Abstract
Fewer than 100 cases of amiodarone-induced thyrotoxicosis (AIT) managed surgically have been reported worldwide. This study aims to assess the outcome of thyroidectomy under general anesthesia in a relatively large case series. A retrospective analysis of the clinical records of 53 patients who underwent thyroidectomy for AIT between 1995 and 2019 was conducted. There were 48 (90%) males and 5 females with an average age of 63.7 years. Type 1 and 2 AIT were present in 35 (66%) and 18 (34%) of patients, respectively. The mean preoperative ejection fraction (EF) was 45 ± 13%. Salvage surgery was performed in 6 (11%) patients due to decompensating heart failure and/or malignant arrhythmias. 35 (66%) patients underwent urgent surgery due to a predicted late response to medical therapy and/or the need to discontinue it. Elective surgery was performed in the remainder. A considerable improvement in mean EF occurred 12 months post-surgery (44% vs. 49%; p < 0.001). The overall survival rate following thyroidectomy was 96% at 12 months, and 83% at 5 years. No survival differences were observed based on systolic function. Cardiac-specific mortality was 11%, and these patients demonstrated a considerably shorter survival post-surgery compared to those who died of a non-cardiac cause (27 ± 18 vs. 77.5 ± 54 months; p < 0.05). Total thyroidectomy can be safely performed under general anesthesia despite severe cardiac disease. It considerably improves cardiac function and confers a survival advantage. Therefore, it should be considered early in the treatment plan of select cases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Amiodarone is a potent antiarrhythmic drug that is used to treat ventricular and supraventricular tachyarrhythmias [1]. It is an iodine-rich compound with some structural similarity to thyroxine (T4). Iodine comprises 37% of its weight and with the usual daily maintenance doses of 200–600 mg, amiodarone provides 50–100 times the daily requirement of iodine [1,2,3]. In addition to its use in cases of serious ventricular arrhythmia, amiodarone is used in controlling paroxysmal supraventricular tachycardia, atrial flutter, and fibrillation [4].
The three main complications of long-term amiodarone use are pulmonary toxicity, thyroid disease and liver toxicity. Amiodarone-induced thyrotoxicosis (AIT) occurs more frequently in countries where iodine deficiency is prevalent with an incidence rate of up to 23% [5, 6]. Its occurrence in a patient population with underlying cardiac dysrhythmias that do not tolerate the cardiac effects of hyperthyroidism can lead to cardiac decompensation [7].
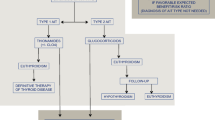
The diagnosis, classification and management of AIT are challenging and have been covered in guidelines published by the European Thyroid Association (ETA) [8]. There are two proposed mechanisms for thyrotoxicosis: type 1 is an iodine-induced hyperthyroidism (Jod–Basedow effect). It usually affects patients with latent or pre-existing thyroid disorders and is more common in areas of low iodine intake. Type 2 is related to the direct toxic effect of amiodarone and its metabolites on the thyroid, causing follicular destruction and apoptosis with the release of thyroid hormone into the circulation [9].
When AIT represents an imminent risk for cardiac function, it should be managed accordingly and without delay. Accordingly, thyroidectomy in these circumstances may represent the most effective and rapid treatment modality [8]. However, thyroidectomy in these cases has been demonstrated to result in considerable perioperative morbidity and mortality [10]. Only a few cases series in the literature with a sum of 88 cases have demonstrated the safety of thyroidectomy under general anesthesia in AIT [11,12,13,14,15,16,17].
We herein present a single-center study reporting the outcomes of surgical management of AIT in 53 cases. This case series, up to our knowledge, represents the largest to be reported in the literature to date.
