Abstract
Breast cancer (BC) is the most commonly diagnosed cancer among women. Chemo-, immune- and photothermal therapies are employed to manage BC. However, the tumor microenvironment (TME) prevents free drugs and nanocarriers (NCs) from entering the tumor premises. Formulation scientists rely on enhanced permeation and retention (EPR) to extravasate NCs in the TME. However, recent research has demonstrated the inconsistent nature of EPR among different patients and tumor types. In addition, angiogenesis, high intra-tumor fluid pressure, desmoplasia, and high cell and extracellular matrix density resist the accumulation of NCs in the TME. In this review, we discuss TME normalization as an approach to improve the penetration of drugs and NCSs in the tumor premises. Strategies such as normalization of tumor vessels, reversal of hypoxia, alleviation of high intra-tumor pressure, and infiltration of lymphocytes for the reversal of therapy failure have been discussed in this manuscript. Strategies to promote the infiltration of anticancer immune cells in the TME after vascular normalization have been discussed. Studies strategizing time points to administer TME-normalizing agents are highlighted. Mechanistic pathways controlling the angiogenesis and normalization processes are discussed along with the studies. This review will provide greater tumor-targeting insights to the formulation scientists.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Breast cancer (BC) is the most commonly diagnosed cancer among women. A total of 2,230,000 diagnoses and 685,000 deaths were reported in the year 2020. Chemotherapy has been employed to shrink or manage the tumor pre-surgery and promote tumor-free survival post-surgical ablation of breast tumors. Conventional chemotherapy suffers from toxic effects such as cytotoxicity at non-tumor sites, low blood count, hair loss, nausea and vomiting, etc. [1]. The advent of nanocarriers (NCs) has eased the delivery of hydrophobic chemotherapeutic agents. For example, albumin-bound paclitaxel (Abraxane®) has been able to promote tumor-free survival and decrease side effects in breast cancer patients. Despite the boom in the NC-based research for tumor drug delivery, the clinical translation is poor. Various factors such as short systemic half-life, poor tumor penetration, high tumor interstitial pressure, macrophage entrapment, and poor tumor extravasation have been implicated in the same [2, 3].
The development of fresh blood vessels from the prevailing blood vessels in an uncontrolled fashion is called angiogenesis. A tumor is characterized by hyperplasia and an increase in metabolism. To meet the high nutrition demands of the tumor cells, angiogenesis is required to meet the increased nutrition requirements of the tumor cells [4]. Pro-angiogenic signaling results in uncontrolled growth of new blood vessels which enhances the micro-vessel density of the tumor [5]. Rapid pro-angiogenic signaling leads to the development of anomalous blood vessels with excessive branching, bulging, defective basement membrane, blind ends, excessive fenestrations, ineffective pericyte coverage, and discontinuous epithelial layer [6]. The tips of the projections are responsible for creating intercellular openings in the blood vessel. These openings are responsible for the leaky tumor vasculature. Moreover, these vessels may demonstrate uneven and multi-layered epithelia [7]. Such vessel characteristics lead to poor blood perfusion and hypoxia in the Tumor microenvironment (TME) [6]. The TME is a complex environment that consists not only of cancerous cells but stromal cells and extracellular matrix, too. Cancer cells manipulate this environment to promote cell growth, for metastasis, to prevent itself against immune action. The presence of pores in the tumor epithelium leads to “Enhanced permeation and Retention” (EPR) of plasma proteins such as albumin and nanocarriers in the TME. The EPR effect causes an increase in the infiltration of nano-sized carriers in the tumor microenvironment, while the lack of lymphatic drainage leads to the retention of infiltered NCs in the tumor [8].
The hypoxic microenvironment leads to the selection of robust, aggressive, and invasive cancer subpopulations, and hampers the anti-tumor action of immune cells [9]. Hypoxic cells secrete vascular endothelial growth factor A (VEGFA) which binds to the VEGF receptor 2 (VEGFR2) on the local endothelial cells (ECs) and promote their proliferation [10].
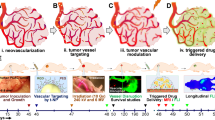
The tumor vessels developed due to pro-angiogenesis signaling are hypo-perfused and leaky (Fig. 1). These vessels, in combination with poor lymphatic flow, are responsible for generating hypoxia, acidosis, and high intra-tumor pressure [11]. The loss of nutrient supply further potentiates angiogenesis, pushing the TME into a never-ending loop of non-productive angiogenesis. The hostile environment inside the tumor forces the tumor cells to metastasize and disseminate to other organs [12]. NCs, owing to their nano-range particle size, have been hypothesized to penetrate tumors through these pores. However, studies have shown that NCs cannot permeate through more than two layers of cells. Desmoplasia and high intratumor pressure prevent the accumulation of NCs in the TME. In addition, the diffusion of drugs in TME is restricted by the presence of extracellular matrix.
(A) Represents the state of vessels in the tumor post-angiogenesis. The vessels in the tumor are tortuous and irregular. (B) Shows the restoration of the normal vascular system in the normalization window post-treatment. (C) Represents the effects of vascular normalization on the intensity of intra-tumor hypoxia, intratumor interstitial pressure, and drug perfusion in the tumor
The desmoplastic microenvironment of solid tumors, especially breast tumors, adds to the poor permeation of drugs and NCs in the TME premises. Higher than normal concentrations of extracellular matrix and cancer cells increase the density of the TME. Thus, the permeation of drugs and NCs is limited to areas in close proximity to blood vessels [13]. In addition, dense TME blocks the blood vessels by physical pressure. The blocking of vessels further exaggerates the poor nourishment inside the TME.
In this review, we have discussed the mechanism of angiogenesis in tumors and the impact of angiogenesis on the flow of fluid. We have discussed the effect of excessive extracellular matrix deposition and stiffness on drug perfusion and NC permeation. The various factors impacting the accumulation of therapeutic moieties and the efficacy of immune therapies are discussed. Studies discussing the positive effects of normalizing the blood vessels and TME have been discussed.
Mechanisms of angiogenesis and potential therapeutic targets
Angiogenesis is a prerequisite for the development of both primary tumors and metastatic tumors from the circulating tumor clusters [14]. Folkman and co-workers reported that the malignant cells could not form a solid tumor without vascularization for obvious nourishment [15]. Angiogenesis can take place by various methods. Sprouting angiogenesis is the development of fresh blood vessels from preexistent vessels. Such vascularization is stimulated by growth factors such as VEGF [16]. Intussusceptive angiogenesis or splitting angiogenesis is the splitting of a pre-existing vessel into two daughter vessels by the formation of transvascular pillars. This process takes place in both normal physiological functions such as wound healing and pathological conditions such as cancer and fibrosis. Factors responsible for intussusceptive angiogenesis comprise platelet-derived growth factor B (PDGFB), angiopoietin, Eph receptors, and ephrins [17]. Another method wherein tumor cells mimic ECs to form blood vessels is known as vasculogenic mimicry. Vasculogenic mimicry has been linked to poor prognosis in malignant cancers. Vasculogenesis is the vascularization process that mimics the vascularization process witnessed during embryo development. It creates new blood vessels from endothelial progenitors [18]. Approximately 30 growth factors, mainly VEGF secreted by the TME, are responsible for the creation of new blood vessels [19]. The origin of the new ECs has been an area of uncertainty. It has always been assumed that the new ECs and pericytes originate from the neighboring blood vessels [11]. A study on 1238 BC patients showed that 24% of breast tumors demonstrated vasculogenic mimicry. This phenomenon was positively associated with tumor size > 2 cm, lymph node metastasis, and low overall survival [20].
VEGFA transforms ECs into migratory tip cells which degrade the connective tissues to develop space for movement [21]. Tip cells are present at the tip of the growing vessels to initiate sprouting and guide the following endothelial cells for the successful development of new vessels. Tip cells are motile, invasive, and have filopodia, which react to the growth factors and other regulators [22]. The signaling molecules such as angiopoietin-2 and delta ligand-like 4 (DLL4) are crucial for transforming ECs [23]. The ECs that follow tip cells are called the stalk cells, and they contribute by proliferation and maintenance of functional and structural aspects of the new vessel [24]. The NOTCH pathway regulates the tip/stalk cell conversion. The stimulation of VEGFR2 by VEGF up-regulates the NOTCH ligand DLL4. DLL4 binds to the NOTCH1 in the neighboring ECs to release the NOTCH1 intracellular domain (NCID). NCID is responsible for reducing neuropilin-1, vegfr2 and vegfr3 expression and simultaneously up-regulating the vegfr1 expression in the ECs [25]. JAGGED1 mediated signaling prevents the NOTCH1 signaling in the tip cells. It prevents DLL4 produced in the stalk cells from stimulating the NOTCH1 in the tip cells and maintaining phenotype differences between tip and stalk cells [26].
The hypoxia created by irregular vasculature in the first place is mainly responsible for promoting molecular signaling for angiogenesis. The prolyl hydroxylase domain (PHD) substrates reduce the hydroxylation of HIFα in the hypoxic environment. The HIFα is central in various molecular signaling pathways triggering metastasis, angiogenesis, and therapy resistance. HIFα binds to HIFβ to form a dimer, which further binds to hypoxia response elements [27]. Tumor cells also secrete pro-angiogenic factors such as VEGF [28], and TGFβ3 [29], further bending the balance in favor of increased angiogenesis. These factors are responsible for increased proliferation, migration, and adhesion in endothelial cells, which increases tube formation (Fig. 2) [30].
Linc-RNAs such as linc-OIP5 have been recently reported to be an upstream target of YAP1/NOTCH/JAG1 signaling axes. These signaling axes are important for the proliferation, migration, and tube formation of HUVEC cells. BC cells over-express linc-OIP5, YAP1 transcription co-activator, and JAG1 protein. Knockdown of these proteins inhibited the tube-formation ability of the co-cultured HUVEC cells [31]. Hence, we conclude that the inflammatory mediators and chemokines play a significant part in angiogenesis and in the continuation of the invasion and migration. Epithelial-to-mesenchymal transition inhibitors and invasion inhibitors such as TGF-β inhibitors, COX-2 inhibitors, Wnt/β-catenin inhibitors, and YAP/TAZ inhibitors should be explored to inhibit the overall metastatic event in mammary tumors.
Tumor vessel-mediated hindrance to drug and nanocarrier-based therapies
Chemotherapy or carrier-mediated chemotherapy is effective when it can be perfused through the maximum volume of BC tumors [32]. Conditions such as high intra-tumor pressure and stiff TME make it difficult for the therapeutic entity (TE) to perfuse homogeneously [33]. The altered vasculature and stiff TME increase intra-tumor pressure, which restricts the diffusion and flow of plasma and, ultimately, TEs in the TME [34].
