Abstract
Background
Bacterial ghosts (BGs) are empty cell envelopes derived from bacteria, making them safe and non-replicative, and BGs have shown great potential as a vaccine platform. Specifically, chemically induced BGs are generated by selectively removing the cytoplasmic content of bacterial cells while preserving the structural integrity of the cell envelope.
Objective
Generally, BGs are genetically engineered, but this is limited to Gram-negative bacteria. However, the utilization of chemically induced BGs can be extended to Gram-positive bacteria, resulting in empty bacterial envelopes that hold potential as a platform for drug delivery.
Results
Chemically induced BGs offer several advantages, including improved safety profile and immunogenicity, and efficient antigen presentation. Preclinical studies have yielded promising results, exhibiting enhanced immune responses and protection against diverse pathogens.
Conclusion
Chemically induced BGs represent a novel and promising approach for vaccine development, holding the potential for advancing disease prevention and public health.
Purpose of review
In this review, we discuss key aspects of chemically induced BGs, including their production principles, mechanisms of formation, characterization techniques, immunogenicity, and medical applications. We also discuss the challenges and direction of future research for optimizing production methods for chemically induced BGs, evaluating long-term safety, and undertaking clinical trials to assess their efficacy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bacteria are prokaryotes, lacking a nucleus and membrane-bound organelles (Gitai 2005). They have a simple cellular structure consisting of a cell membrane, cytoplasm, genetic material (DNA or RNA), and often a cell wall (Lewis 2004). Bacteria have wide applications in various aspects of human life, including in medicine (Sender et al. 2016; Eckhardt et al. 2013). While some bacteria cause infections and diseases (van Elsland and Neefjes 2018; Fernebro 2011; Bunchorntavakul et al. 2016), many beneficial bacteria have important medical applications (Taylor et al. 2002; Hinchliffe et al. 2021). Beneficial bacteria have several noteworthy medical applications (Yan et al. 2023). First, live microorganisms provide health benefits when consumed in adequate amounts as probiotics (Garbacz 2022; Mu and Cong 2019). Probiotics, such as certain strains of Lactobacillus and Bifidobacterium, are used to prevent and treat various gastrointestinal conditions, including diarrhea, irritable bowel syndrome, and inflammatory bowel disease (Ringo et al. 2020; Fioramonti et al. 2003). They help maintain a healthy balance of microorganisms in the gut microbiota, which is crucial for digestion, nutrient absorption, immune system modulation, and overall gut health (de Moreno de LeBlanc and LeBlanc 2014; Wallace et al. 2011; Abenavoli et al. 2019). In addition, numerous antibiotics are derived from bacteria or their metabolic products (Anke and Zahner 1978; Gunnarsson et al. 2004; Korp et al. 2016). Antibiotics are used to treat bacterial infections by inhibiting the growth or killing bacteria causing infections (Hutchings et al. 2019; Kummerer 2003). Certain bacteria can break down or degrade environmental pollutants (Segura and Ramos 2013; Le Borgne et al. 2008). Moreover, bacteria are employed in various industrial processes. For instance, bacteria are used in the production of enzymes, such as amylase or cellulase, which find applications in the food, textile, and detergent industries (Awasthi et al. 2018; Mohammad et al. 2017; Dai et al. 2020). Additionally, bacteria like Escherichia coli (E. coli) have been genetically engineered to produce therapeutic proteins like insulin, growth factors, and vaccines (Waegeman and Soetaert 2011; Goldstein and Thomas 2004). Bacteria are also important in medical diagnostics. They can be isolated from patient samples and cultured to identify the causative agents of bacterial infections. Although bacteria have numerous medical applications, certain infectious strains cause deadly diseases. It is crucial to maintain a balance between the beneficial and harmful aspects of bacteria and undertake appropriate measures, such as hygiene practices and responsible antibiotic use, to effectively manage health issues related to bacteria.
Bacterial ghosts (BGs) are empty cell envelopes derived from bacteria. BGs generated through controlled biological or chemical treatment offer unique advantages such as retained antigenicity, structural integrity, and versatile cargo loading capacity (Abenavoli et al. 2019; Li et al. 2021; Kang et al. 2022). Herein, we discuss the mechanisms, characterization, and diverse applications of chemically induced BGs in vaccine development, drug delivery, and diagnostics. Additionally, we highlight the potential of chemically induced BGs in advancing biomedical strategies and contributing to innovative biomedical approaches.
Principles of BG production
BGs, which are primarily hollow cell envelopes, are produced from Gram-negative bacteria by controlled expression of the PhiX174 lysis gene E (Park et al. 2016; Lee et al. 2008; Witte et al. 1990). This gene encodes a small protein with hydrophobic regions at its N-terminal (Hajam et al. 2017). Upon binding to the inner membrane of the bacterial cell wall, the hydrophobic N-terminal of lysis protein E enhances internal osmotic pressure, leading to the formation of transmembrane structures (Park et al. 2016). These structures facilitate the release of cellular contents, including ribosomes and nucleic acids, generating BGs (Hajam et al. 2017). The resulting transmembrane tunnels have a diameter of approximately 40–200 nm and allow the expulsion of the cytoplasm while preserving the integrity and morphology of the cellular envelope (Lubitz et al. 2009; Witte et al. 1992). Electron microscopy revealed that these empty bacterial envelopes retain the surface structures of native bacteria, such as outer membrane proteins, adhesions, lipopolysaccharides (LPS), and the peptidoglycan layer (Li et al. 2021; Chen et al. 2021). Therefore, BGs are promising candidates for vaccine development and as delivery systems for antigens in humans and other animal.