Materials and methods
Between the years 1995 and 2019, 53 patients underwent thyroidectomy under general anesthesia for AIT at an academic tertiary referral center. The diagnosis of AIT was based on clinical grounds (clinical manifestations of thyrotoxicosis), and laboratory results (elevated serum free T3 and T4, suppressed TSH, and increased urinary iodine excretion). The first thyroidectomy was performed in Feb. 1997, and the last in Sept. 2019. The clinical records of the study cohort (n = 53) were retrospectively reviewed. Data collection and analysis were performed according to the institutional guidelines and the ethical standards of the Helsinki Declaration. The requirement for informed consent was waived because of the retrospective nature of the study.
Data collection was performed under two broad headings 1. Pre-operative data (demographics, data regarding amiodarone therapy, endocrinological and craniological data), and 2. Post-operative data (measures of surgical outcome). The former included patient demographics, body mass index (BMI), the indication and duration of amiodarone therapy, the type of AIT, pre-existing thyroid disorders and the sonographically estimated thyroid volume (SETV), the preoperative cardiac assessment of patients, and patients’ American Society of Anaesthesiologists (ASA) physical status classification, and the precise indication for thyroidectomy [18, 19]. According to the indication for thyroidectomy, patients were classified into three groups: 1. The salvage surgery group which represented patients with a life-threatening deterioration of cardiac function and/or malignant arrhythmia mandating emergency thyroidectomy as a salvage treatment 2. The urgent surgery group which represented patients in whom the underlying heart condition may become risky owing to a predicted late response to medical therapy and/or patients with side effects to glucocorticoids or thionamides who require their withdrawal 3. The elective surgery group which represents patients who gained resolution of thyrotoxicosis but require a thyroidectomy as a definitive treatment for AIT or to correct the underlying thyroid disease.
Postoperative data included operative time (OT), post-operative cardiac assessment at 12 months, post-operative morbidity, and the requirement and length of intensive care unit (ICU) stay. Post-operative morbidity included complications that occurred within 30 days of the index surgery. Hypoparathyroidism was defined as a serum calcium level below 7.5 mg/dL, or below 8.5 mg/dL in the presence hypocalcaemia-related symptoms. The persistence of the condition at 12 months post-surgery labeled it as permanent. Vocal fold palsy was documented by laryngoscopy and was labeled as permanent if persisted more than 12 months post-surgery [20].
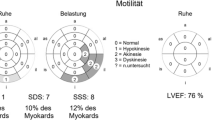
The pre- and post-operative cardiac status evaluation was conducted through echocardiography-measured left ventricular ejection fraction (EF). EF values were used to define three systolic function classes: normal left ventricular systolic function (EF ≥ 50%), mild systolic dysfunction (40% ≤ EF < 50%), and moderate-to-severe systolic dysfunction (EF < 40%) [21].
Lastly, survival data were established in Dec. 2019 by phone interview or clinic visit. Survival time was calculated from the date of the index procedure up to Dec. 2019.
Results
The study cohort consisted of 48 males (90%) and 5 females (10%) with a male to female ratio of around 10:1. The average age was 63.7 years (range: 46–81 years), and the average BMI was 26 kg/m2 (range: 18–39 kg/m2). 35 patients (66%) had type 1 AIT, and 18 patents (34%) had type 2 AIT. According to the physical status classification of the ASA, patients with ASA class 1–4 included: none, 7 (13%), 33 (62%), and 13 (25%), respectively. (Table 1).
Endocrinological data
Almost half (45%) of the study cohort (24 patients) had pre-existing nodular and/or multinodular goiter. The mean SETV was 47.5 ± 40 ml (range 10–187). The mean preoperative TSH was 0.211 ± 0.510 mU/L (range 0.001 – 2.330), mean free T4 was 39.7 ± 25.2 pg/mL (range 2.81–67.1 pg/mL), and mean free T3 was 6.3 ± 3.9 pg/ml (range 2.2–21.2). 32 (60%) patients were operated in a thyrotoxic state. (Table 1).
