Abstract
We investigated patterns of species richness and co-occurrence in a montane carnivore community within a forested landscape in Bhutan that ranged in altitude from 2000 to 3760 m above sea level, and covered an area of approximately 140 km2. Species were detected by unbaited camera traps set along animal trails and baited camera traps set away from trails. During the 6-month study, we gathered 1,329 independent mammal events from 67 camera-trap locations, of which, 145 (10.9%) were of 13 different carnivore species from five different families. Four carnivores were IUCN red-listed threatened species: tiger (Panthera tigris), marbled cat (Pardofelis marmorata), dhole (Cuon alpinus), and Asiatic black bear (Ursus thibetanus). For most camera stations where carnivores were detected, only a single carnivore species was captured on camera and there was evidence of temporal partitioning of activity between large (tiger and leopard, Panthera pardus) and small (marbled cat, golden cat Catopuma temminckii, and leopard cat Prionailurus bengalensis) felids, and between two common mustelids, the Siberian weasel (Mustela sibirica) and yellow-throated marten (Martes flavigula). Furthermore, we detected significant non-random spatial co-occurrence for most pairwise comparisons of carnivores despite the short timeframe of our study. This, combined with temporal patterns in activity, facilitates localized species co-occurrence in a diverse montane carnivore community.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Carnivores are important members of most mammal communities and despite existing at naturally low densities, can often exert strong influence on other species in the community. Mammalian carnivores tend to exert top-down influence on herbivores through direct predation (Andersen et al. 2006) with profound effects on the flow of nutrients and the structure of food webs (Ripple et al. 2014; Roemer et al. 2009). Large carnivores exert further population pressure on smaller mesocarnivores through intraguild competition (Ripple et al. 2014) encompassing direct predation (Mohammadi et al. 2017; Palomares and Caro 1999), or influencing changes in mesocarnivore behavior and resource use to avoid antagonistic interactions (Thinley et al. 2018). Avoidance mechanisms include using different habitat types (Ramesh et al. 2017; Schaller 1967; Steinmetz et al. 2013), having different activity schedules (Steinmetz et al. 2013), and utilizing fine-scale movement patterns (Ramesh et al. 2017) to achieve avoidance in a shared environment. Avoidance mechanisms are not mutually exclusive. For example, Steinmetz et al. (2013) showed that the common leopard (Panthera pardus) and dhole (Cuon alpinus) spatially avoided habitats with tigers (Panthera tigris), and were active at times that tigers were not. Similarly, Ramesh et al. (2017) showed that common leopards used temporal means to avoid lions (Panthera leo), but spatial means to avoid hyenas (Crocuta crocuta).
Carnivores reach their highest richness in two global biogeographical regions: the Afrotropical Region of sub-Saharan Africa and the Indo-Malaysian Region extending from insular South East Asia to the Eastern Himalayans (Crooks et al. 2008; Procheş and Ramdhani 2012). Here, carnivore communities are typically dominated by the families Felidae, Canidae, Mustelidae, and Viverridae (Chutipong et al. 2017; Kauffman et al. 2007; Kawanishi and Sunquist 2004; Schuette et al. 2013). Although regional and global patterns of mammal diversity in Asia are relatively well understood (Chi et al. 2020; Dorji et al. 2018), local patterns of species richness and community structure remain poorly known due to costly field surveys, difficulty in detecting uncommon or cryptic species, and difficulty distinguishing species from indirect signs like pug-marks and scats (Sangay et al. 2014). However, rapid advancement of heat-in-motion camera trap technology has revolutionized wildlife ecology (Meek et al. 2015) with some camera trap studies in Asia revealing localized carnivore community richness comparable to broader regional scales, with as many as 18–20 co-occurring carnivore species detected within relatively small areas (Chutipong et al. 2017; Johnson et al. 2006; Kawanishi and Sunquist 2004). Camera traps also represent a non-invasive method of gathering spatiotemporal data on animals, even when they occur at low densities (Karanth et al. 2017).
Bhutan, situated in the Eastern Himalayas at the intersection of the Palearctic and Indo-Malaysian biogeographical regions (Procheş and Ramdhani 2012), has an altitudinal range encompassing a diversity of habitats from low-altitude tropical rainforest, mid-altitude broadleaf and mixed conifer forest, to high-altitude alpine and scree (Tharchen 2013). Correspondingly, Bhutan has a rich mammal diversity that includes at least 39 species of carnivores (Wangchuk et al. 2004), contributing to its listing as a global biodiversity hotspot (Mittermeier et al. 2011). Localized carnivore richness is also high. For example, a camera-trapping study by Tempa et al. (2013) in the tropical lowlands of Royal Manas National Park revealed 16 co-occurring carnivore species, including six felid species. However, little information exists on carnivore diversity in mid-to high-altitude forests in Bhutan.
