Abstract
For a century, fingermark analysis has been one of the most important and common methods in forensic investigations. Modern chemical analysis technologies have added the potential to determine the molecular composition of fingermarks and possibly identify chemicals a suspect may have come into contact with. Improvements in analytical detection of the molecular composition of fingermarks is therefore of great importance. In this regard, matrix-assisted laser desorption ionization (MALDI) and laser desorption ionization (LDI) imaging mass spectrometry (IMS) have proven to be useful technologies for fingermark analysis. In these analyses, the choice of ionizing agent and its mode of deposition are critical steps for the identification of molecular markers. Here we propose two novel and complementary IMS approaches for endogenous and exogenous substance detection in fingermarks: sublimation of 2-mercaptobenzothiazol (2-MBT) matrix and silver sputtering.

ᅟ
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fingerprint evidence is one of the most common methods of biometric identification. There are a variety of techniques for the visualization of latent fingerprints on porous (paper, cotton, wood), semi-porous (plastic, waxy surfaces), and non-porous (glass, metal, paint) surfaces, including powder methods [1], chemical methods (ninhydrin, DFO) [2], spectroscopic imaging approaches (IR, Raman) [3, 4], and nanotechnology-based technologies [5]. Interestingly, the molecular composition of a latent fingermark can lead to additional forensic evidence and may assist law enforcers in the case of fingerprint matching process failure. [6–8]. The detection of latent fingermarks is a considerably challenging analytical problem. Fingermarks are complex, composed of a mixture of contaminants derived from the environment, secretion of metabolites from external uptake, and natural secretions from the body, including proteins, peptides, amino acids, vitamins, salts, fatty acids, wax esters, diglycerides (DAGs), triglycerides (TAGs), cholesterol, squalene, and more [9, 10]. Further, latent fingermark deposits behave differently on different substrate types. In this regard, mass spectrometry (MS) has proven to be a useful technology to monitor the molecular composition of fingermarks. Much of the prior research for the detection and identification of endogenous substances in fingermarks has been carried out using gas chromatography-mass spectrometry (GC-MS). Despite the fact that this technique is destructive and limited to a small class of compounds (<500 Da), it allows the detection of endogenous substances, such as amino acids, fatty acids, wax esters, squalene, and cholesterol, as well as the detection of some exogenous substances coming from hair product, parfum residue, cosmetics, and skin lotion [11–16]. Many nondestructive MS techniques have been developped for the detection of endogenous and exogenous substances from fingermarks. As an exemple, desorption electrospray ionization (DESI) MS has been successfully applied to several exogenous substances and external uptake detection [17]. Surface assisted-laser desorption/ionization-time-of-flight (SALDI-TOF) MS has been carried out for the detection of some fatty acids, dehydrated cholesterol, squalene, as well as the identification of explosives, drugs, and nicotine residues [8, 18, 19]. More recently, matrix-assisted-laser desorption/ionization (MALDI) TOF MS has been reported for the detection of endogenous substances, such as amino acids, fatty acids, peptides, dehydrated cholesterol, squalene, DAGs, and TAGs, as well as surfactants, polymers, drugs, pharmaceuticals, and explosives [20–22].
Imaging mass spectrometry (IMS) [23–25] is particularly well suited for investigating fingermarks because it combines suspect identification by visualizing the ridge pattern defined by the molecular images, and the chemical composition for further forensic evidence. Despite proven capabilities in fingermark analysis, for the detection and mapping of endogenous and exogenous markers, MALDI IMS critically relies on the nature of the matrix and its mode of deposition that may need tailoring to the chemistry of the interested species. In this regard, extensive research has been performed to improve the analytical detection for numerous samples by minimizing the analyte delocalization, to improve the sensitivity and the imaging spatial resolution. The first application of IMS has been reported by Ifa et al. for the detection of drugs of abuse and explosives on latent fingermark by DESI MS [7]. MALDI IMS introduced by Francese et al. has also proven to be a powerful tool for fingermark analysis. Initially, fingermark IMS was performed by depositing the MALDI matrix, α-cyano-4-hydroxycinnamic acid (CHCA), by spray-coating [26]. This procedure allows both visual and chemical information to be obtained in one analysis. Subsequently, the search for more efficient protocols for matrix deposition has resulted in the development of a novel two-step procedure, named “dry-wet” that consist of dusting the MALDI matrix CHCA onto the fingermark followed by solvent spray deposition [27]. This method has proven to be more efficient on fingermarks by improving the imaging spatial resolution and increasing the ion abundance and intensity. Moreover, they demonstrated the versatility and the robustness of fingermark IMS methods for different forensic situations, contributing to additional evidences [28], including the determination of the presence of blood for criminal investigation [29].