Data regarding amiodarone therapy, and preoperative cardiological data
The indication for amiodarone therapy included atrial fibrillation 26/53 (49%), ventricular extrasystoles 12/53 (23%), ventricular fibrillation 9/53 (17%), ventricular tachycardia 2/53 (4%), supraventricular tachycardia 1/53 (2%), AV-block 1/53 (2%), and an unknown cause 1/53 (2%). The mean duration of amiodarone therapy up to the diagnosis of AIT was 874 ± 990 days (range: 3–6116 days).
The mean preoperative EF was 45 ± 13% (range 15–73%). EF values were normal in 23 (43%) patients, mild in 14 (26.4%) patients, and moderate-to-severe in 16 (20%) patients. Cardiac function analysis demonstrated that the salvage surgery group had a significantly lower EF compared to both the urgent and elective surgery groups (p = 0.03). 13 (25%) patients had an implantable cardioverter-defibrillator device (ICD) at the time of surgery. While 42 patients (79%) were under anticoagulant or antiplatelet therapy at the time of thyroidectomy.
The indication for thyroidectomy
6 (11%) patients underwent salvage surgery, 35 (66%) patients underwent urgent surgery, and 12 (23%) had an elective thyroidectomy. In the salvage surgery group (n = 6), 3 (50%) patients presented with an acutely decompensating heart failure which was followed by the development of malignant arrhythmia in one the cases. 2 (33%) patients presented with malignant arrhythmias. The sixth patient underwent salvage surgery due to the development of agranulocytosis while still being thyrotoxic [22]. Table 2 summarizes the indications for thyroidectomy in the study cohort. The median time after which total thyroidectomy was performed in the entire cohort was 30 days post diagnosis of AIT. This was 12.5 days for salvage surgery, 18 days for urgent surgery, and 35 days for elective surgery. The differences in the timing of surgery were statistically significant among all three groups (p = 0.008).
Postoperative data
The mean OT was 56 ± 12 min (range 25–100 min). All patients were admitted to the ICU for a median time of one day (range: 1- 4 days) postoperatively. Perioperative complications included 3 (6%) cases of intraoperative arrhythmias two of which required intravenous medical therapy, and a case of successfully managed paroxysmal atrial fibrillation developing in the ICU during the first 24 h after surgery. Postoperative complication specific to thyroid surgery included: permanent hypoparathyroidism (3/5.7%), transient vocal fold palsy (1/1.8%), post-operative hematoma and/or seroma (none). None of the patients required further hospitalization 60 days post-surgery. Differences between patients who developed a perioperative complication (7.5%) and those who did not were analyzed in relation to the following variables: sex, age, type of AIT, preoperative thyroid function test, SETV, surgical group (elective, urgent, and salvage), and time to thyroidectomy. No positive association and/or correlation was found.
A considerable improvement in the mean EF of the study cohort occurred 12 months post-surgery (mean preoperative EF: 44% vs. mean EF 12-moths postoperatively: 49%; p < 0.001). When reporting the improvement in EF according to the class of left ventricular function, the groups that demonstrated considerable improvement were the normal systolic function and the moderate-to-severe systolic dysfunction groups (57% vs. 60%; p = 0.007, and 27% vs. 36%; p = 0.003, respectively).
Survival data
By Dec. 2019, 15 (28%) patients were dead. 6 (11%) patients died of a cardiac cause (cardiac-specific mortality). Cancer was the cause of death in 3 (6%) patients. 3 (6%) patients died from a medical cause other than cardiac- or cancer-related. Trauma was the cause of death in 1 (2%) patient, and no specific cause was identified in 2 (4%) patients.
The overall survival rate following thyroidectomy was 96% at 12 months, and 83% at 5 years. The mean survival time following thyroidectomy in patients who died of a cardiac-related cause was 27 ± 18 months. This was considerably higher in patient with a non-cardiac-specific mortality (77.5 ± 54 months; p < 0.05).
No survival differences were observed comparing the three groups of left ventricular systolic function: normal vs. mild; p = 0.876, normal vs. moderate-to-severe; p = 0.420, and mild vs. moderate-to-severe; p = 0.412).
