Abstract
Tryptophan metabolites in plasma samples from 20 male subjects with type 2 diabetes mellitus (T2DM) and 20 nondiabetic reference males were analyzed by ultra high performance liquid chromatography. Tryptophan levels in the diabetic subjects were significantly lower than those in nondiabetic subjects. The concentrations of 5-hydroxytryptophan, 5-hydroxyindoleacetic acid, kynurenic acid, 3-hydroxykynurenine, 3-hydroxyanthranilic acid, and xanthurenic acid were found to be higher in the diabetic patients. When the diabetic patients were divided into higher- and lower-tryptophan groups, the concentrations of 5-hydroxytryptophan, indole-3-acetic acid, kynurenine, 5-hydroxykynurenine, and kynurenic acid were found to be higher in the diabetic patients with higher tryptophan levels. However, diabetic patients with lower plasma tryptophan levels had higher levels of 5-hydroxyindoleacetic acid than the patients with higher tryptophan levels. These results suggest that tryptophan was metabolized more in T2DM patients than in nondiabetic subjects. In the kynurenine pathway, the degradation of tryptophan seems to be accelerated in patients with higher plasma levels of tryptophan than in patients with lower levels of tryptophan. In the serotonin pathway, when the level of tryptophan is low, the conversion of serotonin to 5-hydroxyindoleacetic acid appears to be accelerated. In conclusion, our results suggest that T2DM patients may be exposed to stress constantly.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Recently, tryptophan metabolites such as kynurenic acids were found to be linked to the risk of coronary heart disease. Eussen [1] indicated that kynurenine and 3-hydroxykynurenine were associated with an increased risk of acute coronary events, and suggested that the kynurenine pathway is involved in the early development of coronary heart disease. In patients with suspected stable angina pectoris, elevated plasma kynurenines predicted an increased risk of acute myocardial infarction [2].
Serotonin, another metabolite of tryptophan, has been implicated in mediating diverse physiologic and psychiatric processes. Stress produces a number of changes in the metabolism of central transmitters, particularly noradrenaline and serotonin [3, 4]. A number of studies have demonstrated that increased plasma (and therefore brain) levels of tryptophan not only augment brain levels of serotonin, but also kynurenine synthesis [5].
We have shown that applying foot shock to rats results in increases in not only brain serotonin levels but also kynurenine levels in plasma, kidney, liver, and every part of the brain [6–8].
Several tryptophan metabolites have been shown to exhibit neuroexcitatory, convulsant, and toxic properties [9, 10]
In the periphery, only 1 % of dietary tryptophan was converted to serotonin and more than 95 % was metabolized to kynurenines [4, 5].
We thought that tryptophan metabolism may be altered in diabetes mellitus (DM) patients due to the many stresses to which DM patients are exposed, so we measured the plasma levels of serotonin and many tryptophan metabolites in DM and non-DM old men.
Methods
We asked acquaintances older than 50 years old to enrol in the study, and checked their health carefully. We recruited them if they had no health problems such as diabetes and hypertension, had not suffered from any serious diseases in the past, and had not been smokers previously. We also asked the DM patients to provide blood samples for assays of blood parameters and amino acids. We obtained informed consent from the patients prior to conducting the protocol, which had been approved by the Ethical Committee of Showa Women’s University and Saiseikai Main Hospital.
Healthy participants were given self-administered diet history questionnaires, and provided answers for each item by recollecting their diets. Using the data from these questionnaires, we calculated their energy, carbohydrate, fat, and protein intakes. We unfortunately did not obtain self-administered diet history questionnaire data from DM patients.
Measurements of blood parameters
The plasma from the blood samples provided by the patients was obtained by centrifugation, and levels of lipids, amino acids, and insulin were measured as background information for these participants.
The plasma samples were sent to SRL Inc. to assay amino acids and lipids. Thirty-eight amino acids and related substances such as taurine as well as the total amino acids, total essential amino acids, and total nonessential amino acids were measured. In short, the amino acids were measured by high-speed liquid chromatography, and cholesterol was measured by homogeneous methods. Triglycerides were measured by GK/GPO methods. Insulin was measured by a CLEIA (chemiluminescent immunoassay) method.
