Abstract
Respiratory infections are quite challenging due to their complexity in ailments and composition of viral genetic material and their rate of proliferation. In particular, the eradication of viral illness is still a concern, irrespective of advancements in prevention and remedial procedures. The nature of the viral particle with the possibility of rapid transmission is prone to attach on the deposited surface for days together. This antigen expulses due to sneezing or coughing resulted in multiphase turbulent flow, contaminates the surroundings and is carried away by simple touch or inhalation and find newer hosts for instance, SARSCoV-2 aerosols remain viable for about an hour leading to infection. The present review focuses on the remedial aspects of respiratory infections through a knowledge-based approach towards nanosystems. The complete understanding of standard antiviral drugs and the remodelling of these drugs through nanosystems still is the need of the hour. The genetic material and epidemiology of viral antigen, help in redefining standard drugs along with nanocarriers to achieve more feasible and hour-based approach. The main goal of this review is to elaborate on the repurposing of existing standard antiviral drugs and ways to accelerate their mode of action to promote a feasible and hour-based approach. The consolidated three-dimensional approaches aimed at sustained, targeted and optimized levels of drug concentration in the circulating system along with bioactive nanocarriers which could effectively pass the cell membrane were reported. The platforms for nanomaterial evolution depend on nature of source, size, structure, and their unique functionalities (Stable, speedy, and long-lasting recovery procedure). However, the research activities and literature on coronavirus have been overwhelming but the information on the sustainability of nanotherapy in SARS-CoV-2 is still in the developmental stage. Hereby, the clinical aspects of SARS-CoV-2 and the eradication strategy developed for antiviral infections through nanotechnology will pave the way ahead for treating upcoming new variants or other pandemics.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
COVID-19 is a pandemic disease spread through human–human transmission. As per WHO to date, in India about ~ 45 million COVID-19-positive cases have been reported with about 0.5 million deaths. (World Health Organization 2023). The epidemiology of COVID-19 involves the transmission of viral antigens among humans via touching or handling the surfaces of the aerosols (World Health Organization 2014). Coughing, sneezing and talking cause the transmission of droplets with viral particles among individuals (Liu et al. 2020). Larger droplets of pathogens settle on the host surface and are carried through dried microdroplets (~ 100 μm). More focus to be provided on these microdroplets for eradicating respiratory infections on the larger scale. The particle size distribution may shift even lower when the airborne droplets are evaporated to form droplet nuclei. The droplet nuclei formation is dependent on the ambient temperature and humidity (Seminara et al. 2020). Sustainable technology may be developed to fight these microdroplets to address both environmental hygiene and human well-being. One such sustainable process that attracted researchers in designing nanomaterials from biopolymers. These materials of the nanometre scale attribute to significant features such as their high surface area, increased volume ratio, strength, and reactivity conductance. Varied forms of particles, capsules, composites, sensors and scaffolds were purposed for imaging, tracking, tracing, and drug delivery mechanisms. In the real-time approach, the role of nanomaterials is reviewed against inflammatory responses or post-stages of viral infections (Banerjee et al. 2016). Exploring current clinical trials for COVID-19, the number of registries such as intervention, methodology, patient groups, and outcome measures have been considered (Li et al. 2020). The swiftness in genomic sequencing of SARS-CoV-2 in comparison with MERS-CoV, SARS-CoV, and Mac Viruses is greater (Lythgoe and Middleton 2020). The conserved regimes of the viral enzymes involved in pathogenic coronavirus were also investigated (Lu et al. 2020).
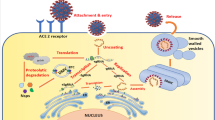
The mode of viral entry, its replication inside the host, and release through exocytosis are represented in Fig. 1. The interventional studies focused on preventive strategies and treatment directions among COVID-19 patients. Chloroquine, lopinavir, ritonavir, favipiravir, dexamethasone, tocilizumab, remdesivir, hydroxychloroquine and ibuprofen are prescribed for treatment practices (Ledford 2020a). Severe SARS CoV-2 patients are suggested with hydroxychloroquine with sofosbuvir and ledipasvir (Wang et al. 2020).
In some cases, interferon-alpha 2a is delivered through a nebulizer for clearing respiratory conjunctions. The interferon,α1b with placebo eye drops were used for eye infections (Haasnoot et al. 2007). Antimalarial drugs such as dihydroartemisinin with piperaquine are restricting the replication of SARS CoV-2 (Barbaro et al. 2005). Methylprednisolone also acts as an immunosuppressant (Kamal and Jusko 2004). Cytokine removal, convalescent plasma-based therapy, monoclonal antibodies, antiangiogenic, and microbiomes are also employed for COVID-19 therapeutics (Zhang et al. 2008). The immunogenicity of SARS-CoV-2 is achieved through cell-mediated and humoral immunity. The impact of drugs on therapeutic systems is explored with the possibility of understanding viruses through molecular and cellular components. It leads to the development of multi-functionalized carriers or vehicles based on the localization and targeting of ligands for drug delivery systems in vitro (Yoo et al. 2019). Nanotherapeutic systems are more reproducible, easy to handle, non-toxic, stress-free repositories and cost-effective to treat the full range of viral infections in the heterogeneous populations (Davis et al. 2008). The sustainable and unconventional nanodelivery system with nil or less toxicity, biodegradability, and efficient excretion mechanism are in use for pulmonary application. Regulating technological advancement and evaluating the potential pitfalls are essential in establishing the nanomedicine as a therapeutic choice in anti-COVID-19 treatment. The current review focuses on the nanotherapeutics options for delivering antiviral drug-loaded carriers for treating respiratory infections and their symptoms.