Mechanisms of chemically induced BG formation
While gene E-induced BGs have proven effective in conferring protective immunity against specific infections, their formation has been limited to Gram-negative bacteria due to the lack of suitable inner and outer membranes in Gram-positive bacteria (Park et al. 2016). However, a novel protocol has been developed to overcome this constraint and expand the scope of BGs to use Gram-positive bacteria (Amara et al. 2013). This innovative approach involves the application of various chemical agents, including sodium hydroxide (NaOH), hydrochloric acid (HCl), sulfuric acid (H2SO4), sodium dodecyl sulfate (SDS), nitric acid (HNO3), hydrogen peroxide (H2O2), and Tween-80, at their minimum inhibitory concentrations (MIC) or minimum growth concentrations (MGC) (Li et al. 2021; Park et al. 2016; Ji et al. 2022; Rabea et al. 2018, 2022; Sheweita et al. 2022). These chemically induced BGs by specific chemical agents disrupt the cell membrane and release the cytoplasmic contents, selectively removing the cellular contents of bacterial cells while preserving the cell envelope. This multi-step process begins with the growth of bacterial cells under suitable conditions to promote cell envelope production. Subsequently, the cells are treated with a chemical agent that selectively disrupts the cytoplasmic membrane while leaving the cell envelope intact (Fig. 1). The resulting BGs retain the structural and functional properties of the original cell, including the cell wall and outer membrane, while maintaining its antigenicity. These BGs interact with immune cells, eliciting both innate and adaptive immune responses (Lubitz et al. 2009; Vinod et al. 2015, 2017). Additionally, their well-preserved morphology and high loading capacity make BGs excellent vehicles for delivering subunit vaccines, DNA vaccines, as well as chemotherapeutics and antibiotics (Szostak et al. 1996; Liu et al. 2006; Hensel et al. 2000; Mayr et al. 2005a). In conclusion, BGs represent a versatile platform for the development of vaccines and therapeutic carriers for a wide range of infectious and non-infectious diseases.
Characterization of chemically induced BGs
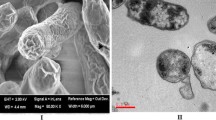
Chemically derived BGs undergo morphological and structural changes compared to their original bacterial cells. Morphological analysis of chemically derived BGs is commonly conducted using electron microscopy such as scanning electron microscopy (SEM) and transmission electron microscopy (TEM) (Chaudhari et al. 2012; Wu et al. 2017; Ma et al. 2021). SEM reveals that chemically induced BGs retain the surface features and structures of the parent bacterial cells, including outer membrane proteins, adhesions, and other surface components, maintaining morphological similarity (Park et al. 2016; Jawale et al. 2014) (Fig. 2). TEM allows for the examination of the internal structures of chemically derived BGs, displaying empty cell envelopes with intact cell walls and outer membranes devoid of internal components (Wu et al. 2017). Apart from morphological analysis, various techniques help assess the structural characteristics of chemically derived BGs. These include analysis of their biochemical composition through techniques like Fourier-transform infrared spectroscopy (FTIR) (Huang et al. 2017; Parvulescu et al. 2021). Furthermore, the integrity and stability of BGs can be assessed through techniques like dynamic light scattering (DLS) (Go et al. 2019; Myers et al. 2022) and atomic force microscopy (AFM) (Eslaminejad et al. 2022; Anderson et al. 2004) by determining their size distribution, surface charge, and stability. As the antigenic properties of chemically derived BGs are preserved, immunological techniques like enzyme-linked immunosorbent assay (ELISA) and immunofluorescence can confirm the presence and accessibility of specific surface antigens, providing insight into their immunogenicity and antigen presentation capabilities. These comprehensive analyses help characterize BGs by providing details about their morphology, surface features, biochemical composition, integrity, stability, and antigenicity, as well as, highlight the potential of chemically derived BGs for various biomedical applications.
Roles of chemically induced BGs in immunogenicity and immune response modulation
Chemically induced BGs play a significant role in immunogenicity and immune response modulation. They retain the original bacterial cells’ structural components and surface antigens, such as LPS, outer membrane proteins, and other surface molecules (Li et al. 2021). These components are recognized by the immune system as foreign antigens, triggering immune responses (Kang et al. 2022). The presence of intact surface structures in BGs enhances their immunogenicity, eliciting robust immune responses. BGs can activate innate immune responses through the recognition of pathogen-associated molecular patterns (PAMPs) on their surface (Akira et al. 2006; Schnare et al. 2001). Components such as LPS and other bacterial surface molecules are recognized by pattern recognition receptors (PRRs) on immune cells, such as macrophages and dendritic cells (Medzhitov and Janeway 2000; Kawai and Akira 2011; Akira and Hemmi 2003). This recognition leads to the secretion of pro-inflammatory cytokines and further recruitment of immune cells, promoting the immune response against the target antigen (Quevedo-Diaz et al. 2010; Steinhagen et al. 2011).