In this paper, we demonstrate the potential of two novel MALDI- and LDI-based imaging methods for the detection of endogenous and exogenous substances from fingermarks. Recently, matrix sublimation was demonstrated to be a powerful approach for very homogeneous matrix deposition that allows for the detection and IMS of small molecules and proteins with high sensitivity and high spatial resolution [30–32]. In the first approach proposed herein, we provide an alternative to the “dry-wet” method using sublimation for homogeneous matrix deposition followed by automated solvent spray deposition. To this end, we screened several matrix candidates and found that 2-mercaptobenzothiazole (2-MBT) offered similar and complementary results to this method. In the second approach, we tested the potential of silver assisted laser desorption ionization (AgLDI) for IMS of fingermarks. Previous research has demonstrated the potential of metal sputtering, such as gold for the detection of deprotonated fatty acids and hazardous substrates [33] and the use of silver nanoparticles for the detection of exogenous drug compounds [34] from fingermarks. AgLDI IMS was recently proposed by our laboratory and allows the imaging of cholesterol, fatty acids, and other olefin-containing molecules from thinly cut tissue sections with high specificity and sensitivity [35]. Surprisingly, AgLDI has demonstrated to be a powerful tool for fingermark analysis. The deposition of metallic silver by sputtering on fingermark allows high-resolution IMS and higher sensitivity of several class of biomolecules and some exogenous substances. Ultimately, we found that both approaches provide complementary results that could significantly contribute to forensic investigations.
Experimental
Chemicals and Materials
1,5-Diaminonaphtalene (DAN), 2,5-dihydroxybenzoic acid (DHB), α-cyano-4-hydroxycinnamic acid (CHCA), 2′,4′,6′-trihydroxyacetophenone (THAP), 2-mercaptobenzothiazole (2-MBT), L-amino acids kit, squalene, dimethyldioctadecylammonium bromide, oleic acid, stearic acid, 1,2-dioleoyl-sn-glycerol, trifluoroacetic acid (TFA), and liquid chromatography grade solvents were purchased from Sigma-Aldrich (St. Louis, MO, USA). 1,2-Dipalmitoyl-sn-glycero-3-phosphate was purchased from Avanti Polar Lipids (Alabaster, AL, USA). 1-Palmitoyl-2-oleoyl-3-linoleoyl-rac-glycerol was purchased from Cayman Chemical (Orlando, FL, USA). 9-Aminoacridine (9-AA) was purchased from Acros Organics (Morris Plains, NJ, USA). Ethanol (EtOH) was purchased from Greenfield Ethanol Inc. (Boucherville, QC, Canada). Double-sided carbon conductive tape was purchased from Ted Pella, Inc. (Redding, CA, USA).
Fingermark Preparation
The slides were washed in acetone to ensure complete cleanness prior to fingermark deposition. Latent groomed and ungroomed fingermarks were prepared on ITO coated slides (Delta Technologies, Loveland, CO, USA) according to published preparation methods [26]. The same procedure was followed for fingermark deposition on sheets of paper. Fingermarks were obtained in a de-identified manner. All procedures were approved by the Health Canada Research Ethics Board and comply with the Ethical Principles set out in Tri-Council Policy Statement: Ethical Conduct for Research Involving Humans guidelines.