From 2001 to 2014, iopanoic acid (IopAc) was added to the treatment of AIT in 32 (60%) patients prior to surgery as a potent inhibitor of thyroid hormone release and/or peripheral conversion of T4 to T3 [23]. The median duration of iopanoic administration prior to surgery was 12 days (range 5 to 30 days). In 8 patients, treatment with iopanoic acid was extended for a median of 6 days after surgery. The use of IopAc did not confer a survival benefit (p = 0.359), nor was there a difference in the time of normalization of thyroid hormone levels preoperatively with its use (free T4 levels; p = 0.640, and free T3 levels; p = 0.894).
Discussion
The incidence rate of AIT has been estimated to be as high as 23% [6]. Data on the optimal management of AIT are scarce. The main factors that make the management of AIT challenging include the difficulty differentiating between its types, the frequent absence of a prompt response to medical therapy, and the common coexistence of severe heart disease [24,25,26]. Patients with AIT are considered high-risk patients for surgery because of both their underlying cardiac condition, and poorly tolerated thyrotoxic state. In fact, thyroidectomy, as a therapeutic modality, has been demonstrated to be associated with considerable perioperative morbidity and mortality [10]. Nevertheless, others have considered it as a rapid means for restoring euthyroidism, improving cardiac function and potentially reducing mortality [12, 15]. A recent study compared total thyroidectomy to medical therapy in the management of AIT [27]. It demonstrated the non-inferiority of thyroidectomy to medical therapy in patients with normal or mild systolic dysfunction. However, thyroidectomy conferred a considerable survival benefit in patents with moderate-to-severe systolic dysfunction. Therefore, the study concluded that thyroidectomy could be considered the treatment of choice for AIT in patients with moderate-to-severe systolic dysfunction. Our results show a considerable improvement in the mean EF at 12 months post-surgery among the entire study cohort (mean preoperative EF: 44% vs. mean EF 12 months postoperatively: 49%; p < 0.001). Although this improvement was most striking among those with moderate-to-severe systolic dysfunction, the survival advantage was not limited to this group of patients. The universal improvement of cardiac function following thyroidectomy translated into a survival advantage among all patients regardless of their class of systolic function. The overall survival rate following thyroidectomy was 96% at 12 months, and 83% at 5 years. At this point, it is worth mentioning that a relatively short time from diagnosis to surgery potentially affects outcome favorably. The median time to surgery for the entire study cohort was 30 days. This probably reflected in a more favorable surgical outcome compared to the results obtained by Conen et al. who performed surgery after a median of 112 days [28]. It is also worth highlighting that the perioperative complication rate following surgery was relatively low (7.5%) and after accounting for all potential confounders, they represented independent happenings. Furthermore, post-operative bleeding was not reported despite almost 80% of the study cohort being on anticoagulation and/or antiplatelet therapy. All this emphasizes the importance of referring such cases to high-volume centers with high-volume surgeons. As Surgeon’s experience reflected by volume is considered the most important denominator in thyroid surgery [29]. The consensus statement of the American Head and Neck Society on the surgical management of the recurrent laryngeal nerve in thyroidectomy reported that compared to high-volume surgeons, lower-volume surgeons had considerably higher complication rates and lengthier hospital stays [30].
With the intent of minimizing perioperative morbidity and mortality, some advocate performing thyroidectomy in AIT patients under local and/or regional anesthesia [31, 32]. Our results are concordant with the results of others that demonstrate the safety of thyroidectomy under general anesthesia regardless of systolic function [13]. Furthermore, our results have demonstrated the safety of thyroidectomy under general anesthesia in the unprepared (thyrotoxic) patient.
Although IopAc is no longer available commercially, we evaluated its benefit as part of the treatment protocol prior to surgery. Since more than half (60%) of the study cohort were treated with IopAc, the number of patients is considered enough to make valid conclusions regarding its use in the preoperative setting. Our results did not show that the use of IopAc conferred a survival advantage, nor did it significantly alter the time of normalization of thyroid hormone levels.
