Abstract
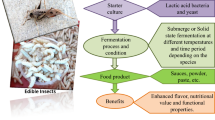
Edible insects (EI) are also becoming as a part of the diet due to their nutritional value and health benefits in many regions of the world. These EI are inexhaustible sources accessible by garnering from the wild with high feed conversion efficiency. Appreciating the budding of EI in justifiable food production, enlightening food security and biodiversity conversion, is promising a sufficient supply of the insect resource for future food to the world. These insects are processed to develop new products, improve organoleptic and nutritional parameters as well as the extension of shelf life. In this review, we discuss the edible insect characteristics, the potential application of EI in food industry, processing, pretreatments, drying, extraction of edible compounds like protein, lipid and chitin various food products formulation, safety regulation. Availability of broad nutritional spectrum of EI includes protein, mono and poly unsaturaturated fatty acids, amino acids, vitamins, amino aids and minerals has been used as an ingredient in development of various forms of food products such as flours in the form of whole insect powder, protein isolate, canned products, extruded products, hard candies, spreads, liquor infusion, cookies and other products.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
From recent surveys, it is believed that the population of the world will hit around 9 billion by 2025 which in turn insists on doubling the current food production, scarcity of land for agriculture, and over exploitation of fisheries practices in addition to climatic changes could have an adverse effect on satisfying the world’s nourishment and nutrition challenges (Liceaga and Andrea 2021). Due to rapid population growth, environmental pressures, food and feed insecurity, and alternative solutions to conventional livestock and feedstocks, the consumption of insects as human food, animal feed, and poultry feed emerges as an alarming contribution to the environment, health, and livelihoods. However, consuming insects as food is very rare in India due to the influence of cultural and religious practices. According to Food and Agriculture Organization, 2100 species has been identified as EI (Table 1) have been in diet of among 3071 cultural groups in 130 countries throughout the world (Deka et al. 2021). Most of the insects are inexhaustible resource obtainable by harvesting from wild with high feed conversion efficiency. With high protein, essential amino acids, vitamins, essential fatty acids, lipids, fiber and minerals insects are considered to be predominantly nutritious and healthy source of food. These species are known for having a high nutritional content, and they can be eaten whole or ground, as well as included into other processed meals. They can be fried, cooked, stewed, steamed, roasted, or boiled, or being heat processed and flavoured with various spices as seasoning. EI have a wide nutritional value due to their high protein content and mono- and polyunsaturated fatty acid content. In addition, insects contain adequate vitamins (including pantothenic acid, riboflavin, biotin and folic acid) and minerals (including iron, copper, magnesium, phosphorus, selenium, manganese and zinc) to help meet the recommended daily intake of these nutrients, according to Food and Agriculture Organization (FAO) and World Health Organization (WHO) nutritional guidelines. The quantity of energy delivered by insects varies by species, with estimations ranging from 293 to 776 kcal per 100 g (g) of dry matter (Kinyuru et al. 2015). Insects are mostly made up of proteins, which account for 38 to 77% of their entire dry mass. Insect proteins are also of excellent quality and easily digested with less digestible rate than egg protein, but higher digestible rate when compared to plant derived proteins (Finke 2015). The proteins contained in edible bug species contain a large percentage of important amino acids, such as lysine, tyrosine, tryptophan, and phenylalanine, which are required for optimal body growth and activity. There are some exceptions, since some species or orders lack certain amino acids; for example, Diptera lack leucine and cysteine, while Hemiptera lack lysine, phenylalanine, tyrosine, and valine. Most bug species, on the other hand, meet the World Health Organization’s essential amino acid content guidelines. As a result, insects are being regarded as a potential solution to the nutritional deficits that plague developing countries, as well as a viable and sustainable feedstock for the livestock industry (Raheem et al. 2019). The variation in nutritional content of insects is significant between species and