Matrix Deposition
Solvent-free matrix deposition on fingermarks was carried out in a sublimation apparatus (Chemglass Life Science, Vineland, NJ, USA) as previously described [30]. The temperature and the time of sublimation were optimized for each matrix to ensure the best coverage and MALDI efficiency (number of signals detected and signal to noise). To enhance MS signals, recrystallization of the matrix was performed by spraying the surface with a solution of 70:30 EtOH:water at 30°C using a TM-Sprayer (HTX LC-Transform). Ten passes of solvent over the surface of the fingermarks were deposited on the sublimated 2-MBT matrix with a lateral travel velocity and debit of 1.2 m/min and 13.5 mL/min, respectively. Each solvent pass was separated by 5 mm and for every even numbered pass an offset of 2.5 mm was applied leading to a homogenous solvent layer deposition. Recrystallization of the sublimated matrix CHCA and DHB have also been performed by spraying the surface with a solution of 70:30 ACN:water and 70:30 MeOH:water at 30°C, respectively, using the same instrument parameters.
Matrix was also deposited on fingermarks using the ImagePrep spray system (Bruker Daltonics, Billerica, MA, USA) starting from the default CHCA method and optimized for each matrix studied. 2-MBT was prepared at a concentration of 20 mg/mL in 70:30 EtOH:water, CHCA was prepared at 7 mg/mL in 50:50 acetonitrile:water, DAN was prepared at 5 mg/mL in 70:30 EtOH:water, and DHB was prepared at 25 mg/mL and THAP at 10 mg/mL, both in 50:50 MeOH:water. Finally, 9-AA was prepared at 10 mg/mL in 50:50 EtOH:water. For fingermarks analyzed in positive ionization mode, 0.1% of TFA was added to the matrix solution.
Silver Sputter Coating
Metallic silver was sputtered on top of fingermarks using a Cressington 308R sputter coater (Ted Pella, Inc., Redding, CA, USA) as previously described [35]. For optimal results, the deposited silver layer thickness was optimized at 14 ± 2 nm corresponding to 30 s of deposition.
IMS of Fingermarks on Paper
Double-sided tape was first applied to a glass slide (Supplementary Figure 8). The piece of paper containing the fingermark was then mounted on the tape using an aluminum sheet to press down on the piece of paper to minimize alteration of the fingermark. Subsequent silver deposition was performed as described above.
Mass Spectrometry Instrumentation
Profiling and IMS of fingermarks were performed on either a MALDI TOF/TOF Ultraflextreme mass spectrometer or a MALDI FT/ICR SolariX XR 7 T mass spectrometer both equipped with SmartBeam II Nd:YAG 355 nm lasers operating at 1000 Hz or at 250 Hz for porous surface analyses (Bruker Daltonics). For IMS data acquisition, 200 shots were summed per array position with a lateral resolution of 75 μm using the “medium” focus setting or with a lateral resolution of 10 μm using the “minimum” focus setting. Using these parameters, we achieved imaging speeds of ~70 pixels/min. For a whole fingermark at 75 μm of spatial resolution, IMS acquisition required approximately 5 h. Profiling and IMS data from the Ultraflextreme were performed in reflectron geometry under optimized delayed extraction conditions with a source accelerating voltage of +25 kV, in a mass range of 100–1100 Da. For MALDI IMS, external calibration was performed using a homemade mix of standards (lysine, stearic acid, 1,2-dioleoyl-sn-glycerol, and 1-palmitoyl-2-oleoyl-3-linoleoyl-rac-glycerol). Subsequently, internal calibration was performed with known species including the [M + H]+ and [2 M – H]+ matrix signal (168.00 and m/z 332.96, respectively) and the [M]+ DTDMAC ion at m/z 550.63 (see below). For AgLDI IMS, internal calibration was carried out in quadratic mode using the silver clusters (Ag1 to Ag9, see Fig. 4) present in all the spectra. IMS data were reconstructed and visualized without normalization using the FlexImaging 3.0 software (Bruker Daltonics). Lipid identification was performed by MALDI/AgLDI tandem MS on the Ultraflextreme mass spectrometer in combination with the LIPID MAPS prediction tool by comparing accurate mass measurements (www.lipidmaps.org/tools/index.html).