High hydraulic conductivity due to the leaky nature of tumor vessels leads to an overflow of plasma into the TME and other blood traversing colloid particles such as albumin and nano-carrier systems [35]. This leads to a rise in intra-tumor pressure. High inflow and unavailability of lymphatic drainage cease the plasma flow in the tumor [36]. TEs travel via convection currents of flowing plasma over large distances. The stagnant fluid reservoir inside the tumor prevents homogeneous delivery of high molecular weight drugs and nano-carriers. Low molecular weight TEs still travel over shorter distances via diffusion; while high molecular weight TEs and carriers struggle to make any distance from vessels [34].
The tumor also shows aberrant microvascular structures with high tortuosity, which reduces blood flow significantly. In contrast to healthy tissues, the vessel diameters do not decrease with increased vessel branch order. This leads to a loss of relationship between vessel diameters and blood flow [37]. Furthermore, the inability to remove waste materials from TME due to the absence of a functional lymphatic system increases fluid viscosity [38]. This prevents the diffusion of low molecular weight drugs over short distances in TME.
The role of TME in hindering the NCs-based therapies
Breast tumors are characterized by a high density of extracellular matrix (ECM) [13]. ECM such as hyaluronic acid and collagen, surround BC cell colonies inside tumors to provide structural support. ECM prevents the transportation of TEs by being a mechanical barrier to diffusion and convection [39]. It increases the distance that TEs have to cover to reach target cells. Further, the binding of drugs to ECM reduces the concentration of the free drug. Furthermore, dense ECM mechanically compresses the vessels in TME [40]. The hyperplasia continuously increases the population of cells in the tumor, which further adds to compressive stress on vessels, leading to the partial or complete collapse of the flow system [41]. The sum total of all the pathological processes discussed in this section is reduced drug flow and increased fluid pressure along with other tumor-associated pathological conditions, mainly hypoxia and immune suppression [42].
Moreover, stiff ECM has positively correlated with invasion, migration, and poor overall survival in BC patients owing to the activation of Yes-associated protein (YAP) and transcriptional coactivator with PDZ-binding motif (TAZ) [43]Therefore, pre-treatment with pharmacological agents with the ability to disintegrate the components of ECM, such as hyaluronic acid and proteins, will promote drug accumulation in the tumors and prevent BC cell invasion. However, degrading the ECM may prove to be a dual-edged sword as it is necessary to move cancer cell clusters across TME and adjacent tissues. Such clusters are responsible for the release of circulating tumor cell clusters with high metastatic potential. Thus, exhaustive studies are a prerequisite for the successful clinical translation of such therapeutic strategies.
The role of tumor vessels and TME in immunosuppression
Immunotherapy has gained popularity in the management of metastatic disease over the last 10 years [44]. However, these therapies fail in a large population of patients mainly due to the “tolerogenic” or immunogenically “cold” tumors. Immunosuppressive TME demonstrates intrinsic low sensitivity towards immunotherapies due to low infiltration rates of the immune cells, mainly cytotoxic CD8 + T-cells, natural killer cells, and macrophages, and the presence of a high ratio of immunosuppressive myeloid-derived cells [45]. Other factors, such as the up-regulation of programmed cell death protein-1 (PD-1) [46] and cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) [47], prevent the survival and multiplication of immune cells in the TME. Various strategies such as immune checkpoint inhibition [48], immunotherapies [49], and vaccines [50] are used to promote immune action against tumors. For triple-negative breast cancer (TNBC) tumors, the response rate for anti-PD-1 immunotherapy lies between a meager 5.2 – 18.5% [51]. Hence, it is high time to develop a strategy to sensitize BC cells to immunotherapy.
Faulty vessels affect the proliferation, survival, infiltration, and functioning of immune cells in TME. Lack of blood perfusion in the TME results in hypoxia, acidosis, and decreased entry of drugs and immune cells [52]. It leads to the prevention of natural immune response against tumors and failure of immunotherapy. Poor perfusion checks the penetration of cytotoxic T cells in the TME [53]. Hypoxia is associated with preventing immune cell infiltration from the vascular system to the TME and decreasing the cytotoxic potential of immune effector cells. Hypoxic TME is also responsible for the up-regulation of immune checkpoints and the transformation of macrophages from immune-responsive (M1) to immunosuppressive (M2) [54]. Hypoxic TME tends to infiltrate and recruit immunosuppressive regulatory T cells (Tregs) and myeloid-derived suppressor cells [55]. Additionally, VEGF inhibits antigen presentation, and dendritic cell maturation and triggers pro-apoptotic signaling in T-cells via FAS ligand (FASL) expression [56]. Tumor vasculature is characterized by the absence of intercellular cell adhesion molecule (ICAM) and vascular cell adhesion molecule (VCAM), which causes poor infiltration of leukocytes (Table 1) [57].
Along with hypo-perfused vessels, the stiff nature of breast TME prevents the penetration of immune cells in the TME. The mechanical forces due to uninhibited growth of tumor cells and dense ECM in confined space compress the vessels; which further complicates the admission of immune cells into the TME. Furthermore, a hypoxic microenvironment renders immune-resistant phenotypes to the BC cells [34].
Thus, along with chemotherapy-based drug delivery, the natural anti-tumor action of the host suffers due to dire changes in the TME. The normalization of such tumor-specific changes can provide anti-tumor therapeutic capabilities. Normalizing tumor vasculature can prevent the loss of nutrients and oxygen homeostasis, thus promoting the proliferation and functioning of immune cells. Normalizing the tumor microenvironment by decreasing the connective tissues can reduce the tumor density and, thus, promote the infiltration of immune cells.
Vascular and TME normalization as a strategy to alleviate chemotherapy failures
Normalization of tumor vessels to improve chemotherapy
The thought of vascular normalization was introduced by Dr. Rakesh Kumar Jain and his research group [85]. The excessive pruning of tumor vessels in traditional anti-angiogenesis treatments leads to an aggravation of the hostile environment in TME [86]. The loss of nutrients and gas translocation aggravate the hypoxic and acidic environment in the TME. This triggers the advent of chemo-resistance, radio-resistance, and photodynamic therapy resistance in the tumor cells [87, 88]. Several studies have reported that the anti-angiogenesis treatment initially improved the penetration of therapeutics in the TME. The continuation of anti-angiogenesis therapy led to resistance to various therapies. The initial improvement in the tumor flow was credited to normalizing the tumor vasculature by eliminating the immature vessels [89]. The elimination of the immature tumor vessels repairs the structural and functional faults of the tumor vessels; thus improving [90]. Normalization restores the functions and structure of the tumor-associated blood vessels, which prevents metastasis [91], hypoxia [92], and therapy resistance [87, 93].
The restoration of the balance between pro-angiogenic and anti-angiogenic signaling holds the key to normalizing the faulty vessels in the TME. Anti-VEGF antibodies are also known to hold a normalization window before the complete pruning of the tumor vessels [89]. This window is associated with the pruning of immature vessels and curing the defects in the fundamental vessels via uniform pericyte covering [94].
Systemic delivery of chemotherapies during the normalization window increases the entry of drugs into the tumor increasing its efficacy. As the oxygen saturation levels in tumors improve and most of the anti-tumor therapies kill tumor cells via an increase in the concentration of oxygen radicals, the normalization window improves the effectiveness of the cytotoxic therapies [93, 95]. The transient nature of normalization can be attributed to further pruning of major tumor vessels or activation of other pro-angiogenic axes. Thus, tracking the normalization process is critical in delivering the chemo- or immune-therapeutics [96].
Yu and co-workers delivered endostar, a derivative of endostatin (endogenous angiogenesis inhibitor) to 4-T1 mammary tumor-bearing mice to study the molecular mechanism of normalization. Studies demonstrated the normalization capabilities of endostar. 2-D gel electrophoresis and mass spectrometry analysis demonstrated up-regulation of SRC kinase signaling inhibitor 1 (SRCIN1) protein in the normalized TME. Immunoblotting studies revealed that the SRCIN1 bound and regulated SRC activation to normalize the mammary tumor normalization. The co-delivery of endostar and doxorubicin led to increased tumor volume shrinkage (34%) and decreased metastatic nodule formation (29%) in comparison to the control group [97].
Normalization of TME
The breast tumors are generally characterized by immunosuppressive TME. Immunosuppressive tumor microenvironment demonstrates a low response rate to immunotherapeutics. It is credited to the inability of immune cells such as T cells, natural killer cells, and macrophages to enter, activate, and multiply in the TME. TME normalization mainly involves a reduction in the density of the cell population in the TME to alleviate the mechanical compression of the vascular system (Fig. 3) [98]. Decreasing the ECM in and around the tumor (desmoplastic tumors) further improves the drug and immune cell penetration inside the otherwise immunosuppressive tumor environment [99]. Metronomic chemotherapy has been able to lower the cancer cells and, hence, density in the TME [100]. Other strategies, such as anti-ECM therapies, have recently been tested to normalize the TME [99]. The knockout of biglycan in E0771 tumor-bearing mice demonstrated normalization by inhibition of pro-angiogenetic TNF-α/ANG-2 signaling. The mammary tumors also demonstrated a decrease in lung metastasis and desmoplasia. An improvement in the immune microenvironment with increased cytotoxic CD8 + T-cell infiltration was witnessed [101].
(A) depicts the vascular compression in the breast tumors due to the high density of cancer cells and ECM. The presence of ECM also limits the perfusion of therapeutic entities and carriers in the tumor. (B) Pictorially represents the effect of mechanotherapeutics in the normalization of breast tumors. Reduction in cancer cells and ECM density restore the normal shape of the vessels. It also alleviates high intra-tumor interstitial pressure leading to improvement in drug perfusion
In this subsection, we conclude that the atypical and hostile conditions in the TME and tumor-blood vessels are mainly responsible for the failure of anti-tumor therapies. Meanwhile, restoration of normal physiological conditions improved the efficacy of various therapies discussed above. However, the potential challenge is to gauge when to stop the normalization therapy once appropriate vascular and ECM conditions are attained. Hence, extensive preclinical and clinical studies must identify the correct anti-angiogenesis dosage regimens.