Epidemic treatment frequency has been brought out through homeopathy, allopathy, and ayurvedic medicine (Natarajan et al. 2021). Besides, the National Institute of Siddha and Central Council for Research in Siddha from India recently adopted effective strategies to prevent the virus from spreading, using natural sources to regulate the mortality rate (Maideen 2021). Traditional Indian medicines developed as an integrated approach for managing COVID-19 by incorporating the Siddha formulations for profound preventive and adjuvant effects. However, these studies need to be validated using large clinical trials (Teixeira 2021). In some places, the arsenicum album is a preventative medicine against primary infection (Gautret et al. 2020).
The biopharmaceutical industry has proceeded with developing and distributing vaccines throughout the world. Social impressions offset vaccines and their risks over time. Vaccines are developed and circulated with clinical efficacy by addressing the immune response to protect the medical challenges. Multiple vaccines are administered across countries (Agrawal 2021). Vaccines such as BNT162 / mRNA-1273 / ChAdOx1nCoV-191 / Ad26.CoV2.5 / Sputnik /BBIBP-CorV / PiCoVacc / Covaxin / NVX-CoV2373 /AS03 /Ad5-nCoV / ZyCoV-D / CVnCoV / INO-4800 / ZF 2001 have been manufactured in coordination with the private agencies, governments, researchers, clinicians and the pharmaceutical companies.
The challenges of this nanodelivery system revolve around the proclamation of uniform distribution of the aerosol particles with a complete ventilation system. This standard approach depends on the factors influencing the quantification and inhibition of virulence activity of viral infectious agents (Gunathilake et al. 2021). The complete eradication of infection depends on the source of the material and its physiochemical and biological characteristics based on its functional moiety.
Nanodrug carrier towards infectious diseases
In the case of conventional medications, the solubility, adsorption, and resistance behaviour of conventional medications seem to be a concern for the human health system (Yang et al. 2020). As an alternative solution, the nanotherapeutics method for treating viral infections has evolved during the rise of influenza, hepatitis and other mosaic diseases. Nanosystems aim to increase drug activity efficiency, dosage minimization, administration, and sustainability (Grenha et al. 2005). The main advantages of these nanosystems are their size, enabling them to have site-directed action and increased availability of surface area. These nanoformulations are tenable to surface functionalization, ensuring cellular activity across the membrane (Kumar et al. 2017). The encapsulation of drugs in nanoparticles and the formation of stable structures are the essential elements to make them a potential antiviral therapeutic tool (Müller et al. 2000). Besides this, the repurposing of drugs, dosing pattern, improved stability, and retention time remain exceptional (Courrier et al. 2002). The site-targeted delivery in host systems (cells, tissues, and their sub-cellular locations) depends on the interaction of nanoparticle–functional sites of the drug moieties.
Antiviral nanotherapeutics
Antiviral drugs directed for targeted treatment procedures may be less specific and face rapid clearance in the host system. Studies evaluated to increase the action of drugs against the stressed immune system through the nanoparticle carriers (Molina et al. 2020). Nanoparticles are explicitly designed depending on the drug specificity and the biodegradability capacity. For example, Dexamethasone, a low-cost steroid used for treating severe respiratory complications ensures a low mortality rate among populations (Ledford 2020b). The therapeutic utility in coronavirus treatment involves numerous drugs specific to symptomatic treatments, as listed in Table 1. In recent times, apart from drugs like lopinavir-ritonavir and ribavirin, baloxavir marboxil, favipiravir, remdesvir, umifenovir, chloroquine, hydroxychloroquine, azithromycin, corticosteroids, losartan, statins, interferons, nitric oxide, epoprostenol, tocilizumab, siltuximab, sarilumab anakinra, and ruxolitinib were repurposed for the treatment of COVID-19 (Drożdżal et al. 2020). The research on the future therapeutic regimens extended based on molecular and genomic discoveries. In addition, the antiviral drugs were chosen and repurposed to form lopinavir, ritonavir, ribavirin, baloxavir marboxil, favipiravir, remdesvir, and umifenovir (arbidol) specific to COVID antigen. Extension of the therapeutic system also included potential drugs such chloroquine, hydroxychloroquine, azithromycin, corticosteroids, losartan, statins, interferons, nitric oxide, and epoprostenol specific to COVID-19 regimen and do not have antiviral property (Wu et al. 2020).
Antiviral nanoprevention
In case of prevention strategies for viral activity, the interventions such as vaccines (COVID-19/aAPC /Ad-nCoV), antiviral (Darunavir/Cobicistat/Umifenorin), antimicrobial (Chloroquine/Placebo) (Cortegiani et al. 2020), personal protective equipment (Gastroscope/Medical masks/N95 respiratory Mask) and others (Nitric oxide/PUL-042, rehabilitation, and lung segment service) have been used. The development of the pandemic flu (HINI/09) helped in devising strategy for COVID-19 treatment (Shi et al. 2007a). The research groups at the National Institute of Allergy and Infectious Diseases have initiated a vaccine development from a modified mRNA vaccine against COVID-19 encoding viral spike protein. The vaccination process seems fruitful over the classical and traditional treatments to bypass the conventional vaccines (Fang et al. 2022). The ultimate goal of the preventive strategies is to overcome the factors such as weak immunogenicity, intrinsic in vivo instability, toxicity, and repeated dosage patterns.
