Abstract
Introduction
Sodium-dependent glucose cotransporter 2 (SGLT2) inhibitors ameliorate blood glucose levels in patients with type 2 diabetes mellitus (T2DM) by inhibiting the reabsorption of glucose from the kidneys, thus increasing urinary glucose excretion. Most SGLT2 inhibitors have been reported to exert dose-dependent effects. However, little is known about the benefits of increasing the dose of SGLT2 inhibitors in clinical use. The aim of the present study was to investigate the effect of increasing the dose of the SGLT2 inhibitor empagliflozin in T2DM.
Methods
We collected 52 subjects with T2DM with inadequate glycemic control. The dose of empagliflozin was increased from 10 to 25 mg, taken once daily, and the alterations in glycemic control and several other clinical parameters were evaluated.
Results
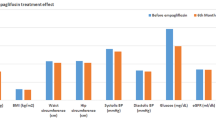
The increased dose of empagliflozin significantly ameliorated glycemic control. In addition, body weight (BW), body mass index (BMI), triglyceride (TG), and γ-glutamyltranspeptidase (GGT) were significantly decreased and hematocrit (Hct) was increased. Multivariate logistic regression analyses revealed that baseline diastolic blood pressure (DBP) (odds ratio 1.093, 95% CI 1.019–1.156, P = 0.012) and baseline TG (odds ratio 1.012, 95% CI 1.001–1.023, P = 0.026) were retained as independent predictors for the improvement of hemoglobin A1c (HbA1c) levels. Moreover, multivariate stepwise regression analyses revealed that changes in high-density lipoprotein cholesterol (β − 0.264, 95% CI − 1.217 to 0.000, P = 0.049) and HbA1c (β 0.302, 95% CI 0.077–1.096, P = 0.025) were retained as independent predictors for changes in BMI.
Conclusion
Increasing the dose of empagliflozin significantly ameliorated BW, BMI, GGT, TG, fasting plasma glucose and HbA1c and increased Hct in patients with T2DM. Moreover, baseline DBP and TG were independent predictors for the improvement of HbA1c. These findings may provide useful information when considering increasing the dosage of SGLT2 inhibitors in patients with T2DM who have inadequate glycemic control.
Trial Registration
UMIN Clinical Trials Registry (UMIN000041543).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Although sodium-dependent glucose cotransporter 2 (SGLT2) inhibitors have been reported to exert dose-dependent effects in clinical use, the benefits of increasing the dose of SGLT2 inhibitors are not well known. |
In this study, we investigated the beneficial effect of increasing the dose of empagliflozin, an SGLT2 inhibitor, in patients with type 2 diabetes mellitus (T2DM). |
What was learned from the study? |
Increasing the dose of empagliflozin significantly ameliorated HbA1c, and baseline diastolic blood pressure and triglyceride were independent predictors for the improvement of HbA1c. |
When we are considering increasing the dose of SGLT2 inhibitors in patients with T2DM who have inadequate glycemic control, these findings may provide beneficial information. |
Introduction
Sodium-glucose cotransporter 2 (SGLT2) inhibitors, a novel class of antidiabetes reagents, have a unique mechanism of action. In the nondiabetic state, glucose can pass through the renal glomeruli, but is completely reabsorbed in the renal tubules by sodium-glucose co-transporter mechanisms [1]. Glycosuria occurs when the renal threshold for glucose (a blood glucose level of approximately 10 mmol/l) is exceeded. Hyperglycemia increases the filtrated and reabsorbed glucose up to two- to threefold [1]. SGLT2 is the main transporter responsible for glucose reabsorption, which is found in the proximal kidney tubule [1]. SGLT2 inhibitors block the SGLT2 system and reduce the reabsorption of renal filtrated glucose, thereby lowering blood glucose levels [1].