Impact of vessel normalization in BC management
Strategies mainly focus on developing a normalization window for increased perfusion of anticancer drugs in the TME. Early anti-VEGF therapies focused on the inhibition of angiogenesis and pruning of already existing vessels to starve tumors [102]. Bevacizumab, an anti-VEGF antibody showed promise against BC in pre-clinical studies [103]. Bevacizumab also demonstrated excellent capabilities to improve the structural integrity of blood vessels by increasing pericyte coverage in BC patients [104]. Motivating clinical results in metastatic BC patients led to approval for commercialization by the FDA in 2008 [105]. Alone, anti-VEGF therapies could not eradicate the tumor in the clinical setup. Vessel pruning led to excessive hypoxia due to the rarefaction of microvessels and further metastasis into neighboring and distant tissues and therapy resistance. This led to the withdrawal of FDA approval for the commercialization of bevacizumab [106]. However, the anti-angiogenesis therapies were effective only in combination with chemotherapies; hence, the concept of vascular normalization came into the light [104]. The establishment of dosage regimen of anti-angiogenic drugs to achieve persistent normalization is the need of the hour.
Such response was replicated in a phase 2 trial in TNBC patients. A single dose of bevacizumab has been reported to improve the infiltration of cytotoxic CD8 + T-cells, CD4 + T-helper cells, and mature dendritic cells in TNBC tumors. They also reported the down-regulation of ANG2 in TNBC tumors, which led to the better structural integrity of tumor vessels and normalization of vessels. A single dose of bevacizumab improved the population of mature dendritic cells in the TNBC tumors [107]. However, continuous use of anti-angiogenesis agents still demonstrates negative results. Thus, the combinations of pre-treatment of anti-angiogenesis antibodies and anti-cancer therapies should be studied comprehensively.
A comparative study between sunitinib and bevacizumab was done in a recent clinical trial on 62 HER2-negative BC patients. Sunitinib demonstrated a better vascular normalization index than bevacizumab (p < 0.001). Sunitinib also showed a better response against lymphangiogenesis. Thus, it is a potential agent to alleviate the problem of high pressure and fluid plugging in mammary tumors [108]. The combination of two agents can be used to reduce the intra-tumor plugging.
Mollard and co-workers reported an ideal regimen for the combination of bevacizumab and paclitaxel (PTX). They found that pre-treatment with bevacizumab demonstrated better results than co-administration of the antibody along with PTX. Studies in the orthotopic MDA-MB-231luc+ BC mouse model demonstrated that pre-treatment with bevacizumab (3 days before PTX administration) showed improved tumor suppression activity with 48% more reduction in tumor size in comparison to concomitant drug administration (p < 0.05) [109]. In a clinical study on 8 patients with BC-induced refractory brain metastasis, single dose of bevacizumab induced normalization within 1 h of administration. However, peak normalization was achieved after 24 h of the dose administration [110]. Another phase 2 clinical trial reported that neoadjuvant bevacizumab therapy promoted pathological complete response in 52% of patients with TNBC. However, estrogen receptor-positive tumors did not demonstrate significant improvement. The authors reported decreased interstitial fluid pressure and homogenous pericyte covering around tumor vasculature [111]. Hence, the aforementioned clinical studies conclude that the pre-treatment of the anti-angiogenesis agent is important to induce normalization in the TME.
Among newer strategies bone morphogenetic protein-9 (BMP9)/activin receptor-like kinase 1 (ALK1) signaling axes have been explored. BMP9 is a cytokine belonging to the TGFβ superfamily of proteins. The BMP signaling occurs via smad1, 4, 8 in contrast to smad2, 3 pathway in TGFβ signaling. The BMP9 cytokine is encoded by the GDF2 gene [112]. BMP9 is a vascular quiescence factor inhibiting the movement and proliferation of ECs [113]. A recent study on the GDF2 knock-out orthotopic mice model of metastatic BC (E0771 cell line) showed vasculature normalization and reduction of lung metastases [114].
Chen and co-workers administered erlotinib (50 mg/kg), an epidermal growth factor receptor (EGFR) inhibitor, orally to attain vascular normalization in the subcutaneous BC mice model at days 0, 2, 4, and 6 prior to treatment of PTX-loaded NPs (13 mg/kg) at 4, 6, 8 and 10 days. The treatment led to better uptake of PTX-loaded NPs in the TME. Erlotinib also reduced intra-tumor fluid pressure and hypoxia which transformed tolerogenic TME into immune-supportive. The transformed TME improved the retention and efficacy of the anti-PDL1 antibody. Thus, vascular normalization improved both chemo- and immune therapies [115].
Vessel-normalization-induced improvement in immunotherapeutic potential
Recently, Mirando and co-workers reported that a novel peptide AXT201 demonstrated vasculature normalization in the TNBC mice model by potent inhibitory effects on VEGF signaling. The peptide demonstrated various positive alterations in BC TME such as an increase in the population of infiltrating cytotoxic CD8 + T-cells and an increase in the expression of interferon. Lower populations of Tregs and myeloid-derived suppressor cells, along with down-regulation of PD-L1, were also observed, therefore improving the tumor’s susceptibility to the body’s immune system [116].
Li and co-workers reported that the low-dose anti-VEGFR2 antibody (DC101) normalized tumor vessels, which led to the infiltration of cytotoxic CD8 + T-cells in the TME at a rapid rate and promoted the secretion of osteopontin (OST). OST sensitized the BC cells to anti-PD-1 therapy (SHR-1210) by promoting PD-L1 expression on tumor cells via the TGFβ pathway. This strategy showed improved anti-PD-1 immune therapy results in TNBC patients. Therapy-induced higher OST and TGFβ levels correlated with better therapy results [117]. Melaiu et al. reviewed the improvement in the therapeutic potential of immune checkpoint inhibitors when co-administered with tumor vessel normalizing agents [118].
The use of PI3K inhibitors as vessel-normalizing agents
Kim et al. employed PI3K inhibitors to normalize the tumors. BEZ235 and HS-173 alleviated hypoxia and promoted BC cell death in the mice models. Microscopic studies revealed improved morphology with a regular and flat monolayer of ECs in the tumor vessels. Further, an increase in ECs maturity, pericyte coverage, junctions, and thickness of the basement membrane was observed. Tissue histology revealed a decrease in vascular density, tortuosity, and vessel diameter, leading to an improvement in blood flow in the tumor. Mechanistic studies revealed that the PI3K inhibitors orchestrated anti-angiogenic signaling via NOTCH signaling. HS-173 even inhibited the migration of BC cells to the lungs [119].
Anti-apelin therapy prevents metastasis associated with traditional anti-angiogenic therapy in BC
Apelin is an adipokine, mainly released by the adipose tissues. It binds to the g-protein-coupled apelin receptor (APJ) expressed by endothelial cells and vascular smooth muscle cells [120]. High apelin expressions in the BC murine model (MMTV-Neut) had been positively correlated with poor prognosis [121]. Combining anti-apelin therapy with traditional anti-angiogenic kinase inhibitors (sunitinib) has led to a decrease in the BC growth and normalization of tumor vessels [90, 121]. Uribesalgo and co-workers reported that high apelin expression in the BC led to a higher risk of metastasis. They found that inhibition of apelin reduced tumor growth, angiogenesis, and infiltration of immuno-suppressive myeloid cells into the TME. The inhibition of apelin also prevented the resistance to receptor tyrosine kinase inhibitors and prevented hypoxia as well as metastasis [122].
Effect of physical exercise on vascular normalization in BC cancer
Jones and co-workers observed the normalization of BC vasculature in exercising animals. The sedentary group did not show any improvement in the tumor vasculature. Meanwhile, no significant difference in the tumor size was noticed between the two groups. However, HIF-1 levels were over-regulated in the exercising groups [123]. Later, Schadler and co-workers reported that the mechanical stimuli exerted on ECs due to physical work improve vascular integrity. Mechanistic pathways studies demonstrated that the activation of calcineurin-NFAT-TSP1 signaling in ECs was responsible for vascular remodeling. The authors achieved better penetration of chemotherapeutic drugs into the mammary tumors with a moderate exercise routine in mice models [124]. Practically, it may not be feasible to get advanced-stage cancer patients to exercise. However, calcineurin-NFAT-TSP1 axes must be explored to find new therapeutic targets.
Novel strategies to normalize the tumor microenvironment for better BC management
Mechano-therapeutics to normalize TME
Breast tumors demonstrate anomalous mechanical forces acting on the cancer cells owing to the factors such as high interstitial fluid pressure, dense ECM, etc. Such mechanical factors have been reported to promote tumorigenic pathways. Mechanical strain can result in the activation of embryonic developmental biochemical pathways. High interstitial fluid pressure promotes hypoxia that ultimately leads to metastasis. Stiff tumor tissues promote cell multiplication rate, epithelial-to-mesenchymal transition, and cell invasion [125]. Thus, strategies aimed at normalizing the extreme mechanical conditions in TME can be useful in treating tumors. Martin et al. demonstrated the delivery of dexamethasone relieved the intra-tumor fluid pressure, stiffness, and solid stress upon the tumor vasculature. Therapy achieved a lower density of hyaluronic acid in the TME, which relieved the pressure and stiffness. Normalization of vasculature was also noticed. Cumulative normalization effects led to an increase in the infiltration of nanostructures with diameters of 13 and 32 nm inside the tumor. Dexamethasone improved the accumulation of cisplatin-loaded micelles (diameter 30 nm) into the primary BC tumors and secondary lung metastasis in the mice model [126].
Panagi and co-workers repurposed FDA-approved antifibrotic and anti-histaminic agent TGFβ inhibitor tranilast for normalizing the tumor vessels. The co-delivery of tranilast and Doxil reduced the concentration of extracellular matrix, thus reducing the mechanical pressure on tumor vessels and alleviating the intratumoral high pressure. Further, the TGFβ inhibitor improved the structural integrity of vessels by improving the pericyte coverage around vessels, which reduced the plasma plugging in the TME. An increase in the recruitment of anticancer lymphocytes was achieved. The activity of immunostimulatory M1 macrophages was significantly improved, leading to improved efficacy in anti-PD-L1 and anti-CTLA4 therapies. Thus, tranilast improved the immune environment in mammary tumors, and this technique improved the positioning of Doxil in the TME [127]. Mpekris and co-authors also achieved similar results in TNBC lung metastases. The mechano-therapeutic tranilast was able to decompress the lung metastases blood vessels by alleviating intra-tumor pressure and ultimately alleviating hypoxia. The TGFβ inhibitor improved the perfusion of Doxil and immune checkpoint inhibitors. The alleviation of immunosuppressive TME and improvement in the kinetics of therapies re-sensitized the lung metastases to the immuno- and chemotherapy [128].
The delivery of the above-mentioned mechanotherapeutics achieved a common goal of relieving the intra-tumor pressure and improving the accumulation of NCs and immune cells in the TME. The use of neoadjuvant chemotherapy may have demonstrated promising results, but the authors do not support the use of low-concentration chemotherapy pre-treatment, as it may lead to the emergence of multidrug resistance in the cancer cells.