The host immunity may be enhanced with the peptide subunit vaccines as adjuvants incorporated with novel delivery vehicles such as liposomes, polymers, virosomes, etc. (Sarkar et al. 2019) to fight against SARS-CoV-2. Some of the adjuvants like monophosphoryl lipid A (MPLA) and cytosine-phosphorothioate-guanine oligodeoxynucleotides (CpG ODN) are used as alternative therapies in COVID-19. In the case of liposomes, 1, 2-dioleoy-l-3-trimethylammoniumpropane (DOTAP) was used for specificity with antigen-presenting cells enabling the CD4 + and CD8 + immune response. Specifically, viral proteins such as virosomes are used as subunit vaccines which are highly degradable, nontoxic, and do not form antiphospholipid antibodies (Schwendener 2014). In comparison to liposomes, virosomes enhance the active substances from proteolytic degradation. Additionally, it is also reported that RNA vaccines are formulated using lipid nanoparticles to increase their stability and immune response (Bolhassani 2023).
The most important prevention techniques highlighted based on person-to-person transmission of SARS-CoV-2 by defining the symptoms of COVID-19 patients and their host immune response towards SARS-CoV-2.
Technical advancements in nanomaterials for antiviral treatments
The emerging field in the delivery of drugs through nanotechnology remains inevitable. The development of nanomaterials involved varied formulation techniques that kept evolving. The refinement of experimentation is mainly engaged in enhancing the bulk production of nanomaterials (Sharma et al. 2015). The top-down and bottom-up approaches are employed to prepare nanomaterials, as shown in Fig. 2. Solvent diffusion and emulsification are the most commonly used method for preparing defined nanomaterials. Polymeric nanomaterials for antiviral treatments endure promising material formulated using solvent evaporation (Li et al. 2004). Approaches involved in designing the nanomaterials are optimized based on the following factors: carrier material, mechanism of drug release, targeting ability, administration route, drug choice, delivery duration, and biocompatibility nature.
The regeneration and repurposing of existing antiviral drugs targeting lung health have contributed dosage lot in the treatment of SARS-CoV-2. For example, remdesivir through oral delivery has less bioavailable compared to nebulized drugs to prevent the spreading of the virus from the lungs (Li et al. 2021). The nebulization of nanodrugs have achieved 100% recovery compared to intravenous administration; remained a boon for health centre and provided stress-free experience while self-administration during COVID-19 pandemic.
Formulation platforms
Nanomedicine fascinates the medical fraternity with diagnosis and treatment. The underlying principle of the interaction system of particle–drug complex, particle–host, and particle–drug–host complexes in targeted drug delivery systems determines the efficiency of therapeutics (Grove and Marsh 2011). The size acts as a determinant for antiviral, antioxidant, and antimicrobial treatments for the active drug delivery system, as listed in Table 2. The in-depth analysis of nanocarriers for antiviral therapy and their breakthroughs is expanding. The nanocarriers with surface modifications play a major role in improving conventional antiviral drugs and enhanced drug delivery systems. The surface functionalization or coatings retain the uniform dispersity and conserve the drug–carrier molecule properties (Mitchell et al. 2021). Nanocarrier’s surface functional moiety may be subjected based on the hydrophilicity and hydrophobicity of the drug. The surface charge of the nanocarriers are governed by the addition of drug molecule or carrier molecule (polymer/lipids, etc.) thereby influencing its cell viability, biodegradability, drug encapsulation and drug elimination. For example, anionic liposomes were formulated to enhance the entrapment efficiency, whereas the polymeric nanocarriers prefers positive charges to induce phagocytosis for effective antiviral activity (Maus et al. 2021). The surface properties of the nanoparticles are inevitable for determining the mode of action, delivery route and the recovery rate. The research findings have confirmed that the application of these carrier-coated nanosystems could block specific viral proteins and disrupt the replication process in the infected cells (Maus et al. 2021).
Evolution of nanomaterial solutions
The functionalized nanomaterial improved efficacy with reduced side effects. Innovative delivery systems distribute numerous therapeutic agents (drug/gene) in the different developmental stages through productive ways. Antiretroviral therapy, gene therapy, and immunotherapy are still used for sustainable delivery (Teixeira 2020; Shi et al. 2007b). The gold nanoparticles are loaded using drugs such as SDC-1721, fullerene, and dendrimers. Immunotherapy using mannose-targeted polyethyleneimine polymers encoded with plasmid DNA for targeted delivery (Yao et al. 2020; Ioerger et al. 2013). In recent times, fabricated nanomaterials using both inorganic and organic constituents for antiviral treatments. In the case of inorganic materials, silica and gold nanoparticles evolved for innovative delivery systems (Fig. 3). The amino group functionalized and carboxymethylated nanoparticles were used for promising biomedical applications (Diebold et al. 1999). Currently, researchers are exploring a novel nanocarrier platform for the sustainable delivery of antiviral drugs. Achievable tunable size and surface functionalization for antiviral treatments prepared using polymeric nanoparticles, for example, polyethylene glycol (Patil et al. 2009). WHO and FDA agencies approved polylactide, polyglycolide, and poly (lactide-co-glycolides) for the nano-preparations for antiviral treatments. Hydrophilic polymers are used as nanocapsules or nanospheres to decrease the susceptibility to body functions. They defer the uptake of phagocytosis by prolonging the drug life (Fig. 3) (Pang 2015). Additionally, the nanoforms of micelles and dendrimers are also involved in treating viral infections.