Both large and small Asian carnivores are highly threatened by habitat destruction, agricultural expansion, overhunting, and poaching (Ashrafzadeh et al. 2020; Marneweck et al. 2021). This is especially evident in the Eastern Himalayas (Dorji et al. 2018) with its high rates of deforestation and habitat fragmentation (Pandit et al. 2007) in conjunction with high hunting pressure (Velho et al. 2012) and a high human population density (Mittermeier et al. 2011). Such substantial anthropogenic pressure necessitates a better understanding of Asian carnivore community ecology for conservation planning (Dalerum et al. 2009). In particular, Marneweck et al. (2021) recently encouraged the need for more research on the ecology and demography of small Asian carnivores to inform conservation, given current and future threats from global change. Our investigation on carnivore co-occurrence in a montane zone of central Bhutan in the Eastern Himalayas addresses this premise toward conserving threatened carnivores, and supplements the lack of information on carnivore community richness at this elevation zone.
Materials and methods
Study area
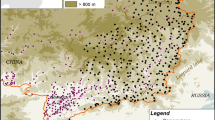
We undertook a camera trap mammal survey over 6 months between November 2012 and April 2013 in Corridor 8 of Bhutan’s Biological Corridor Complex (Fig. 1), which connects Jigme Singye National Park in central Bhutan with Wangchuk Centennial and Jigme Dorji National Parks in the north of the country. Our study area covered approximately 140 km2 ranging in altitude from 2000 to 3760 m above sea level (asl). Lower elevation (2000–2700 m asl) zones comprised cool broadleaf forest; mid-elevation (2700–3500 m asl) zones were either cool broadleaf forest or mixed conifer forest (including spruce Picea spinulosa, hemlock Tsuga dumosa, Blue pine Pinus wallichiana, and larch Larix spp.); and high-elevation zones (above 3500 m asl) were sub-alpine forest that was either pure stands of fir (Abies densa) with a bamboo dominated understorey, or a mix of species including various conifers, juniper (Juniperus recurva), and rhododendron (Rhododendron spp.). Habitat across the study area was generally intact and only moderately impacted by some cattle grazing, and sustainable collection of firewood, timber, and non-wood forest products by local people. Narrow footpaths used by people, livestock, and wildlife traversed most spurs and ridgelines, while small grazing meadows and livestock herder huts dotted the predominantly forested landscape. The east–west National Highway (at the time a narrow single-lane road) bisected the corridor.
The study area in central Bhutan. This corridor, known as “Corridor 8” in the Bhutan Biological Corridors Complex (B2C2), links Jigme Singye Wanchuck National Park in the south to Jigme Dorji National Park and Wangchuck Centennial National Park in the north. Outlines of other corridors in the B2C2 and their relationship to protected areas, is also indicated
Survey methods
We surveyed 16 × 1000-m-long transects along footpaths and game trails, with a random starting point for each transect. Starting at 0 m along each transect, we established a 50 × 50-m survey plot at 500-m intervals that was placed 100 m away from each transect along a randomly generated compass heading. We recorded all signs of vertebrate fauna (both direct sightings and indirect evidence) in the plot. We additionally undertook a detailed vegetation survey in a nested 20 × 20-m subplot. A Reconyx Hyperfire PC850 white-flash camera trap, set to take 5 photos per trigger with no “quiet time,” was placed at the center of each survey plot and positioned 30 cm from the ground and 1.5 m from a bait canister containing cotton wool soaked in truffle oil as a lure. These cameras, hereafter referred to as “plot cameras,” aimed to capture carnivores that avoided main trails. We set Cuddeback Attack white-flash camera traps on the main transect (trail) at intervals not less than 100 m from one another, to detect carnivore species using the trail. These camera traps (hereafter called “trail cameras”) were not baited, and their deployed locations were non-random and selected on the evidence of pronounced mammal activity (scats, tracks, scrapes, etc.) most notably at the convergence of two or more game trails and/or forest paths. We affixed trail camera traps to tree trunks at a height of approximately 0.5–1 m. These camera traps faced the main trail at an angle of approximately 45° relative to the direction of the trail. Camera traps ran for 36–51 days (average ± S.D. = 43 ± 5 days) before being recovered and redeployed at new transects. In total, we set 40 plot cameras (20 in cool broadleaf forest, 15 in mixed conifer forest, and five in sub-alpine forest) and 27 trail cameras (14 in cool broadleaf forest, seven in mixed conifer forest, and six in sub-alpine forest) for a total of 3,085 trap days and nights.
We sorted photos of carnivore species at each camera location and processed them using the “CameraSweet” camera trapping analysis software (Sanderson and Harris 2013). We considered independent events to be those where 30 or more minutes had elapsed between successive photos of the same species at the same camera. A similar 30-min cut-point for independent events has been commonly used in other carnivore studies (Dorji et al. 2019; Edwards et al. 2015; Kennedy et al. 2018; Vernes et al. 2015).
