Abstract
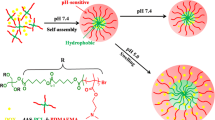
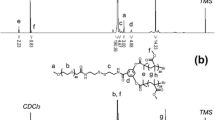
The four-arm star copolymers poly(methacrylic acid)-poly(2-hydroxyethyl methacrylate-disulfide~)-poly(poly(ethylene glycol) methyl ether methacrylate) (4AS-PMAAx-(PHEMA-SS~)y-PPEGMAz) with four different block ratios were synthesized and could self-assembled into cross-linked polymer micelles for the exploration of the structure-property relationship. The cross-linked polymer micelles in aqueous solution had low critical micelle concentration (CMC) values (1.9-4.6 mg/L), which exhibited better stability than non-cross-linked micelles. The CMC value decreased with the increase of the length of inner PMAA core and hydrophobic PHEMA cross-linked middle layer. The blank and doxorubicin (DOX)-loaded micelles with different block ratios were prepared by dialysis with the particle sizes of 120-240 nm. The longer inner PMAA core and cross-linked middle layer enhanced the drug loading content (DLC) results and led to relatively bigger particle sizes of polymer micelles. The in vitro DOX release data revealed that DOX-loaded micelles had low DOX cumulative release percentages of 18-37% after 110 h at pH 7.4, but up to 83-90% when introducing reductant GSH at pH 5.0. The 4AS-PMAA21.2-(PHEMASS≈)13.1-PPEGMA5.1 micelles with the longest PMAA core had the largest cumulative release of 90.1%. The DOX release process and mechanism of the micelles at different conditions fitted well with the semi-empirical equation. Overall, the results demonstrated that the block ratios and pH/redox-responsiveness of these four-arm star copolymers could be well-controlled and their self-assembled cross-linked micelles as anticancer drug carrier system could be improved by optimizing the different ratios.

Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
A. N. Lukyanov and V. P. Torchilin, Adv. Drug Deliv. Rev., 56, 1273 (2004).
C. Sun, X. Li, X. Du, and T. Wang, Int. J. Biol. Macromol., 112, 65 (2018).
L. Xiao, L. Huang, F. Moingeon, M. Gauthier, and G. Yang, Biomacromolecules, 18, 2711 (2017).
Y. Deng, H. Yuan, and W. Yuan, J. Mater. Chem. B, 7, 286 (2019).
Y. Q. Yang, W. J. Lin, L. J. Zhang, C. Z. Cai, W. Jiang, X. D. Guo, and Y. Qian, Macromol. Res., 21, 1011 (2013).
D. Xiong, N. Yao, H. Gu, J. Wang, and L. Zhang, Polymer, 114, 161 (2017).
B. Pandey, N. G. Patil, G. S. Bhosle, A. V. Ambade, S. S. Gupta, Bioconjugate Chem., 30, 633 (2019).
Q. Chen, F. Han, C. Lin, X. Wen, and P. Zhao, Polymer, 146, 378 (2018).
T. Sim, S. M. Han, C. Lim, W. R. Won, E. S. Lee, Y. S. Youn, K. T. Oh, Macromol. Res., 27, 795 (2019).
N. Wang, X.-C. Chen, R.-L. Ding, X.-L. Yang, J. Li, X.-Q. Yu, K. Li, and X. Wei, RSC Adv., 9, 2371 (2019).
X. Ma, J. Liu, L. Lei, H. Yang, and Z. Lei, J. Appl. Polym. Sci., 136, 47946 (2019).
Z. Zhou, G. Li, N. Wang, F. Guo, L. Guo, and X. Liu, Colloid. Surface. B, 172, 136 (2018).
Z. Su, Y. Xu, Y. Wang, W. Shi, S. Han, and X. Shuai, Biomater. Sci., 7, 3821 (2019).
X. Shan, J. Mao, M. Long, K. S. Ahmed, C. Sun, L. Qiu, and J. Chen, J. Appl. Polym. Sci., 136, 47854 (2019).
Y. Li, K. Xiao, W. Zhu, W. Deng, K. S. Lam, Adv. Drug Deliv. Rev., 66, 58 (2014).
S. Mura, J. Nicolas, and P. Couvreur, Nat. Mater., 12, 991 (2013).
L. Bu, H. Zhang, K. Xu, B. Du, C. Zhu, and Y. Li, Drug Deliv., 26, 300 (2019).
L. Li, D. Li, M. Zhang, J. He, J. Liu, and P. Ni, Bioconjugate Chem., 29, 2806 (2018).
Y. Qu, B. Chu, X. Wei, M. Lei, D. Hu, R. Zha, L. Zhong, M. Wang, F. Wang, and Z. Qian, J. Control. Release, 296, 93 (2019).
X. Sang, Q. Yang, G. Shi, L. Zhang, D. Wang, and C. Ni, Mater. Sci. Engi. C, 91, 727 (2018).
Z. Guo, K. Zhao, R. Liu, X. Guo, B. He, J. Yan, and J. Ren, J. Mater. Chem. B, 7, 334 (2019).
B. Ma, W. Zhuang, Y. Wang, R. Luo, and Y. Wang, Acta Biomater., 70, 186 (2018).
Z. Zhang, Q. Qu, J. Li, and S. Zhou, Macromol. Biosci., 13, 789 (2013).
Y. Q. Yang, B. Zhao, Z. D. Li, W. J. Lin, C. Y. Zhang, X. D. Guo, J. F. Wang, L. J. Zhang, Acta Biomater., 9, 7679 (2013).
Y. Q. Yang, W. J. Lin, B. Zhao, X. F. Wen, X. D. Guo, L. J. Zhang, Langmuir, 28, 8251 (2012).
Y. Q. Yang, X. D. Guo, W. J. Lin, L. J. Zhang, C. Y. Zhang, and Y. Qian, Soft Matter, 8, 454 (2012).
Y.-N. Xue, Z.-Z. Huang, J.-T. Zhang, M. Liu, M. Zhang, S.-W. Huang, and R.-X. Zhuo, Polymer, 50, 3706 (2009).
J. Zeng, P. Du, L. Liu, J. Li, K. Tian, X. Jia, X. Zhao, and P. Liu, Mol. Pharm., 12, 4188 (2015).
Tian, X. Jia, X. Zhao, and P. Liu, Mol. Pharm., 13, 2683 (2016).
P. L. Ritger and N. A. Peppas, J. Control. Release, 5, 23 (1987).
J. Siepmann and N. A. Peppas, Adv. Drug Deliv. Rev., 64, 163 (2012).
W. Lin, S. Nie, Q. Zhong, Y. Yang, C. Cai, J. Wang, and L. Zhang, J. Mater. Chem. B, 2, 4008 (2014).
P. L. Ritger and N. A. Peppas, J. Control. Release, 5, 37 (1987).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher’s Note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Acknowledgments: The authors gratefully acknowledge the financial support from the National Natural Science Foundation of China (No. 51873042, No. 21808039), the Natural Science Foundation of Guangdong Province (No. 2018A030310525), the Science and Technology Planning Project of Guangdong Province (No. 2017B090915004), and the Guangdong Provincial Key Laboratory of Advanced Coatings Research and Development (No. 2017B030314105).
Rights and permissions
About this article
Cite this article
Huang, Y., Li, Y., Tang, Z. et al. Dual-Responsive Cross-Linked Micelles from Amphiphilic Four-Arm Star Copolymers with Different Block Ratios for Triggering DOX Release. Macromol. Res. 28, 762–771 (2020). https://doi.org/10.1007/s13233-020-9094-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13233-020-9094-0