Polymeric nanomaterials are mostly natural which portrays various advantages of being inert and biodegradable material; rapid adsorption and trouble free elimination for improved patient compliance. These materials have improved chemical flexibility and functional charge properties leading to host–drug interaction (Han et al. 2018). The intranasal delivery of these polymeric nanoparticles has led to the preferred condition for therapeutic moieties. Along with polymeric organic matter (chitosan/starch/cellulose, etc.), lipids (liposomes), niosomes and dendrimers have convenient modes of delivery, degree of functionalization, side chain rotation and biocompatibility (Chenthamara et al. 2019).
In the case of inorganic materials (gold/zinc) and carbon quantum dots, the theranostic approach has reached a pinnacle with maximum adaptability and drug diffusibility in the targeted delivery. As a novel approach, researchers have developed virus-like nanoparticles (Self-assembling protein) from viral capsids without genetic materials for antiviral treatment (Xue et al. 2022). This has enhanced the immune response and with minimum enzymatic degradation. On the other hand, siRNA has been developed along with polymers or lipids for inhibition of RNA interference on SARS-CoV-2. Similarly, mRNA-based vaccines prepared using lipid nanoparticles were developed as beneficial, and in preparation for the future pandemic (Wilson and Geetha 2022).
Hybridization of mesoporous materials for antiviral treatment
Mesoporous nanomaterials are known for their enhanced pore size, volume, surface charge functionalization and degradation profile which makes them exciting for therapeutics (Lembo and Cavalli 2010). In addition, the flow property in the porous material for adsorption or diffusion is greatly influenced by its pore structure, structural network, and physical characteristics. The crucial part of drug encapsulation is dependent on (a) liquid penetration into the porous material (Baas et al. 1994), (b) molecular and bulk properties of the liquid and (c) the surface property of the porous medium. The large surface area with high pore volume of mesoporous particles possesses unique drug-delivery properties and maintains constant drug circulation in the blood. The design of the mesoporous nanoparticles is crucial for localized delivery in lymph nodes and other pulmonary infections (Bharti et al. 2015). In recent times, many formulations have been carried out in combination with natural bioactive compounds in the preparation of mesoporous nanoparticles for loading antiviral drugs.
Challenges for nano-based delivery
The most significant problem in the use of nanosystems is reduced permeability through biological membranes. After the delivery to the host systems, cells undergo endocytosis pathways, limiting the uptake and utility of nanoparticles (Fig. 4). In some cases, the uptake of therapeutic agents is limited through non-specific cells such as macrophages and organs of the reticuloendothelial system (e.g., liver and spleen) (Singh et al. 2017). However, surface modification, dosage administration, and biodistribution play a significant role, whereas toxicity remains crucial. The occurrence of toxicity depends on the type of nanoparticle, accumulation system, circulation time, and excretion phenomenon. These efforts induce acute pulmonary toxicity, renal toxicity, hepatotoxicity, neurotoxicity, and genotoxicity (Panyam and Labhasetwar 2003).
The structural moiety of SARS-CoV-2 keep changing and the variants of interest keeps evolving such as XBB.1.5, XBB.1.16 and EG.5. This remain the most challenging among healthcare units in developing drugs or vaccines sensitivity towards the variants (Sharma et al. 2023). The side effects of acute respiratory distress syndrome (ARDS) in COVID-19 patients lead to associated complications in cardiovascular regions such as arrhythmias. The recovery rate and the treatment regimen vary among normal patients and patients in immunocompromised situations such as Type 2 diabetes, hypertension, hyperlipidemia, and hypothyroidism.
Biodegradation and elimination of nanoforms in antiviral treatments
The nanoparticle preparation and its application process are designed based on its biodegradability processes. The biodegradation process in the host system determines the sustained release and biodistribution profiles. This biodegradation profile depends on polymer composition, hydrophobic–hydrophilic sites, size, and molecular weight (Kumar Malik et al. 2007). Till date, the evidence about the nanoparticle degradation in the host system remains untangled. Non-biodegradable nanosystems exit the cell through an exocytosis system. The ionic strength of the nanosystem seems foremost in excretion from the body. In terms of cationic particles, they tend to agglomerate and seem to retard their elimination within the host. Whereas anionic particles happen to be eliminated more rapidly from the system (Jain et al. 2008).
Similarly, particle size is a crucial factor for the excretion system. The mutation rate remains complicated in coronavirus, and they tend to infect and reside in cells such as macrophages (Fig. 4). The nanoparticles are prepared and modified by increasing their affinity towards macrophages (Meuleman et al. 2012). Continuous improvement in the functionalization of typical drugs with improved specificity requires a stringent strategy. Depending on the size and functional groups, the release mechanism are varied such as sustained, on-site directed, pulsative, or simple diffusion mechanisms (Patra et al. 2018).