BGs also stimulate adaptive immune responses through the activation of B and T cells. The presence of antigens on the surface or within the BGs facilitates antigen presentation to immune cells, leading to the generation of antigen-specific antibodies by B cells (Hajam et al. 2017; Szostak et al. 1996) and the activation of T cells (Felnerova, et al. 2004; Kudela et al. 2010; Langemann et al. 2010; Mayr et al. 2005b; Dobrovolskiene et al. 2018). This ultimately results in the production of specific antibodies, memory B cells, and cytotoxic T lymphocytes, contributing to long-term immunity. BGs possess intrinsic adjuvant properties, meaning that they can enhance the immune response to co-administered antigens (Abenavoli et al. 2019; Langemann et al. 2010; Riedmann et al. 2007; Szostak et al. 1997; Jalava et al. 2002). The presence of bacterial surface components, such as LPS, can activate immune cells and promote the secretion of pro-inflammatory cytokines (Ciesielska et al. 2021; Yang et al. 2015). This adjuvant effect helps to amplify the immune response and improve the effectiveness of the vaccine (Zariri and van der Ley 2015; Kumar et al. 2019; Melssen et al. 2019). Depending on the properties of the bacterial components retained in the BGs and the type of antigen presented, they can skew the immune response in a particular direction. For example, BGs containing specific bacterial components can promote a Th1 response characterized by cellular immunity (Hensel et al. 2000; Cao et al. 2018; Won et al. 2017), or a Th2 response characterized by antibody production (Ebensen et al. 2004; Cai et al. 2010; Senevirathne et al. 2021). Therefore, chemically induced BGs can enhance immunogenicity, activate innate and adaptive immune responses, exhibit adjuvant properties, and modulate the immune response. These features make them valuable tools in vaccine development and immunotherapy to improve the effectiveness of vaccines and tailor immune responses to specific pathogens or diseases.
Discussion
Chemically induced BGs have shown great potential in vaccine development due to their unique properties and immunogenicity (Chen et al. 2021; Ebensen et al. 2004; Chiang et al. 2005). BGs serve as efficient carriers for delivering antigens to the immune system (Ebensen et al. 2004). They can be loaded with specific antigens by incorporating them into the inner spaces of the ghost or by presenting them on the surface of the cell envelope (Li et al. 2021; Lubitz et al. 2009). This allows for the targeted delivery of antigens to antigen-presenting cells, leading to effective immune responses. In terms of adjuvant properties, BGs possess intrinsic adjuvant properties, meaning that they enhance the immune response to co-administered antigens (Szostak et al. 1997). Chemically induced BGs are produced from various bacterial species of both Gram-negative and Gram-positive bacteria and have wide applications. This expands the scope of potential antigens and their applications in vaccine development, targeting a wide range of infectious diseases (Batah and Ahmad 2020). Furthermore, BGs have improved stability compared to live bacterial vaccines (Szostak et al. 1996). Chemically induced BGs offer numerous advantages in vaccine development, including efficient antigen delivery, enhanced immunogenicity, intrinsic adjuvant properties, safety, and scalability. Moreover, the production of BGs can be easily scaled up, making them highly suitable for large-scale vaccine manufacturing (Amara et al. 2013). The flexibility of BGs has been exploited to generate foreign antigens in a biologically active form that are firmly attached to the envelope. It has been effective as a vaccine delivery or immune modulator in animal studies (Chen et al. 2021; Riedmann et al. 2007; Jalava et al. 2002). In in vivo studies, they have successfully used BGs to deliver antigens form various pathogens, such as Salmonella, Escherichia coli, Klebsiella pneumonia, and Shigella (Mayr et al. 2005a; Kim et al. 2016; Hur et al. 2015; Osorio et al. 2007; Hoseini Shahidi et al. 2019). The administration of these ghost-based vaccines induced specific immune responses and provided protection against subsequent infections in the animal. These desirable features make chemically induced BGs compelling candidates for the development of vaccines against a wide range of infectious diseases.