Empagliflozin, a potent, highly selective SGLT2 inhibitor, is an effective and generally well-tolerated antidiabetes agent approved for the treatment of diabetes mellitus (DM) [2, 3] in the European Union, United States, Japan and other countries. Empagliflozin is also recognized to have weight- and blood-pressure-lowering effects and improves liver disorders, as evaluated by aspartate aminotransferase (AST), alanine aminotransferase (ALT) and/or γ-glutamyltranspeptidase (GGT) measurements in patients with type 2 DM (T2DM) [4, 5]. Moreover, clinical trials of empagliflozin have demonstrated efficacy in lowering the incidence of cardiovascular disease (CVD) in T2DM patients, independent of its glucose-lowering ability [6], and have shown reduced progression of diabetic kidney disease (DKD) [7]. Therefore, empagliflozin is considered an antidiabetic drug that is beneficial in the management of T2DM with obesity, hypertension, metabolic syndrome, fatty liver, and a high risk of CVD and/or DKD.
Almost all SGLT2 inhibitors have been reported to have dose-dependent effects. For example, the recommended dosage of empagliflozin is 10 mg once daily before or after breakfast, and combination therapy with several antihyperglycemic drugs has been approved. When its efficacy is insufficient, an increased dose of up to 25 mg once daily is allowed. However, little is known about the clinical benefits of increasing the dose of empagliflozin. Therefore, the aim of the present study was to investigate the benefit of increasing the dose of empagliflozin in T2DM.
Methods
Study Subjects
This was a retrospective, longitudinal study of patients with T2DM who presented to Nishinihon Hospital between January 2019 and June 2020. We retrospectively reviewed the medical records and collected various information on participants whose dose of empagliflozin was increased from 10 mg once daily to 25 mg once daily, and who did not receive any additional medications such as antihypertensive or antihyperlipidemic drugs, for 6 months. We used the following inclusion criteria: (1) patients who were continuing to visit the hospital for blood glucose control; (2) patients who were already prescribed 10 mg of empagliflozin once daily; and (3) patients from whom blood samples had been collected early in the morning in the fasting state. Participants meeting the following criteria were excluded: (1) < 20 years of age; (2) having type 1 diabetes; (3) pregnancy; (4) active infectious disease including urinary tract infection; (5) severe hepatic disease; and (6) malignancy.
Age, sex, height, weight, blood pressure, and pre-existing use of hypoglycemic agents, anti-hypertensive agents or anti-hyperlipidemic agents were recorded. Body mass index (BMI) was calculated as weight divided by height squared (kg/m2). Kidney function was estimated via the estimated glomerular filtration rate (eGFR) on the basis of age, sex and serum creatinine using the equation for Japanese subjects developed by Matsuo et al. [8].
Compliance with Ethics Guidelines
All procedures in this study involving human participants were performed in accordance with the ethical standards of the institutional and/or national research committee and the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study protocol was approved by the Human Ethics Review Committee of Nishinihon Hospital (protocol number: R2-7) and registered at UMIN-CTR (UMIN000041543). All subjects provided written informed consent.
Measurements of Blood and Urinary Parameters
To avoid the influence of external factors on glucose homeostasis measurement, morning blood samples were selected from fasted participants only. Fasting plasma glucose (FPG), hemoglobin A1c (HbA1c), serum total cholesterol (TC), triglyceride (TG) and high-density lipoprotein cholesterol (HDL-C) concentrations as well as AST, ALT and GGT were measured using a Hitachi 7600 analyzer (Hitachi Ltd., Tokyo, Japan). Low-density lipoprotein cholesterol (LDL-C) concentration was determined using the Friedewald formula [9]. The eGFR was derived using the formula recommended by the Japanese Society of Nephrology [10, 11]. The baseline and changes (Δ) in several factors were evaluated over a period of 24 weeks.