Measurements of tryptophan metabolites
Reagents
The standard materials of 15 major tryptophan metabolites—tryptophan, L-5-hydroxytryptophan, serotonin, L-kynurenine (KYN), 3-hydroxykynurenine (3-HKYN), 5-hydroxytryptophol (5-HTOL), tryptophol (TOL), kynurenic acid (KA), quinaldic acid (QA), xanthurenic acid (XA), 5-hydroxyindole-3-acetic acid (HIAA), indole-3-acetic acid (IAA), 3-hydroxyanthranilic acid (HAA), anthranilic acid (AA), and indole-3-lactic acid (ILA)—were special-grade or biochemical-grade reagents purchased from Wako Pure Chemical Industries, Ltd. (Osaka, Japan). The formic acid and acetonitrile used were LC/MS-grade reagents, and other major chemicals such as methanol were special-grade reagents purchased from Wako Chemicals too. Water was obtained as needed from the installed Milli-Q water purification system (Merck–Millipore, Billerica, MA, USA).
Each standard “mother” solution was prepared to a concentration of 1000 mg/L from the specified reagent in water and/or an aqueous 0.1 mol/L solution of sodium hydroxide. The standard mixture solution for each concentration level used to construct the calibration curve was prepared by diluting the mother solution with water.
Instrumentation and analytical conditions
Tryptophan metabolites were analyzed using a Shimadzu (Kyoto, Japan) LCMS-8060 or 8050 system (with an electrospray interface) coupled to a Nexera UHPLC system, including solvent delivery units, an autosampler, and a column oven. The HPLC column used was an L-column2 ODS (2.1 mm × 150 mm, 3 µm; CERI: Chemical Evaluation and Research Institute, Tokyo, Japan) with a gradient elution of 0.1 % formic acid/acetonitrile. Before analyses, the multiple reaction monitoring (MRM) conditions were optimized using a standard solution. The optimized MRM transitions are shown in Table 1.
Gradient elution was performed using a high-pressure binary gradient program: 5 % of an aqueous 0.1 % formic acid solution for 3 min with acetonitrile, then 5–95 % of an aqueous 0.1 % formic acid solution for 6 min, followed by 95 % of an aqueous 0.1 % formic acid solution for another 3 min at a flow rate of 0.4 mL/min at 40 °C. Ionization was performed in electrospray (ESI) positive mode at 150 °C (the desolvation line) and 400 °C (the interface) with 3 L/min nebulizing and 5 L/min drying gas. Chromatographic data were obtained in MRM mode with optimized transitions.
Sample pretreatment
The samples were stored at −80 °C.
A fifty-microliter aliquot of the blood plasma sample of interest was placed in a 1.5-mL test tube, and then 25 µL of a 0.1 % aqueous formic acid solution, 150 µL of acetonitrile, and an additional 75 µL of 0.1 % aqueous formic acid solution were added, respectively. The solution was vortexed for 30 s and then left to stand for 5 min at a cooling temperature. After this, the sample was centrifuged at 3000 rpm at 4 °C for 10 min. Following the centrifugation, a 120-µL aliquot of the resulting supernatant in the 1.5-mL test tube was transferred to another 1.5-mL test tube and an additional 80 µL of 0.1 % aqueous formic acid solution were added before the sample was vortexed for 30 s. The final solution was diluted tenfold with respect to the original blood plasma sample. One microliter of the final solution was injected into the LC/MS system by an autosampler.
Statistics
The standard ANOVA methodology was used, and p < 0.05 was considered to indicate a significant difference. In the figures, bars represent standard deviations.
Results
Table 2 lists the values of basic parameters for the healthy adults and DM patients. Patients were treated by either diet alone or with drugs such as biguanide and sulfonylurea and with a minimum dose of insulin as BOT (basal supported oral therapy). The patients had not taken any drugs for hypertension or high lipidemia when the research was carried out.
Plasma levels of HDL cholesterol and total cholesterol were higher in healthy adults than in DM patients. Arachidonic acid levels were also higher in healthy adults than in DM patients.
Table 3 provides the amino acid profiles of the healthy adults and DM patients. Plasma levels of amino acids such as isoleucine, leucine, aspartic acid, glutamic acid, and aspartic acid were significantly higher in the DM patients. Levels of tryptophan, cystine, arginine, aspargine, and glutamine were lower in the DM patients.
Although there were no differences in AA (all amino acids) and EAA (essential amino acids) levels between the healthy adults and DM patients, BCAA (branched-chain amino acids) levels were higher in the DM patients than in the healthy adults.
Table 4 shows that the concentrations of various tryptophan metabolites such as L-hydroxytryptophan, 5-hydroxyindoleacetic acid, kynurenic acid, 3-hydroxykynurenine, and 3-hydroxyanthranilic acid were higher in the DM patients than in the healthy adults. Although serotonin levels tended to be higher in the DM patients than in the healthy adults, the difference was not statistically significant. Levels of L-5-hydroxytryptophan, 5-hydroxytryptophol, tryptophol, and anthranilic acid in the two groups were too low to compare.