Analytical methods
For carnivore species with adequate number of records for meaningful comparisons (tiger, leopard, golden cat Catopuma temminckii, leopard cat Prionailurus bengalensis, Asiatic black bear Ursus thibetanus, dhole, Siberian weasel Mustela sibirica, and yellow-throated marten Martes flavigula), we investigated carnivore assemblage structure at our 67 camera trap stations by examining pairwise relationships between species using the probabilistic model of species co-occurrence developed by Veech (2013) within the R package “cooccur” (Griffith et al. 2016). We assumed potential avoidance between species based on inter-specific competition for resources, i.e., leopard cat avoiding golden cat (Rasphone et al. 2020) and Siberian weasel avoiding yellow-throated marten (Zhao et al. 2020), or antagonistic interactions such as leopard and dhole avoiding tiger (Seidensticker 1976; Steinmetz et al. 2013; Thinley et al. 2018), dhole avoiding leopard (Karanth et al. 2017), and Siberian weasel avoiding yellow-throated marten (Zhao et al. 2020). We tested these interactions explicitly for subsets of camera data that detected these species, rather than examining all possible pairwise combinations. Prior to analyses, we created a “site mask” (see Griffith et al. 2016) to exclude sites at which particular mammal species would not be expected to occur based upon the elevational range for each species. Information on elevation ranges was drawn from a nation-wide wildlife camera trapping dataset (Sangay et al. 2019) which yielded minimum and maximum elevation recorded for each species in our study area. That dataset comprised approximately 10 million photographs obtained from 148,598 trap-nights across 1858 camera traps deployed within 1129 5-km × 5-km grids across Bhutan.
Results
Sampling effort
During our 6-month survey, we gathered approximately 16,000 images from 67 camera-trap locations (40 plot cameras; 27 trail cameras) and recorded 1,329 independent mammal events from these images, of which, 145 (10.9%) were carnivore species. Carnivores were detected at 44 (65.7%) of the 67 camera traps. There was no significance difference in carnivore detections between plot (61 detections; 42.4%) and trail (83 detections; 57.6%) cameras (t = 1.02; df = 12; p < 0.5), although some species were more likely to occur at one type of camera station over the other. For example, dhole were only detected on trail cameras (n events = 16) while Siberian weasels were mostly detected on plot cameras (n events = 15) compared to trail cameras (n events = 5).
Species richness
We detected thirteen species of mammalian carnivores from five families (Table 1). Families comprised Felidae (5 species), Canidae (2 species), Ursidae (1 species), Mustelidae (3 species), and Viverridae (2 species). Four were IUCN red-listed threatened species: tiger (endangered; 7 records), dhole (endangered; 16 records), common leopard (vulnerable; 12 records), and Asiatic black bear (vulnerable; 23 records) (Table 1). Two additional cat species (marbled cat Pardofelis marmorata; 4 records and Asiatic golden cat; 13 records) are listed as near-threatened by the IUCN (Table 1).
We detected the yellow-throated marten at the highest number of camera trap sites (15 sites; 22% of total) followed equally by the Siberian weasel and Asiatic black bear (12 sites; 18% of total), and then the leopard cat (10 sites; 15% of total). Three species (Himalayan palm civet Paguma larvata; spotted linsang Prionodon pardicolor; and yellow-bellied weasel Mustela kathiah) were each detected at a single site, and only once (Table 1). Fifty percent (n = 22) of our camera traps detected only a single carnivore species while 22.7% (n = 10) and 18.2% (n = 8) detected two and three species, respectively. Only 4.5% (n = 2) of camera traps detected the presence of ≥ 4 carnivore species.
Temporal and spatial partitioning
Most pairwise comparisons had co-occurrences which were significantly different from the expected co-occurrence (Table 2). Strongest differences were found among the small carnivores. For species that occurred at more than four cameras (Asiatic golden cat, leopard cat, Siberian weasel, and yellow-throated marten), all pairwise comparisons returned co-occurrences which were significantly lower than would be expected by chance (Table 2). For large carnivores, significant negative co-occurrences were detected between Asiatic black bear and tiger, Asiatic black bear and dhole, tiger and dhole, and common leopard and dhole, but not between tiger and common leopard, or Asiatic black bear and common leopard (Table 2).
Tigers exhibited strong diurnal activity overlap with leopards (55% overlap; Fig. 2a) and dholes (74% overlap; Fig. 2b). For other carnivore species with sufficient activity data, yellow-throated martens were strongly diurnal with activity peaking at mid-afternoon whereas Siberian weasels were primarily nocturnal, but with activity peaks that extended from early evening through to mid-morning (40% overlap; Fig. 2c). Despite a 45% activity overlap, Asiatic golden cats were predominantly diurnal compared to leopard cats (Fig. 2d).
Temporal overlap between species and species groupings from camera trap data. a Tiger (Panthera tigris) and leopard (Panthera pardus), b tiger and dhole (Cuon alpinus), c yellow-throated marten (Martes flavigula) and Siberian weasel (Mustela sibirica), d Asiatic golden cat (Catopuma temminckii) and leopard cat (Prionailurus bengalensis), e small cat group (golden cat, leopard cat and marbled cat, Pardofelis marmorata) and large cats and dhole group (tiger, leopard and dhole), and f Himalayan black bear (Ursus thibetanus) and large cat and dhole group
When differentiated by size, there was evidence of temporal activity partitioning between the large felid and canid guild (tiger, leopard, dhole) and the small felid (marbled, golden, and leopard cats) guild despite a 46% overlap (Fig. 2e). Large predators were more diurnal than small felids with their activity being broadly crepuscular, while small felids were predominantly nocturnal. Asiatic black bears exhibited both diurnal and nocturnal behaviors with a 60% overlap with the large felid and canid guild (Fig. 2f).
