Abstract
In metastatic renal cell carcinoma (mRCC), the prognostic role of several tumor tissue biomarkers has been evaluated, but the results were controversial. This study aims to verify the prognostic importance of selected tumor tissue biomarkers in patients with mRCC. The clinicopathological features, immunohistochemical staining and scoring for select tissue biomarkers, treatment, and outcome of patients with mRCC treated with vascular endothelial growth factor receptor (VEGFR) tyrosine kinase inhibitors (TKIs) between July 2006 and March 2011 at Asan Medical Center in Seoul, South Korea, were reviewed. In total, 123 patients met the inclusion criteria. Most patients had clear-cell carcinoma (107 patients, 87.0 %). First-line VEGFR TKIs were sunitinib (97 patients, 78.9 %), sorafenib (23 patients, 18.7 %), and pazopanib (3 patients, 2.4 %). With a median follow-up period of 60.0 months (95 % confidence interval (CI), 56.3–63.6), median overall survival (OS) and progression-free survival (PFS) were 25.6 months (95 % CI, 19.2–32.0) and 12.2 months (95 % CI, 8.1–16.3), respectively. In the multivariable analysis for OS, carbonic anhydrase IX (CAIX; 47.5 % or less vs. more than 47.5 %, p = 0.014), sarcomatoid change (40 % or less vs. more than 40 %, p < 0.001), tumor necrosis (20 % or less vs. more than 20 %, p = 0.006), and Heng’s risk group (good vs. intermediate vs. poor, p = 0.011) were identified as independent prognostic factors. In the multivariable analysis for PFS, CAIX (p < 0.001), phosphatase and tensin homolog (PTEN; 45 % or less vs. more than 45 %, p = 0.004), sarcomatoid change (p = 0.002), and tumor necrosis (p = 0.001) were identified as independent factors affecting PFS. CAIX and PTEN had prognostic importance for mRCC patients receiving first-line VEGFR TKI. Future validation and mechanistic studies are required.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Until early 2000, immunotherapy using interferon or interleukin was the de facto standard treatment for metastatic renal cell carcinoma (mRCC). Unfortunately, immunotherapy achieved only occasional responses, whereas substantial toxicities occurred invariably [1]. Limited treatment options and nonspecific mechanisms of action behind immunotherapy retarded vigorous biomarker investigations, preventing discrimination between patients who are likely to respond and those who are unlikely to respond.
Recent advances in understanding the molecular pathobiology underlying RCC has lead to the introduction of agents that target the vascular endothelial growth factor (VEGF) and mammalian target of rapamycin (mTOR) signaling pathways, resulting in huge changes in the management of this disease. There are currently several effective approaches for treating metastatic RCC [2–7]. Although VEGF receptor (VEGFR) tyrosine kinase inhibitors (TKIs) are rationally designed and are most commonly used as first-line treatment for mRCC, biomarkers that predict or prognosticate the treatment outcome have not been well elucidated.
In addition to VEGF and mTOR signaling pathway, several molecules have been investigated for their role in RCC pathogenesis. Von Hippel–Lindau (VHL) gene mutation leads to hypoxia-inducible factor (HIF) disregulation and resultant overexpression of VEGF that can lead to tumor angiogenesis [8]. Hepatocyte growth factor receptor (HGFR, also known as MET) is a receptor tyrosine kinase functions for cell proliferation, differentiation, angiogenesis, and tissue repair. It is also a proto-oncogene with implication in various cancers [9]. MET expression was higher in all RCC subtypes than in adjacent normal renal tissues in a series of more than 300 patients [10]. Carbonic anhydrases are metalloenzymes whose function in solid tumors is a neutralization of the intracellular milieu with a concomitant acidification of the extracellular milieu [11]. Although carbonic anhydrase IX (CAIX) is not expressed in healthy renal tissue, it is expressed in almost all clear cell RCCs and its expression is influenced by activation of the HIF-1α and the inactivation of the VHL gene [12, 13].