The use of anti-HER2 trastuzumab as a normalization agent in HER2 + BC cells
A study reported that trastuzumab treatment in the HER2 + murine BC model demonstrated perfusion and permeability of the drug in the TME. The authors credited this improvement to the killing of the HER2 + tumor cells which led to the decrease in the cell density in TME. The decrease in cell density freed up space, which decreased interstitial fluid pressure and relieved space for diffusion and convection of fluid (and drugs). On day 4 after antibody treatment, an increase in the expression of α–SMA was reported; indicating an increase in pericyte cells covering the tumor vessels. Hence, due to all these corrections in pathological HER2 + BC TME, the intensity of doxorubicin treatment was improved [129].
Oxygen microbubbles to achieve normalization in the tumor
Ho and co-workers employed oxygen microbubbles with ultrasound to deliver oxygen gas locally inside mammary tumors. Continuous use of conventional anti-angiogenic therapeutic moieties leads to vessel pruning, which worsens the intra-tumor hypoxic environment. Hypoxia increases the hypoxia-inducible factor (HIF-1α) levels in the tumor, leading to increased angiogenesis and metastasis [130]. The authors normalized up to 8 days after a single treatment with oxygen microbubbles. The treatment blocked the HIF-1α/VEGF pathway to restore normalization in tumor vessels. The pericyte coverage increased with up to a four-fold increase in the doxorubicin accumulation in the BC TME [131]. Low oxygen levels lead to lower levels of therapy-induced ROS, which leads to the development of resistance against various therapies, especially photodynamic therapy. A coupling of oxygen microbubbles therapy along with chemo- and photodynamic therapies can demonstrate excellent results.
Potential strategy to achieve immune environment makeover in immune-suppressive BC
Wang et al. studied the effects of neoadjuvant chemotherapy on the infiltration of lymphocytes and vascular normalization in 75 BC patients. They concluded that the patients with complete remission demonstrated higher infiltration of cytotoxic T-cells after neoadjuvant chemotherapy. The number of immunosuppressive FOXP3 + Tregs was reduced in the mammary tumor after neoadjuvant therapy. Tumor-free survival in patients was found to be better in patients with lower FOXP3 + Treg accumulation in the TME. The vascular pericyte coverage and cytotoxic T cell levels were higher in the primary tumors rather than in the metastatic lymph nodes [132].
Recent studies on vascular normalization have been found to significantly improve the immune microenvironment in the tumor. The normalization of tumor vasculature has been associated with developing tertiary lymphoid structures (TLS) in the TME. TLS is the ectopic lymphoid organs, which mimic secondary lymphoid structures in structure, cell population diversity, and functions. The difference between secondary lymphoid organs and TLS is the development of TLS in non-lymphoid tissues [133]. TLS does not develop prenatally or in a normal physiological environment but in pathologically inflamed locations such as tumors [134]. The presence of TLSs in a mammary tumor is associated with a positive prognosis [135]. However, the spatial position of TLS significantly defines its functions. Intra-invasive margin TLSs have been exploited for lymphocyte infiltration and priming for tumor-specific antigen response [136]. Matured TLSs are enriched in B-cells, T-cells, dendritic cells, and high endothelial venules [137]. The priming and infiltration of cytotoxic T-cells are associated with poor tumor immunotherapy and patient outcomes. TLS can be used to raise anti-tumor immunity by infiltrating naive lymphocytes in the TME via high endothelial venules and priming them against tumors [138]. A recent study demonstrated the ability of TNFS14, an inflammatory cytokine, to normalize the tumor vasculature at lower concentrations and the formation of intra-tumor TLSs at higher concentrations (2–20 ng/mL) by binding to lymphotoxin-β-receptor. The generation of TLSs in TME improved the infiltration of lymphocytes to reverse the tolerogenic TME [139]. Thus, such cytokines or other therapeutic moieties must be employed along with immunotherapies for better management of BC.
The STING pathway is known for its role in linking innate immunity to adaptive immunity. It has been studied to induce immune responses against tumors [140]. Yang et al. reported that endothelial levels of STING are positively correlated with increased infiltration of cytotoxic T-cells in BC patients [141]. STING activation with RR-CDA or cGAMP normalized the blood vessels in the TME BC mice models. The normalization effects were carried out via up-regulation of vessel stabilizing genes such as Pdgfrb, Angpt1, and Col4a. STING activation orchestrated TME normalization via type-1 interferon signaling [142]. The co-administration of STING agonists along with VEGFR2 blockade and anti-immune checkpoint inhibitor therapies led to an increase in the immune-therapy potential and re-sensitization of immunotherapy-resistant mammary tumors [143].
Nano-scale carrier-based strategies to attain normalization in breast TME
The last decade has witnessed a boom in the use of NCs for the delivery of anticancer drugs. Different nanosystems, such as nanoemulsion [144], nanoparticles [145]liposomes [146], polymeric micelles [147], dendrimers [148], etc., have been used as nanocarriers for BC therapy. Nanocarriers demonstrate various advantages such as increased surface area, high surface-to-volume ratio, increased avidity, particle size tunability, surface functionalization, and site-specificity over conventional delivery systems [149, 150]. Entrapment of drugs in such systems leads to desired pharmacokinetic changes, increased stability of sensitive drugs such as proteins, and site-specific drug release [151, 152]. The ability of surface functionalization has enabled us to specifically target a wide range of cancer cells among various cancer subtypes [153, 154]. Thus, researchers over time have employed nanocarriers to deliver normalizing agents to the tumor vessels and microenvironment. However, entrapment of synthetic NCs by the macrophages in the liver and spleen shortens their plasma half-life [155].
Photodynamic therapy (PT) has gained substantial attention as a cancer treatment in the past decade. Characteristics such as economic, efficacy, safety, and synergism with other therapies make PT ideal for BC management [156]. However, the lack of O2 in hypoxic TME makes tumors resistant to PT [157]. Xu et al. developed mesoporous polydopamine-based NPs for the co-delivery of photosensitizer indocyanine green (ICG) and PFKFB3 kinase inhibitor 3-(3-pyridyl)-1-(4-pyridinyl)-2-propen-1-one (3PO). 3PO functions by inhibiting the VEGF-induced internalization of vascular endothelial cadherin. The inhibition of cadherin internalization leads to structural normalization of the tumor vessels, leading to a reduction of hypoxia in the TME. The NPs were surface-functionalized with sialic acid for selective binding to the E-selectins over-expressed on the tumor endothelial cells. The tremendous properties of polydopamine nanoparticles such as biocompatibility, pH-responsive drug release, high surface functionalization potential, photothermal conversion efficiency, and biodegradability made them ideal carriers. The designed NPs demonstrated a combination of activities including vascular normalization, PT, and photothermal therapy. The sialic acid confirmed the local accumulation of NPs in the angiogenic vessels. Initially, the NPs showed cytotoxicity due to the combination of PT and photothermal therapy. As the PT went on, the consumption of O2 gas aggravated the hypoxic conditions leading to the over-expression of VEGF in the mammary tumor. The controlled release of 3PO prevented the VEGF-mediated internalization of vascular endothelial cadherin. This led to the normalization of tumor vessels and the prevention of tumor metastasis [158].
Another research group developed anionic liposomes to normalize tumor vessels and hypoxic TME. They entrapped hydrophobic chemotherapeutic drug topotecan in the hydrophobic bilayers of the liposomes, with photosensitizer ICG in the hydrophilic core. Cationic normalizing agent erlotinib was adsorbed on the anionic liposomal surface. The prepared system was able to normalize the tumor vessels and controlled the release of topotecan ensuring the killing of BC cells. Along with the cytotoxic action, topotecan ensured down-regulation of HIF-1α, further promoting vascular normalization for longer periods. The prolonged normalization of vasculature ensured the perfusion of O2, thus, resistance against PT was prevented [159].
Zoledronate is known for its anti-tumor properties, which inhibit angiogenesis. It decreased the plasma levels of VEGF in patients with bone metastasis [160]. However, its low plasma half-life of 15 min and intrinsic nature to accumulate in bones make untargeted delivery unsuccessful [161, 162]. Cai et al. developed actively targeted cationic liposomes to improve the specific availability of zoledronate to target BC vasculature. They surface-functionalized the liposomes with alanine-proline-arginine-proline-glycine (APRPG) peptides to target VEGFR-1 expressed by the ECs in tumor vasculature. The anti-angiogenic drug was successfully accumulated in tumor vasculature to achieve normalization. The intra-tumor fluid pressure was successfully decreased, leading to increased diffusion of cisplatin to deep cancer tissues. The formulation was able to decrease the HIF1-α levels, increase thrombospondin levels, and decrease the migration, viability, and tube formation ability of HUVEC cells [163].
NogoB receptor regulates the epithelial-to-mesenchymal transition, angiogenesis, and vascular development in the TME [164]. Wang et al. developed acid environment-responsive, charge-switchable polymeric nanostructures for the delivery of siRNA against NogoB receptors. The nanostructures were prepared by functionalizing 2,3-dimethyl maleic acid on the surface of micelles formed by the block copolymer of PLGA and poly (ethyleneimine). The prepared nanostructure remained negatively charged in the physiological pH while converting to the positive charge in the acidic TME, thus, releasing siRNAs specifically in the tumor. Site-specific nucleic acid release inhibited the NogoB receptors leading to inhibition of metastasis and normalization of tumor vessels [165].
We conclude that the co-delivery of anti-angiogenesis agents, along with anticancer therapies such as PT or chemotherapy, was able to attain the normalization and increased nanocarrier accumulation in the TME. The use of nanocarriers enabled the researchers to target ligands such as sialic acid to the tumor-specific ECs and prevent side effects. As the majority of the anti-cancer drugs are hydrophobic in nature, the use of nanoparticles could improve the pharmacokinetics of the anti-cancer drugs.
Obstacles towards bench to bedside translation
From the available literature, it is evident that the characteristics of the tumor barrier pose hindrances towards nanoparticulate penetration and payload accumulation within the tumor site. Leaky tumor vasculature could result in non-specific accumulation of nanocarriers, which could further hamper their anticancer potential. Vascular normalization could help accomplish adequate accumulation of nanocarriers within the TME and promote payload delivery within the deeper tumor tissues, which is absent in the conventional EPR effect. The tricky part lies in the chronology of treatment to be administered to the patient in the real-time scenario. It is clear that a vascular normalizing agent is to be administered prior to the anticancer drug. However, its pharmacokinetic and bio-distribution profile, onset and duration of action, potency, and accumulation at the active site, as well as its effect, must be studied in-depth for its successful translation. From a plethora of existing strategies (Table 2), the most appropriate strategy needs to be screened out, based on the pathophysiology and tumor progression at the site. This situation demands the amalgamation of expertise from oncologists, biopharmaceutical, and formulation scientists to design a dosage regimen to cater to the existing needs associated with vascular normalization. The site-specific action of vascular normalizing agents is another concern that could potentially be addressed by delivery experts. Additionally, successful tumor-mimicking organoids and tumor-on-a-chip model development play a key role in evaluating the strategy in situ, which could be translated in vivo. Fluorescent dye-labeled NCs can be utilized to check the improvement in the penetration of nano-formulations in the tumor post-normalization therapy. Combined rigorous efforts from biopharmaceutical scientists, oncologists, and formulation experts could circumvent the barriers associated with the clinical translation.