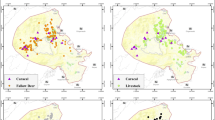
Despite being present across all forest types, most carnivore species were associated with a particular forest type and elevation range (Fig. 3). For example, Asiatic black bears were most common in low-elevation cool broadleaf forest (2000–2700 m; Fig. 3a) whereas dhole mostly occurred in mid-elevation cool broadleaf forest (2700–3500 m) and high-elevation alpine habitat (> 3500 m; Fig. 3b). Tiger and common leopard largely occupied separate forest types. Leopards were found mostly in low-elevation cool broadleaf forest (Fig. 3c) at elevations < 2800 m whereas tigers were common to mid-elevation mixed conifer forest and high-elevation alpine habitat (Fig. 3d) up to 3760 m. Leopard cats and yellow-throated martens were mostly found in low-elevation cool broadleaf forest (Fig. 3f, g) whereas Siberian weasels were found at high-elevation mixed conifer forest and alpine habitat (Fig. 3h).
Number of independent events per camera trap for cameras set in cool broadleaf forest (CBL) at low (2000–2700 m) elevation (“CBL(Low)”), cool broadleaf forest at mid (2700–3500 m) elevation (“CBL(High)”), mixed conifer forest at mid (2700–3500 m) elevation (“MCF”), and sub-alpine vegetation at high (> 3500 m) elevation (“Alpine”) for the eight most commonly detected mammals in the study area
Discussion
Species richness
The Eastern Himalayan region supports rich carnivore diversity at regional scales (Dorji et al. 2018) and camera trapping (Bu et al. 2016; Johnson et al. 2009) has revealed high species richness at local scales. Although our study timeframe was short and within a relatively small study area (140 km2), we successfully detected the presence of 13 co-occurring carnivore species. Another carnivore, the red panda (Ailurus fulgens), has since been detected by us (unpublished data) in the same study area as part of ongoing work. Although some camera trapping studies in the Eastern Himalayas have reported local site-specific carnivore richness as high as 16 species (Datta et al. 2008; Naniwadekar et al. 2013; Tempa et al. 2013), these studies focused on the species-rich tropical lowland forest. Our study indicates that local carnivore richness in mid- to high-elevation (2000–3760 m) montane forests of the Eastern Himalayas is comparable to that found in the region’s tropical lowlands, demonstrating that these montane forests are equally important landscapes in supporting a diverse carnivore community.
Of the 13 carnivore species detected in our study, four species are globally threatened on the IUCN Red List. These comprise the endangered tiger and dhole, and the vulnerable common leopard and Asiatic black bear. Although the Asiatic golden cat and marbled cat are listed as near threatened, their global populations are deemed to be declining. Global populations are also declining for three species of least concern detected in our study, which are the yellow-throated marten, Himalayan palm civet, and spotted linsang. Given the overall species richness combined with the presence of both threatened and declining populations of near threatened species, the montane forest zone in Bhutan is important for regional carnivore conservation in the Eastern Himalayas.
Large carnivore patterns
Although large carnivores had overlapping distributions, they appeared to restrict spatial occurrence by rarely overlapping at camera sites. Tiger, leopard, and dhole are large, wide-ranging carnivores that occur throughout much of Bhutan (Sangay and Vernes 2008), and each species was detected at between six (tiger and leopard) and seven (dhole) camera sites. All three species never overlapped at any one camera site, while overlap between tiger and leopard, leopard and dhole, and tiger and dhole only occurred at one camera station, with non-random statistical support for comparisons. Significant non-random co-occurrences were also detected between the Asiatic black bear and most other large carnivores. Tiger and leopard co-occurrence was not significantly less than expected. However, because these species occupied different ends of the elevation gradient and typically occupied different forest types, they appeared to be largely segregated at the landscape scale in our study area and spatially avoided each other. At another study site in central Bhutan, Thinley et al. (2018) found evidence that large predators were sympatric at the landscape scale, but were spatially segregated at finer scales. In that study, tiger, leopard, and dhole were never captured on a camera trap at the same location, on the same day, or during the same 2-week sampling occasion despite each species occurring throughout the study area. The ecological dominance of tigers over leopards is well documented (Morse 1974; Steinmetz et al. 2013) and is probably responsible for the patterns we observed. For example, Seidensticker (1976) showed that leopards avoided areas frequented by tigers in Nepal’s Chitwan National Park. Similarly, Odden et al. (2010) found significantly higher leopard signs near the periphery of a tiger’s home range in Nepal’s Bardia National Park rather than closer to the tiger’s home range core area, while Steinmetz et al. (2013) showed that tiger occurrence in Thailand’s Kuiburi National Park was driven by prey availability with leopard and dhole distribution concurrently influenced by their avoidance of tigers. However, Allen et al. (2020) showed high temporal overlap between the dhole and tiger in Sumatra, and attributed it to lack of fear by pack-hunting dholes toward the tiger.