The flow of viral antigen activity starts from attachment to penetration and is further exposed and preceded by RNA-directed RNA synthesis (Modrow et al. 2013). Later, it assembles and releases the intact virion (Fig. 4). Virus-nano drugs complex seems to undergo four different ways of clearance, such as pulmonary (lungs), renal (kidney), hepatic (liver), and RES clearance (spleen) mechanism (Milovanovic et al. 2017). The size of the nanosystems plays a significant role in the excretion. The nanoparticles are excreted in the bloodstream and not engulfed by the phagocytic cells when administered intravenously. These fundamental behaviours of novel viruses explain the imperative need for nanosystems for antiviral therapeutic applications.
The biodegradation of natural materials are irreversible, leading to change in mechanical properties, damage, and depolymerisation. The inert materials inside the host systems are monitored for the degree of hydrolysis and degradation of enzymes (Kamaly et al. 2016). The interaction with water molecule initiates the material decomposition, and vary with the nature of particles (amorphous and crystallinity).
Management of post-respiratory infections
Patients who acquired COVID-19 may be prone to post-infection complications and drug- induced side effects. The immune response of these sufferers play a crucial role in the management of post-complications. Most common symptoms rely on breathing complications and the stats of the lung functions get reduced (Desai et al. 2022). Patients with immunocompromised disorders need regular monitoring and follow-ups for betterment. The dose percentage of vaccine administration by the common people has reached more than half of the whole population across the globe. The effects of these vaccine administered are also under research by health professionals and clinicians (Yarlagadda et al. 2022). Overall treatment systems move towards having a better environment with the development of new and novel drugs (with changes in size and dimension), effective for complete recovery and able to fight against upcoming new variants or other pandemic infections.
Conclusion
Severe complications of pneumonia occurred in late 2019 in Wuhan, China. This review has emphasized the importance of nanotechnology in COVID-19 interventions and treatment measures through sustainable technology. A complete understanding of the various classical nanoplatforms to treat viral infections remains promising for exploring the new model for treating SARS-CoV-2 disorders. Moreover, the nanomaterial specificity, the size and determinants of targeting help reach the site of infection and inflammation for a speedy recovery. The pyramidal response involving drug, host, and nanomaterials plays a significant role in nanotherapeutics. In the future, nanosystems and their treatments will help expand our study for viral therapeutics with zero or significantly less toxicity towards the ecosystem. Thereby, the value-added therapeutic systems helps the clinical fraternity to facade other new disease or pandemic infections.
References
Agrawal G (2021) On pins and needles: tracking COVID-19 vaccines and therapeutics
Baas J, Senninger N, Elser H (1994) Das retikuloendotheliale System. Eine Ubersicht über Funktion, Pathologie und neuere Messmethoden [The reticuloendothelial system. An overview of function, pathology and recent methods of measurement]. Zeitschrift fur Gastroenterologie 32(2):117–123
Banerjee A, Qi J, Gogoi R, Wong J, Mitragotri S (2016) Role of nanoparticle size, shape and surface chemistry in oral drug delivery. J Control Release 238:176–185. https://doi.org/10.1016/j.jconrel.2016.07.051
Barbaro G, Scozzafava A, Mastrolorenzo A, Supuran CT (2005) Highly active antiretroviral therapy: current state of the art, new agents and their pharmacological interactions useful for improving therapeutic outcome. Curr Pharm Des 11(14):1805–43
Bharti C, Nagaich U, Pal AK, Gulati N (2015) Mesoporous silica nanoparticles in target drug delivery system: a review. Int J Pharma Investig 5(3):124–133. https://doi.org/10.4103/2230-973X.160844
Bolhassani A (2023) Lipid-based delivery systems in development of genetic and subunit vaccines. Mol Biotechnol 65(5):669–698. https://doi.org/10.1007/s12033-022-00624-8
Chenthamara D, Subramaniam S, Ramakrishnan SG, Krishnaswamy S, Essa MM, Lin FH, Qoronfleh MW (2019) Therapeutic efficacy of nanoparticles and routes of administration. Biomater Res 23:20. https://doi.org/10.1186/s40824-019-0166-x