Results and Discussion
Evaluation of Matrix Candidates for Sublimation and IMS
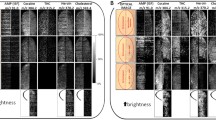
Initial sample preparation tests were performed with several MALDI matrices using two different modes of deposition; direct matrix spray coating (Image Prep system) or sublimation followed by solvent spray deposition. The solvent system employed in matrix spray coating enables a better co-crystallization of some analytes, enhancing their signal. However, depending on the matrix and solvent used, we found that this mode of deposition produces some analyte delocalization, which in many cases prevented the proper visualization of the fingerprint motif (Supplementary Figure 3). Similar comparisons between the dry-wet method and spray coating have also been performed, leading to the conclusion that the initial dry deposition on fingermarks allowed higher ion signal intensity and enhanced clarity of the ridge details [36]. Sublimation is an interesting approach for matrix deposition on fingermarks because of its high reproducibility, the absence of analyte delocalization, and capability to detect numerous small molecules by MALDI IMS. Six MALDI matrices that were previously investigated by sublimation for lipid IMS analyses from tissue sections were tested on latent fingermarks [37]. IMS analyses were performed in positive and negative ionization modes. As shown in Table 1, for each matrix tested, the sublimation time and temperature were optimized to obtain the best MS signals. Their performance was evaluated using the following analytical criteria: (1) MS signal quality: intensity and number of compounds detected based on existing literature for fingermark analysis; (2) uniformity and crystal size for optimal IMS spatial resolution (Supplementary Figure 4); and (3) number of background signals. Overall, 2-MBT showed the best performance and reproducibility for fingermark IMS analysis in positive ionization mode, when considering the sum of MS signals observed from fingermarks and the intensity of background signals (Supplementary Figures 5 and 6). More importantly, its sublimation on latent fingermarks allows the detection of numerous exogenous and endogenous substances, possibly making 2-MBT a valuable matrix for forensic screening (Fig. 1). For example, the ions detected at m/z 494.6, 522.6, and 550.6 have previously been identified as a ditallowdimethylammonium ions (DTDMAC) and originate from personal care and domestic products [38]. In a previous publication, polymer analysis by MALDI MS has been performed on fingermarks using dithranol, instead of CHCA, allowing a better detection efficiency [39]. Polymeric substances such as polyethylene glycol (PEG) and polypropylene glycol (PPG) can also be easily observed on ungroomed fingermarks using the matrix 2-MBT and often originate from hand lotions and other beauty products (Fig. 1). For endogenous substance detection, previous studies have demonstrated the efficiency of 2-MBT as a MALDI matrix for tissue analysis of lipids after application by spray deposition [40]. Its deposition by sublimation on fingermarks allows the detection of amino acids, sodiated wax esters, diglycerides (DAGs), triglycerides (TAGs), and some fatty acids, such as oleic and stearic acids (Supplementary Table 1). Figure 2 shows IMS results of the most abundant exogenous and endogenous substances detected in fingermarks. With data acquisition at a spatial resolution of 75 μm, the fingermark ridge pattern is clearly visible when comparing the relative intensities of the displayed ions and is more clearly observed when looking at the total ion chromatogram (TIC) image (sum of signal intensities between m/z 300 and 1100).