Statistical Analysis
All statistical analyses were performed using SPSS version 27.0 for Windows (IBM Corp., Armonk, NY, USA). A normality test was performed for all continuous variables using the Shapiro–Wilk test. All data are given as the mean ± standard error of the mean for normally distributed continuous variables. Categorical data are expressed as percentages. The laboratory parameters at baseline and at 4, 12, or 24 weeks after SGLT2 inhibitor treatment were compared using the paired t test or the Wilcoxon signed-rank test. The Spearman correlation coefficient was calculated to examine the relationship between ΔHbA1c and ΔBMI with several plasma factors at baseline and 24 weeks. Multivariate logistic regression analysis was used to investigate independent predictors of improvement in HbA1c after 24 weeks of treatment (≤ 0.0%), and this analysis was constructed by adjusting for factors such as diastolic blood pressure (DBP; baseline), TG (baseline), AST (baseline), GGT (baseline) and HbA1c (baseline). To investigate independent predictors of ΔBMI, a multivariate stepwise regression analysis was constructed by adjusting for factors such as ΔHbA1c, ΔGTT, ΔHDL-C and TG (baseline). All P values < 0.05 were considered statistically significant.
Results
Characteristics of the Study Participants
A total of 52 participants were recruited in this study, and their baseline characteristics are summarized in Table 1. The median age and HbA1c of the study participants were 64.9 years and 7.46%, respectively. More than half of the patients were being treated with biguanides, dipeptidyl peptidase-4 (DPP-4) inhibitors and statins, and there were no further changes to medications after the dose of empagliflozin was increased during the 6-month trial period. During the observation period, no adverse events were observed.
Switching to a High Dose of Empagliflozin Ameliorates Several Parameters in Patients with Type 2 DM
Changes in clinical parameters were investigated after the dose of empagliflozin was increased from 10 to 25 mg for 6 months (Table 2, Fig. 1). Diabetic markers including FPG and HbA1c were significantly improved by –12.7 mg/dL and –0.13%, respectively. In addition, body weight (BW), BMI, TG and GGT were significantly decreased by –0.6 kg, –0.2 kg/m2, –22.1 mg/dL and –6.6 U/L, respectively, and hematocrit (Hct) was significantly increased by 0.9% after 3 months.
Independent Predictors for the Improvement of HbA1c and the Reduction of BMI
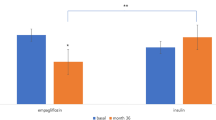
Univariate and multivariate logistic regression analyses for predicting the improvement of HbA1c with the increased dose of empagliflozin are presented in Table 3. Univariate analyses revealed that baseline DBP, baseline LDL-C, baseline TG, ΔTG, ΔAST, ΔALT, and baseline GGT and ΔGGT were significantly correlated with improvement of HbA1c. Multivariate logistic regression analyses revealed that baseline DBP (odds ratio 1.093, 95% CI 1.019–1.156, P = 0.012) and baseline TG (odds ratio 1.012, 95% CI 1.001–1.023, P = 0.026) were retained as independent predictors for the improvement of HbA1c. Next, univariate and multivariate stepwise regression analyses for predicting the ΔBMI with the increased dose of empagliflozin are presented in Table 4. Univariate analyses revealed that ΔHDL-C, baseline TG, ΔHbA1c and ΔGGT were significantly correlated with ΔBMI. Multivariate stepwise regression analyses revealed that ΔHDL-C (β − 0.264, 95% CI − 1.217–0.000, P = 0.049) and ΔHbA1c (β 0.302, 95% CI 0.077–1.096, P = 0.025) were retained as independent predictors for the ΔBMI. Since the baseline HbA1c and BMI were thought to be typically the factors that are the most powerful predicting variables, we reperformed these analyses while adding in these factors. However, there was no change in each explanatory variable (data not shown).
Discussion
To our knowledge, this is the first study to investigate the clinical benefits of increasing the dose of the SGLT2 inhibitor empagliflozin in patients with T2DM. In this retrospective, observational study, we revealed that increasing the dose of empagliflozin from 10 to 25 mg once daily significantly decreased BW, BMI, GGT, FPG and HbA1c and increased Hct in Japanese patients with T2DM. Moreover, multivariate logistic regression analyses revealed that baseline DBP and TG were independent predictors for the improvement of HbA1c, and multivariate stepwise regression analyses indicated that ΔHDL-C and ΔHbA1c were independent predictors for ΔBMI.