On the other hand, some proteins are shown to be involved in inducing resistance to VEGF inhibitors. Interleukin-8 (IL-8) is a member of the CXC family of chemokines and is a potent proangiogenic factor. In a recent study, IL-8 was upregulated in sunitinib-resistant tumor xenograft and inhibition of IL-8 re-sensitized these resistant tumors to sunitinib. Also, IL-8 expression was elevated in human clear cell RCC having intrinsic resistance to sunitinib therapy [14]. There are some evidences that upregulation of fibroblast growth factor (FGF) and FGF receptor (FGFR) may have a role in introducing resistance to anti-VEGF therapy [15]. FGF2 (FGF-basic) can suppress sunitinib-induced retraction of endothelial tubules and thus regulates endothelial sensitivity to sunitinib [16].
Several tumor tissue biomarkers such as von Hippel–Lindau protein, HIF-1α/HIF-2α, VEGF (and related proteins), or CAIX have been evaluated, but the results were controversial and defective with respect to retrospective design, small sample size, and lack of validation [17].
Some prognostic models comprising several clinical characteristics are widely used in practice [18, 19], but they are population-based models that categorize patients into a few prognostic groups (good, intermediate, or poor risk group); are composed of only clinical and laboratory characteristics, which are indicators of the general condition of the patients and the burden of the disease; and are missing molecular pathologic characteristics reflecting tumor biology. Therefore, to provide precision treatment, avoid futile treatment, and educate patients, the development of reliable biomarkers, which can be integrated into established prognostic models, is eagerly required.
To verify the prognostic importance of selected tumor tissue biomarkers in patients with mRCC, we performed immunohistochemical (IHC) staining in tumor samples and statistical analyses of the results.
Patients and methods
Patient selection
The medical records of 520 consecutive patients with histologically confirmed recurrent or metastatic RCC who were treated with VEGFR TKIs at the Department of Oncology, Asan Medical Center (Seoul, South Korea) from January 2006 to December 2013 were retrospectively reviewed. Among these patients, the selection criteria for study inclusion were as follows: patients who received VEGFR TKI as a first-line systemic treatment, patients for whom pre-treatment tissue samples were available, and patients who had undergone proper imaging, including baseline imaging with at least one follow-up image. We set liberal inclusion criteria in terms of performance status, prognostic risk group, and histology. Patients were excluded if the patients received immunotherapy, cytotoxic chemotherapy, or molecular targeted agents other than VEGFR TKI prior to VEGFR TKI; patients had inadequate clinicopathological data; insufficient tissue amount to perform pre-specified IHC; patients cannot be included in survival information due to lost to follow-up.
Clinical data collection
Clinicopathological data, including age, sex, Karnofsky performance status (KPS), tumor histology, pathological stage, laboratory data, date of the initial diagnosis, disease status (recurrent or metastatic), metastatic site, surgery (if performed), and survival status, were collected. A comprehensive review was performed on baseline and follow-up radiographic images, which were obtained every 6–8 weeks during treatment. Target lesion selection and response evaluations were performed retrospectively according to Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1. The study was approved by the institutional review board of Asan Medical Center.
Pathology review
Pre-treatment pathologic materials from each patient were reviewed for diagnostic reassessment and histologic subtyping according to the 2004 World Health Organization Tumor Classification. Each tumor was graded according to the Fuhrman grading system [20]. Pathologic materials, including immunostained tissue microarray (TMA) slides, were assessed by a single pathologist (YMC) who was unaware of the clinical variables and therapeutic outcomes.
TMA construction
A TMA construct with 1-mm-diameter cores was generated from formalin-fixed paraffin-embedded tissue blocks of renal cell carcinoma specimens using a tissue microarrayer (Beecher Instruments, Silver Spring, MD). Three representative cores were used to access each case.
IHC
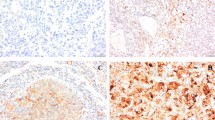
The expression of FGFR 1, 2, 3, and 4, FGF-basic, HIF-1α and HIF-2α, CAIX, mTOR, phosphatase and tensin homolog (PTEN), IL8, phosphor-S6 (pS6), and HGFR/MET was analyzed by IHC staining, which was performed using an automated staining system (BenchMark XT, Ventana Medical Systems, Tucson, AZ) and the ultraView universal DAB detection kit (Ventana Medical Systems). The primary antibodies used in the study, their dilutions, and the subcellular localization of each antigen are summarized in supplementary Table 1. Nuclei were counterstained with hematoxylin.