Typical MALDI-TOF mass spectra acquired after 2-MBT deposition by sublimation on ungroomed fingermarks in the positive ionization mode. Numerous endogenous compounds such as DAGs and TAGs can be observed along with exogenous compounds such as DTDMAC. In some cases, polypropylene glycol (PPG) differing by 58 mass units as potassium adducts (★) and polyethylene glycol (PEG) differing by 44 mass units for sodium (✦) and potassium (✱) adducts, respectively, can be observed
MALDI-TOF IMS analysis of a latent fingermark acquired at 75 μm spatial resolution after 2-MBT matrix deposition by sublimation and recrystallization. Top left corner, photomicrograph of the fingermark after matrix deposition. TIC = total ion current (sum of all MS signal intensities between m/z 300 and 1100)
Fingermark IMS after Silver Sputtering
Silver sputter deposition is a novel method developed for IMS that allows the privileged detection of olefin containing molecules observed as [M + Ag]+ ions directly from tissue sections [35]. Silver adducts are easily determined by the presence of the 107Ag and 109Ag isotopic pattern, with relative abundances of 52% and 48%, respectively. After silver layer deposition on fingermarks and analysis by LDI-TOF MS, numerous [M + Ag]+ ions have been detected such as cholesterol, squalene, wax esters, fatty acids, and TAGs (Supplementary Table 2). In addition, the presence of Na+ and K+ in fingermark residues allow us to detect sodium and potassium adducts of DAGs and TAGs. Some exogenous substances were also observed, including DTDMAC and polymeric substances, such as PEG (Supplementary Figure 7). Figure 3a shows some endogenous and exogenous ion images acquired from a latent fingermark by AgLDI-TOF MS. The whole fingermark was imaged again with a lateral resolution of 75 μm, allowing a good definition of the fingermark pattern. As expected, most of the compounds were detected from the ridge of the fingerprint. Silver sputter deposition, however, also allows IMS acquisition at high spatial resolution [35]. As demonstrated in Fig. 3b, a small area of a fingermark was imaged at 10 μm of lateral resolution, revealing information such as the minutiae points, pores, and ridge shape. The optical image after silver deposition is enough to provide this forensic information, but it is interesting to look at the molecular distribution of compounds, such as oleic acid (OA) at m/z 391.2 and the [TAG(50:1) + Na]+ at m/z 855.7, with the latter displaying some correlation with the fingerprint pores. Fingermark IMS after silver sputter deposition was also performed using a MALDI FT/ICR mass spectrometer, providing higher mass resolving power and increased sensitivity compared with the TOF MS instrument. The high mass resolution scan provides an exact mass measurement for compound identification. As a result, the detection of even more endogenous compounds, including fatty acids, wax esters, DAGs, and TAGs, was possible (Fig. 4). Moreover, with the high sensitivity of the FTMS instrument, small fatty acids with odd numbers of carbons were also observed and imaged (Fig. 5). Such species are likely to come from bacteria since the human body typically produces fatty acids with even numbers of carbons. It is also interesting to note that odd number fatty acids were predominately observed in the valley of the fingermarks. The fingerprint valleys being less prone to friction with respect to the ridges may create a better environment for bacterial proliferation. This observation opens the possibility of identifying pathogens or other biological agents carried or manipulated by suspects directly from fingermarks.
(a) AgLDI-TOF IMS of a latent fingermark acquired at 75 μm spatial resolution. Top left corner, photomicrograph of the fingermark after silver sputter deposition. (b) AgLDI-TOF IMS of a small area from a latent fingermark acquired at 10 μm spatial resolution. (Top) Oleic acid [OA + 109Ag]+ at m/z 391.17 and (Bottom) [TAG(50:1) + Na]+ at m/z 855.74. *Composite image of both oleic acid [OA + 109Ag]+ and stearic acid [SA + 107Ag]+. Stearic acid (SA) is typically observed in much lower intensity than oleic acid in fingermarks
AgLDI-FTICR IMS of a latent fingermark after silver deposition acquired at 75 μm spatial resolution. (a) Hierarchical clustering using ClinPro Tool (Bruker Daltonics). Composite images of the distribution of arachidonic acid [FA(20:4) + 107Ag]+ with (b) margaric acid [FA(17:0) + 107Ag]+, (c) tridecylic acid [FA(13:0) + 107Ag]+ and (d) pentadecylic acid [FA(15:0) + 107Ag]+
Ideally, for IMS analyses the fingermarks are deposited under controlled conditions on (ITO-coated conductive) glass slides. However, in forensic investigations, fingermarks can potentially be found on a variety of nonconductive surfaces. By using layers of sputtered silver, the target surface is rendered conductive, which then allows analyses of latent fingermarks made on nonconductive surfaces by TOF IMS. To demonstrate this potential, AgLDI IMS of fingermarks deposited on a sheet of paper was investigated. Interestingly, the fingerprint motif was clearly observed on the paper after silver deposition, and numerous compounds such as squalene, wax esters, fatty acids, DAGs, TAGs, as well as polymeric substances were detected by IMS (Fig. 6). However, many sample preparation issues remain for IMS of latent fingermarks on porous surfaces such as paper. First, the paper needs to be properly applied on the double-sided tape without damaging the fingermark. Second, the ideal amount of silver deposited will vary depending on the amount of fingermark residue on the surface. Nevertheless, this method opens the opportunity for using silver sputtering on many nonconductive surfaces, such as cartbord, glass, or plastic, and analyzing lifted fingermarks directly from the tape.