Chemically induced BGs have promising applications in the field of diagnostics. They can be engineered to present specific surface antigens, derived from various pathogens such as bacteria, viruses, or parasites. BGs carrying these antigens can be used for developing diagnostic assays to detect specific antibodies or immune responses in patient samples, enabling the sensitive and specific detection of infectious agents or disease biomarkers. BGs can also be employed in serological assays, including ELISA or immunoblotting, where they serve as capture agents to detect corresponding antibodies in patient sera. This allows for the diagnosis of infectious diseases and the identification of specific antibody responses. Furthermore, BGs can be incorporated into point-of-care diagnostic devices, facilitating rapid and on-site testing. These devices use BGs as capture agents for target analytes, enabling the direct detection of pathogens or disease markers at the point of patient care. This approach offers the advantage of timely and convenient diagnostics, particularly in resource-limited settings. The integration of bacterial BGs into biosensor platforms enables the detection of a wide range of analytes. The integration of BGs with specific receptors or ligands can serve as highly sensitive and selective sensing elements. This allows for the detection of target molecules, including nucleic acids, proteins, or small molecules, in patient samples. Owing to the unique characteristics of BGs, such as their antigenicity, stability, and cargo-carrying capacity, they can be used in diverse diagnostic strategies for the accurate detection of infectious agents, disease biomarkers, and other analytes of interest. These applications can significantly enhance diagnostic accuracy through the development of efficient and reliable diagnostic tools and improve patient management.
Despite the wide applicability of chemically induced BGs, several challenges impede their use and some aspects require further improvement. One challenge is optimizing the production process to enhance efficiency, yield, and reproducibility. This involves identifying safe and effective chemical agents, determining optimal concentrations and exposure times, and refining purification and characterization methods. Another challenge is improving the stability and storage of BGs. Developing suitable preservation techniques, such as lyophilization or freeze-drying, and identifying stabilizing agents can enhance their long-term stability, facilitating storage, transportation, and distribution. Strategies to enhance the immunogenicity of BGs can be explored, including surface modification techniques to increase antigen loading or incorporation of immune-enhancing molecules like adjuvants or immunomodulatory agents. Additionally, optimizing formulation and delivery methods can improve the immune response elicited by BG-based vaccines. The efficiency of chemically induced BG production may vary depending on bacterial species and the chemical agents used. The stability of BG envelopes is also a concern, as they may have less practical application due to reduced stability and shelf life. Furthermore, using BGs as immune enhancers has challenges, such as achieving a comparable immune response to that of live bacterial infection and the risk of undesired immune responses to immunogenic peptides or proteins resulting from the chemically induced process. Moreover, BGs may not be as efficient in triggering the adaptive immune response as a live bacterial infection, which could lead to inadequate protection against future infections. BGs might still retain some residual components that could trigger an immune response in the host. This could potentially impact their use in vaccine production, as the immune response could be undesirable or interfere with the desired immune response generated by the vaccine. Taken together, addressing these challenges and focusing on areas such as production efficiency, stability, immunogenicity, expansion to Gram-positive bacteria, targeted delivery, improved cargo loading strategies, and conducting clinical studies will promote the development of safe, effective, and versatile BG-based technologies for various biomedical applications.
Conclusion
Chemically induced BGs have emerged as safe and non-replicative vaccine platform. These BGs are created by selectively removing the cytoplasmic content of bacterial cells while preserving the structural integrity of the cell envelope through chemical treatments. This process results in empty bacterial envelopes that possess key advantages for various biomedical applications, including drug delivery and vaccine development. Chemically induced BGs offer several desirable properties, such as safety, effective antigen presentation, immunogenicity, and structural integrity. The key features of chemically derived BGs (Fig. 3) make them highly promising tools for vaccine development, drug delivery, and other biomedical applications. Considering their unique attributes, chemically induced BGs provide a novel and safe avenue for improving disease diagnosis and treatment.