Our study is limited by the relatively small sample size despite being the largest to be reported in literature to date, and its retrospective nature. Furthermore, the population included in the study comes from an iodine-deficient area which might influence accurate generalizability to other populations. Finally, although we adjusted for confounding factors regarding perioperative morbidity, additional confounders cannot be excluded.
Conclusion
Total thyroidectomy can be safely performed under general anesthesia in AIT regardless of systolic function. It considerably improves cardiac function and confers a survival advantage. Therefore, it should be considered early in the treatment plan of select cases. Furthermore, a shorter time to surgery seems to favorably affect surgical outcome.
Data availability
The data supporting the findings of this study are available from the corresponding author, upon request.
References
Martino E, Bartalena L, Bogazzi F et al (2001) The effects of amiodarone on the thyroid. Endocr Rev 22:240–254
Harjai KJ, Licata AA (1997) Effects of amiodarone on thyroid function. Ann Intern Med 1(126):63–73
Reiffel JA, Estes NA 3rd, Waldo AL, Prystowsky EN, DiBianco R (1994) A consensus report on antiarrhythmic drug use. Clin Cardiol 17:103–116
Ceremuzynski L, Kleczar E, Krzeminska-Pakula M et al (1992) Effect of amiodarone on mortality after myocardial infarction: a double-blind, placebo-controlled, pilot study. J Am Coll Cardiol 20:1056–1062
Trip MD, Wiersinga W (1991) Incidence, predictability, and pathogenesis of amiodarone-induced thyrotoxicosis and hypothyroidism. Am J Med 91:507–511
Guyetant S, Rousselot MC, Wion-Barbot N et al (1995) Hyperthyroidism induced by amiodarone and hyperthyroidism induced by iodine. Histologic, immunohistochemical and ultrastructural aspects. Ann Pathol 15:431–437
Osuna PM, Udovcic M, Sharma MD (2017) Hyperthyroidism and the heart. Methodist DeBakey Cardiovasc J 13:60–63
Bartalena L, Bogazzi F, Chiovato L et al (2018) 2018 European thyroid association (ETA) guidelines for the management of amiodarone-associated thyroid dysfunction. Eur Thyroid J 7:55–66
El-Shirbiny AM, Stavrou SS, Dnistrian A et al (1997) Jod-Basedow syndrome following oral iodine and radioiodinated-antibody administration. Int J Nucl Med 38:1816–1817
Houghton SG, Farley DR, Brennan MD et al (2004) Surgical management of amiodarone-associated thyrotoxicosis: Mayo clinic experience. World J Surg 28:1083–1087
Hamoir E, Meurisse M, Defechereux T et al (1998) Surgical management of amiodarone-associated thyrotoxicosis: too risky or too effective? World J Surg 22:537–542
Mulligan DC, McHenry CR, Kinney W et al (1993) Amiodarone-induced thyrotoxicosis: clinical presentation and expanded indications for thyroidectomy. Surgery 114:1114–1119
Gough J, Gough IR (2006) Total thyroidectomy for amiodarone-associated thyrotoxicosis in patients with cardiac disease. World J Surg 30:1957–1961
Sutherland J, Robinson B, Delbridge L (2001) Anesthesia for amiodarone induced thyrotoxicosis: a case review. Anaesth Intensiv Care 29:24–29
Tomisti L, Materazzi G, Bartalena L et al (2012) Total thyroidectomy in patients with amiodarone-induced thyrotoxicosis and severe left ventricular systolic dysfunction. J Clin Endocrinol Metab 97:3515–3521
Meerwein C, Vital D, Greutmann M et al (2014) Total thyroidectomy in patients with amiodarone-induced hyperthyroidism: when does the risk of conservative treatment exceed the risk of surgery? HNO 62:100–105
Kotwal A, Clark J, Lyden M et al (2018) Thyroidectomy for amiodarone-induced thyrotoxicosis: Mayo clinic experience. J Endocr Soc 2:1226–1235