development phase. Crude protein in a species does not fluctuate with phase of development, despite caloric values (Kim et al. 2019). Lipids are the insect's second most prevalent dietary component, accounting for anything from 4 to 77% dry weight, depending on life stage. In general, insect lipid content was increased in larval stages than those in adult stages, with adults typically containing 80 percent triglycerides and less than 20% phospholipids. Insects also typically contain fatty acids, such as monounsaturated, and polyunsaturated fatty acids. These acids include oleic, linoleic, lauric, α-linolenic, and palmitic acids, all of which are beneficial to human health (Nadeau et al. 2015). The most variable nutritional component of edible insects is micronutrients, such as minerals and vitamins, which can be available in significant or low amounts depending on the insects' diet (Baiano 2020). In terms of vitamin content, EI are good providers of pantothenic acid, riboflavin, and biotin, while vitamin A, C, and E are lacking. Vitamin content is influenced by the insect’s diet, just like mineral content, and is often higher in insects raised in commercial farms where their food is enriched with vitamins. This nutrient-rich meal improves the nutrient benefit of the insect and has health benefits for humans (Churchward-Venne et al. 2017). Chitin, a component of the exoskeleton of insects, is a dietary fiber that typically ranges from 11.6 mg to 137.2 mg / kg of dry matter. Despite its difficulty in digestion, chitin has been studied as a viable source of dietary fiber in the human diet, and it has been found to enhance the human immune system, protecting against parasite infections and allergies (Wang et al. 2019). Insects have a diverse microbiota in both their digestive system and on their external surfaces, which is molded by the insect's upbringing and living environments, as well as during processing for human nutrition (Frigerio et al. 2020). Because insects are vectors for the spread of dangerous bacteria, it is critical to ensure the microbiological safety of edible insects, as the total microbial load of a food determines its safety and shelf life (Garofalo et al. 2019). Wild insects are more likely to be contaminated by microbes, whereas growing insects in fields allows the farmer to know, control, (Table 2 and 3)and ensure the production conditions, lowering the danger of microbial contamination (Megido et al. 2017). The industrial sector is increasingly involved in the development, processing, and commercialization of edible insects as a result of the benefits connected with their usage in human meals. Realizing the potential of EI in sustainable food production,civilizing food security and bio diversity conversion, is assuring sufficient supply of the insect resource for future food to the world.
EI of India
Insects deliver a congregation of ecological services such as plant reproduction, waste biodegradation, pest control, fertilizer, as important bio-converters by change of low quality and modest biomass into nutritional rich and innovative economical products. Diversified EI has been highly consumed in traditional diets in the regions of Asia, subsequently Africa and South America. These are also hotspots where populations living in dearth leading to the risk of undernutrition, malnutrition, micronutrient deficiencies and adult obesity. However, these populations are largely unaware of the nutritional role of EI to their diets because of the lacking in fundamental data research and subtle information on insect consumption and supply chain. The major gap and lacking in statistical record of in global level of EI is evident that insects are still belonging to the informal sector of food processing, and thereby did not get its legal positions such as national regulations and legistlations. In India, rearing of EI in rural areas has been carried out for income generation besides functional and nutritional values. EI are consumed by members of various tribes according to their traditional beliefs, regional, taste and seasonal availability. Being a tropical country diversity and abundance of insects are greater in India and considered to be a great history of human-insect-interaction. Around 255 edible species from 10 different states of the country have been recorded in India, few of them includes orders of Coleoptera-34%, Orthoptera-24%, Odonata -8%, Hemiptera-17%, Hymenoptera-0%, Lepidoptera-4%, Isoptera-2% and Ephemeroptera-1%. Common names of the species were termites, locusts, ants, weaver ants, bees, cinnamon bugs, silkworm pupae, mealworm, grasshoppers (short & long-horned), crickets, mole crickets, pentatomid bugs, etc.(Lange et al. 2021).