Assessment of IHC results
IHC staining was assessed using the semiquantitative “eyeball” measurement, and each core was scored based on the percentage of positive cell staining and the intensity, which was scored as negative, weak, moderate, or strong. Cases with moderate staining (FGFR1, FGF base, and HIF-1α) or strong staining (CAIX, mTOR, PTEN, FGFR2, FGFR3, FGFR4, IL8, pS6, HIF2α, and HGFR/MET) were regarded as positive, and the average percentage of positive cells in all cores was recorded.
Statistical analysis
The primary and secondary objectives of the present study were to identify prognostic biomarkers for overall survival (OS) and progression-free survival (PFS). OS was defined as the time from the date of initiation of the first-line line treatment to the date of death from any cause, and PFS was defined as the date of disease progression or death from any cause, whichever comes first. Patient characteristics, histologic findings, and IHC results were summarized descriptively, and continuous clinical variables were dichotomized for convenience. Maxstat, a maximally selected rank statistics in R 3.0.3 (R Development Core Team, Vienna, Austria, http://www.R-project.org), was used to identify optimal cutting points for each marker [21]. We performed univariate analyses for survival using the log-rank test, and the Cox proportional hazard model was performed for factors that were significant in univariate analysis. Statistical analyses were performed using PASW statistics (version 20; IBM Co., Armonk, NY) and R. p values less than 0.05 (two-sided) were considered statistically significant.
Results
Patient characteristics
In total, 123 patients met the inclusion criteria (Table 1). The median age was 57 years (range 17–85), and 88 patients (71.5 %) were male. Most patients had clear-cell carcinoma (107 patients, 87.0 %). Fuhrman’s nuclear grade (NG) was 2 in 21 (17.1 %), 3 in 49 (39.8 %), and 4 in 52 patients (42.3 %). Sarcomatoid change and coagulative necrosis were found in 51 patients (41.5 %) and 58 patients (47.2 %), respectively. Seventy-two patients (57.7 %) had initially metastatic disease, whereas 52 patients (42.3 %) had recurrent disease. Nephrectomy was performed in 120 patients (97.6 %), and metastasectomy in 33 patients (26.8 %). Most patients had a relatively good performance status, as 11 (8.9 %), 51 (41.5 %), and 50 patients (40.7 %) had 100, 90, and 80 % of KPS. Common sites of metastases were the lung (74.8 %), lymph node (24.4 %), bone (20.3 %), and liver (11.4 %). Using Heng’s criteria, 20 (16.3 %), 87 (70.7 %), and 16 patients (13.0 %) belonged to the favorable, intermediate, and poor risk groups, respectively. First-line VEGFR TKIs prescribed were sunitinib (97 patients, 78.9 %), sorafenib (23 patients, 18.7 %), and pazopanib (3 patients, 2.4 %).
The best responses to first-line VEGFR TKI were complete response (CR) in 1 patient (0.8 %), partial response (PR) in 44 (35.8 %), stable disease (SD) in 58 (47.2 %), and progressive disease (PD) in 20 patients (16.3 %). Thus, the objective response rate (CR or PR) was 36.6 %.
Impact of biomarkers on OS
For the primary endpoint, we analyzed OS with known clinical prognostic factors and IHC staining results. Cutoff values for statistical analyses were obtained by the maximally selected rank statistics. With a median follow-up duration of 60.0 months (95 % confidence interval (CI), 56.3–63.6 months), OS for all patients was 25.6 months (95 % CI, 19.2–32.0 months, Fig. 1). In the univariate analysis (Table 2), CAIX (less than 47.5 vs. 47.5 % or more, p = 0.001), HIF-2α (negative vs. positive, p < 0.001), mTOR (20 % or less vs. more than 20 %, p = 0.0032), Heng’s risk group (good vs. intermediate vs. poor, p < 0.001), sarcomatoid change (40 % or less vs. more than 40 %, p < 0.001), tumor necrosis (20 % or less vs. more than 20 %, p < 0.001), and Furhman NG (2 vs. 3 vs. 4, p = 0.037) were statistically significant. In the multivariable analysis (Table 3), CAIX, Heng’s risk group, sarcomatoid change, and tumor necrosis were identified as independent prognostic factors related to OS.