Conclusion and future perspectives
The hostile vascular and tumor microenvironment leads to the failure of chemo- and immune therapies. We conclude that for better perfusion of therapeutic moieties inside the TME, angiogenesis, and high intra-tumor pressure are the main barriers. Various strategies, such as vascular normalization, neoadjuvant chemotherapy, and anti-ECM agents, show promise. However, the design of dosing regimens plays an important role. Discussed studies have shown that the delivery of an anti-angiogenesis agent 1 to 3 days before chemo- or immunotherapy can normalize the TME and increase the accumulation of TE inside the tumor. However, extensive efforts are required for the clinical translation of such strategies. New formulations such as targeted nano-carriers should be developed to specifically target immature tumor vessels. As targeted nano-carriers improve the pharmacokinetic properties of the drugs, such formulations may help to attain rapid normalization.
Scientists all over the world rely on EPR for the accumulation of nanocarriers inside the TME. However, EPR presents itself as a double-edged sword. On one side, it increases the infiltration of nanocarriers inside the TME; while on the other hand, it increases the intra-tumor pressure. Reduction of ECM density increases the chemo- and immunotherapies by allowing the diffusion of drugs and infiltration of nano-carriers immune cells inside the TME. In our manuscript, we have discussed the advantages of co-administering TGF inhibitors along with chemotherapeutic agents. Tranilast was able to show tumor normalization and reduction of ECM. However, decreasing the ECM density may aid in the metastatic escape of the tumor cells. Hence, critical evaluations are required to gauge the pros and cons while employing anti-ECM agents. However, TGF inhibitors are known for their anti-metastatic potential. Their role may prove to be central in designing the improved therapies for BC management.
The use of metronomic therapy should also be extensively explored for the effective management of BC. As per reports, such therapies can reduce the number of cancer cells inside the tumor. However, the use of lower drug concentrations may lead to the emergence of more aggressive tumors. Lower drug concentrations may lead to the emergence of resistant tumor cells, which will re-populate the tumor. Furthermore, it may trigger extrinsic resistance pathways in the cancer cells, leading to the emergence of multi-drug resistance in cancer cells.
Hence, normalization of the tumor makes BC cells more prone to chemotherapy, immune-therapy and photodynamic therapies. The amalgamation of the said strategies along with nanomedicine and normalization principles and its clinical translation could help open novel avenues for researchers in cancer therapy. The transient nature of the normalization window is the stumbling block in achieving improved results. Strategies such as LIGHT, which may extend the normalization window, are needed at the hour.
Data Availability
Not applicable.
Abbreviations
- BC:
-
Breast Cancer
- NCs:
-
Nanocarriers
- TME:
-
Tumor Microenvironment
- ECs:
-
Endothelial Cells
- VEGF:
-
Vascular Endothelial Growth Factor
- DLL4:
-
Delta Ligand Like-4
- NCID:
-
Notch 1 Intracellular Domain
- PHD:
-
Propyl Hydroxylase Domain
- TE:
-
Therapeutic Entity
- ECM:
-
Extracellular Matrix
- YAP:
-
Yes-Associated Protein
- TAZ:
-
Transcriptional Coactivator with PDZ-Binding Motif
- PD-1:
-
Programmed Cell Death Protein-1
- CTLA-4:
-
Cytotoxic T lymphocyte Associated Protein-4
- TNBC:
-
Triple Negative Breast Cancer
- FASL:
-
FAS ligand
- ICAM:
-
Intercellular Adhesion Molecule 1
- VCAM:
-
Vascular Cell Adhesion Molecule
- SRCIN1:
-
SRC Kinase Signaling Inhibitor 1
- PTX:
-
Paclitaxel
- BMP-9:
-
Bone Morphogenetic Protein-9
- ALK1:
-
(Activin Receptor-Like Kinase 1)
- EGFR:
-
Epithelial Growth Factor Receptor
- MDSC:
-
Myeloid-derived Suppressor cells
- HIF-1α:
-
Hypoxia-Inducible Factor
- TLS:
-
Tertiary Lymphoid Structures
- PT:
-
Photodynamic Therapy
References
Famta P, Shah S, Jain N, Kumar KC, Bagasariya D, Khatri DK, et al. Tumor-promoting aftermath post-chemotherapy: A focus on breast cancer. Life Sci. 2022;310:121125.
Famta P, Shah S, Jain N, Srinivasarao DA, Murthy A, Ahmed T, et al. Albumin-hitchhiking: Fostering the pharmacokinetics and anticancer therapeutics. J Control Release. 2023;353:166–85.
Famta P, Shah S, Jain N, Srinivasarao DA, Vambhurkar G, Shahrukh S, et al. Nanocarrier-based drug delivery via cell-hitchhiking: Emphasizing pharmacokinetic perspective towards taming the “big-old” tumors. J Drug Deliv Sci Technol. 2023;89:105050.
Teleanu RI, Chircov C, Grumezescu AM, Teleanu DM. Tumor angiogenesis and anti-angiogenic strategies for cancer treatment. J Clin Med. 2020;9:84.
Ludwig N, Rubenich DS, Zaręba Ł, Siewiera J, Pieper J, Braganhol E, et al. Potential roles of tumor cell-and stroma cell-derived small extracellular vesicles in promoting a pro-angiogenic tumor microenvironment. Cancers (Basel). 2020;12:1–15.
Lugano R, Ramachandran M, Dimberg A. Tumor angiogenesis: causes, consequences, challenges and opportunities. Cell Mol Life Sci. 2020;77:1745–70.
Zeng A, Wang SR, He YX, Yan Y, Zhang Y. Progress in understanding of the stalk and tip cells formation involvement in angiogenesis mechanisms. Tissue Cell. 2021;73:101626.
A new concept for macromolecular therapeutics in cancer chemotherapy: mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs - PubMed [Internet]. [cited 2024 Jun 25]. Available from: https://pubmed.ncbi.nlm.nih.gov/2946403/
Schito L. Hypoxia-dependent angiogenesis and lymphangiogenesis in cancer. Adv Exp Med Biol. 2019;1136:71–85.
Rahma OE, Hodi FS. The intersection between tumor angiogenesis and immune suppression. Clin Cancer Res. 2019;25:5449–57.
Majidpoor J, Mortezaee K. Angiogenesis as a hallmark of solid tumors - clinical perspectives. Cell Oncol. 2021;44:715–37.
Yehya AHS, Asif M, Petersen SH, Subramaniam AV, Kono K, Majid AMSA, et al. Angiogenesis: Managing the culprits behind tumorigenesis and metastasis. Medicina (Lithuania). 2018;54:8.
Jena MK, Janjanam J. Role of extracellular matrix in breast cancer development: A brief update. F1000Res. 2018;7.
Zuazo-Gaztelu I, Casanovas O. Unraveling the role of angiogenesis in cancer ecosystems. Front Oncol. 2018;8:248.
Folkman J, Kalluri R. Beginning of angiogenesis research. In: Kufe DW, Pollock RE, Weichselbaum RR et al. editors. H-FCMedicine 6th edition. H (ON): BD. Beginning of angiogenesis research. Available from: https://www.ncbi.nlm.nih.gov/books/NBK13877/. 2003;1–31.
Hillen F, Griffioen AW. Tumour vascularization: Sprouting angiogenesis and beyond. Cancer Metastasis Rev. 2007;26:489–502.
Saravanan S, Vimalraj S, Pavani K, Nikarika R, Sumantran VN. Intussusceptive angiogenesis as a key therapeutic target for cancer therapy. Life Sci. 2020;252:117670.
Wei X, Chen Y, Jiang X, Peng M, Liu Y, Mo Y, et al. Mechanisms of vasculogenic mimicry in hypoxic tumor microenvironments. Mol Cancer. 2021;20:1–18.
Jiang X, Wang J, Deng X, Xiong F, Zhang S, Gong Z, et al. The role of microenvironment in tumor angiogenesis. J Exp Clin Cancer Res. 2020;39:1–19.
Shen Y, Quan J, Wang M, Li S, Yang J, Lv M, et al. Tumor vasculogenic mimicry formation as an unfavorable prognostic indicator in patients with breast cancer. Oncotarget. 2017;8:56408–16.
Lamalice L, Le Boeuf F, Huot J. Endothelial cell migration during angiogenesis. Circ Res. 2007;100:782–94.
Dallinga MG, Boas SE, Klaassen I, Merks RH, van Noorden CJ, Schlingemann RO. Tip Cells in Angiogenesis. eLS. 2015;1–10.
Huang CC, Kuo HM, Wu PC, Cheng SH, Chang TT, Chang YC, et al. Soluble delta-like 1 homolog (DLK1) stimulates angiogenesis through Notch1/Akt/eNOS signaling in endothelial cells. Angiogenesis. 2018;21:299–312.
Hida K, Maishi N, Annan DA, Hida Y. Contribution of tumor endothelial cells in cancer progression. Int J Mol Sci. 2018;19:1272.
Akil A, Gutiérrez-García AK, Guenter R, Rose JB, Beck AW, Chen H, et al. Notch Signaling in Vascular Endothelial Cells, Angiogenesis, and Tumor Progression: An Update and Prospective. Front Cell Dev Biol. 2021;9:177.
Lin J, Lin Y, Su L, Su Q, Guo W, Huang X, et al. The role of Jagged1/Notch pathway-mediated angiogenesis of hepatocarcinoma cells in vitro, and the effects of the spleen-invigorating and blood stasis-removing recipe. Oncol Lett. 2017;14:3616–22.
Fong GH, Takeda K. Role and regulation of prolyl hydroxylase domain proteins. Cell Death Differ. 2008;15:635–41.
Zhu Z, Hou Q, Guo H. NT5DC2 knockdown inhibits colorectal carcinoma progression by repressing metastasis, angiogenesis and tumor-associated macrophage recruitment: A mechanism involving VEGF signaling. Exp Cell Res. 2020;397: 112311.
Mezu-Ndubuisi OJ, Maheshwari A. The role of integrins in inflammation and angiogenesis. Pediatr Res. 2021;89:1619–26.