The results of randomized clinical trials using empagliflozin are summarized in Table 5. Several trials [12,13,14,15,16,17,18,19,20,21,22,23,24] have shown significant reductions of HbA1c and BW with empagliflozin. Increasing the empagliflozin dose from 10 to 25 mg consistently caused a further reduction in HbA1c (13/16 trials, see Table 5). However, this increase in the empagliflozin dose caused a further reduction of BW in only half the studies (8/16 trials, see Table 5). A meta-analysis of randomized controlled trials of empagliflozin reported that the reduction of HbA1c by 25 mg empagliflozin [weighted mean difference (WMD) − 0.66%; 95% confidence interval (CI) − 0.76 to − 0.57%] was greater than that caused by 10 mg empagliflozin [WMD − 0.62%; 95% CI − 0.68 to − 0.57%] [25]. However, it was also reported that the reduction of BW by 25 mg empagliflozin [WMD − 1.84 kg; 95% CI − 2.30 to − 1.38 kg] was almost the same as that caused by 10 mg empagliflozin [− 1.85 kg; 95% CI − 2.09 to − 1.60 kg] [25].
Recently, many network meta-analyses have been performed to distinguish the effects of different agents. For example, Shi et al. found the difference in the effect on heart failure between 10 mg dapagliflozin and 10 mg empagliflozin using network meta-analyses, and they concluded that 10 mg dapagliflozin may be more beneficial for heart failure compared to 10 mg empagliflozin [26]. On the other hand, there are two reports of network meta-analyses based on randomized control trials of empagliflozin. Wu et al. reported that the reduction of HbA1c by 25 mg empagliflozin was greater than that caused by 10 mg empagliflozin, but this was not a significant difference [25 mg vs. 10 mg: effect size − 0.04%; 95%CI − 0.19 to 0.10)] [27]. They also reported that the reduction of BW by 25 mg empagliflozin was significantly greater than that caused by 10 mg empagliflozin [25 mg vs. 10 mg: effect size − 0.24 kg; 95%CI − 0.39 to − 0.09)] [27]. Zaccardi et al. reported in the other network meta-analysis that the reduction of HbA1c by 25 mg empagliflozin was greater than that caused by 10 mg empagliflozin, but, as in the above reports, the difference was not significant [25 mg vs. 10 mg: effect size − 0.05%; 95%CI − 0.13 to 0.02)] [28]. The reduction of BW by 25 mg empagliflozin was reported to be greater than that caused by 10 mg empagliflozin, but unlike the above report, the difference was not significant [25 mg vs. 10 mg: effect size − 0.11 kg; 95%CI − 0.38 to 0.15)] [28]. Although there are differences in the clinical conditions, in our study, increasing the dose of empagliflozin from 10 to 25 mg significantly decreased both HbA1c and BW (Table 2). Overall, these results suggest that increasing the dose of empagliflozin from 10 to 25 mg is a reliable means of enhancing the hypoglycemic effect of this antidiabetic drug. However, since the changes in HbA1c and BMI are significant but minimal, they are not clinically significant.
A network meta-analysis performed by Zaccardi et al. demonstrated the results of using two other SGLT2 inhibitors, canagliflozin and dapagliflozin. In that report, the reduction of HbA1c by 300 mg canagliflozin was significantly greater than that caused by 100 mg canagliflozin [300 mg vs. 100 mg: effect size − 0.10%; 95%CI − 0.20 to − 0.00)] [28]. The reduction of BW by 300 mg canagliflozin was also reported to be significantly greater than that caused by 100 mg canagliflozin [300 mg vs. 100 mg: effect size − 0.61 kg; 95%CI − 0.99 to − 0.23)] [28]. On the other hand, the reduction of HbA1c by 10 mg dapagliflozin was greater than that caused by 5 mg dapagliflozin, but this was not significant difference [10 mg vs. 5 mg: effect size − 0.10%; 95%CI − 0.21 to 0.02)] [28]. The reduction of BW by 10 mg dapagliflozin was reported to be significantly greater than that caused by 5 mg dapagliflozin [10 mg vs. 5 mg: effect size − 0.60 kg; 95%CI − 1.00 to − 0.19)] [28]. To summarize the above, the effects of increasing the dose of the SGLT2 inhibitor on HbA1c tends to differ with each agent, but the effect on body weight may be consistent across agents.