Kaplan–Meier curves for overall survival (OS) and progression-free survival (PFS) with first-line VEGFR TKI (n = 123). With a median follow-up period of 60.0 months (95 % CI, 56.3–63.6 months), median OS and PFS were 25.6 months (95 % CI, 19.2–32.0 months) and 12.2 months (95 % CI, 8.1–16.3 months), respectively
Impact of biomarkers on PFS and response
Median PFS was 12.2 months (95 % CI, 8.1–16.3 months, Fig. 1) for all patients. Using the same cutoff value as in OS analyses, we performed univariate analysis on PFS (Table 4). PTEN (40 % or less vs. more than 40 %, p = 0.012), CAIX (less than 47.5 % vs. 47.5 % or more, p = 0.001), Heng’s risk group (good vs. intermediate vs. poor, p < 0.014), sarcomatoid change (40 % or less vs more than 40 %, p < 0.001), tumor necrosis (20 % or less vs more than 20 %, p < 0.001), and Furhman NG (2 vs. 3 vs. 4, p = 0.003) were statistically significantly associated with longer PFS. In the multivariable analysis (Table 5), CAIX, PTEN, sarcomatoid change, and tumor necrosis were identified as independent factors affecting PFS.
Discussion
In the present study, we examined the prognostic role of tissue biomarkers in patients with mRCC treated with first-line VEGFR TKIs. We found that a lower expression of CAIX, a higher percentage of sarcomatoid change, abundant tumor necrosis, and belonging to the poor Heng’s risk group were independent adverse prognostic factors for OS. In addition, lower expression of CAIX, higher expression of PTEN, higher percentage of sarcomatoid change, and abundant tumor necrosis were independent adverse prognostic factors for PFS.
Heng’s risk group [18], sarcomatoid change [22], and tumor necrosis [23] are well-known prognostic factors in mRCC. In our study, all three factors were statistically significant in multivariable analysis for OS. However, CAIX, which was identified as a statistically significant prognostic factor for OS in our study, has been controversial. CAIX is a cytosolic transmembrane protein that regulates cell proliferation under hypoxic conditions and is expressed in the vast majority of clear-cell RCCs but is absent in non-tumor kidney tissue [24]. CAIX expression is mediated by the HIF transcriptional complex and correlates with von Hippel–Lindau gene (VHL) inactivation, which has a central role in the pathogenesis of clear-cell RCC [25]. In the pre-VEGFR TKI era, high CAIX expression (>85 %) as determined by IHC was initially reported to be associated with a higher response rate and a better survival in mRCC patients undergoing immunotherapy [26, 27], but in a prospective validation study, the SELECT trial failed to validate its role as a predictive factor [28]. In the VEGFR TKI era, CAIX was shown to be neither a predictive factor for response nor a prognostic factor for survival in patients with mRCC receiving VEGFR targeted therapy (mainly sunitinib or sorafenib), in both retrospective [29] and prospectively maintained datasets [30, 31] from the TARGET or renal EFFECT trial [2, 32].
Contrary to those studies that evaluated CAIX in the VEGFR TKI era [29–31], CAIX was an independent prognostic factor for both OS and PFS in the present study. It is uncertain why the prognostic role of CAIX differs between studies, but there were some differences between our study and previous studies. Our study only included patients who were administered VEGFR TKI as a first-line systemic treatment, whereas many patients in the study by Choueiri et al. (2010) had received previous systemic therapy. In total, 40 % of patients in the retrospective dataset received cytokines, vaccines, chemotherapy, or thalidomide, and all patients in the prospectively maintained dataset received cytokines [30]. In addition, the cutoff value for dichotomizing patients was different. Both studies by Choueiri et al. used 85 % as the cutoff point based on a previous study [27], in which the cutoff value was calculated by using survival tree analysis. Because the 85 % cutoff point was derived from patients who underwent immunotherapy [27], it might be unreliable to adopt this cutoff point for patients receiving VEGFR TKI. To solve this problem, we determined a new cutoff value of 47.5 % by using the maximally selected rank statistics which can estimate and provide a simple cutoff point, even for censored or tied observations [21]. Recently, a more complicated scoring system of CAIX, such as individual scores (percent of tumor expression, predominant intensity, focal score, and focal intensity) and composite scores, was used, and four-group categorization also failed to show any statistical and clinical significance [31].