Prey availability can influence the degree of spatial and temporal separation in carnivore guilds. Karanth et al. (2017) demonstrated that local prey resource availability influenced the behavior of tigers, leopards, and dhole in Southern India. At low prey density, spatial overlap between all three species was high as each sought to maximize prey acquisition against the trade-off of an antagonistic encounter. However, when prey density was high, the three species avoided encounters through fine-scale spatial segregation. We recorded several prey species on camera traps but it was not possible to determine empirically whether prey density at our study site was higher or lower relative to other regions. Nevertheless, Bhutan’s forests are largely intact landscapes supporting diverse mammal communities not overtly impacted by high livestock densities or human hunting pressure. Although livestock predation is present (Sangay and Vernes 2008), predation rates are considered tolerable (Sangay and Vernes 2014), perhaps indicating that Bhutan’s large predators are subsisting largely on abundant native prey species. Furthermore, native ungulates (principally barking deer Muntiacus muntjac, sambar Rusa unicolor, and wild pig Sus scrofa) were detected three times more often on our camera traps than livestock, indicating relatively high native prey densities.
Small carnivore patterns
Small felids in our study also temporally avoided larger felids and the dhole. Pasanen-Mortensen et al. (2013) indicated a negative correlation between relative abundance of apex and mesocarnivores due to direct dominance by apex predators (Oliveira and Pereira 2014). Mesocarnivore species subsequently adopt spatiotemporal segregation to allow coexistence (Bitetti et al. 2010). Zhao et al. (2020) discovered a similar pattern in China whereby mesocarnivores (leopard cat, red fox, Asian badger Meles leucurus, Siberian weasel, Himalayan palm civet, and yellow-throated marten) spatially and seasonally avoided leopards. Similarly, Allen et al. (2020) found that leopard cats and Asian golden cats temporally avoid the tiger in Sumatra.
Smaller mesopredators in our study also displayed interspecific avoidance, possibly based on intra-guild aggression and/or resource competition. Three small felids (leopard cat, Asiatic golden cat, and marbled cat) common to Bhutan’s mid-altitudinal forests were detected at our study site but interspecific overlap at a camera trap station was rare, with strong statistical support for this non-random pattern. The Asiatic golden cat, the largest of the small felids in the study area, had a diurnal activity schedule whereas the smaller leopard cat was strongly nocturnal. Leopard cats are typically nocturnal (Bu et al. 2016; Johnson et al. 2009) to maximize encounters with small rodents, which are their primary prey (Austin et al. 2007; Grassman et al. 2005; Rajaratnam et al. 2007). Similar temporal activity partitioning has also been detected for Asiatic golden cats and leopard cats in Northern Laos to help facilitate coexistence (Rasphone et al. 2020).
The yellow-throated marten and Siberian weasel were the most common mustelids in our study. However, both species exhibited strong spatial partitioning, occurring together at only 3 of the 27 camera trap sites utilized by this species pair. They also exhibited little temporal overlap. Yellow-throated martens were diurnal while Siberian weasels were largely nocturnal. Yellow-throated martens are considered to be dominant over the smaller Siberian weasel, and there is evidence from other studies that spatial and temporal partitioning occurs. Bu et al. (2016) showed that the diurnal yellow-throated marten in China switched to feeding on small mammals in winter due to lack of fruit. Concurrently, Siberian weasels which until then had no clear diel activity pattern, switched to being nocturnal. Bu et al. (2016) argued that because Siberian weasels also prey predominantly on small mammals (Bu et al. 2016; Chiang et al. 2012), they switched activity to minimize interactions with the larger, dominant yellow-throated marten. Similarly, Chiang et al. (2012) found that Siberian weasels were mostly nocturnal in southern Taiwan where they co-occurred with yellow-throated martens, while Wong (1997) found weasels were more diurnal at a Taiwanese site where yellow-throated martens were absent. Contrastingly, Zhao et al. (2020) revealed at another site in China that Siberian weasels and yellow-throated martens were mainly diurnal, but spatially avoided each other and were temporally segregated from other mesopredators. Similar temporal shifts in activity have been recorded for other mesopredators. For example, Harrington (2007) showed invasive mink (Mustela vison) in the UK switched activity from predominantly nocturnal to predominantly diurnal in the presence of a larger aggressive competitor, the Eurasian otter (Lutra lutra).