AgLDI-TOF IMS of a latent fingermark on a piece of paper acquired at 75 μm spatial resolution. (a) and (b) Photomicrograph of the fingermark after silver sputter deposition. The box in panel a (enlarged in panel b) represents the area analyzed by IMS. (c) Ion image of oleic acid [OA + 109Ag]+ at m/z 391.2
Conclusions
We propose herein two novel and alternative means of preparation for investigating the molecular composition of fingermarks by IMS. The first approach is an extension of the “dry-wet” method proposed by the Francese laboratory where finely crushed dry matrix is manually deposited on the fingermarks using a specialized brush [27]. Based on the matrix crystal size, such an approach may not achieve 100% surface coverage and may also be limiting when used to investigate lower abundance fingermark residues [36]. We, however, chose here to deposit the matrix by sublimation, since it allows controlled deposition of very homogeneous matrix coatings without analyte delocalization. Indeed, no direct contact is made with the fingermark during the sublimation process. Automated solvent spray deposition is then performed to locally dissolve the matrix and allow sample co-crystallization. Several different matrices were tested and 2-MBT was found to be the most efficient matrix for IMS of exogenous (DTDMAC and polymers) and endogenous (some fatty acids, amino acids, sodiated wax esters, DAGs, and TAGs) substances from latent fingermarks. The second approach investigated is AgLDI IMS. In this case, a nanometer thin layer of silver is deposited on the fingermarks, which allows the detection and imaging of numerous endogenous compounds such as cholesterol, squalene, wax esters, DAGs, TAGs, fatty acids, as well as other exogenous substances. With AgLDI IMS, odd carbon number fatty acids presumably coming from skin bacteria were also detected and localized to the fingerprint valley. This opens the possibility to detect biological agents carried or manipulated by suspects directly from fingermarks. We also demonstrated that AgLDI IMS of fingermarks can also be performed on nonconductive surfaces such as paper. Ultimately, both IMS approaches give complementary information on the molecular composition of fingermarks, which could be of use in forensic science.
References
Sodhi, G.S., Kaur, J.: Powder method for detecting latent fingerprints: a review. Forensic Sci. Int. 120, 172–176 (2001)
Payne, G., Reedy, B., Lennard, C., Comber, B., Exline, D., Roux, C.: A further study to investigate the detection and enhancement of latent fingerprints using visible absorption and luminescence chemical imaging. Forensic Sci. Int. 150, 33–51 (2005)
Chen, T., Schultz, Z.D., Levin, I.W.: Infrared spectroscopic imaging of latent fingerprints and associated forensic evidence. Analyst 134, 1902–1904 (2009)
Tahtouh, M., Scott, S.A., Kalman, J.R., Reedy, B.J.: Four novel alkyl 2-cyanoacylate monomers and their use in latent fingermark detection by mid-infrared spectral imaging. Forensic Sci. Int. 207, 223–238 (2011)
Choi, M.J., McDonagh, A.M., Maynard, P., Roux, C.: Metal-containing nanoparticles and nano-structured particles in fingermark detection. Forensic Sci. Int. 179, 87–97 (2008)
Bradshaw, R., Wolstenholme, R., Ferguson, L.S., Sammon, C., Mader, K., Claude, E., Blackledge, R.D., Clench, M.R., Francese, S.: Spectroscopic imaging based approach for condom identification in condom contaminated fingermarks. Analyst 138, 2546–2557 (2013)
Ifa, D.R., Manicke, N.E., Dill, A.L., Cooks, R.G.: Latent fingerprint chemical imaging by mass spectrometry. Science 321, 805 (2008)
Benton, M., Chua, M.J., Gu, F., Rowell, F., Ma, J.: Environmental nicotine contamination in latent fingermarks from smoker contacts and passive smoking. Forensic Sci. Int. 200, 28–34 (2010)
