Abstract
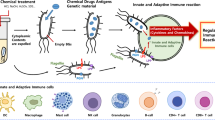
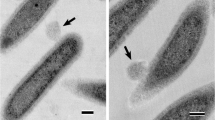
Bacteria are known to release nanometer scale proteoliposomes termed bacterial membrane vesicles (MVs), and it is considered that native and bioengineered MVs would be applicable for development of acellular vaccines and novel drug delivery systems in medical settings. However, important considerations for manufacturing purposes include the varied productivity of MV among bacterial species and strains, as well as endotoxicity levels due to the lipopolysaccharide component. The method for MV induction using glycine described here is simple and provides a solution to these problems. Glycine weakens bacterial peptidoglycans and significantly increases bacterial MV formation, while the relative endotoxin activity of glycine-induced MVs is extremely reduced as compared to that of noninduced MVs. Nevertheless, glycine-induced MVs elicit strong immune responses at levels nearly equivalent to those of noninduced MVs. Taken together, the present method for induction by glycine is convenient for research studies of bacterial MVs and has potential for use in medical applications including vaccine development.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Schwechheimer C, Kuehn MJ (2015) Outer-membrane vesicles from gram-negative bacteria: biogenesis and functions. Nat Rev Microbiol 13(10):605–619
McBroom AJ, Johnson AP, Vemulapalli S, Kuehn MJ (2006) Outer membrane vesicle production by Escherichia coli is independent of membrane instability. J Bacteriol 188(15):5385–5392
Turnbull L, Toyofuku M, Hynen AL, Kurosawa M, Pessi G, Petty NK, Osvath SR, Cárcamo-Oyarce G, Gloag ES, Shimoni R (2016) Explosive cell lysis as a mechanism for the biogenesis of bacterial membrane vesicles and biofilms. Nat Commun 7(1):1–13
Toyofuku M (2019) Bacterial communication through membrane vesicles. Biosci Biotechnol Biochem 83(9):1599–1605
Hirayama S, Nakao R (2020) Glycine significantly enhances bacterial membrane vesicle production: a powerful approach for isolation of LPS-reduced membrane vesicles of probiotic Escherichia coli. Microb Biotechnol 13(4):1162–1178
Vollmer W, Blanot D, De Pedro MA (2008) Peptidoglycan structure and architecture. FEMS Microbiol Rev 32(2):149–167
Hammes W, Schleifer K, Kandler O (1973) Mode of action of glycine on the biosynthesis of peptidoglycan. J Bacteriol 116(2):1029–1053
Rembacken B, Snelling A, Hawkey P, Chalmers D, Axon A (1999) Non-pathogenic Escherichia coli versus mesalazine for the treatment of ulcerative colitis: a randomised trial. Lancet 354(9179):635–639
Kruis W, Frič P, Pokrotnieks J, Lukáš M, Fixa B, Kaščák M, Kamm M, Weismueller J, Beglinger C, Stolte M (2004) Maintaining remission of ulcerative colitis with the probiotic Escherichia coli Nissle 1917 is as effective as with standard mesalazine. Gut 53(11):1617–1623
Behnsen J, Deriu E, Sassone-Corsi M, Raffatellu M (2013) Probiotics: properties, examples, and specific applications. Cold Spring Harb Perspect Med 3(3):a010074
Ichinohe T, Watanabe I, Ito S et al (2005) Synthetic double-stranded RNA poly(I:C) combined with mucosal vaccine protects against influenza virus infection. J Virol 79(5):2910–2919
Hirayama S, Nakao R (2021) Intranasal vaccine study using Porphyromonas gingivalis membrane vesicles: isolation method and application to a mouse model. Methods Mol Biol 2210:157–166. https://doi.org/10.1007/978-1-0716-0939-2_15
Smith PK, Krohn RI, Hermanson GT, Mallia AK, Gartner FH, Provenzano M, Fujimoto EK, Goeke NM, Olson BJ, Klenk D (1985) Measurement of protein using bicinchoninic acid. Anal Biochem 150(1):76–85
Acknowledgments
The authors wish to thank Michiyo Kataoka for technical assistance with the transmission electron microscope observations. This study was supported by JSPS KAKENHI (JP18K15160, JP19K22644, JP20K18492, JP20H03861) and the Japan Agency for Medical Research and Development (AMED) (JP18fk0108124).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Hirayama, S., Nakao, R. (2022). Glycine Induction Method: Effective Production of Immunoactive Bacterial Membrane Vesicles with Low Endotoxin Content. In: Bidmos, F., Bossé, J., Langford, P. (eds) Bacterial Vaccines. Methods in Molecular Biology, vol 2414. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-1900-1_13
Download citation
DOI: https://doi.org/10.1007/978-1-0716-1900-1_13
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-1899-8
Online ISBN: 978-1-0716-1900-1
eBook Packages: Springer Protocols