Our study has shown that 13 different carnivores are able to co-occur within a relatively small area through non-random spatial and temporal partitioning to facilitate co-occurrence. Leopards and dhole appeared to avoid tiger, smaller cats and mustelids avoided one another, and the smaller Siberian weasel avoided the much larger yellow-throated marten. Our work was, however, undertaken over a relatively small timeframe in only one small area of the Himalaya. Nevertheless, this initial understanding of spatial and temporal segregation in a montane carnivore guild can inform future carnivore conservation especially through its applicability to alleviate potential human-wildlife conflict. Livestock in Bhutan are commonly grazed untended in a landscape shared by sympatric predators, resulting in opportunistic livestock depredation (Rajaratnam et al. 2016; Sangay and Vernes 2008) by the four IUCN-listed threatened large carnivore species (tiger, common leopard, dhole, and Asiatic black bear) detected in our study. Despite being deemed as tolerable (Sangay and Vernes 2014), livestock predation has socioeconomic repercussions on rural agropastoralists which can possibly lead to retaliation (Din et al. 2017). As such, knowledge of carnivore spatial and temporal activity can guide improved livestock husbandry by modifying grazing patterns and timing, including provisions for tethering in villages (Katel et al. 2015; Rajaratnam et al. 2016; Sangay and Vernes 2008). Our study lays the foundation to further investigate spatiotemporal patterns within Himalayan carnivore guilds over seasonal time frames, and across a larger geographical area. This expanded research scope can help determine if carnivore species are modifying behavior or populations are under stress from anthropogenic impacts.
Change history
12 January 2022
A Correction to this paper has been published: https://doi.org/10.1007/s13364-022-00618-6
References
Allen ML, Sibarani MC, Utoyo L, Krofel M (2020) Terrestrial mammal community richness and temporal overlap between tigers and other carnivores in Bukit Barisan Selatan National Park, Sumatra. Anim Biodivers Conserv 43:97–107
Andersen R, Linnell J, Solberg E (2006) The future role of large carnivores in terrestrial trophic interactions: the northern temperate view. In “Large Herbivore Ecology, Ecosystem Dynamics and Conservation.”. (Eds. D K, P Duncan, Bergstrom R and P J) pp. 413–448. Cambridge University Press, Cambridge
Ashrafzadeh MR, Khosravi R, Adibi MA, Taktehrani A, Wan HY, Cushman SA (2020) A multi-scale, multi-species approach for assessing effectiveness of habitat and connectivity conservation for endangered felids. Biol Conserv 245:108523
Austin SC, Tewes ME, Grassman LI, Silvy NJ (2007) Ecology and conservation of the leopard cat Prionailurusbengalensis and clouded leopard Neofelisnebulosa in Khao Yai National Park, Thailand. Acta Zoologica Sinica 53:1–14
Bitetti MSD, Angelo CDD, Blanco YED, Paviolo A (2010) Niche partitioning and species coexistence in a Neotropical felid assemblage. Acta Oecologia 36:403e412
Bu H, Wang F, McShea WJ, Lu Z, Wang D, Li S (2016) Spatial co-occurrence and activity patterns of mesocarnivores in the temperate forests of Southwest China. PloS one 11(10):e0164271
Chi Y, Wang J, Xi C, Qian T, Sheng C (2020) Spatial pattern of species richness among terrestrial mammals in China. Diversity 12:96
Chiang P-J, Pei KJ-C, Vaughan MR, Li C-F (2012) Niche relationships of carnivores in a subtropical primary forest in Southern Taiwan. Zoological Studies 51:500–511
Chutipong W, Steinmetz R, Savini T, Gale GA (2017) Assessing resource and predator effects on habitat use of tropical small carnivores. Mammal Research 62(1):21–36
Crooks KR, Grigione M, Scoville A, Scoville G (2008) Exploratory use of track and camera surveys of mammalian carnivores in the Peloncillo and Chiricahua mountains of southeastern Arizona. Southwest Nat 53:510–517
Dalerum F, Cameron EZ, Kunkel K, Somers MJ (2009) Diversity and depletions in continental carnivore guilds: implications for prioritizing global carnivore conservation. Biol Let 5(1):35–38
Datta A, Anand MO, Naniwadekar R (2008) Empty forests: large carnivore and prey abundance in Namdapha National Park, north-east India. Biol Cons 141(5):1429–1435
Din J, Ali H, Alli A, Younus M, Mehmood T, Norma-Rashid Y, Nawaz M (2017) Pastoralist-Predator Interaction at the Roof of the World: Conflict Dynamics and Implications for Conservation. Ecol and Soc 22(2):32
Dorji S, Rajaratnam R, Falconi L, Williams S, Sinha P, Vernes K (2018) Identifying conservation priorities for threatened Eastern Himalayan mammals. Conserv Biol 32:1162–1173