Farming and consumption advantages
EI are primary potential natural resources categorized under products belonging to non-wood forest type. In spite of its identification, among which few are consideration for its valuable products, insect ecology has been limited. EI are considered to be inexhaustible resources that provide essential ecosystem services. Recently, researchers found that many species of EI are under potential threats and in peril due to direct competition with predators, over exploitation by utilizing mature insects before mating, climate changes, unsustainable collection methods, damage to habitats such as pollution, wildfire and deforestation. On the other hand, the role of insects in the conservation of ecological services and their habitats is receiving more attention; as a result, Flagship species and Umbrella species have been evolved in the promotion of protecting species In addition to compensating for rising demand for animal protein and avoiding deforestation for pasture usage, edible insects have a high feed conversion ratio compared to conventional cattle and generate comparatively low greenhouse gas and ammonia emissions (Poma et al. 2017). Insects have a good nutritional composition, not only because of their greater concentration of amino acids compared to other sources, but also because of their ability to satisfy principles that are sustainable, healthful, accessible, and delicious (Ynsect 2018). A drastic increase in production and consumption of livestock and fish imparts increased adverse effects on environmental costs needs an urgent alternative resource to overcome the demand in the world food production system. As a consequence, rearing and farming of EI has been evolved with promising advantages such as; high feed conversion efficiency, bio-waste of organic side streams into value added products, reduction in emission of green-house gases and ammonia, less water demand, low risk of transmitting zoonotic infection, life cycle analysis, bio-security, requires less space, create cash inflow in short period and high reproductive rate. Insects are substantially more effective than mammals in converting plant proteins to insect proteins (Deroy et al. 2015). Crickets, for example, have been determined to require less than 2 kg of feed for 1 kg of bodyweight increase. In comparison, for a 1 kg rise in bodyweight (feed-to-meat conversion rate), 2.5 kg of feed is commonly required for chicken, 5 kg for pig, and up to 10 kg for beef. When these values are adjusted for edible weight, the benefits of eating insects become further clearer. It is claimed as up to 80% of a cricket may be consumed and digested, compared to 55% for hens and pigs and just 40% for cattle (Collavo et al. 2002). The theory of farming insects is quite new to development circle which includes rearing in a designated area with proper environmental conditions and diet. Contrast to it, rearing insects under confined environment may impulse effect on population of insects by affecting genetic characters through inbreeding depression, laboratory adaptation and founder effect.
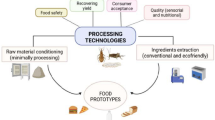
Processing of EI
EI has been proved to have potential nutritious food source as its rich in fat, protein, chitin and certain amount of minerals and vitamins. The typical protein content of dried insect matter has been shown to range from 35% (termites) to 61% (crickets, grasshoppers, and locusts), with some of the species reaching as high as 77% (Rumplod et al. 2013). Insects' total fat content has been estimated to vary between 2% and 62 percent. (Figs. 1 and 2) While the fatty acid profile of EI looks to be comparable to that of vegetable oils and animal fats, insects have a high concentration of unsaturated fatty acids, indicated that up to 75% of total fatty acid content (Williams et al. 2016). Chitin concentration varies by species and developmental level, but it accounts for around 10% of dry weight. Purified chitin is made up of around 90% dietary fibre that can be metabolized by humans (da et al. 2021). The need for edible insect processing is staggering at high speed in order to overcome the demand as an novel ingredient in new product formulations. Processing of functional compounds from insects involves standard protocol as explained in Fig. 3.
Consumption pattern of EI (number of species) in different parts of the world (Mitsuhashi 2016)
Status of EI among different states of India (Chakravorty 2014)
Pre-treatment
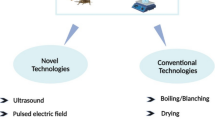
The most common and prior step in edible insect processing starts with inactivation (or) killing of live insects as a pretreatment method. These techniques are majorly focused on microbial inactivation such as Enterobacteriaceae, Staphylococcus, Bacilli, yeasts, and molds (Grabowski et al. 2017) and degradation of the phenol oxidase enzymatic mechanism (Janssen et al. 2019) responsible for food poisoning and spoilage. Blanching or wet thermal treatment is one of the majorly used pretreatments for killing, enzyme inactivation and microbial reduction. It’s a process of immersing live insects into boiling water for 2 to 3 min and rapidly cooling it under cold water. The combined effect of blanching assisted with microwave drying of T.molitor has been investigated and reported the increased moisture content with a negligible significant difference in their chemical composition (Selaledi et al. 2021). Blanching of edible insects in combination with indirect plasma for better surface contamination to ensure microbial safety (Vandeweyer et al. 2017). In the future, there is great demand for suitable pretreatment techniques such as non-thermal technologies that could be adopted to provide zero sacrifices in physical properties and chemical composition.