In an analysis for PFS, we demonstrated that lower CAIX expression, abundant PTEN expression, sarcomatoid change, and abundant tumor necrosis were independent adverse prognostic factors for PFS. PTEN inhibits phosphoinositide 3-kinase (PI3K)/Akt signaling via its lipid phosphatase activity, thereby controlling cell growth, survival, and metabolic processes [33]. In addition, the PI3K/Akt/mTOR signaling pathway positively regulates HIF-1α protein in certain cancer cells [34–36]. Therefore, PTEN theoretically inhibits HIF-1α via inhibition of PI3K/Akt/mTOR pathway. In patients with advanced RCC, positive PTEN was indicative of a good prognosis with sunitinib (PFS, 15.1 vs. 6.5 months; p = .003) [37]. Therefore, our result that higher PTEN expression (>45 %) is an independent adverse prognostic factor for PFS is contradictory to that reported previously.
Although canonical role of PTEN with relation to inhibition of PI3K/Akt/mTOR pathway is plain, it is not clear how PTEN correlates with HIFs. In recent years, it has emerged that among HIFs, HIF-2α is more relevant in the development and progression of RCC [38]. However, most studies have focused on the relationship between the PI3K/Akt/mTOR pathway and HIF-1α [34, 35]. On the other hand, the PI3K/Akt/mTOR pathway has not been clearly shown to correlate with HIF-2α [36]. One study suggested that HIF-2α is upregulated by PTEN at both the transcriptional and posttranscriptional levels under hypoxic conditions in the embryonic stem cell–derived embryoid body [39]. Although the exact mechanisms underlying the regulation of HIF-2α by PTEN have not been elucidated, yin yang 1 (YY1), a noble corepressor of HIF-2α, may play a role in this pathway [40]. Thus, the role of PTEN with respect to its relationship with HIF-1α and HIF-2α under VHL inactivation conditions in the prognosis of mRCC patients receiving VEGFR TKI needs further evaluation.
Despite the considerable amount of effort that has been expended by clinicians and researchers investigating tissue biomarkers for RCC, unfortunately, no tissue biomarker has been universally accepted as a predictive or prognostic factor for mRCC [17]. Recently, the concept of intratumoral heterogeneity and branched evolution has arisen [41]. This concept purports that genetic alterations or the protein expression profile of only a part of a tumor might not be shared with and representative of the whole cancer burden in the patient. This constitutes a major challenge for the development of tissue biomarkers, which usually reflect a limited part of the tumor. For the time being at least, perhaps the priority should not be tissue biomarker development or validation but, rather, the invention of novel techniques that overcome intratumoral heterogeneity and clonal evolution.
There are some limitations in this study of note. First, this is a retrospective study which included a limited number of patients who were treated at single institution over several years and had available pre-treatment tissue sample. Therefore, there could be potential biases inherent to this type of study, such as information or selection bias. Second, pathologic examination and immunohistochemical staining were performed on only limited part of tumor mass. Some of them were prior nephrectomy specimen for the patients with recurrent disease, while others are debulking nephrectomy specimen or core needle biopsy specimen for the patients with initially metastatic patients. Considering intratumoral heterogeneity and branched evolution, our result may not reflect overall characteristics of whole tumor burden.
Conclusions
Lower expression of CAIX, higher expression of PTEN, higher percentage of sarcomatoid change, and abundant tumor necrosis were independent adverse prognostic tissue biomarkers for OS and/or PFS. Future validation and mechanistic studies are required, and consideration should be given to novel techniques for overcoming intratumoral heterogeneity and branched evolution.
References
Rini BI, Campbell SC, Escudier B. Renal cell carcinoma. Lancet. 2009;373(9669):1119–32. doi:10.1016/S0140-6736(09)60229-4.
Escudier B, Eisen T, Stadler WM, Szczylik C, Oudard S, Siebels M, et al. Sorafenib in advanced clear-cell renal-cell carcinoma. N Engl J Med. 2007;356(2):125–34. doi:10.1056/NEJMoa060655.