Dorji S, Rajaratnam R, Vernes K (2019) Mammal richness and diversity in a Himalayan hotspot: the role of protected areas in conserving Bhutan’s mammals. Biodivers Conserv 28(12):3277–3297
Edwards S, Gange AC, Wiesel I (2015) Spatiotemporal resource partitioning of water sources by African carnivores on Namibian commercial farmlands. J Zool 297(1):22–31
Grassman LI, Tewes ME, Silvy NJ, Kreetiyutanont K (2005) Spatial organization and diet of the leopard cat (Prionailurusbengalensis) in north-central Thailand. J Zool 266:45–54
Griffith D, Veech J, Marsh C (2016) cooccur: probabilistic SPECIES CO-OCCURRENCE ANALYSIS In R. J Stat Softw 69(2):1–17
Harrington LA (2007) The American mink, Mustelavison: its management and interactions with two native mustelids, the European polecat, M putorius, and the Eurasian otter. University of Oxford, Oxford, UK, Lutra lutra
Johnson A, Vongkhamheng C, Hedemark M, Saithongdam T (2006) Effects of human-carnivore conflict on tiger (Pantheratigris) and prey populations in Lao PDR. Anim Conserv 9(4):421–430
Johnson A, Vongkhamheng C, Saithongdam T (2009) The diversity, status and conservation of small carnivores in a montane tropical forest in northern Laos. Oryx 43(4):626–633
Karanth KU Srivathsa A, Vasudev D, Puri M, Parameshwaran R, Kumar NS (2017) Spatio-temporal interactions facilitate large carnivore sympatry across a resource gradient. Proc Royal Soc B: Biol Sci 284:20161860
Katel O, Pradhan S, Schmidt-Vogt D (2015) A survey of livestock losses caused by Asiatic wild dogs, leopards and tigers, and of the impact of predation on the livelihood of farmers in Bhutan. Wildl Res 41:300–310
Kauffman MJ, Sanjayan M, Lowenstein J, Nelson A, Jeo RM, Crooks KR (2007) Remote camera-trap methods and analyses reveal impacts of rangeland management on Namibian carnivore communities. Oryx 41(1):70–78
Kawanishi K, Sunquist ME (2004) Conservation status of tigers in a primary rainforest of Peninsular Malaysia. Biol Cons 120(3):329–344
Kennedy B, Brown WY, Vernes K, Kortner G, Butler JRA (2018) Dog and cat interactions in a remote aboriginal community. Animals 8(5):65
Marneweck C, Butler AR, Gigliotti LC, Harris SN, Jensen AJ, Muthersbaugh M, Newman BA, Saldo EA, Shute K, Titus KL, Yu SW, Jachowski DS (2021) Shining the spotlight on small mammalian carnivores: global status and threats. Biol Conserv 255:1009005
Meek PD, Ballard G-A, Vernes K, Fleming PJ (2015) The history of wildlife camera trapping as a survey tool in Australia. Australian Mammalogy 37(1):1–12
Mittermeier RA, Turner WR, Larsen FW, Brooks TM, Gascon C (2011) Global biodiversity conservation: the critical role of hotspots. In “Biodiversity Hotspots.” (Eds. F Zachos and J Habel) pp. 3–22. Springer, Berlin, Heidelberg
Mohammadi A, Kaboli M, López-Bao JV (2017) Interspecific killing between wolves and golden jackals in Iran. Eur J Wildl Res 63(4):61
Morse DH (1974) Niche breadth as a function of social dominance. Am Nat 108:818–830
Naniwadekar R, Shukla U, Viswanathan A, Datta A (2013) Records of small carnivores from in and around Namdapha Tiger Reserve, Arunachal Pradesh, India. Small Carnivore Conservation 49:1–8
Odden M, Wegge P, Fredriksen T (2010) Do tigers displace leopards? If so, why? Ecol Res 25:875–881
Oliveira TGD, Pereira JA (2014) Intraguild predation and interspecific killing as structuring forces of carnivoran communities in south America. J Mamm Evol 21:427e436
Palomares F, Caro TM (1999) Interspecific killing among mammalian carnivores. Am Nat 153:492–508
Pandit MK, Sodhi NS, Koh LP, Bhaskar A, Brook BW (2007) Unreported yet massive deforestation driving loss of endemic biodiversity in Indian Himalaya. Biodiversity Conservation 16:153–163
Pasanen-Mortensen M, Pyykönen M, Elmhagen B (2013) Where lynx prevail, foxes will fail - limitation of a mesopredator in Eurasia. Glob Ecol Biogeogr 22:868e877
Procheş Ş, Ramdhani S (2012) The world’s zoogeographical regions confirmed by cross-taxon analyses. Bioscience 62:260–270
Rajaratnam R, Sunquist M, Rajaratnam L, Ambu L (2007) Diet and habitat selection of the leopard cat (Prionailurusbengalensisborneoensis) in an agricultural landscape in Sabah, Malaysian Borneo. J Trop Ecol 23:209–217
Rajaratnam R, Vernes K, Sangay T (2016) A review of livestock predation by large carnivores in the Himalayan Kingdom of Bhutan. In Angelici F (ed) Problematic Wildlife. Springer, Cham, pp 143–171
Ramesh T, Kalle R, Downs CT (2017) Staying safe from top predators: patterns of co-occurrence and inter-predator interactions. Behav Ecol Sociobiol 71(2):41