We divided the DM patients into those with higher than the mean level of tryptophan and those with lower than the mean level of tryptophan. Plasma levels of L-5-hydroxytryptophan, indole-3-acetic acid, kynurenic acid, 3-hydroxykynurenine, and xanthurenic acid were higher in the DM patients with higher tryptophan levels than in those with lower tryptophan levels, and levels of 3-hydroxyanthranilic acid appeared to be almost the same in the DM patients with higher and lower tryptophan levels. Levels of 5-hydroxyindoleacetic acid were higher in DM patients with lower tryptophan levels than in DM patients with higher tryptophan levels.
Discussion
It has been shown that dysregulation of the kynurenine pathway may be involved in the pathogenesis of cardiovascular disease [11] and DM [12]. Tryptophan metabolites are involved in balancing the activation and inhibition of the immune system [13].
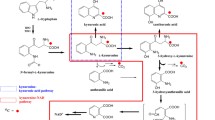
Since the metabolism of tryptophan is a complicated process, Fig. 1 shows the kynurenine and serotonin pathways for tryptophan catabolism.
Tryptophan metabolites are not only involved in the regulation of the immune system but they also exert many neurotoxic activities. One such neuroactive metabolite is 3-hydroxykynurenine [10], the synthesis of which is catalyzed by kynurenine 3-hydroxylase. Highly increased levels of 3-hydroxykynurenine may lead to nerve dysfunction [14]. Kynurenic acid has been shown to be an endogenous antagonist of an excitatory amino acid receptor, and may serve to modulate excitatory nerve transmission [5, 15]
Although there are several reports of the involvement of tryptophan metabolites such as kynurenine or kynurenic acid in disease, there has been no systematic research into the role of tryptophan metabolites in the pathogenesis of DM.
In the present study, we found that tryptophan levels were decreased in plasma from DM patients and that levels of tryptophan metabolites such as 5-hydroxytryptophan, 5-hydroxyindoleacetic acid, kynurenic acid, 3-hydroxykynurenine, and 3-hydroxyanthranilic acid were increased in plasma from DM patients, indicating that the metabolism of tryptophan is accelerated in DM patients.
We recently obtained data indicating that plasma samples from patients with acute coronary syndrome that showed low tryptophan levels also had high levels of almost all tryptophan metabolites (paper submitted for publication). We hypothesized that the metabolism of tryptophan is accelerated in patients with lower plasma tryptophan levels. We were therefore interested to determine if more tryptophan is consumed in DM patients, leading to lower levels of tryptophan and higher levels of tryptophan metabolites in plasma from these patients. We found that, in DM patients, tryptophan appears to be degraded more when plasma tryptophan levels are high (Table 5), except in the degradation to serotonin pathway. 5-Hydroxyindoleacetic acid levels were found to be higher in DM patients with lower plasma levels of tryptophan that in those with higher tryptophan levels (Table 5). This suggests that the conversion of serotonin to 5-hydroxyindoleacetic acid is enhanced when the plasma tryptophan level is low.
There have been many papers describing a relationship between stress and tryptophan degradation. Chronic stress increased blood and brain kynurenine pathway activity in mouse [16], and acute psychological stress has been shown to change tryptophan metabolite levels in mouse [17]. We also showed that stress increases the metabolism of tryptophan in rats [6–8]. However, all of those results were obtained using animal models.
We previously found that sucrose or glucose uptake does not influence fasting glucose levels, insulin levels, and BMI [18]. These results suggest that factors other than food intake may be involved in the pathogenesis of diabetes mellitus. We believe that stress may be one of these factors, and that the relationship between changes in the kynurenine pathway and the pathogenesis of metabolic syndrome such as DM is worthy of further investigation.
Finally, the present results imply that DM patients are constantly exposed to stress.
References
Eussen SJ, Ueland PM, Vollset SE, Nygård O, Midttun Ø, Sulo G, Ulvik A, Meyer K, Pedersen ER, Tell GS. Kynurenines as predictors of acute coronary events in the Hordaland Health Study. Int J Cardiol. 2015;189:18–24.
Pedersen ER, Tuseth N, Eussen SJ, Ueland PM, Strand E, Svingen GF, Midttun Ø, Meyer K, Mellgren G, Ulvik A, Nordrehaug JE, Nilsen DW, Nygård O. Associations of plasma kynurenines with risk of acute myocardial infarction in patients with stable angina pectoris. Arterioscler Thromb Vasc Biol. 2015;35:455–62.
Adell A, Garcia-Marquez C, Armario A, Gelpi E. Chronic stress increases serotonin and noradrenaline in rat brain and sensitizes their responses to a further acute stress. J Neurochem. 1988;5016:78–81.