Because several reports have demonstrated that treatment with SGLT2 inhibitors ameliorated not only HbA1c and BW but also blood pressure, lipid profile and liver injury in patients with T2DM [3, 4], we expected that switching to higher doses of empagliflozin would improve these parameters. Indeed, we found in the present study that increasing the dose of empagliflozin had additive effects on the reduction of serum TG and GGT levels. To clarify the causal relationship between these changes and the effects on HbA1c, we divided the patients into two groups: an improved HbA1c group and a nonimproved HbA1c group, and performed multivariate logistic regression analysis of various factors we suspected of being related. Interestingly, our study revealed that improvement of HbA1c was positively correlated with baseline DBP, baseline TG, ΔTG, ΔAST, ΔALT, baseline GGT and ΔGGT, and that baseline DBP and baseline TG were independent predictors for the improvement of HbA1c. As these factors are components of metabolic syndrome, increasing the dose of empagliflozin may be effective in T2DM patients with insulin resistance. We also revealed that ΔBW was correlated with ΔHDL-C, ΔHbA1c and ΔGGT, and that the independent predictors for the improvement of BMI were ΔHDL-C and ΔHbA1c, suggesting that HbA1c and HDL-C are related factors that respond well to ΔBW.
This study has some limitations. First, our study was conducted in a single center and the sample size was relatively small. Second, because this was a retrospective study, the dosing of antidiabetic agents was uncontrolled. However, we selected patients who did not change their intakes of other drugs, including antidiabetes agents, statins, fenofibrate, antihypertensive agents, and other agents during the follow-up period. Third, this study consisted only of Japanese patients. Further studies are needed to apply the results of this study to other populations.
Conclusions
We demonstrated in the present study that increasing the dose of empagliflozin from 10 to 25 mg once daily significantly decreased BW, BMI, GGT, FPG and HbA1c and slightly increased Hct in patients with T2DM. Moreover, baseline DBP and TG were independent predictors for the improvement of HbA1c, and ΔHDL-C and ΔHbA1c were independent predictors for ΔBMI. These findings may provide useful information when considering whether to increase the dose of SGLT2 inhibitors in patients with type 2 diabetes who have inadequate glycemic control.
References
Vallon V. The mechanisms and therapeutic potential of SGLT2 inhibitors in diabetes mellitus. Annu Rev Med. 2015;66:255–70.
Heise T, Seewaldt-Becker E, Macha S, Hantel S, Pinnetti S, Seman L, et al. Safety, tolerability, pharmacokinetics and pharmacodynamics following 4 weeks’ treatment with empagliflozin once daily in patients with type 2 diabetes. Diabetes Obes Metab. 2013;15:613–21.
Ferrannini E, Seman L, Seewaldt-Becker E, Hantel S, Pinnetti S, Woerle HJ. A Phase IIb, randomized, placebo-controlled study of the SGLT2 inhibitor empagliflozin in patients with type 2 diabetes. Diabetes Obes Metab. 2013;15:721–8.
Roden M, Weng J, Eilbracht J, Delafont B, Kim G, Woerle HJ, et al. EMPA-REG MONO trial investigators. Empagliflozin monotherapy with sitagliptin as an active comparator in patients with type 2 diabetes: a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Diabetes Endocrinol. 2013;1:208–19.
Sattar N, Fitchett D, Hantel S, George JT, Zinman B. Empagliflozin is associated with improvements in liver enzymes potentially consistent with reductions in liver fat: results from randomised trials including the EMPA-REG OUTCOME trial. Diabetologia. 2018;61:2155–63.
Zinman B, Wanner C, Lachin JM, Fitchett D, Bluhmki E, Hantel S, et al. EMPA-REG OUTCOME Investigators. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373:2117–28.
Wanner C, Inzucchi SE, Lachin JM, Fitchett D, von Eynatten M, Mattheus M, et al. Empagliflozin and progression of kidney disease in type 2 diabetes. N Engl J Med. 2016;375:323–34.