Cortegiani A, Ingoglia G, Ippolito M, Giarratano A, Einav S (2020) A systematic review on the efficacy and safety of chloroquine for the treatment of COVID-19. J Crit Care 57:279–83
Courrier HM, Butz N, Vandamme TF (2002) Pulmonary drug delivery systems: recent developments and prospects. Crit Rev Ther Drug Carrier Syst 19(4–5):425–498. https://doi.org/10.1615/critrevtherdrugcarriersyst.v19.i45.40
Davis ME, Chen Z, Shin DM (2008) Nanoparticle therapeutics: an emerging treatment modality for cancer. Nat Rev Drug Discovery 7(9):771–782
Desai AD, Lavelle M, Boursiquot BC, Wan EY (2022) Long-term complications of COVID-19. Am J Physiol Cell Physiol 322(1):C1–C11. https://doi.org/10.1152/ajpcell.00375.2021
Diebold SS, Kursa M, Wagner E, Cotten M, Zenke M (1999) Mannose polyethylenimine conjugates for targeted DNA delivery into dendritic cells. J Biol Chem 274(27):19087–19094. https://doi.org/10.1074/jbc.274.27.19087
Drożdżal S, Rosik J, Lechowicz K, Machaj F, Kotfis K, Ghavami S, Łos MJ (2020) FDA approved drugs with pharmacotherapeutic potential for SARS-CoV-2 (COVID-19) therapy. Drug Resist Updat 53:100719. https://doi.org/10.1016/j.drup.2020.100719
Fang E, Liu X, Li M, Zhang Z, Song L, Zhu B, Wu X, Liu J, Zhao D, Li Y (2022) Advances in COVID-19 mRNA vaccine development. Sig Transduct Target Ther 7(1):94. https://doi.org/10.1038/s41392-022-00950-y
Gautret P, Lagier JC, Parola P, Meddeb L, Sevestre J, Mailhe M, Doudier B, Aubry C, Amrane S, Seng P, Hocquart M (2020) Clinical and microbiological effect of a combination of hydroxychloroquine and azithromycin in 80 COVID-19 patients with at least a six-day follow up: a pilot observational study. Travel Med Infect Dis 34:101663
Grenha A, Seijo B, Remunán-López C (2005) Microencapsulated chitosan nanoparticles for lung protein delivery. Eur J Pharm Sci 25(4–5):427–37
Grove J, Marsh M (2011) Host-pathogen interactions: the cell biology of receptor-mediated virus entry. J Cell Biol 195(7):1071–1082
Gunathilake TMSU, Ching YC, Uyama H, Chuah CH (2021) Nanotherapeutics for treating coronavirus diseases. J Drug Deliv Sci Technol 64:102634. https://doi.org/10.1016/j.jddst.2021.102634
Haasnoot J, Westerhout EM, Berkhout B (2007) RNA interference against viruses: strike and counterstrike. Nat Biotechnol 25(12):1435–43
Han J, Zhao D, Li D, Wang X, Jin Z, Zhao K (2018) Polymer-based nanomaterials and applications for vaccines and drugs. Polymers 10:31. https://doi.org/10.3390/polym10010031
Ioerger TR, O’Malley T, Liao R, Guinn KM, Hickey MJ, Mohaideen N, Murphy KC, Boshoff HIM, Mizrahi V, Rubin EJ, Sassetti CM, Barry CE, Sherman DR, Parish T, Sacchettini JC (2013) Identification of new drug targets and resistance mechanisms in mycobacterium tuberculosis. PLoS One 8:1–13. https://doi.org/10.1371/journal.pone.0075245
Jain AK, Khar RK, Ahmed FJ, Diwan PV (2008) Effective insulin delivery using starch nanoparticles as a potential trans-nasal mucoadhesive carrier. Eur J Pharm Biopharm 69(2):426–35
Kamal MA, Jusko WJ (2004) Interactions of prednisolone and other immunosuppressants used in dual treatment of systemic lupus erythematosus in lymphocyte proliferation assays. J Clin Pharmacol 44(9):1034–1045
Kamaly N, Yameen B, Wu J, Farokhzad OC (2016) Degradable controlled-release polymers and polymeric nanoparticles: mechanisms of controlling drug release. Chem Rev 116(4):2602–2663. https://doi.org/10.1021/acs.chemrev.5b00346
Kumar V, Sharma N, Maitra SS (2017) In vitro and in vivo toxicity assessment of nanoparticles. Int Nano Lett 7:243–256. https://doi.org/10.1007/s40089-017-0221-3
Kumar Malik D, Baboota S, Ahuja A, Hasan S, Ali J (2007) Recent advances in protein and peptide drug delivery systems. Curr Drug Deliv 4(2):141–51
Lawrence MJ, Rees GD (2000) Microemulsion-based media as novel drug delivery systems. Adv Drug Deliv Rev 45(1):89–121. https://doi.org/10.1016/s0169-409x(00)00103-4
Ledford H (2020) Chloroquine hype derails coronavirus drug trials. Nature 580(7805):573
Ledford H (2020b) Coronavirus breakthrough: dexamethasone is first drug shown to save lives. Nature 582(7813):469. https://doi.org/10.1038/d41586-020-01824-5
Lembo D, Cavalli R (2010) Nanoparticulate delivery systems for antiviral drugs. Antivir Chem Chemother 21(2):53–70
Li ZZ, Wen LX, Shao L, Chen JF (2004) Fabrication of porous hollow silica nanoparticles and their applications in drug release control. J Control Release 98(2):245–254. https://doi.org/10.1016/j.jconrel.2004.04.019
Li L, Li R, Wu Z, Yang X, Zhao M, Liu J, Chen D (2020) Therapeutic strategies for critically ill patients with COVID-19. Ann Intensive Care 10:1–9