Price LH, Charney DS, Delgado PL, Goodman WK, Krystal JH, Woods SW, Heninger GR. Clinical studies of 5-HT function using i.v. l-tryptophan. Prog Neuropsychopharmacol Biol Psychiatry. 1990;14:459–72.
Stone TW. Neuropharmacology of quinolinic and kynurenic acids. Pharmacol Rev. 1993;45:309–79.
Malyszko J, Urano T, Yan D, Serizawa K, Kozima Y, Takada Y, Takada A. Foot shock-induced changes in blood and brain serotonin and related substances in rats. Jpn J Physiol. 1994;44:35–47.
Malyszko J, Urano T, Takada Y, Takada A. Amino acids, serotonin, and 5-hydroxyindoleacetic acid following foot shock in rats. Brain Res Bull. 1995;36:137–40.
Pawlak D, Takada Y, Urano T, Takada A. Serotonergic and kynurenic pathways in rats exposed to foot shock. Brain Res Bull. 2000;53:197–206.
Okuno E, Nishikawa T, Nakamura M. Kynurenine aminotransferases in the rat. Localization and characterization. Adv Exp Med Biol. 1996;398:455–64.
Niinisalo P, Oksala N, Levula M, Pelto-Huikko M, Järvinen O, Salenius JP, Kytömäki L, Soini JT, Kähönen M, Laaksonen R, Hurme M, Lehtimäki T. Activation of indoleamine 2,3-dioxygenase-induced tryptophan degradation in advanced atherosclerotic plaques: Tampere Vascular Study. Ann Med. 2010;42:55–63.
Oxenkrug G. Insulin resistance and dysregulation of tryptophan-kynurenine and kynurenine-nicotinamide adenine dinucleotide metabolic pathways. Mol Neurobiol. 2013;48:294–301.
Moffett JR, Price RA, Anderson SM, Sipos ML, Moran AV, Tortella FC, Dave JR. DNA fragmentation in leukocytes following subacute low-dose nerve agent exposure. Cell Mol Life Sci. 2003;60:2266–71.
Nakagami Y, Saito H, Katsuki H. 3-Hydroxykynurenine toxicity on the rat striatum in vivo. Jpn J Pharmacol. 1996;2:183–6.
Okuda S, Nishiyama N, Saito H, Katsuki H. Basic fibroblast growth factor and brain-derived neurotrophic factor promote survival and neuronal circuit formation in organotypic hippocampal culture. J Neurochem. 1998;70:299–307.
Schwarcz R, Du F. Quinolinic acid and kynurenic acid in the mammalian brain. Adv Exp Med Biol. 1991;294:185–99.
Fuertig R, Azzinnari D, Bergamini G, Cathomas F, Sigrist H, Seifritz E, Vavassori S, Luippold A, Hengerer B, Ceci A, Pryce CR. Mouse chronic social stress increases blood and brain kynurenine pathway activity and fear behaviour: both effects are reversed by inhibition of indoleamine 2,3-dioxygenase. Brain Behav Immun. 2016;54:59–72.
Vecchiarelli HA, Gandhi CP, Hill MN. Acute psychological stress modulates the expression of enzymes involved in the kynurenine pathway throughout corticolimbic circuits in adult male rats. Neural Plast. 2016;2016:7215684. doi:10.1155/2016/7215684 (Epub 2015 Dec 27).
Takao T, Ogawa M, Ishii Y, Shimizu F, Takada A. Different glycemic resposes to sucrose and glucose in old and young male adults. J Nutr Food Sci. 2016;6:460. doi:10.4172/2155-9600.1000460.
Acknowledgments
The experiments were designed and performed by all of the authors. AT wrote the manuscript. Statistical analyses were done by TT. All of the authors read and approved the final manuscript, and take responsibility for the final content.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Financial support
This study was supported by grants from the Ito Memorial Foundation and the NPO International Projects on Food and Health.
Conflict of interest
M. Ogawa, T. Takao, Y. Ishii, F. Shimizu, J. Masuda, and T. Takada declare that they have no conflict of interest.
Ethics
This work was approved by the ethical committees of Showa Women’s University and the NPO International Projects on Food and Health, and was carried out in accordance with the code of ethics of the World Medical Association (Declaration of Helsinki) for experiments.
About this article
Cite this article
Matsuoka, K., Kato, K., Takao, T. et al. Concentrations of various tryptophan metabolites are higher in patients with diabetes mellitus than in healthy aged male adults. Diabetol Int 8, 69–75 (2017). https://doi.org/10.1007/s13340-016-0282-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13340-016-0282-y