Xu S, Zhang H, Chong Y, Guan B, Guo P. YAP Promotes VEGFA Expression and Tumor Angiogenesis Though Gli2 in Human Renal Cell Carcinoma. Arch Med Res. 2019;50:225–33.
Zhu Q, Li J, Wu Q, Cheng Y, Zheng H, Zhan T, et al. Linc-OIP5 in the breast cancer cells regulates angiogenesis of human umbilical vein endothelial cells through YAP1/Notch/NRP1 signaling circuit at a tumor microenvironment. Biol Res. 2020;53:1–12.
Yeldag G, Rice A, Hernández A del R. Chemoresistance and the self-maintaining tumor microenvironment. Cancers (Basel). 2018;10.
Trédan O, Galmarini CM, Patel K, Tannock IF. Drug resistance and the solid tumor microenvironment. J Natl Cancer Inst. 2007;99:1441–54.
Dewhirst MW, Secomb TW. Transport of drugs from blood vessels to tumour tissue. Nat Rev Cancer. 2017;17:738–50.
Rasouli SS, Jolma IW, Friis HA. Impact of spatially varying hydraulic conductivities on tumor interstitial fluid pressure distribution. Inform Med Unlocked. 2019;16:100175.
Garnier L, Gkountidi AO, Hugues S. Tumor-associated lymphatic vessel features and immunomodulatory functions. Front Immunol. 2019;10.
Less JR, Skalak TC, Sevick EM, Jain RK. Microvascular network architecture in a mammary carcinoma. EXS. 1992;61:74–80.
Baxter LT, Jain RK. Transport of fluid and macromolecules in tumors. II. Role of heterogeneous perfusion and lymphatics. Microvasc Res. 1990;40:246–63.
Baxter LT, Jain RK. Transport of fluid and macromolecules in tumors. I. Role of interstitial pressure and convection. Microvasc Res. 1989;37:77–104.
Raavé R, van Kuppevelt TH, Daamen WF. Chemotherapeutic drug delivery by tumoral extracellular matrix targeting. J Control Release. 2018;274:1–8.
Purkayastha P, Jaiswal MK, Lele TP. Molecular cancer cell responses to solid compressive stress and interstitial fluid pressure. Cytoskeleton. 2021;78:312–22.
Secomb TW, Hsu R, Dewhirst MW, Klitzman B, Gross JF. Analysis of oxygen transport to tumor tissue by microvascular networks. Int J Radiat Oncol Biol Phys. 1993;25:481–9.
Ishihara S, Haga H. Matrix Stiffness Contributes to Cancer Progression by Regulating Transcription Factors. Cancers (Basel). 2022;14:1049.
Keenan TE, Tolaney SM. Role of immunotherapy in triple-negative breast cancer. JNCCN Journal of the National Comprehensive Cancer Network. 2020;18:479–89.
Deshmukh SK, Srivastava SK, Poosarla T, Dyess DL, Holliday NP, Singh AP, et al. Inflammation, immunosuppressive microenvironment and breast cancer: opportunities for cancer prevention and therapy. Ann Transl Med. 2019;7:593–593.
Abaza A, Sid Idris F, Anis Shaikh H, Vahora I, Moparthi KP, Al Rushaidi MT, et al. Programmed Cell Death Protein 1 (PD-1) and Programmed Cell Death Ligand 1 (PD-L1) Immunotherapy: A Promising Breakthrough in Cancer Therapeutics. Cureus. 2023
Ghahremani Dehbokri S, Alizadeh N, Isazadeh A, Baghbanzadeh A, Abbaspour-Ravasjani S, Hajiasgharzadeh K, et al. CTLA-4: As an Immunosuppressive Immune Checkpoint in Breast Cancer. Curr Mol Med. 2023;23:521–6.
Swoboda A, Nanda R. Immune Checkpoint Blockade for Breast Cancer. 2018. p. 155–65.
Debien V, De Caluwé A, Wang X, Piccart-Gebhart M, Tuohy VK, Romano E, et al. Immunotherapy in breast cancer: an overview of current strategies and perspectives. NPJ Breast Cancer. 2023;9:7.
Alhawary SIS, Saleh EAM, Mamajanov NA, S. Gilmanova N, Alsaab HO, Alghamdi A, et al. Breast cancer vaccines A comprehensive and updated review. Pathol Res Pract. 2023;249:154735.
Dirix LY, Takacs I, Jerusalem G, Nikolinakos P, Arkenau HT, Forero-Torres A, et al. Avelumab, an anti-PD-L1 antibody, in patients with locally advanced or metastatic breast cancer: A phase 1b JAVELIN solid tumor study. Breast Cancer Res Treat. 2018;167:671–86.
Chen W, Shen L, Jiang J, Zhang L, Zhang Z, Pan J, et al. Antiangiogenic therapy reverses the immunosuppressive breast cancer microenvironment. Biomark Res. 2021;9:1–16.
Meng S, Li LI, Zhou M, Jiang W, Heng NIU, Yang K. Distribution and prognostic value of tumor-infiltrating T cells in breast cancer. Mol Med Rep. 2018;18:4247–58.
Vito A, El-Sayes N, Mossman K. Hypoxia-Driven Immune Escape in the Tumor Microenvironment. Cells. 2020;9.
Lee CT, MacE T, Repasky EA. Hypoxia-driven immunosuppression: A new reason to use thermal therapy in the treatment of cancer? Int J Hyperth. 2010;26:232–46.
Motz GT, Santoro SP, Wang LP, Garrabrant T, Lastra RR, Hagemann IS, et al. Tumor endothelium FasL establishes a selective immune barrier promoting tolerance in tumors. Nat Med. 2014;20:607–15.
Harjunpää H, Asens ML, Guenther C, Fagerholm SC. Cell adhesion molecules and their roles and regulation in the immune and tumor microenvironment. Front Immunol. 2019;10:1078.
Tilley SL, Coffman TM, Koller BH. Mixed messages: modulation of inflammation and immune responses by prostaglandins and thromboxanes. J Clin Invest. 2001;108:15–23.
Strassmann G, Patil-Koota V, Finkelman F, Fong M, Kambayashi T. Evidence for the involvement of interleukin 10 in the differential deactivation of murine peritoneal macrophages by prostaglandin E2. J Exp Med. 1994;180:2365–70.
Siegel PM, Massagué J. Cytostatic and apoptotic actions of TGF-β in homeostasis and cancer. Nat Rev Cancer. 2003;3:807–20.
Derynck R, Akhurst RJ, Balmain A. TGF-β signaling in tumor suppression and cancer progression. Nat Genet. 2001;29:117–29.
Dumont N, Arteaga CL. Transforming growth factor-beta and breast cancer: Tumor promoting effects of transforming growth factor-β. Breast Cancer Res. 2000;2:125.
Mouri H, Sakaguchi K, Sawayama T, Senoh T, Ohta T, Nishimura M, et al. Suppressive effects of transforming growth factor-beta1 produced by hepatocellular carcinoma cell lines on interferon-gamma production by peripheral blood mononuclear cells. Acta Med Okayama. 2002;56:309–15.
Zhang S, Regan K, Najera J, Grinstaff MW, Datta M, Nia HT. The peritumor microenvironment: physics and immunity. Trends Cancer. 2023;9:609–23.
Friese MA, Wischhusen J, Wick W, Weiler M, Eisele G, Steinle A, et al. RNA Interference Targeting Transforming Growth Factor-β Enhances NKG2D-Mediated Antiglioma Immune Response, Inhibits Glioma Cell Migration and Invasiveness, and Abrogates Tumorigenicity In vivo. Cancer Res. 2004;64:7596–603.
Lee J-C, Lee K-M, Kim D-W, Heo DS. Elevated TGF-β1 Secretion and Down-Modulation of NKG2D Underlies Impaired NK Cytotoxicity in Cancer Patients. J Immunol. 2004;172:7335–40.
Chitadze G, Kabelitz D. Immune surveillance in glioblastoma: Role of the <scp>NKG2D</scp> system and novel cell‐based therapeutic approaches. Scand J Immunol. 2022;96.
Beck C, Schreiber H, Rowley DA. Role of TGF-? in immune-evasion of cancer. Microsc Res Tech. 2001;52:387–95.
Hanks BA, Holtzhausen A, Evans KS, Jamieson R, Gimpel P, Campbell OM, et al. Type III TGF-β receptor downregulation generates an immunotolerant tumor microenvironment. J Clin Investig. 2013;123:3925–40.
Yang AS, Lattime EC. Tumor-induced interleukin 10 suppresses the ability of splenic dendritic cells to stimulate CD4 and CD8 T-cell responses. Cancer Res. 2003;63:2150–7.
Sharma S, Stolina M, Lin Y, Gardner B, Miller PW, Kronenberg M, et al. T cell-derived IL-10 promotes lung cancer growth by suppressing both T cell and APC function. J Immunol. 1999;163:5020–8.
Bhat AA, Goyal A, Thapa R, almalki WH, Kazmi I, Alzarea SI, et al. Uncovering the complex role of interferon-gamma in suppressing type 2 immunity to cancer. Cytokine. 2023;171:156376.
Menetrier-Caux C, Montmain G, Dieu MC, Bain C, Favrot MC, Caux C, et al. Inhibition of the Differentiation of Dendritic Cells From CD34+ Progenitors by Tumor Cells: Role of Interleukin-6 and Macrophage Colony-Stimulating Factor. Blood. 1998;92:4778–91.
Wang Y, Xiang Y, Xin VW, Wang X-W, Peng X-C, Liu X-Q, et al. Dendritic cell biology and its role in tumor immunotherapy. J Hematol Oncol. 2020;13:107.
Vitiello PF, Shainheit MG, Allison EM, Adler EP, Kurt RA. Impact of tumor-derived CCL2 on T cell effector function. Immunol Lett. 2004;91:239–45.
Peng L, Shu S, Krauss JC. Monocyte chemoattractant protein inhibits the generation of tumor-reactive T cells. Cancer Res. 1997;57:4849–54.
Pozzi S, Satchi-Fainaro R. The role of CCL2/CCR2 axis in cancer and inflammation: The next frontier in nanomedicine. Adv Drug Deliv Rev. 2024;209: 115318.
Renschler MF. The emerging role of reactive oxygen species in cancer therapy. Eur J Cancer. 2004;40:1934–40.
Bogdan C. Nitric oxide and the immune response. Nat Immunol. 2001;2:907–16.
Thomsen LL, Miles DW. Role of nitric oxide in tumour progression: lessons from human tumours. Cancer Metastasis Rev. 1998;17:107–18.
Mantovani A, Allavena P, Marchesi F, Garlanda C. Macrophages as tools and targets in cancer therapy. Nat Rev Drug Discov. 2022;21:799–820.