Knowles, A.M.: Aspects of physicochemical methods for the detection of latent fingerprints. J. Phys. E 11, 713–721 (1978)
Ramotowski, R.S.: Composition of Latent Print Residue. In: Lee, H.C., Gaensslen, R.E. (eds.) Advances in fingerprint technology, 2nd edn, pp. 63–104. CRC Press, Boca Raton, FL (2001)
Archer, N.E., Charles, Y., Elliott, J.A., Jickells, S.: Changes in the lipid composition of latent fingerprint residue with time after deposition on a surface. Forensic Sci. Int. 154, 224–239 (2005)
Croxton, R.S., Baron, M.G., Butler, D., Kent, T., Sears, V.G.: Variation in amino acid and lipid composition of latent fingerprints. Forensic Sci. Int. 199, 93–102 (2010)
Croxton, R.S., Baron, M.G., Butler, D., Kent, T., Sears, V.G.: Development of a GC-MS method for the simultaneous analysis of latent fingerprint components. J. Forensic Sci. 51, 1329–1333 (2006)
Girod, A., Ramotowski, R., Weyermann, C.: Composition of fingermark residue: a qualitative and quantitative review. Forensic Sci. Int. 223, 10–24 (2012)
Girod, A., Weyermann, C.: Lipid composition of fingermark residue and donor classification using GC/MS. Forensic Sci. Int. 238, 68–82 (2014)
Weyermann, C., Roux, C., Champod, C.: Initial results on the composition of fingerprints and its evolution as a function of time by GC/MS analysis. J. Forensic Sci. 56, 102–108 (2011)
Morelato, M., Beavis, A., Kirkbride, P., Roux, C.: Forensic applications of desorption electrospray ionisation mass spectrometry (DESI-MS). Forensic Sci. Int. 226, 10–21 (2013)
Rowell, F., Hudson, K., Seviour, J.: Detection of drugs and their metabolites in dusted latent fingermarks by mass spectrometry. Analyst 134, 701–707 (2009)
Rowell, F., Seviour, J., Lim, A.Y., Elumbaring-Salazar, C.G., Loke, J., Ma, J.: Detection of nitro-organic and peroxide explosives in latent fingermarks by DART- and SALDI-TOF-mass spectrometry. Forensic Sci. Int. 221, 84–91 (2012)
Kaplan-Sandquist, K., LeBeau, M.A., Miller, M.L.: Chemical analysis of pharmaceuticals and explosives in fingermarks using matrix-assisted laser desorption ionization/time-of-flight mass spectrometry. Forensic Sci. Int. 235, 68–77 (2014)
Sundar, L., Rowell, F.: Detection of drugs in lifted cyanoacrylate-developed latent fingermarks using two laser desorption/ionisation mass spectrometric methods. Analyst 139, 633–642 (2014)
Francese, S., Bradshaw, R., Ferguson, L.S., Wolstenholme, R., Clench, M.R., Bleay, S.: Beyond the ridge pattern: multi-informative analysis of latent fingermarks by MALDI mass spectrometry. Analyst 138, 4215–4228 (2013)
Chughtai, K., Heeren, R.M.: Mass spectrometric imaging for biomedical tissue analysis. Chem. Rev. 110, 3237–3277 (2010)
Chaurand, P.: Imaging mass spectrometry of thin tissue sections: a decade of collective efforts. J. Proteome. 75, 4883–4892 (2012)
Norris, J.L., Caprioli, R.M.: Analysis of tissue specimens by matrix-assisted laser desorption/ionization imaging mass spectrometry in biological and clinical research. Chem. Rev. 113, 2309–2342 (2013)
Wolstenholme, R., Bradshaw, R., Clench, M.R., Francese, S.: Study of latent fingermarks by matrix-assisted laser desorption/ionisation mass spectrometry imaging of endogenous lipids. Rapid Commun. Mass Spectrom. 23, 3031–3039 (2009)
Ferguson, L., Bradshaw, R., Wolstenholme, R., Clench, M., Francese, S.: Two-step matrix application for the enhancement and imaging of latent fingermarks. Anal. Chem. 83, 5585–5591 (2011)
Bradshaw, R., Bleay, S., Wolstenholme, R., Clench, M.R., Francese, S.: Towards the integration of matrix assisted laser desorption ionisation mass spectrometry imaging into the current fingermark examination workflow. Forensic Sci. Int. 232, 111–124 (2013)