Mak PH, Campbell RC, Irwin MG, American Society of A (2002) The ASA physical status classification: inter-observer consistency. Anaesth Intensiv Care 30:633–640
Bogazzi F, Tomisti L, Bartalena L, Martino E (2018) Amiodarone-induced thyrotoxicosis. In: Matfin G (ed) Endocrine and metabolic medical emergencies. John Wiley & Sons Ltd
ReeveT TNW (2000) Complications of thyroid surgery: how to avoid them, how to manage them, and observations on their possible effect on the whole patient. World J Surg 24:971–975
Wang TJ, Evans JC, Benjamin EJ, Levy D, LeRoy EC, Vasan RS (2003) Natural history of asymptomatic left ventricular systolic dysfunction in the community. Circulation 108:977–982
Ibáñez L, Vidal X, Ballarín E, Laporte JR (2005) Agranulocytosis associated with dipyrone (metamizole). Eur J Clin Pharmcol 60:821–829
Bogazzi F, Bartalena L, Cosci C et al (2003) Treatment of type II amiodarone-induced thyrotoxicosis by either iopanoic acid or glucocorticoids: a prospective, randomized study. J Clin Endocrinol Metab 88:1999–2002
Binz K, Burger A, Valloton MB (1998) Amiodarone and thyroid function: clinical implications. Schweiz Med Wochenschr 128:1051–1058
Harjai KJ, Licata AA (1997) Effects of amiodarone on thyroid function. Ann Intern Med 126:63–73
Mulligan DC, McHenry CR, Kinney W, Esselstyn CB (1993) Amiodarone-induced thyrotoxicosis: clinical presentation and expanded indications for thyroidectomy. Surgery 114:1114–1119
Cappellani D, Papini P, Pingitore A et al (2020) Comparison between total thyroidectomy and medical therapy for amiodarone-induced thyrotoxicosis. J Clin Endocrinol Metab. https://doi.org/10.1210/clinem/dgz041
Conen D, Melly L, Kaufmann C, Bilz S, Ammann P et al (2007) Amiodarone-induced thyrotoxicosis: clinical course and predictors of outcome. J Am Coll Cardiol 49:2350–2355
Bakkar S, Papavramidis TS, Aljarrah Q et al (2020) Energy-based devices in thyroid surgery—an overview. Gland Surg 9(Supp 1):S14–S17. https://doi.org/10.21037/gs.2019.08.05
Fundakowski CE, Hales NW, Agrawal N et al (2018) Surgical management of the recurrent laryngeal nerve in thyroidectomy: American head and neck society consensus statement. Head Neck 40:663–675
Klein SM, Greengrass RA, Knudsen N, Leight G, Warner DS (1997) Regional anesthesia for thyroidectomy in two patients with amiodarone-induced hyperthyroidism. Anesth Analg 85:222–224
Williams M, Lo Gerfo P (2002) Thyroidectomy using local anesthesia in critically ill patients with amiodarone-induced thyrotoxicosis: a review and description of the technique. Thyroid 12:523–525
Author information
Authors and Affiliations
Contributions
SB, PP: study concept and design, data interpretation, final approval, and accountability for all aspects of the work. DC, FF, CD, SC, and CA: study concept and design, data collection and interpretation, article drafting, final approval, and accountability for all aspects of the work. FB, PM, and GM: study concept, critical revision, final approval, and accountability for all aspects of the work.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no financial ties or conflicts of interest to declare.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Research involving human participants or animals
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
The need for an informed consent was waived due to the retrospective nature of the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bakkar, S., Cappellani, D., Forfori, F. et al. Early surgery: a favorable prognosticator in amiodarone-induced thyrotoxicosis—a single-center experience with 53 cases. Updates Surg 74, 1413–1418 (2022). https://doi.org/10.1007/s13304-022-01297-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13304-022-01297-3