Kono K, Salazar-Onfray F, Petersson M, Hansson J, Masucci G, Wasserman K, et al. Hydrogen peroxide secreted by tumor-derived macrophages down-modulates signal-transducing zeta molecules and inhibits tumor-specific T cell-and natural killer cell-mediated cytotoxicity. Eur J Immunol. 1996;26:1308–13.
Takahashi A, Kono K, Ichihara F, Sugai H, Amemiya H, Iizuka H, et al. Macrophages in tumor-draining lymph node with different characteristics induce T-cell apoptosis in patients with advanced stage-gastric cancer. Int J Cancer. 2003;104:393–9.
He M, Wang M, Xu T, Zhang M, Dai H, Wang C, et al. Reactive oxygen species-powered cancer immunotherapy: Current status and challenges. J Control Release. 2023;356:623–48.
Jain RK. Normalization of tumor vasculature: An emerging concept in antiangiogenic therapy. Science. 1979;2005(307):58–62.
Cantelmo AR, Pircher A, Kalucka J, Carmeliet P. Vessel pruning or healing: endothelial metabolism as a novel target? Expert Opin Ther Targets. 2017;21:239–47.
Abou Khouzam R, Brodaczewska K, Filipiak A, Zeinelabdin NA, Buart S, Szczylik C, et al. Tumor Hypoxia Regulates Immune Escape/Invasion: Influence on Angiogenesis and Potential Impact of Hypoxic Biomarkers on Cancer Therapies. Front Immunol. 2021;11:3479.
Thews O, Riemann A. Tumor pH and metastasis: a malignant process beyond hypoxia. Cancer Metastasis Rev. 2019;38:113–29.
Li S, Zhang Q, Hong Y. Tumor Vessel Normalization: A Window to Enhancing Cancer Immunotherapy. Technol Cancer Res Treat. 2020;19.
Martin JD, Seano G, Jain RK. Normalizing Function of Tumor Vessels: Progress, Opportunities, and Challenges. Annu Rev Physiol. 2019;81:505–34.
Huynh H, Lee LY, Goh KY, Ong R, Hao HX, Huang A, et al. Infigratinib Mediates Vascular Normalization, Impairs Metastasis, and Improves Chemotherapy in Hepatocellular Carcinoma. Hepatology. 2019;69:943–58.
Viallard C, Larrivée B. Tumor angiogenesis and vascular normalization: alternative therapeutic targets. Angiogenesis. 2017;20:409–26.
Muz B, de la Puente P, Azab F, Azab AK. The role of hypoxia in cancer progression, angiogenesis, metastasis, and resistance to therapy. Hypoxia. 2015;3:83.
Magnussen AL, Mills IG. Vascular normalisation as the stepping stone into tumour microenvironment transformation. Br J Cancer. 2021;125:324–36.
Shi R, Liao C, Zhang Q. Hypoxia-driven effects in cancer: Characterization, mechanisms, and therapeutic implications. Cells. 2021;10:1–26.
Li W, Quan YY, Li Y, Lu L, Cui M. Monitoring of tumor vascular normalization: The key points from basic research to clinical application. Cancer Manag Res. 2018;10:4163–72.
Yu M, Han Y, Zhuo H, Zhang S. Endostar, a Modified Endostatin Induces Vascular Normalization to Improve Chemotherapy Efficacy Through Suppression of Src Signaling Pathway. Cancer Biother Radiopharm. 2018;33:131–8.
Jain RK, Martin JD, Stylianopoulos T. The role of mechanical forces in tumor growth and therapy. Annu Rev Biomed Eng. 2014;16:321–46.
Abyaneh HS, Regenold M, McKee TD, Allen C, Gauthier MA. Towards extracellular matrix normalization for improved treatment of solid tumors. Theranostics. 2020;10:1960–80.
Pasquier E, Kavallaris M, André N. Metronomic chemotherapy: new rationale for new directions. Nat Rev Clin Oncol. 2010;7:455–65.
Cong L, Maishi N, Annan DA, Young MF, Morimoto H, Morimoto M, et al. Inhibition of stromal biglycan promotes normalization of the tumor microenvironment and enhances chemotherapeutic efficacy. Breast Cancer Res. 2021;23:1–17.
Sitohy B, Nagy JA, Dvorak HF. Anti-VEGF/VEGFR therapy for cancer: Reassessing the target. Cancer Res. 2012;72:1909–14.
Drevs J. VEGF and angiogenesis: implications for breast cancer therapy. Eur J Cancer Suppl. 2008;6:7–13.
Sun Z, Lan X, Xu S, Li S, Xi Y. Efficacy of bevacizumab combined with chemotherapy in the treatment of HER2-negative metastatic breast cancer: A network meta-analysis. BMC Cancer. 2020;20:1–17.
Goldfarb SB, Dickler MN. Bevacizumab in metastatic breast cancer: When may it be used? Ther Adv Med Oncol. 2011;3:85–93.
Li Q, Yan H, Zhao P, Yang Y, Cao B. Efficacy and safety of bevacizumab combined with chemotherapy for managing metastatic breast cancer: A meta-analysis of randomized controlled trials. Sci Rep. 2015;5:1–11.
Boucher Y, Kumar AS, Posada JM, Gjini E, Pfaff K, Lipschitz M, et al. Bevacizumab improves tumor infiltration of mature dendritic cells and effector T-cells in triple-negative breast cancer patients. NPJ Precis Oncol. 2021;5:1–6.
Yadav K, Lim J, Choo J, Ow SGW, Wong A, Lee M, et al. Immunohistochemistry study of tumor vascular normalization and anti-angiogenic effects of sunitinib versus bevacizumab prior to dose-dense doxorubicin/cyclophosphamide chemotherapy in HER2-negative breast cancer. Breast Cancer Res Treat. 2022;192:131–42.
Mollard S, Ciccolini J, Imbs DC, El Cheikh R, Barbolosi D, Benzekry S. Model driven optimization of antiangiogenics + cytotoxics combination: Application to breast cancer mice treated with bevacizumab + paclitaxel doublet leads to reduced tumor growth and fewer metastasis. Oncotarget. 2017;8:23087–98.
Bin Chen B, Lu YS, Lin CH, Chen WW, Wu PF, Hsu CY, et al. A pilot study to determine the timing and effect of bevacizumab on vascular normalization of metastatic brain tumors in breast cancer. BMC Cancer. 2016;16:1–8.
Tolaney SM, Boucher Y, Duda DG, Martin JD, Seano G, Ancukiewicz M, et al. Role of vascular density and normalization in response to neoadjuvant bevacizumab and chemotherapy in breast cancer patients. Proc Natl Acad Sci. 2015;112:14325–30.
Salmon RM, Guo J, Wood JH, Tong Z, Beech JS, Lawera A, et al. Molecular basis of ALK1-mediated signalling by BMP9/BMP10 and their prodomain-bound forms. Nat Commun. 2020;11:1–16.
Ayuso-Íñigo B, Méndez-García L, Pericacho M, Muñoz-Félix JM. The dual effect of the BMP9–ALK1 pathway in blood vessels: An opportunity for cancer therapy improvement? Cancers (Basel). 2021;13:5412.
Ouarné M, Bouvard C, Boneva G, Mallet C, Ribeiro J, Desroches-Castan A, et al. BMP9, but not BMP10, acts as a quiescence factor on tumor growth, vessel normalization and metastasis in a mouse model of breast cancer. J Exp Clin Cancer Res. 2018;37:1–12.
Chen Q, Xu L, Chen J, Yang Z, Liang C, Yang Y, et al. Tumor vasculature normalization by orally fed erlotinib to modulate the tumor microenvironment for enhanced cancer nanomedicine and immunotherapy. Biomaterials. 2017;148:69–80.
Mirando AC, Patil A, Rafie CI, Christmas BJ, Pandey NB, Stearns V, et al. Regulation of the tumor immune microenvironment and vascular normalization in TNBC murine models by a novel peptide. Oncoimmunology. 2020;9:1760685.
Li Q, Wang Y, Jia W, Deng H, Li G, Deng W, et al. Low-dose anti-angiogenic therapy sensitizes breast cancer to PD-1 blockade. Clin Cancer Res. 2020;26:1712–24.
Melaiu O, Vanni G, Portarena I, Pistolese CA, Anemona L, Pomella S, et al. The Combination of Immune Checkpoint Blockade with Tumor Vessel Normalization as a Promising Therapeutic Strategy for Breast Cancer: An Overview of Preclinical and Clinical Studies. Int J Mol Sci. 2023;24:3226.
Kim SJ, Jung KH, Son MK, Park JH, Yan HH, Fang Z, et al. Tumor vessel normalization by the PI3K inhibitor HS-173 enhances drug delivery. Cancer Lett. 2017;403:339–53.
Kunduzova O, Alet N, Delesque-Touchard N, Millet L, Castan-Laurell I, Muller C, et al. Apelin/APJ signaling system: a potential link between adipose tissue and endothelial angiogenic processes. FASEB J. 2008;22:4146–53.
Claesson-Welsh L. What is normal? Apelin and VEGFA, drivers of tumor vessel abnormality. EMBO Mol Med. 2019;11:e10892.
Uribesalgo I, Hoffmann D, Zhang Y, Kavirayani A, Lazovic J, Berta J, et al. Apelin inhibition prevents resistance and metastasis associated with anti-angiogenic therapy. EMBO Mol Med. 2019;11:e9266.
Jones LW, Viglianti BL, Tashjian JA, Kothadia SM, Keir ST, Freedland SJ, et al. Effect of aerobic exercise on tumor physiology in an animal model of human breast cancer. J Appl Physiol. 2010;108:343–8.
Schadler KL, Thomas NJ, Galie PA, Bhang DH, Roby KC, Addai P, et al. Tumor vessel normalization after aerobic exercise enhances chemotherapeutic efficacy. Oncotarget. 2016;7:65429–40.
Broders-Bondon F, Nguyen Ho-Bouldoires TH, Fernandez-Sanchez M-E, Farge E. Mechanotransduction in tumor progression: The dark side of the force. J Cell Biol. 2018;217:1571–87.
Martin JD, Panagi M, Wang C, Khan TT, Martin MR, Voutouri C, et al. Dexamethasone Increases Cisplatin-Loaded Nanocarrier Delivery and Efficacy in Metastatic Breast Cancer by Normalizing the Tumor Microenvironment. ACS Nano. 2019.
Panagi M, Voutouri C, Mpekris F, Papageorgis P, Martin MR, Martin JD, et al. TGF-β inhibition combined with cytotoxic nanomedicine normalizes triple negative breast cancer microenvironment towards anti-tumor immunity. Theranostics. 2020;10:1910–22.