Hudes G, Carducci M, Tomczak P, Dutcher J, Figlin R, Kapoor A, et al. Temsirolimus, interferon alfa, or both for advanced renal-cell carcinoma. N Engl J Med. 2007;356(22):2271–81. doi:10.1056/NEJMoa066838.
Motzer RJ, Escudier B, Oudard S, Hutson TE, Porta C, Bracarda S, et al. Efficacy of everolimus in advanced renal cell carcinoma: a double-blind, randomised, placebo-controlled phase III trial. Lancet. 2008;372(9637):449–56. doi:10.1016/S0140-6736(08)61039-9.
Motzer RJ, Hutson TE, Tomczak P, Michaelson MD, Bukowski RM, Rixe O, et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med. 2007;356(2):115–24. doi:10.1056/NEJMoa065044.
Rini BI, Escudier B, Tomczak P, Kaprin A, Szczylik C, Hutson TE, et al. Comparative effectiveness of axitinib versus sorafenib in advanced renal cell carcinoma (AXIS): a randomised phase 3 trial. Lancet. 2011;378(9807):1931–9. doi:10.1016/S0140-6736(11)61613-9.
Sternberg CN, Davis ID, Mardiak J, Szczylik C, Lee E, Wagstaff J, et al. Pazopanib in locally advanced or metastatic renal cell carcinoma: results of a randomized phase III trial. J Clin Oncol: OffJ Am Soc Clin Oncol. 2010;28(6):1061–8. doi:10.1200/JCO.2009.23.9764.
Patel PH, Chadalavada RS, Chaganti RS, Motzer RJ. Targeting von Hippel-Lindau pathway in renal cell carcinoma. Clin Cancer Res: Off J Am Assoc Cancer Res. 2006;12(24):7215–20. doi:10.1158/1078-0432.CCR-06-2254.
Birchmeier C, Birchmeier W, Gherardi E, Vande Woude GF. Met, metastasis, motility and more. Nat Rev Mol Cell Biol. 2003;4(12):915–25. doi:10.1038/nrm1261.
Gibney GT, Aziz SA, Camp RL, Conrad P, Schwartz BE, Chen CR, et al. c-Met is a prognostic marker and potential therapeutic target in clear cell renal cell carcinoma. Ann Oncol. 2013;24(2):343–9. doi:10.1093/annonc/mds463.
Tostain J, Li G, Gentil-Perret A, Gigante M. Carbonic anhydrase 9 in clear cell renal cell carcinoma: a marker for diagnosis, prognosis and treatment. Eur J Cancer. 2010;46(18):3141–8. doi:10.1016/j.ejca.2010.07.020.
Grabmaier K, AdW MC, Verhaegh GW, Schalken JA, Oosterwijk E. Strict regulation of CAIX(G250/MN) by HIF-1alpha in clear cell renal cell carcinoma. Oncogene. 2004;23(33):5624–31. doi:10.1038/sj.onc.1207764.
Ivanov SV, Kuzmin I, Wei MH, Pack S, Geil L, Johnson BE, et al. Down-regulation of transmembrane carbonic anhydrases in renal cell carcinoma cell lines by wild-type von Hippel-Lindau transgenes. Proc Natl Acad Sci U S A. 1998;95(21):12596–601.
Huang D, Ding Y, Zhou M, Rini BI, Petillo D, Qian CN, et al. Interleukin-8 mediates resistance to antiangiogenic agent sunitinib in renal cell carcinoma. Cancer Res. 2010;70(3):1063–71. doi:10.1158/0008-5472.CAN-09-3965.
Lieu C, Heymach J, Overman M, Tran H, Kopetz S. Beyond VEGF: inhibition of the fibroblast growth factor pathway and antiangiogenesis. Clin Cancer Res: Off J Am Assoc Cancer Res. 2011;17(19):6130–9. doi:10.1158/1078-0432.CCR-11-0659.
Welti JC, Gourlaouen M, Powles T, Kudahetti SC, Wilson P, Berney DM, et al. Fibroblast growth factor 2 regulates endothelial cell sensitivity to sunitinib. Oncogene. 2011;30(10):1183–93. doi:10.1038/onc.2010.503.
Maroto P, Rini B. Molecular biomarkers in advanced renal cell carcinoma. Clin Cancer Res: Off J Am Assoc Cancer Res. 2014;20(8):2060–71. doi:10.1158/1078-0432.CCR-13-1351.