Li J, Zhang K, Wu D, Ren L, Chu X, Qin C, Han X, Hang T, Xu Y, Yang L, Yin L (2021) Liposomal remdesivir inhalation solution for targeted lung delivery as a novel therapeutic approach for COVID-19. Asian J Pharm Sci 16(6):772–783. https://doi.org/10.1016/j.ajps.2021.09.002
Liu J, Liao X, Qian S et al (2020) Community transmission of severe acute respiratory syndrome coronavirus 2, Shenzhen China. Emerg Infect Dis 26(6):1320. https://doi.org/10.3201/eid2606.200239
Lu R, Zhao X, Li J, Niu P, Yang B, Wu H, Wang W, Song H, Huang B, Zhu N, Bi Y (2020) Genomic characterisation and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. The lancet 395(10224):565–74
Lythgoe MP, Middleton P (2020) Ongoing clinical trials for the management of the COVID-19 pandemic. Trends Pharmacol Sci 41(6):363–382
Maideen NM (2021) Therapeutic efficacy of kabasura kudineer (siddha formulation), in covid-19–a review of clinical and molecular docking studies. Asian J Adv Res 14:817–824
Malmsten M (2006) Soft drug delivery systems. Soft Matter 2(9):760–769
Mamo T, Moseman EA, Kolishetti N, Salvador-Morales C, Shi J, Kuritzkes DR, Langer R, von Andrian U, Farokhzad OC (2010) Emerging nanotechnology approaches for HIV/AIDS treatment and prevention. Nanomedicine (Lond) 5(2):269–85. https://doi.org/10.2217/nnm.10.1
Maus A, Strait L, Zhu D (2021) Nanoparticles as delivery vehicles for antiviral therapeutic drugs. Eng Regen 2:31–46. https://doi.org/10.1016/j.engreg.2021.03.001
Meuleman P, Catanese MT, Verhoye L, Desombere I, Farhoudi A, Jones CT, Sheahan T, Grzyb K, Cortese R, Rice CM, Leroux-Roels G (2012) A human monoclonal antibody targeting scavenger receptor class B type I precludes hepatitis C virus infection and viral spreadin vitroandin vivo. Hepatology 55(2):364–372
Milovanovic M, Arsenijevic A, Milovanovic J, Kanjevac T, Arsenijevic N (2017) Nanoparticles in antiviral therapy. Antimicrobial nanoarchitectonics. Elsevier, Amsterdam, pp 383–410
Mitchell MJ, Billingsley MM, Haley RM et al (2021) Engineering precision nanoparticles for drug delivery. Nat Rev Drug Discov 20:101–124. https://doi.org/10.1038/s41573-020-0090-8
Modrow S, Falke D, Truyen U, Schätzl H (2013) Viruses with single-stranded, positive-sense RNA genomes. Mol Virol 12:185
Molina JM, Delaugerre C, Le Goff J, Mela-Lima B, Ponscarme D, Goldwirt L, de Castro N (2020) No evidence of rapid antiviral clearance or clinical benefit with the combination of hydroxychloroquine and azithromycin in patients with severe COVID-19 infection. Medecine et Maladies Infectieuses 50(4):384
Müller RH, Mäder K, Gohla S (2000) Solid lipid nanoparticles (SLN) for controlled drug delivery - a review of the state of the art. Eur J Pharm Biopharm 50(1):161–177. https://doi.org/10.1016/s0939-6411(00)00087-4
Natarajan S, Anbarasi C, Sathiyarajeswaran P, Manickam P, Geetha S, Kathiravan R, Prathiba P, Pitchiahkumar M, Parthiban P, Kanakavalli K, Balaji P (2021) Kabasura Kudineer (KSK), a poly-herbal Siddha medicine, reduced SARS-CoV-2 viral load in asymptomatic COVID-19 individuals as compared to vitamin C and zinc supplementation: findings from a prospective, exploratory, open-labeled, comparative, randomized controlled trial, Tamil Nadu, India. Trials 22(1):1–1
Pang J (2015) Fabrication of mesoporous silica nanospheres with radially oriented meso channels by microemulsion templating for adsorption and controlled release of aspirin. RSC Adv 5(9):6599–6606
Panyam J, Labhasetwar V (2003) Biodegradable nanoparticles for drug and gene delivery to cells and tissue. Adv Drug Deliv Rev 55(3):329–347. https://doi.org/10.1016/s0169-409x(02)00228-4
Patil YB, Toti US, Khdair A, Ma L, Panyam J (2009) Single-step surface functionalization of polymeric nanoparticles for targeted drug delivery. Biomaterials 30(5):859–866. https://doi.org/10.1016/j.biomaterials.2008.09.05635
Patra JK, Das G, Fraceto LF, Campos EV, Rodriguez-Torres MD, Acosta-Torres LS, Diaz-Torres LA, Grillo R, Swamy MK, Sharma S, Habtemariam S (2018) Nano based drug delivery systems: recent developments and future prospects. J Nanobiotechnol 16(1):1–33
Sarkar I, Garg R, van Littel-van den Hurk S (2019) Selection of adjuvants for vaccines targeting specific pathogens. Expert Rev Vaccines 18(5):505–521. https://doi.org/10.1080/14760584.2019.1604231
Schwendener RA (2014) Liposomes as vaccine delivery systems: a review of the recent advances. Ther Adv Vaccines 2(6):159–182. https://doi.org/10.1177/2051013614541440
Seminara G, Carli B, Forni G et al (2020) Biological fluid dynamics of airborne COVID-19 infection. Rend Fis Acc Lincei 31:505–537. https://doi.org/10.1007/s12210-020-00938-2