Matsuo S, Imai E, Horio M, Yasuda Y, Tomita K, Nitta K, et al. Revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis. 2009;53:982–92.
Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972;18:499–502.
Matsuo S, Imai E, Horio M, Yasuda Y, Tomita K, Nitta K, et al. Collaborators developing the Japanese equation for estimated GFR. Revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis. 2009;53:982–92.
Silveiro SP, Araújo GN, Ferreira MN, Souza FDS, Yamaguchi HM, Camargo EG. Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation pronouncedly underestimates glomerular filtration rate in type 2 diabetes. Diabetes Care. 2011;34(11):2353–5.
Ferrannini E, Seman L, Seewaldt-Becker E, Hantel S, Pinnetti S, Woerle HJ. A Phase IIb, randomized, placebo-controlled study of the SGLT2 inhibitor empagliflozin in patients with type 2 diabetes. Diabetes Obes Metab. 2013;15(8):721–8.
Roden M, Weng J, Eilbracht J, Delafont B, Kim G, Woerle HJ, et al. EMPA-REG MONO trial investigators. Empagliflozin monotherapy with sitagliptin as an active comparator in patients with type 2 diabetes: a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2013;208–19.
Roden M, Merker L, Christiansen AV, Roux F, Salsali A, Kim G, et al. EMPA-REG EXTEND™ MONO investigators. Safety, tolerability and effects on cardiometabolic risk factors of empagliflozin monotherapy in drug-naïve patients with type 2 diabetes: a double-blind extension of a Phase III randomized controlled trial. Cardiovasc Diabetol. 2015;14:154.
Häring HU, Merker L, Seewaldt-Becker E, Weimer M, Meinicke T, Broedl UC, et al. EMPA-REG MET Trial Investigators. Empagliflozin as add-on to metformin in patients with type 2 diabetes: a 24-week, randomized, double-blind, placebo-controlled trial. Diabetes Care. 2014;37(6):1650–9.
Inzucchi SE, Davies MJ, Khunti K, Trivedi P, George JT, Zwiener I, et al. Empagliflozin treatment effects across categories of baseline HbA1c, body weight and blood pressure as an add-on to metformin in patients with type 2 diabetes. Diabetes Obes Metab. 2021;23(2):425–33.
Rosenstock J, Seman LJ, Jelaska A, Hantel S, Pinnetti S, Hach T, Woerle HJ. Efficacy and safety of empagliflozin, a sodium glucose cotransporter 2 (SGLT2) inhibitor, as add-on to metformin in type 2 diabetes with mild hyperglycaemia. Diabetes Obes Metab. 2013;15(12):1154–60.
Häring HU, Merker L, Seewaldt-Becker E, Weimer M, Meinicke T, Woerle HJ, et al. EMPA-REG METSU Trial Investigators. Empagliflozin as add-on to metformin plus sulfonylurea in patients with type 2 diabetes: a 24-week, randomized, double-blind, placebo-controlled trial. Diabetes Care. 2013;36(11):3396–404.
Haering HU, Merker L, Christiansen AV, Roux F, Salsali A, Kim G, et al. EMPA-REG EXTEND™ METSU investigators. Empagliflozin as add-on to metformin plus sulphonylurea in patients with type 2 diabetes. Diabetes Res Clin Pract. 2015;110(1):82–90.
Kovacs CS, Seshiah V, Swallow R, Jones R, Rattunde H, Woerle HJ, et al. EMPA-REG PIO™ trial. Empagliflozin improves glycaemic and weight control as add-on therapy to pioglitazone or pioglitazone plus metformin in patients with type 2 diabetes: a 24-week, randomized, placebo-controlled trial. Diabetes Obes Metab. 2014;16(2):147–58.
Kovacs CS, Seshiah V, Merker L, Christiansen AV, Roux F, Salsali A, et al. EMPA-REG EXTEND™ PIO investigators. Empagliflozin as add-on therapy to pioglitazone with or without metformin in patients with type 2 diabetes mellitus. Clin Ther. 2015;37(8):1773-1788.e1.