Data availability
The datasets used during the present study are available from the corresponding author upon reasonable request.
References
Abenavoli L et al (2019) Gut microbiota and obesity: a role for probiotics. Nutrients 11. https://doi.org/10.3390/nu11112690
Akira S, Hemmi H (2003) Recognition of pathogen-associated molecular patterns by TLR family. Immunol Lett 85: 85–95. https://doi.org/10.1016/s0165-2478(02)00228-6
Akira S, Uematsu S, Takeuchi O (2006) Pathogen recognition and innate immunity. Cell 124: 783–801. https://doi.org/10.1016/j.cell.2006.02.015
Amara AA, Salem-Bekhit MM, Alanazi FK (2013) Sponge-like: a new protocol for preparing bacterial ghosts. Sci World J 2013: 545741. https://doi.org/10.1155/2013/545741
Anderson RC, Haverkamp RG, Yu PL (2004) Investigation of morphological changes to Staphylococcus aureus induced by ovine-derived antimicrobial peptides using TEM and AFM. FEMS Microbiol Lett 240: 105–110. https://doi.org/10.1016/j.femsle.2004.09.027
Anke H, Zahner H (1978) Metabolic products of microorganisms. 170. On the antibiotic activity of cladosporin. Arch Microbiol 116: 253–257. https://doi.org/10.1007/BF00417848
Awasthi MK et al (2018) Biodegradation of food waste using microbial cultures producing thermostable alpha-amylase and cellulase under different pH and temperature. Bioresour Technol 248: 160–170. https://doi.org/10.1016/j.biortech.2017.06.160
Batah AM, Ahmad TA (2020) The development of ghost vaccines trials. Expert Rev Vaccines 19: 549–562. https://doi.org/10.1080/14760584.2020.1777862
Bunchorntavakul C, Chamroonkul N, Chavalitdhamrong D (2016) Bacterial infections in cirrhosis: a critical review and practical guidance. World J Hepatol 8: 307–321. https://doi.org/10.4254/wjh.v8.i6.307
Cai et al (2010) Intragastric immunization of mice with enterohemorrhagic Escherichia coli O157:H7 bacterial ghosts reduces mortality and shedding and induces a Th2-type dominated mixed immune response. Can J Microbiol 56: 389–398. https://doi.org/10.1139/w10-025
Cao J et al (2018) Identification of fish source Vibrio alginolyticus and evaluation of its bacterial ghosts vaccine immune effects. Microbiologyopen 7: e00576. https://doi.org/10.1002/mbo3.576
Chaudhari AA, Jawale CV, Kim SW, Lee JH (2012) Construction of a Salmonella Gallinarum ghost as a novel inactivated vaccine candidate and its protective efficacy against fowl typhoid in chickens. Vet Res 43: 44. https://doi.org/10.1186/1297-9716-43-44
Chen H et al (2021) Bacterial ghosts-based vaccine and drug delivery systems. Pharmaceutics 13. https://doi.org/10.3390/pharmaceutics13111892
Chiang CL, Sung CS, Wu TF, Chen CY, Hsu CY (2005) Application of superparamagnetic nanoparticles in purification of plasmid DNA from bacterial cells. J Chromatogr B Analyt Technol Biomed Life Sci 822: 54–60. https://doi.org/10.1016/j.jchromb.2005.05.017
Ciesielska A, Matyjek M, Kwiatkowska K (2021) TLR4 and CD14 trafficking and its influence on LPS-induced pro-inflammatory signaling. Cell Mol Life Sci 78: 1233–1261. https://doi.org/10.1007/s00018-020-03656-y
Dai J et al (2020) Production of highly active extracellular amylase and cellulase from Bacillus subtilis ZIM3 and a recombinant strain with a potential application in tobacco fermentation. Front Microbiol 11: 1539. https://doi.org/10.3389/fmicb.2020.01539
de Moreno de LeBlanc A, LeBlanc JG (2014) Effect of probiotic administration on the intestinal microbiota, current knowledge and potential applications. World J Gastroenterol 20: 16518–16528. https://doi.org/10.3748/wjg.v20.i44.16518
Dobrovolskiene N et al (2018) Tumor lysate-loaded bacterial ghosts as a tool for optimized production of therapeutic dendritic cell-based cancer vaccines. Vaccine 36: 4171–4180. https://doi.org/10.1016/j.vaccine.2018.06.016
Ebensen T et al (2004) Bacterial ghosts are an efficient delivery system for DNA vaccines. J Immunol 172: 6858–6865. https://doi.org/10.4049/jimmunol.172.11.6858
Eckhardt S et al (2013) Nanobio silver: its interactions with peptides and bacteria, and its uses in medicine. Chem Rev 113: 4708–4754. https://doi.org/10.1021/cr300288v
Eslaminejad T, Moshafi MH, Hasanpore M, Ayatollahi SA, Ansari M (2022) Evaluation of the anticandidal activity of clotrimazole using Lactobacillus caseie ghosts as biological drug carrier. Daru 30: 67–73. https://doi.org/10.1007/s40199-022-00432-0
Felnerova D et al (2004) T cell-specific immune response induced by bacterial ghosts. Med Sci Monit 10: BR362–BR370
Fernebro J (2011) Fighting bacterial infections-future treatment options. Drug Resist Updat 14: 125–139. https://doi.org/10.1016/j.drup.2011.02.001
Fioramonti J, Theodorou V, Bueno L (2003) Probiotics: what are they? What are their effects on gut physiology? Best Pract Res Clin Gastroenterol 17: 711–724. https://doi.org/10.1016/s1521-6918(03)00075-1
Garbacz K (2022) Anticancer activity of lactic acid bacteria. Semin Cancer Biol 86: 356–366. https://doi.org/10.1016/j.semcancer.2021.12.013