Sharma N, Ojha H, Bharadwaj A, Pathak DP, Sharma RK (2015) Preparation and catalytic applications of nanomaterials: a review. RSC Adv 5:53381–53403
Sharma T, Gerstman B, Chapagain P (2023) Distinctive features of the XBB.1.5 and XBB.1.16 spike protein receptor-binding domains and their roles in conformational changes and angiotensin-converting enzyme 2 binding. Int J Mol Sci 24(16):12586. https://doi.org/10.3390/ijms241612586
Shi L, Plumley CJ, Berkland C (2007) Biodegradable nanoparticle flocculates for dry powder aerosol formulation. Langmuir 23(22):10897–901
Shi L, Plumley CJ, Berkland C (2007b) Biodegradable nanoparticle flocculates for dry powder aerosol formulation. Langmuir 23(22):10897–10901. https://doi.org/10.1021/la7020098
Singh L, Kruger HG, Maguire GEM, Govender T, Parboosing R (2017) The role of nanotechnology in the treatment of viral infections. Ther Adv Infect Dis 4(4):105–131. https://doi.org/10.1177/2049936117713593
Song SW, Hidajat K, Kawi S (2005) Functionalized SBA-15 materials as carriers for controlled drug delivery: influence of surface properties on matrix-drug interactions. Langmuir 21(21):9568–75. https://doi.org/10.1021/la051167e
Teixeira MZ (2020) Clinical research protocol to evaluate the effectiveness and safety of individualized homeopathic medicine in the treatment and prevention of the COVID-19 epidemic. Rev Assoc Med Bras 66(4):405–406. https://doi.org/10.1590/1806-9282.66.4.405
Teixeira MZ (2021) Homeopathy: a preventive approach to medicine? Int J High Dilution Res 8(29):155–172. https://doi.org/10.51910/ijhdr.v8i29.36015
Wang M, Cao R, Zhang L, Yang X, Liu J, Xu M, Shi Z, Hu Z, Zhong W, Xiao G (2020) Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res 30(3):269–271
Wilson B, Geetha KM (2022) Lipid nanoparticles in the development of mRNA vaccines for COVID-19. J Drug Deliv Sci Technol 74:103553. https://doi.org/10.1016/j.jddst.2022.103553
World Health Organization (2014) Infection prevention and control of epidemic- and pandemic-prone acute respiratory infections in health care. Geneva: World Health Organization; https://apps.who.int/iris/bitstream/handle/10665/112656/9789241507134_eng.pdf?sequence=1
World Health Organization (2023) COVID-19 weekly epidemiological update, edition 158, 29 September 2023. World Health Organization
Wu R, Wang L, Kuo HD, Shannar A, Peter R, Chou PJ, Li S, Hudlikar R, Liu X, Liu Z, Poiani GJ, Amorosa L, Brunetti L, Kong AN (2020) An update on current therapeutic drugs treating COVID-19. Curr Pharmacol Rep 6(3):56–70. https://doi.org/10.1007/s40495-020-00216-7
Xue Y, Liu C, Andrews G, Wang J, Ge Y (2022) Recent advances in carbon quantum dots for virus detection, as well as inhibition and treatment of viral infection. Nano Converg 9(1):15. https://doi.org/10.1186/s40580-022-00307-9
Yang SNY, Atkinson SC, Wang C, Lee A, Bogoyevitch MA, Borg NA, Jans DA (2020) The broad spectrum antiviral ivermectin targets the host nuclear transport importin α/β1 heterodimer. Antiviral Res 177:104760. https://doi.org/10.1016/j.antiviral.2020.104760
Yao X, Ye F, Zhang M, Cui C, Huang B, Niu P, Liu X, Zhao L, Dong E, Song C, Zhan S (2020) In vitro antiviral activity and projection of optimized dosing design of hydroxychloroquine for the treatment of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Clin Infect Dis 71(15):732–9
Yarlagadda H, Patel MA, Gupta V, Bansal T, Upadhyay S, Shaheen N, Jain R (2022) COVID-19 vaccine challenges in developing and developed countries. Cureus 14(4):e23951. https://doi.org/10.7759/cureus.23951
Yoo J, Park C, Yi G, Lee D, Koo H (2019) Active targeting strategies using biological ligands for nanoparticle drug delivery systems. Cancers 11(5):640
Zhang L, Gu FX, Chan JM, Wang AZ, Langer RS, Farokhzad OC (2008) Nanoparticles in medicine: therapeutic applications and developments. Clin Pharmacol Ther 83(5):761–769. https://doi.org/10.1038/sj.clpt.6100400. (Epub 2007 Oct 24 PMID: 17957183)
Acknowledgements
Dr NU and Dr NS acknowledge the support of the Director, ICMR-National Institute for Research in Tuberculosis, and the approval from ICMR. Dr NU and Dr SVK acknowledge Director, CSIR-Central Leather Research Institute. A/2021/CHR/NWP100/1514 is the communication number obtained from CLRI.
Author information
Authors and Affiliations
Contributions
All authors contributed to the work and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Usharani, N., Kanth, S.V. & Saravanan, N. Biomimetic nanomaterials for pulmonary infections: A prospective view in drug delivery systems. Appl Nanosci 14, 363–373 (2024). https://doi.org/10.1007/s13204-023-02981-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13204-023-02981-5