Barnett AH, Mithal A, Manassie J, Jones R, Rattunde H, Woerle HJ, et al. EMPA-REG RENAL trial investigators. Efficacy and safety of empagliflozin added to existing antidiabetes treatment in patients with type 2 diabetes and chronic kidney disease: a randomised, double-blind, placebo-controlled trial. Lancet Diabetes Endocrinol. 2014;2(5):369–84.
Rosenstock J, Jelaska A, Zeller C, Kim G, Broedl UC, Woerle HJ, EMPA-REG BASALTM trial investigators. Impact of empagliflozin added on to basal insulin in type 2 diabetes inadequately controlled on basal insulin: a 78-week randomized, double-blind, placebo-controlled trial. Diabetes Obes Metab. 2015;17(10):936–48.
Sone H, Kaneko T, Shiki K, Tachibana Y, Pfarr E, Lee J, et al. Efficacy and safety of empagliflozin as add-on to insulin in Japanese patients with type 2 diabetes: a randomized, double-blind, placebo-controlled trial. Diabetes Obes Metab. 2020;22(3):417–26.
Liakos A, Karagiannis T, Athanasiadou E, Sarigianni M, Mainou M, Papatheodorou K, et al. Efficacy and safety of empagliflozin for type 2 diabetes: a systematic review and meta-analysis. Diabetes Obes Metab. 2014;16(10):984–93.
Shi Z, Gao F, Liu W, He X. Comparative efficacy of dapagliflozin and empagliflozin of a fixed dose in heart failure: a network meta-analysis. Front Cardiovasc Med. 2022;9: 869272.
Wu Q, Liu M, Fang Z, Li C, Zou F, Hu L, et al. Efficacy and safety of empagliflozin at different doses in patients with type 2 diabetes mellitus: A network meta-analysis based on randomized controlled trials. J Clin Pharm Ther. 2022;47(3):270–86.
Zaccardi F, Webb DR, Htike ZZ, Youssef D, Khunti K, Davies MJ. Efficacy and safety of sodium-glucose co-transporter-2 inhibitors in type 2 diabetes mellitus: systematic review and network meta-analysis. Diabetes Obes Metab. 2016;18(8):783–94.
Acknowledgements
Funding
This study was sponsored by Shimazu Corporation, Kyoto, Japan. All publication costs, including the journal’s Rapid Service Fee, were funded by the authors.
Editorial Assistance
We thank John Quayle, PhD, from Edanz (https://jp.edanz.com/ac) for editing a draft of this manuscript. This assistance was funded by the authors.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
Takeshi Matsumura contributed to the study design, data analysis and drafting of the manuscript. Tomoko Makabe, Seiko Ueda, Yuki Fujimoto, Kayo Sadahiro and Shiori Tsuruyama contributed to data collection. Yuma Ookubo and Tatsuya Kondo contributed to data interpretation and the editing of the manuscript. Eiichi Araki contributed to data interpretation and the editing of the manuscript. Eiichi Araki supervised the study. All authors read and approved the final manuscript.
Disclosures
Takeshi Matsumura, Tomoko Makabe, Seiko Ueda, Yuki Fujimoto, Kayo Sadahiro, Shiori Tsuruyama, Yuma Ookubo, Tatsuya Kondo and Eiichi Araki have nothing to disclose.
Compliance with Ethics Guidelines
The study was conducted in accordance with the International Conference on Harmonization of Good Clinical Practice, the Ethical Guidelines for Epidemiological Research of Japan, the ethical standards of the responsible committee on human experimentation (institutional and national), and the Helsinki Declaration of 1964, as revised in 2013. The study protocol was reviewed and approved by institutional ethics committees at Nishinihon Hospital and recorded at the UMIN Clinical Trials Registry (UMIN000041543). All participants provided written informed consent.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Matsumura, T., Makabe, T., Ueda, S. et al. Clinical Benefit of Switching from Low-Dose to High-Dose Empagliflozin in Patients with Type 2 Diabetes. Diabetes Ther 13, 1621–1634 (2022). https://doi.org/10.1007/s13300-022-01296-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-022-01296-y