Heng DY, Xie W, Regan MM, Warren MA, Golshayan AR, Sahi C, et al. Prognostic factors for overall survival in patients with metastatic renal cell carcinoma treated with vascular endothelial growth factor-targeted agents: results from a large, multicenter study. J Clin Oncol: Off J Am Soc Clin Oncol. 2009;27(34):5794–9. doi:10.1200/JCO.2008.21.4809.
Motzer RJ, Mazumdar M, Bacik J, Berg W, Amsterdam A, Ferrara J. Survival and prognostic stratification of 670 patients with advanced renal cell carcinoma. J Clin Oncol: Off J Am Soc Clin Oncol. 1999;17(8):2530–40.
Fuhrman SA, Lasky LC, Limas C. Prognostic significance of morphologic parameters in renal cell carcinoma. Am J Surg Pathol. 1982;6(7):655–63.
Lausen B, Schumacher M. Maximally selected rank statistics. Biometrics. 1992;48(1):73–85. doi:10.2307/2532740.
Shuch B, Bratslavsky G, Linehan WM, Srinivasan R. Sarcomatoid renal cell carcinoma: a comprehensive review of the biology and current treatment strategies. Oncologist. 2012;17(1):46–54. doi:10.1634/theoncologist.2011-0227.
Delahunt B, Cheville JC, Martignoni G, Humphrey PA, Magi-Galluzzi C, McKenney J, et al. The International Society of Urological Pathology (ISUP) grading system for renal cell carcinoma and other prognostic parameters. Am J Surg Pathol. 2013;37(10):1490–504. doi:10.1097/PAS.0b013e318299f0fb.
Dorai T, Sawczuk IS, Pastorek J, Wiernik PH, Dutcher JP. The role of carbonic anhydrase IX overexpression in kidney cancer. Eur J Cancer. 2005;41(18):2935–47. doi:10.1016/j.ejca.2005.09.011.
Patard JJ, Fergelot P, Karakiewicz PI, Klatte T, Trinh QD, Rioux-Leclercq N, et al. Low CAIX expression and absence of VHL gene mutation are associated with tumor aggressiveness and poor survival of clear cell renal cell carcinoma. Int J Cancer J Int Du Cancer. 2008;123(2):395–400. doi:10.1002/ijc.23496.
Atkins M, Regan M, McDermott D, Mier J, Stanbridge E, Youmans A, et al. Carbonic anhydrase IX expression predicts outcome of interleukin 2 therapy for renal cancer. Clin Cancer Res: Off J Am Assoc Cancer Res. 2005;11(10):3714–21. doi:10.1158/1078-0432.CCR-04-2019.
Bui MH, Seligson D, Han KR, Pantuck AJ, Dorey FJ, Huang Y, et al. Carbonic anhydrase IX is an independent predictor of survival in advanced renal clear cell carcinoma: implications for prognosis and therapy. Clin Cancer Res: Off J Am Assoc Cancer Res. 2003;9(2):802–11.
McDermott DF, Ghebremichael MS, Signoretti S, Moargolin KA, Clark J, Sosman JA, et al. The high-dose aldesleukin “SELECT” trial in patients with metastatic renal cell carcinoma. J Clin Oncol: Off J Am Soc Clin Oncol. 2010;28(15s):abstr 4514.
Choueiri TK, Regan MM, Rosenberg JE, Oh WK, Clement J, Amato AM, et al. Carbonic anhydrase IX and pathological features as predictors of outcome in patients with metastatic clear-cell renal cell carcinoma receiving vascular endothelial growth factor-targeted therapy. BJU Int. 2010;106(6):772–8. doi:10.1111/j.1464-410X.2010.09218.x.
Choueiri TK, Cheng S, Qu AQ, Pastorek J, Atkins MB, Signoretti S. Carbonic anhydrase IX as a potential biomarker of efficacy in metastatic clear-cell renal cell carcinoma patients receiving sorafenib or placebo: analysis from the treatment approaches in renal cancer global evaluation trial (TARGET). Urol Oncol. 2013;31(8):1788–93. doi:10.1016/j.urolonc.2012.07.004.
