transient viremia that can seed vascular endothelium (Nagel and Gilden 2016). Endothelial cells are known to produce and secrete PS (Fair et al. 1986). In this case, it is plausible that VZV arteritis allowed the presentation of host antigens, leading to an immunoinflammatory response toward endothelial cells with the production of antibodies to free PS, with subsequent strokes.
The incidence of CVAs is elevated in people with HIV/AIDS. Approximately 1–5% of HIV cases develop strokes; upon autopsy, up to 34% have lesions associated with a past CVA (Gutierrez and Ortiz 2011; Benjamin et al. 2012). Previous studies demonstrated VZV as a risk factor for strokes, and immunocompromised individuals at the greatest risk (Nagel et al. 2010; Gutierrez and Ortiz 2011; Nagel and Gilden 2014, 2016). Therefore, VZV vasculitis should be considered in cases of CVA, especially in immunocompromised individuals. With clinical and laboratory evidence, treatment with an anti-VZV regimen such as intravenous acyclovir or oral valacyclovir should be initiated synchronously with ART. If PS deficiency is concurrently found, anticoagulants should be considered. If related to immune reconstitution inflammatory syndrome (IRIS), prednisone therapy also has been successful (Newsome and Nath 2009; Teo et al. 2014).
Prevention of VZV infection by live viral VZV vaccination in immunocompromised individuals is controversial and not recommended in those with CD4 counts < 200 cells/μL (Shafran 2016). The US Food and Drug Administration has approved an adjuvanted herpes zoster subunit vaccine (Shingrix) for use in immunocompetent patients ≥ 50 years old; however, further research is needed to determine whether it is both safe and effective in the immunocompromised.
References
Benjamin LA, Bryer A, Emsley HC, Khoo S, Solomon T, Connor MD (2012) HIV infection and stroke: current perspectives and future directions. Lancet Neurol 11:878–890. https://doi.org/10.1016/S1474-4422(12)70205-3
Fair DS, Marlar RA, Levin EG (1986) Human endothelial cells synthesize protein S. Blood 67:1168–1171
Ferrara M, Bertocco F, Ferrara D, Copozzi L (2013) Thrombophilia and varicella zoster in children. Hematology 18:119–122. https://doi.org/10.1179/1607845412Y.0000000055
Gilden DH, Kleinschmidt-DeMasters BK, Wellish M, Hedley-Whyte ET, Rentier B, Mahalingam R (1996) Varicella zoster virus, a cause of waxing and waning vasculitis: The New England Journal of Medicine case 5-1995 revisited. Neurology 47:1441–1446. https://doi.org/10.1212/WNL.47.6.1441
Gutierrez J, Ortiz G (2011) HIV/AIDS patients with HIV vasculopathy and VZV vasculitis. Clin Neuroradiol 21:145–151. https://doi.org/10.1007/s00062-011-0087-0
Harbecke R, Oxman MN, Arnold BA, Ip C, Johnson GR, Levin MJ, Gelb LD, Schmader KE, Straus SE, Wang H, Wright PF, Pachucki CT, Gershon AA, Arbeit RD, Davis LE, Simberkoff MS, Weinberg A, Williams HM, Cheney C, Petrukhin L, Abraham KG, Shaw A, Manoff S, Antonello JM, Green T, Wang Y, Tan C, Keller PM, Shingles Prevention Study Group (2009) A real-time PCR assay to identify and discriminate among wild-type and vaccine strains of varicella-zoster virus and herpes simplex virus in clinical specimens, and comparison with the clinical diagnoses. J Med Virol 81:1310–1322. https://doi.org/10.1002/jmv.21506
Josephson C, Nuss R, Jacobson L, Hacker MR, Murphy J, Weinberg A, Manco-Johnson MJ (2001) The varicella-autoantibody syndrome. Pediatr Res 50:345–352. https://doi.org/10.1203/00006450-200109000-00009
Manco-Johnson MJ, Nuss R, Key N, Moertel C, Jacobson L, Meech S, Weinberg A, Lefkowitz J (1996) Lupus anticoagulant and protein S deficiency in children with postvaricella purpura fulminans or thrombosis. J Pediatr 128:319–323
Nagel MA, Gilden D (2014) Update on varicella zoster virus vasculopathy. Curr Infect Dis Rep 16:407. https://doi.org/10.1007/s11908-014-0407-z
Nagel M, Gilden D (2016) Developments in varicella zoster virus vasculopathy. Curr Neurol Neurosci Rep 16:1–8. https://doi.org/10.1007/s11910-015-0614-5
Nagel MA, Mahalingam R, Cohrs RJ, Gilden D (2010) Virus vasculopathy and stroke: an under-recognized cause and treatment target. Infect Disord Drug Targets 10:105–111
Nagel MA, Lenggenhager D, White T, Khmeleva N, Heintzman A, Bover PJ, Gilden D (2015) Disseminated VZV infection and asymptomatic VZV vasculopathy after steroid abuse. J Clin Virol 66:72–75. https://doi.org/10.1016/j.jcv.2015.03.016
Newsome SD, Nath A (2009) Varicella-zoster virus vasculopathy and central nervous system immune reconstitution inflammatory syndrome with human immunodeficiency virus infection treated with steroids. J Neuro-Oncol 15:288–291. https://doi.org/10.1080/13550280902913610
Nguyên P, Reynaud J, Pouzol P, Munzer M, Richard O, François P (1994) Varicella and thrombotic complications associated with transient protein C and protein S deficiencies in children. Eur J Pediatr 153:646–649
Pinninti S, Hough-Telford C, Pati S, Boppana S (2016) Cytomegalovirus and Epstein-Barr virus infections. Pediatr Rev 37:223–234. https://doi.org/10.1542/pir.2015-0072
Shafran SD (2016) Live attenuated herpes zoster vaccine for HIV-infected adults. HIV Med 17:305–310. https://doi.org/10.1111/hiv.12311
Teo SY, Raha D, Warren D, Hassan A, Monteiro E (2014) Central nervous system–immune reconstitution inflammatory syndrome presenting as varicella zoster virus-mediated vasculitis causing stroke. Int J STD AIDS 25:683–685. https://doi.org/10.1177/0956462413518501
Thomson JJ, Retter A, Hunt BJ (2010) Novel management of post varicella purpura fulminans owing to severe acquired protein S deficiency. Blood Coagul Fibrinolysis 21:598–600. https://doi.org/10.1097/MBC.0b013e32833c2b52
van der Ven AJAM, van Oostenbrugge RJ, Kubat B, Tervaert JW (2002) Cerebral vasculitis after initiation antiretroviral therapy. AIDS 16:2362–2364
Acknowledgments
The authors would like to thank Steven Lovrich, Ph.D., Gundersen Health System; Scott Cooper, Ph.D., University of Wisconsin – La Crosse; and Bernadette Taylor, Ph.D., University of Wisconsin – La Crosse.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Deviley, J.A., Oldenburg, D., Berg, L.C. et al. Recurrent strokes, central nervous system vasculitis, and acquired protein S deficiency secondary to varicella zoster in a child with AIDS. J. Neurovirol. 25, 137–140 (2019). https://doi.org/10.1007/s13365-018-0697-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13365-018-0697-2