Rasphone A, Kamler J, Macdonald D (2020) Temporal partitioning by felids, dholes and their potential prey in northern Laos. Mammal Research 65:679–689
Ripple WJ, Estes JA, Beschta RL, Wilmers CC, Ritchie EG, Hebblewhite M, Berger J, Elmhagen B, Letnic M, Nelson MP, Schmitz OJ, Smith DW, Wallach AD, Wirsing AJ (2014) Status and ecological effects of the world’s largest carnivores. Science 343(6167):1241484
Roemer GW, Gompper ME, Valkenburgh BV (2009) The ecological role of the mammalian mesocarnivores. Bioscience 165–173
Sanderson J, Harris G (2013) Automatic data organization, storage, and analysis of camera trap pictures. Journal of Indonesian Natural History 1(1):11–19
Sangay T, Rajaratnam R, Vernes K (2014) Wildlife camera trapping in the Himalayan kingdom of Bhutan with recommendations for the future. In “Camera Trapping: Wildlife Management and Research.” (Eds. G Ballard, P Banks, A Claridge, J Sanderson and D Swann) pp. 87–98. CSIRO Publishing, Collingwood
Sangay T, Vernes K (2008) Human-wildlife conflict in the Kingdom of Bhutan: patterns of livestock predation by large mammalian carnivores. Biol Cons 141(5):1272–1282
Sangay T, Vernes K (2014) The economic cost of wild mammalian carnivores to farmers in the Himalayan Kingdom of Bhutan. Proceedings of the Bhutan Ecological Society 1:98–111
Schaller GB (1967) The Deer and the tiger. Chicago, USA, University of Chicago Press
Schuette P, Wagner AP, Wagner ME, Creel S (2013) Occupancy patterns and niche partitioning within a diverse carnivore community exposed to anthropogenic pressures. Biol Cons 158:301–312
Seidensticker J (1976) On the ecological separation between tigers and leopards. Biotropica 8:225–234
Steinmetz R, Seuaturien N, Chutipong W (2013) Tigers, leopards, and dholes in a half-empty forest: assessing species interactions in a guild of threatened carnivores. Biol Cons 163:68–78
Tempa T, Hebblewhite M, Mills LS, Wangchuk TR, Norbu N, Wangchuk T, Nidup T, Dendup P, Wangchuk D, Wangdi Y, Dorji T (2013) Royal Manas National Park, Bhutan: a hot spot for wild felids. Oryx 47:207–210
Tharchen L (2013) Protected areas and biodiversity of Bhutan. Kolkata, CDC Printers
Thinley P, Rajaratnam R, Lassoie JP, Morreale SJ, Curtis PD, Vernes K, Leki L, Phuntsho S, Dorji T, Dorji P (2018) The ecological benefit of tigers (Pantheratigris) to farmers in reducing crop and livestock losses in the eastern Himalayas: Implications for conservation of large apex predators. Biol Cons 219:119–125
Veech JA (2013) A probabilistic model for analysing species co-occurrence. Glob Ecol Biogeogr 22:252–260
Velho N, Karanth KK, Laurance WF (2012) Hunting: a serious and understudied threat in India, a globally significant conservation region. Biol Cons 148(1):210–215
Vernes K, Sangay T, Rajanathan R, Singye R (2015) Social interaction and cooccurrence of colour morphs of the Asiatic golden cat, Bhutan. Cat News 62:18–20
Wangchuk T, Thinley P, Tshering K, Tshering C, Yonten D, Pema B (2004) A field guide to the mammals of Bhutan. Thimphu, Bhutan, Department of Forestry, Ministry of Agriculture, Royal Government of Bhutan
Wong GJ (1997) The activity pattern and population dynamics of Siberian weasel (Mustela sibirica) at Fushan forest. National Taiwan University, Taipei
Zhao G, Yang H, Xie B, Gong Y, Ge J, Feng., L. (2020) Spatio-temporal coexistence of sympatric mesocarnivores with a single apex carnivore in a fine scale landscape. Glob Ecol Conserv 21:e00897
Acknowledgements
We would like to sincerely acknowledge the survey team members from Jigme Singye Wangchuck National Park (Dorji Duba, Yeshi Wangdi, Rinchen Dorji, Sangay Tshewang, Kado Rinchen, Dhanapati, Tshering, and Sonam Rinchen) for their utmost dedication and hard work in assisting with much of the data collection, and the Department of Forests and Park Services for granting approval to undertake this work. We also thank the University of New England, Royal Government of Bhutan, and WWF Bhutan Program for funding. Dr. Joseph Veech and Dr. Nicolas Gotelli provided advice on some of the analyses, for which, we are most grateful.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: The co-author’s surname the letter “i” was missing. The name should been Sangay Dorji instead of Sangay Dorj.
Rights and permissions
About this article
Cite this article
Vernes, K., Rajaratnam, R. & Dorji, S. Patterns of species co-occurrence in a diverse Eastern Himalayan montane carnivore community. Mamm Res 67, 139–149 (2022). https://doi.org/10.1007/s13364-021-00605-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13364-021-00605-3