Gitai Z (2005) The new bacterial cell biology: moving parts and subcellular architecture. Cell 120: 577–586. https://doi.org/10.1016/j.cell.2005.02.026
Go G, Lee J, Choi DS, Kim SS, Gho YS (2019) Extracellular vesicle-mimetic ghost nanovesicles for delivering anti-inflammatory drugs to mitigate Gram-negative bacterial outer membrane vesicle-induced systemic inflammatory response syndrome. Adv Healthc Mater 8: e1801082. https://doi.org/10.1002/adhm.201801082
Goldstein DA, Thomas JA (2004) Biopharmaceuticals derived from genetically modified plants. QJM 97: 705–716. https://doi.org/10.1093/qjmed/hch121
Gunnarsson N, Eliasson A, Nielsen J (2004) Control of fluxes towards antibiotics and the role of primary metabolism in production of antibiotics. Adv Biochem Eng Biotechnol 88: 137–178. https://doi.org/10.1007/b99260
Hajam IA, Dar PA, Won G, Lee JH (2017) Bacterial ghosts as adjuvants: mechanisms and potential. Vet Res 48: 37. https://doi.org/10.1186/s13567-017-0442-5
Haslberger AG et al (2000) Activation, stimulation and uptake of bacterial ghosts in antigen presenting cells. J Biotechnol 83: 57–66. https://doi.org/10.1016/s0168-1656(00)00298-4
Hensel A et al (2000) Intramuscular immunization with genetically inactivated (ghosts) Actinobacillus pleuropneumoniae serotype 9 protects pigs against homologous aerosol challenge and prevents carrier state. Vaccine 18: 2945–2955. https://doi.org/10.1016/s0264-410x(00)00107-9
Hinchliffe JD, Parassini Madappura A, Syed Mohamed SMD, Roy I (2021) Biomedical applications of bacteria-derived polymers. Polymers (Basel) 13. https://doi.org/10.3390/polym13071081
Hoseini Shahidi R, Hashemi Tabar G, Bassami MR, Jamshidi A, Dehghani H (2019) The design and application of a bacterial ghost vaccine to evaluate immune response and defense against avian pathogenic Escherichia coli O2:K1 serotype. Res Vet Sci 125: 153–161. https://doi.org/10.1016/j.rvsc.2019.06.001
Huang G et al (2017) Interaction between bacterial cell membranes and nano-TiO(2) revealed by two-dimensional FTIR correlation spectroscopy using bacterial ghost as a model cell envelope. Water Res 118: 104–113. https://doi.org/10.1016/j.watres.2017.04.023
Hur J, Kim CS, Eo SK, Park SY, Lee JH (2015) Salmonella ghosts expressing enterotoxigenic Escherichia coli k88ab, k88ac, k99, and fasa fimbrial antigens induce robust immune responses in a mouse model. Vet Q 35: 125–132. https://doi.org/10.1080/01652176.2015.1029598
Hutchings MI, Truman AW, Wilkinson B (2019) Antibiotics: past, present and future. Curr Opin Microbiol 51: 72–80. https://doi.org/10.1016/j.mib.2019.10.008
Jalava K, Hensel A, Szostak M, Resch S, Lubitz W (2002) Bacterial ghosts as vaccine candidates for veterinary applications. J Control Release 85: 17–25. https://doi.org/10.1016/s0168-3659(02)00267-5
Jawale CV, Chaudhari AA, Lee JH (2014) Generation of a safety enhanced Salmonella Gallinarum ghost using antibiotic resistance free plasmid and its potential as an effective inactivated vaccine candidate against fowl typhoid. Vaccine 32: 1093–1099. https://doi.org/10.1016/j.vaccine.2013.12.053
Ji S et al (2022) Protective immunity against listeria monocytogenes in rats, provided by HCl- and NaOH-induced listeria monocytogenes bacterial ghosts (LMGs) as vaccine candidates. Int J Mol Sci 23. https://doi.org/10.3390/ijms23041946
Kang SR, Nguyen DH, Yoo SW, Min JJ (2022) Bacteria and bacterial derivatives as delivery carriers for immunotherapy. Adv Drug Deliv Rev 181: 114085. https://doi.org/10.1016/j.addr.2021.114085
Kawai T, Akira S (2011) Toll-like receptors and their crosstalk with other innate receptors in infection and immunity. Immunity 34: 637–650. https://doi.org/10.1016/j.immuni.2011.05.006
Kim CS, Hur J, Eo SK, Park SY, Lee JH (2016) Generation of Salmonella ghost cells expressing fimbrial antigens of enterotoxigenic Escherichia coli and evaluation of their antigenicity in a murine model. Can J Vet Res 80:40–48
Korp J, Vela Gurovic MS, Nett M (2016) Antibiotics from predatory bacteria. Beilstein J Org Chem 12: 594–607. https://doi.org/10.3762/bjoc.12.58
Kudela P, Koller VJ, Lubitz W (2010) Bacterial ghosts (BGs)—advanced antigen and drug delivery system. Vaccine 28: 5760–5767. https://doi.org/10.1016/j.vaccine.2010.06.087
Kumar S, Sunagar R, Gosselin E (2019) Bacterial protein toll-like-receptor agonists: a novel perspective on vaccine adjuvants. Front Immunol 10: 1144. https://doi.org/10.3389/fimmu.2019.01144
Kummerer K (2003) Significance of antibiotics in the environment. J Antimicrob Chemother 52, 5–7. https://doi.org/10.1093/jac/dkg293
Langemann T et al (2010) The bacterial ghost platform system: production and applications. Bioeng Bugs 1: 326–336. https://doi.org/10.4161/bbug.1.5.12540
Le Borgne S, Paniagua D, Vazquez-Duhalt R (2008) Biodegradation of organic pollutants by halophilic bacteria and archaea. J Mol Microbiol Biotechnol 15: 74–92. https://doi.org/10.1159/000121323
Lee DJ et al (2008) Generation of safety enhanced Edwardsiella tarda ghost vaccine. Dis Aquat Organ 81: 249–254. https://doi.org/10.3354/dao01964