Bradshaw, R., Bleay, S., Clench, M.R., Francese, S.: Direct detection of blood in fingermarks by MALDI MS profiling and imaging. Sci. Justice 54, 110–117 (2014)
Chaurand, P., Cornett, D.S., Angel, P.M., Caprioli, R.M.: From whole-body sections down to cellular level, multiscale imaging of phospholipids by MALDI mass spectrometry. Mol. Cell. Proteomics 10, O110.004259 (2011)
Hankin, J.A., Barkley, R.M., Murphy, R.C.: Sublimation as a method of matrix application for mass spectrometric imaging. J. Am. Soc. Mass Spectrom. 18, 1646–1652 (2007)
Yang, J., Caprioli, R.M.: Matrix sublimation/recrystallization for imaging proteins by mass spectrometry at high spatial resolution. Anal. Chem. 83, 5728–5734 (2011)
Tang, H.W., Lu, W., Che, C.M., Ng, K.M.: Gold nanoparticles and imaging mass spectrometry: double imaging of latent fingerprints. Anal. Chem. 82, 1589–1593 (2010)
Niziol, J., Ruman, T.: Surface-transfer mass spectrometry imaging on a monoisotopic silver nanoparticle enhanced target. Anal. Chem. 85, 12070–12076 (2013)
Dufresne, M., Thomas, A., Breault-Turcot, J., Masson, J.F., Chaurand, P.: Silver-assisted laser desorption ionization for high spatial resolution imaging mass spectrometry of olefins from thin tissue sections. Anal. Chem. 85, 3318–3324 (2013)
Ferguson, L.S., Creasey, S., Wolstenholme, R., Clench, M.R., Francese, S.: Efficiency of the dry-wet method for the MALDI-MSI analysis of latent fingermarks. J. Mass Spectrom. 48, 677–684 (2013)
Thomas, A., Charbonneau, J.L., Fournaise, E., Chaurand, P.: Sublimation of new matrix candidates for high spatial resolution imaging mass spectrometry of lipids: enhanced information in both positive and negative polarities after 1,5-diaminonapthalene deposition. Anal. Chem. 84, 2048–2054 (2012)
Manier, M.L., Cornett, D.S., Hachey, D.L., Caprioli, R.M.: Identification of dimethyldioctadecylammonium ion (m/z 550.6) and related species (m/z 522.6, 494.6) as a source of contamination in mass spectrometry. J. Am. Soc. Mass Spectrom. 19, 666–670 (2008)
Bradshaw, R., Wolstenholme, R., Blackledge, R.D., Clench, M.R., Ferguson, L.S., Francese, S.: A novel matrix-assisted laser desorption/ionisation mass spectrometry imaging based methodology for the identification of sexual assault suspects. Rapid Commun. Mass Spectrom. 25, 415–422 (2011)
Astigarraga, E., Barreda-Gomez, G., Lombardero, L., Fresnedo, O., Castano, F., Giralt, M.T., Ochoa, B., Rodriguez-Puertas, R., Fernandez, J.A.: Profiling and imaging of lipids on brain and liver tissue by matrix-assisted laser desorption/ ionization mass spectrometry using 2-mercaptobenzothiazole as a matrix. Anal. Chem. 80, 9105–9114 (2008)
Acknowledgments
The authors acknowledge Professor Jean-François Masson (Department of Chemistry, Université de Montréal) for giving them unlimited access to the silver sputter system. The authors also acknowledge Bruker Daltonics for giving them access to the FTMS instrument, and Katherine Kellersberger for her help with profiling and IMS data acquisition. The authors also acknowledge funding from the Natural Sciences and Engineering Research Council of Canada (NSERC), and the Canadian Foundation for Innovation (CFI).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 4567 kb)
Rights and permissions
About this article
Cite this article
Lauzon, N., Dufresne, M., Chauhan, V. et al. Development of Laser Desorption Imaging Mass Spectrometry Methods to Investigate the Molecular Composition of Latent Fingermarks. J. Am. Soc. Mass Spectrom. 26, 878–886 (2015). https://doi.org/10.1007/s13361-015-1123-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13361-015-1123-0