Motzer RJ, Hutson TE, Hudes GR, Figlin RA, Martini JF, English PA, et al. Investigation of novel circulating proteins, germ line single-nucleotide polymorphisms, and molecular tumor markers as potential efficacy biomarkers of first-line sunitinib therapy for advanced renal cell carcinoma. Cancer Chemother Pharmacol. 2014;74(4):739–50. doi:10.1007/s00280-014-2539-0.
Motzer RJ, Hutson TE, Olsen MR, Hudes GR, Burke JM, Edenfield WJ, et al. Randomized phase II trial of sunitinib on an intermittent versus continuous dosing schedule as first-line therapy for advanced renal cell carcinoma. J Clin Oncol: Off J Am Soc Clin Oncol. 2012;30(12):1371–7. doi:10.1200/JCO.2011.36.4133.
Wu X, Senechal K, Neshat MS, Whang YE, Sawyers CL. The PTEN/MMAC1 tumor suppressor phosphatase functions as a negative regulator of the phosphoinositide 3-kinase/Akt pathway. Proc Natl Acad Sci U S A. 1998;95(26):15587–91.
Pore N, Jiang Z, Shu HK, Bernhard E, Kao GD, Maity A. Akt1 activation can augment hypoxia-inducible factor-1alpha expression by increasing protein translation through a mammalian target of rapamycin-independent pathway. Mol Cancer Res: MCR. 2006;4(7):471–9. doi:10.1158/1541-7786.MCR-05-0234.
Zhong H, Chiles K, Feldser D, Laughner E, Hanrahan C, Georgescu MM, et al. Modulation of hypoxia-inducible factor 1alpha expression by the epidermal growth factor/phosphatidylinositol 3-kinase/PTEN/AKT/FRAP pathway in human prostate cancer cells: implications for tumor angiogenesis and therapeutics. Cancer Res. 2000;60(6):1541–5.
Blancher C, Moore JW, Robertson N, Harris AL. Effects of ras and von Hippel-Lindau (VHL) gene mutations on hypoxia-inducible factor (HIF)-1alpha, HIF-2alpha, and vascular endothelial growth factor expression and their regulation by the phosphatidylinositol 3′-kinase/Akt signaling pathway. Cancer Res. 2001;61(19):7349–55.
Muriel Lopez C, Esteban E, Berros JP, Pardo P, Astudillo A, Izquierdo M, et al. Prognostic factors in patients with advanced renal cell carcinoma. Clin Genitourin Cancer. 2012;10(4):262–70. doi:10.1016/j.clgc.2012.06.005.
Philips GK, Atkins MB. New agents and new targets for renal cell carcinoma. American Society of Clinical Oncology educational book / ASCO American Society of Clinical Oncology Meeting. 2014:e222-7. doi:10.14694/EdBook_AM.2014.34.e222.
Qi Y, Liu J, Saadat S, Tian X, Han Y, Fong GH, et al. PTEN induces apoptosis and cavitation via HIF-2-dependent Bnip3 upregulation during epithelial lumen formation. Cell Death Differ. 2015;22(5):875–84. doi:10.1038/cdd.2014.185.
Petrella BL, Brinckerhoff CE. PTEN suppression of YY1 induces HIF-2 activity in von-Hippel-Lindau-null renal-cell carcinoma. Cancer Biol Ther. 2009;8(14):1389–401.
Gerlinger M, Rowan AJ, Horswell S, Larkin J, Endesfelder D, Gronroos E, et al. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N Engl J Med. 2012;366(10):883–92. doi:10.1056/NEJMoa1113205.
Acknowledgments
This study was presented in part at the 2015 Genitourinary Cancers Symposium, 26–28 February 2015 in Orlando, FL. This study was supported by a grant (HI12C1788, HI14C1931, HI14C1731) from the Korean Health Technology R&D Project, Ministry of Health and Welfare, Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 91 kb)
Rights and permissions
About this article
Cite this article
Park, I., Cho, Y.M., Lee, JL. et al. Prognostic tissue biomarker exploration for patients with metastatic renal cell carcinoma receiving vascular endothelial growth factor receptor tyrosine kinase inhibitors. Tumor Biol. 37, 4919–4927 (2016). https://doi.org/10.1007/s13277-015-4339-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-4339-5