Lewis PJ (2004) Bacterial subcellular architecture: recent advances and future prospects. Mol Microbiol 54: 1135–1150. https://doi.org/10.1111/j.1365-2958.2004.04343.x
Li Z et al (2021) Chemically and biologically engineered bacteria-based delivery systems for emerging diagnosis and advanced therapy. Adv Mater 33: e2102580. https://doi.org/10.1002/adma.202102580
Liu MA, Wahren B, Karlsson Hedestam GB (2006) DNA vaccines: recent developments and future possibilities. Hum Gene Ther 17: 1051–1061. https://doi.org/10.1089/hum.2006.17.1051
Lubitz P, Mayr UB, Lubitz W (2009) Applications of bacterial ghosts in biomedicine. Adv Exp Med Biol 655: 159–170. https://doi.org/10.1007/978-1-4419-1132-2_12
Ma Y et al (2021) A novel and efficient high-yield method for preparing bacterial ghosts. Toxins (Basel) 13. https://doi.org/10.3390/toxins13060420
Mayr UB et al (2005a) Bacterial ghosts as an oral vaccine: a single dose of Escherichia coli O157:H7 bacterial ghosts protects mice against lethal challenge. Infect Immun 73: 4810–4817. https://doi.org/10.1128/IAI.73.8.4810-4817.2005
Mayr UB et al (2005b) Bacterial ghosts as antigen delivery vehicles. Adv Drug Deliv Rev 57: 1381–1391. https://doi.org/10.1016/j.addr.2005.01.027
Medzhitov R, Janeway Jr C (2000) Innate immune recognition: mechanisms and pathways. Immunol Rev 173: 89–97. https://doi.org/10.1034/j.1600-065x.2000.917309.x
Melssen MM et al (2019) A multipeptide vaccine plus toll-like receptor agonists LPS or polyICLC in combination with incomplete Freund’s adjuvant in melanoma patients. J Immunother Cancer 7: 163. https://doi.org/10.1186/s40425-019-0625-x
Mohammad BT, Al Daghistani HI, Jaouani A, Abdel-Latif S, Kennes C (2017) Isolation and characterization of thermophilic bacteria from Jordanian hot springs: Bacillus licheniformis and Thermomonas hydrothermalis isolates as potential producers of thermostable enzymes. Int J Microbiol 2017: 6943952. https://doi.org/10.1155/2017/6943952
Mu Y, Cong Y (2019) Bacillus coagulans and its applications in medicine. Benef Microbes 10: 679–688. https://doi.org/10.3920/BM2019.0016
Myers C, Hastert MC, Cornwall GA (2022) Host defense functions of the epididymal amyloid matrix. Mol Hum Reprod 28. https://doi.org/10.1093/molehr/gaac038
Osorio M, Bray MD, Walker RI (2007) Vaccine potential for inactivated shigellae. Vaccine 25: 1581–1592. https://doi.org/10.1016/j.vaccine.2006.11.012
Park HJ et al (2016) Characterization of chemically-induced bacterial ghosts (BGs) using sodium hydroxide-induced vibrio parahaemolyticus ghosts (VPGs). Int J Mol Sci 17. https://doi.org/10.3390/ijms17111904
Parvulescu MJS et al (2021) Biomorphic ceramics: synthesis and characterization of preceramic polymer-modified melanin. ACS Biomater Sci Eng 7: 3103–3113. https://doi.org/10.1021/acsbiomaterials.1c00265
Quevedo-Diaz MA et al (2010) Involvement of TLR2 and TLR4 in cell responses to Rickettsia akari. J Leukoc Biol 88: 675–685. https://doi.org/10.1189/jlb.1009674
Rabea S et al (2018) A novel protocol for bacterial ghosts’ preparation using tween 80. Saudi Pharm J 26: 232–237. https://doi.org/10.1016/j.jsps.2017.12.006
Rabea S et al (2022) Immunological characterization of the chemically prepared ghosts of Salmonella Typhimurium as a vaccine candidate. BMC Vet Res 18: 72. https://doi.org/10.1186/s12917-021-03112-4
Riedmann EM, Kyd JM, Cripps AW, Lubitz W (2007) Bacterial ghosts as adjuvant particles. Expert Rev Vaccines 6: 241–253. https://doi.org/10.1586/14760584.6.2.241
Ringo E et al (2020) Probiotics, lactic acid bacteria and bacilli: interesting supplementation for aquaculture. J Appl Microbiol 129: 116–136. https://doi.org/10.1111/jam.14628
Schnare M et al (2001) Toll-like receptors control activation of adaptive immune responses. Nat Immunol 2: 947–950. https://doi.org/10.1038/ni712
Segura A, Ramos JL (2013) Plant–bacteria interactions in the removal of pollutants. Curr Opin Biotechnol 24: 467–473. https://doi.org/10.1016/j.copbio.2012.09.011
Sender R, Fuchs S, Milo R (2016) Revised estimates for the number of human and bacteria cells in the body. PLoS Biol 14: e1002533. https://doi.org/10.1371/journal.pbio.1002533
Senevirathne A, Hewawaduge C, Lee JH (2021) Immunization of chicken with flagellin adjuvanted Salmonella enteritidis bacterial ghosts confers complete protection against chicken salmonellosis. Poult Sci 100: 101205. https://doi.org/10.1016/j.psj.2021.101205
Sheweita SA et al (2022) Bacterial ghosts of Pseudomonas aeruginosa as a promising candidate vaccine and its application in diabetic rats. Vaccines (Basel) 10. https://doi.org/10.3390/vaccines10060910
Steinhagen F, Kinjo T, Bode C, Klinman DM (2011) TLR-based immune adjuvants. Vaccine 29: 3341–3355. https://doi.org/10.1016/j.vaccine.2010.08.002
Szostak MP et al (1996) Bacterial ghosts: non-living candidate vaccines. J Biotechnol 44: 161–170. https://doi.org/10.1016/0168-1656(95)00123-9
Szostak MP et al (1997) Bacterial ghosts as multifunctional vaccine particles. Behring Inst Mitt 191–196.