Mpekris F, Panagi M, Voutouri C, Martin JD, Samuel R, Takahashi S, et al. Normalizing the Microenvironment Overcomes Vessel Compression and Resistance to Nano-immunotherapy in Breast Cancer Lung Metastasis. Advanced Science. 2021;8:2001917.
Sorace AG, Quarles CC, Whisenant JG, Hanker AB, McIntyre JO, Sanchez VM, et al. Trastuzumab improves tumor perfusion and vascular delivery of cytotoxic therapy in a murine model of HER2+ breast cancer: preliminary results. Breast Cancer Res Treat. 2016;155:273–84.
Jain RK. Antiangiogenesis Strategies Revisited: From Starving Tumors to Alleviating Hypoxia. Cancer Cell. 2014;26:605–22.
Ho YJ, Chu SW, Liao EC, Fan CH, Chan HL, Wei KC, et al. Normalization of tumor vasculature by oxygen microbubbles with ultrasound. Theranostics. 2019;9:7370–83.
Wang Q, Xiang Q, Yu L, Hu T, Chen Y, Wang J, et al. Corrigendum: Changes in Tumor-Infiltrating Lymphocytes and Vascular Normalization in Breast Cancer Patients After Neoadjuvant Chemotherapy and Their Correlations With DFS (Front. Oncol, (2020), 9, (1545), Front Oncol. 2021;11:1545.
Engelhard VH, Rodriguez AB, Mauldin IS, Woods AN, Peske JD, Slingluff CL. Immune Cell Infiltration and Tertiary Lymphoid Structures as Determinants of Antitumor Immunity. J Immunol. 2018;200:432–42.
Munoz-Erazo L, Rhodes JL, Marion VC, Kemp RA. Tertiary lymphoid structures in cancer – considerations for patient prognosis. Cell Mol Immunol. 2020;17:570–5.
Dieu-Nosjean MC, Goc J, Giraldo NA, Sautès-Fridman C, Fridman WH. Tertiary lymphoid structures in cancer and beyond. Trends Immunol. 2014;35:571–80.
Sautès-Fridman C, Petitprez F, Calderaro J, Fridman WH. Tertiary lymphoid structures in the era of cancer immunotherapy. Nat Rev Cancer. 2019;19:307–25.
Pimenta EM, Barnes BJ. Role of tertiary lymphoid structures (TLS) in anti-tumor immunity: Potential tumor-induced cytokines/chemokines that regulate TLS formation in epithelial-derived cancers. Cancers (Basel). 2014;6:969–97.
Thompson ED, Enriquez HL, Fu YX, Engelhard VH. Tumor masses support naive T cell infiltration, activation, and differentiation into effectors. J Exp Med. 2010;207:1791–804.
Johansson-Percival A, He B, Li ZJ, Kjellén A, Russell K, Li J, et al. De novo induction of intratumoral lymphoid structures and vessel normalization enhances immunotherapy in resistant tumors. Nat Immunol. 2017;18:1207–17.
Pantelidou C, Jadhav H, Kothari A, Liu R, Guerriero JL, Shapiro GI. STING agonism enhances anti-tumor immune responses and therapeutic efficacy of PARP inhibition in BRCA-associated breast cancer. bioRxiv. 2021;2021.01.26.428337.
Rivera Vargas T, Benoit-Lizon I, Apetoh L. Rationale for stimulator of interferon genes–targeted cancer immunotherapy. Eur J Cancer. 2017;75:86–97.
Corrales L, Glickman LH, McWhirter SM, Kanne DB, Sivick KE, Katibah GE, et al. Direct Activation of STING in the Tumor Microenvironment Leads to Potent and Systemic Tumor Regression and Immunity. Cell Rep. 2015;11:1018–30.
Yang H, Lee WS, Kong SJ, Kim CG, Kim JH, Chang SK, et al. STING activation reprograms tumor vasculatures and synergizes with VEGFR2 blockade. J Clin Investig. 2019;129:4350–64.
Tarik Alhamdany A, Saeed AMH, Alaayedi M. Nanoemulsion and Solid Nanoemulsion for Improving Oral Delivery of a Breast Cancer Drug: Formulation, Evaluation, and a Comparison Study. Saudi Pharmaceutical Journal. 2021;29:1278–88.
Kumari M, Sharma N, Manchanda R, Gupta N, Syed A, Bahkali AH, et al. PGMD/curcumin nanoparticles for the treatment of breast cancer. Sci Rep. 2021;11:3824.
Park JW. Liposome-based drug delivery in breast cancer treatment. Breast Cancer Res. 2002;4:95.
Chaudhuri A, Ramesh K, Kumar DN, Dehari D, Singh S, Kumar D, et al. Polymeric micelles: A novel drug delivery system for the treatment of breast cancer. J Drug Deliv Sci Technol. 2022;77: 103886.
Zenze M, Daniels A, Singh M. Dendrimers as Modifiers of Inorganic Nanoparticles for Therapeutic Delivery in Cancer. Pharmaceutics. 2023;15:398.
Shah A, Aftab S, Nisar J, Ashiq MN, Iftikhar FJ. Nanocarriers for targeted drug delivery. J Drug Deliv Sci Technol. 2021;62: 102426.
Edis Z, Wang J, Waqas MK, Ijaz M, Ijaz M. Nanocarriers-Mediated Drug Delivery Systems for Anticancer Agents: An Overview and Perspectives. Int J Nanomedicine. 2021;16:1313–30.
Yusuf A, Almotairy ARZ, Henidi H, Alshehri OY, Aldughaim MS. Nanoparticles as Drug Delivery Systems: A Review of the Implication of Nanoparticles’ Physicochemical Properties on Responses in Biological Systems. Polymers (Basel). 2023;15:1596.
Patra JK, Das G, Fraceto LF, Campos EVR, Rodriguez-Torres MDP, Acosta-Torres LS, et al. Nano based drug delivery systems: recent developments and future prospects. Journal of Nanobiotechnology 2018 16:1 [Internet]. 2018 [cited 2024 Jun 25];16:1–33. Available from: https://jnanobiotechnology.biomedcentral.com/articles/https://doi.org/10.1186/s12951-018-0392-8
Siddique S, Chow JCL. Recent Advances in Functionalized Nanoparticles in Cancer Theranostics. Nanomaterials. 2022;12:2826.
Luiz MT, Dutra JAP, De Araújo JTC, Di Filippo LD, Duarte JL, Chorilli M. Functionalization of Nanosystems in Cancer Treatment. Cancer Nanotechnol. Cham: Springer International Publishing; 2023. p. 71–101.
Haripriyaa M, Suthindhiran K. Pharmacokinetics of nanoparticles: current knowledge, future directions and its implications in drug delivery. Futur J Pharm Sci. 2023;9:113.
Dos Santos AF, De Almeida DRQ, Terra LF, Baptista MS, Labriola L. Photodynamic therapy in cancer treatment - an update review. J Cancer Metastasis Treat. 2019;2019:25.
Shen Z, Ma Q, Zhou X, Zhang G, Hao G, Sun Y, et al. Strategies to improve photodynamic therapy efficacy by relieving the tumor hypoxia environment. NPG Asia Mater. 2021;13:1–19.
Xu XL, Chen MX, Lou XF, Du YY, Shu GF, Qi J, et al. Sialic acid-modified mesoporous polydopamine induces tumor vessel normalization to enhance photodynamic therapy by inhibiting VE-cadherin internalization. Chem Eng J. 2021;414:128743.
Li Y, Liu Y, Du B, Cheng G. Reshaping Tumor Blood Vessels to Enhance Drug Penetration with a Multistrategy Synergistic Nanosystem. Mol Pharm. 2020;17:3151–64.
Wang L, Fang D, Xu J, Luo R. Various pathways of zoledronic acid against osteoclasts and bone cancer metastasis: a brief review. BMC Cancer. 2020;20:1–10.
Ishtiaq S, Edwards S, Sankaralingam A, Evans BAJ, Elford C, Frost ML, et al. The effect of nitrogen containing bisphosphonates, zoledronate and alendronate, on the production of pro-angiogenic factors by osteoblastic cells. Cytokine. 2015;71:154–60.
Clézardin P, Ebetino FH, Fournier PGJ. Bisphosphonates and cancer-induced bone disease: Beyond their antiresorptive activity. Cancer Res. 2005;65:4971–4.
Jun Cai X, Dong Fei W, Ying XuY, Xu H, Yi Yang G, Wei Cao J, et al. Liposome-Encapsulated Zoledronate Favors Tumor Vascular Normalization and Enhances Anticancer Efficacy of Cisplatin. AAPS PharmSciTech. 2020;21:1–14.
Teng RJ, Rana U, Afolayan AJ, Zhao B, Miao QR, Konduri GG. Nogo-B receptor modulates angiogenesis response of pulmonary artery endothelial cells through eNOS coupling. Am J Respir Cell Mol Biol. 2014;51:169–77.
Wang B, Ding Y, Zhao X, Han X, Yang N, Zhang Y, et al. Delivery of small interfering RNA against Nogo-B receptor via tumor-acidity responsive nanoparticles for tumor vessel normalization and metastasis suppression. Biomaterials. 2018;175:110–22.
Acknowledgements
The authors would like to acknowledge the research funding support by the Department of Pharmaceuticals (DoP), Ministry of Chemicals and Fertilizers, Govt. of India to “Pharmaceutical Innovation and Translational Research Lab” (PITRL), National Institute of Pharmaceutical Education and Research (NIPER) Hyderabad, INDIA.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
Paras Famta, Saurabh Shah: Conceptualization; Manuscript writing- original draft. Ganesh Vambhurkar, Giriraj Pandey, Deepkumar Bagasariya, Kondasingh Charan Kumar: Data Curation, Manuscript writing – editing. Sajja Bhanu Prasad, Akshay Shinde, Suraj Wagh, Dadi A. Srinivasarao, Rahul Kumar: Data Curation. Dharmendra Kumar Khatri, Amit Asthana: Supervision. Saurabh Srivastava: Conceptualization, Manuscript writing-editing, Supervision. Corresponding author: Correspondence to Dr. Saurabh Srivastava
Corresponding author
Ethics declarations
Ethics approval and Consent to participate
Not applicable.
Consent for publication
All authors approved for publication.
Competing interests
The authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Famta, P., Shah, S., Vambhurkar, G. et al. Amelioration of breast cancer therapies through normalization of tumor vessels and microenvironment: paradigm shift to improve drug perfusion and nanocarrier permeation. Drug Deliv. and Transl. Res. (2024). https://doi.org/10.1007/s13346-024-01669-9
Accepted:
Published:
DOI: https://doi.org/10.1007/s13346-024-01669-9