Taylor PW, Stapleton PD, Paul Luzio J (2002) New ways to treat bacterial infections. Drug Discov Today 7: 1086–1091. https://doi.org/10.1016/s1359-6446(02)02498-4
van Elsland D, Neefjes J (2018) Bacterial infections and cancer. EMBO Rep 19. https://doi.org/10.15252/embr.201846632
Vinod N et al (2015) Generation of a novel Staphylococcus aureus ghost vaccine and examination of its immunogenicity against virulent challenge in rats. Infect Immun 83: 2957–2965. https://doi.org/10.1128/IAI.00009-15
Vinod N et al (2017) A Salmonella typhimurium ghost vaccine induces cytokine expression in vitro and immune responses in vivo and protects rats against homologous and heterologous challenges. PLoS One 12: e0185488. https://doi.org/10.1371/journal.pone.0185488
Waegeman H, Soetaert W (2011) Increasing recombinant protein production in Escherichia coli through metabolic and genetic engineering. J Ind Microbiol Biotechnol 38: 1891–1910. https://doi.org/10.1007/s10295-011-1034-4
Wallace TC et al (2011) Human gut microbiota and its relationship to health and disease. Nutr Rev 69: 392–403. https://doi.org/10.1111/j.1753-4887.2011.00402.x
Witte A et al (1990) Endogenous transmembrane tunnel formation mediated by phi X174 lysis protein E. J Bacteriol 172: 4109–4114. https://doi.org/10.1128/jb.172.7.4109-4114.1990
Witte A, Wanner G, Sulzner M, Lubitz W (1992) Dynamics of PhiX174 protein E-mediated lysis of Escherichia coli. Arch Microbiol 157: 381–388. https://doi.org/10.1007/BF00248685
Won G, Hajam IA, Lee JH (2017) Improved lysis efficiency and immunogenicity of Salmonella ghosts mediated by co-expression of lambda phage holin-endolysin and ɸX174 gene E. Sci Rep 7: 45139. https://doi.org/10.1038/srep45139
Wu X et al (2017) Production of bacterial ghosts from Gram-positive pathogen listeria monocytogenes. Foodborne Pathog Dis 14: 1–7. https://doi.org/10.1089/fpd.2016.2184
Yan X, Liu X, Zhao C, Chen GQ (2023) Applications of synthetic biology in medical and pharmaceutical fields. Signal Transduct Target Ther 8: 199. https://doi.org/10.1038/s41392-023-01440-5
Yang J, Zhao Y, Shao F (2015) Non-canonical activation of inflammatory caspases by cytosolic LPS in innate immunity. Curr Opin Immunol 32: 78–83. https://doi.org/10.1016/j.coi.2015.01.007
Zariri A, van der Ley P (2015) Biosynthetically engineered lipopolysaccharide as vaccine adjuvant. Expert Rev Vaccines 14: 861–876. https://doi.org/10.1586/14760584.2015.1026808
Acknowledgements
We thank Nayeon Kim for helping in the figure. This work was supported by the Ministry of Science and ICT (MSIT), Korea, under the Innovative Human Resource Development for Local Intellectualization Support Program (IITP-2023-RS-2022-00156334) supervised by the Institute for Information and Communications Technology Planning and Evaluation (IITP), and by the National Research Foundation (NRF) grant funded by the MSIT (2021R1A2C1008317).
Author information
Authors and Affiliations
Contributions
Shin-Young Park supervised the manuscript and project and acquired research funding.
Corresponding author
Ethics declarations
Conflict of interest
Author Shin-Young Park declares that she has no conflict of interest. All the authors approved the manuscript.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Park, SY. Chemically induced bacterial ghosts: a novel approach for advancing biomedical applications. Mol. Cell. Toxicol. 19, 657–665 (2023). https://doi.org/10.1007/s13273-023-00389-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13273-023-00389-4