Drying
Drying is the next step to pretreatment before processing the whole insect into different functional products. Drying strategies range from conventional methods (sun-drying) to current techniques (freeze-drying, microwave-drying, etc.). Drying provides reduced moisture content and water activity; hence, it increases the shelf life, keeping quality and storage constancy of respective products by eliminating the enzymatic reactions and microbial metabolism. Reduction in free water enhances the percentage concentration of dry matter and physical properties of raw material without damaging themand is a major advance for food ingredient extraction. Sun-drying, hot air oven drying, freeze-drying are the favored and universally used methods for drying EI, apart from these microwaves drying, freeze-drying, Radio-frequency, Infrared drying and other non-conventional drying are also being used. This dried and grounded product with an extended shelf-life is well suited for distribution and storage. Drying potentially affects pathogenic microbes, reduces harmful compounds such as neurotoxins and causes inactivation of protease enzyme thus improves the nutritional quality of the product (Musundire et al. 2016). Poor sanitary conditions, unhygienic practices, contamination through contact with soil and air and lack of identifying critical control points such as moisture content and water activity (Megido et al. 2018). Freeze-drying has been one of the majorly used drying technique in a laboratory scale to yield high end quality product with well preserved nutritional profile over an extended shelf life for further processing and characterisation of EI. In products of T. molitor with high lipid content could have more possibility of lipid oxidation with an orderly decreased solubility of protein (Megido et al. 2018; Kröncke et al. 2018). Oven-dried food products are highly compatible because of their lower energy input costs of production, high protein solubility and reduced lipid oxidation (Purschke et al. 2017;Kröncke et al. 2018;Oliveira, Jantzen, and Cadaval 2017). Microwave-assisted dried T.molitor product shows reduced water activity of below 0.6 after 16 min, with least significant changes in fat, protein and mineral contents (Kröncke et al. 2018)by comparing the effect of various drying processes such as microwave-drying, freeze-drying, vacuum-drying, fluidized-bed and conventional-hot air drying and reported < 70% moisture content and < 0.6 water activity in T. molitor larvae with negligible changes in protein, fibre and fat content. Khampakool et al. (2020) investigated the function of Infrared assisted freeze-drying technology on Protaetia brevitarsis larvae. Drying time was reduced to 237 times and reduced electrical energy consumption up to 90% in contrast with the normally used freeze drying technique. It also provides products with less hardness, high protein level, better chewiness and better preservation of other functional compounds. Blanched and milled mealworm slurry has been spray dried under the condition of inlet port at 170–190 ℃ and outlet port at 80–90 °C for producing whole insect powder with high fat and protein content (Kim et al. 2017). Solar drying of blanched whole insect has been done in a specially designed solar tent under processing condition of temperature 50–60℃ and relative humidity of 15–16% followed by milling into the whole insect and reported its moisture sorption capacity (Kamau et al. 2018). The application of medium-wave infrared and hot-air drying techniques for the manufacture of meal worm powder. Beyond the reduction in moisture content and water activity by vast application of different drying technologies prone to have slight to an adverse effect on lipid oxidation, protein solubility, physical properties such as color and texture. As a result, the selection of suitable drying technology mustbe taken into account of the deliberate use of edible insect species and its consumption patterns among the population (Kaur et al. 2019).
Lipid and its extraction
Lipid constitutes the second chief portion of the physicochemical and nutritional value of EI as a source of energy and essential fatty acids. The higher content of lipids has been observed during the larval stage ranges from 10–50%. The deviation in lipid content is due to species, metamorphosis stage, environment, sex and diet. EIinstitute hugequantity of unsaturated fatty acids; ω-3-α-linolenic acid, ω-6-linoleic acid, ω-9-oleic acid, and saturated fatty acids; palmitic acid and stearic acid. Thus improve its functional properties and an alternative fat source extracted from plants and animals (Bubler et al. 2016). A combination of various novel technologies with conventional methods and usage of green solvents as an alternative has to be employed to provide better extraction yield with high fatty acid profile, phospholipids and other functional properties. Extraction of lipids from black soldier fly larvae has been performed by wet extraction method using 2-methyltetrahydrofuran and hexane as solvents. The oil yielded by 2-methyltetrahydrofuran has been reported as 95% with higher content of fatty acids and phospholipids compared to hexane with 70% (Smets et al. 2020). Comparative study on lipid extraction from Acheta domesticus and T.molitor by ultra-sound assisted and high pressurized liquid solvent extraction by with ethanol and ethanol water as common solvents has been conducted. Regardless of the extraction method, insect species and solvent used pressurized liquid method tend to have the highest oil yield of mean value 24% compared to ultra-sound extraction methods of mean value 19%. On the other hand, oil extracts obtained by the ultra-sound assisted method have improved fatty acid profile irrespective of the species and solvent when compared to the pressurized liquid method (Otero et al. 2020). Lipid extracts were obtained from T.molitor and Pachymorus nucleorum by solid–liquid extraction technique using hydro-ethanolic as solvent. The oil extracted from T.molitor contains 0.79% linoleic and 0.23% α-linolenic acid, whereas, P.nucleorum oil contains 42.26% saturated fatty acid and 48.95% unsaturated fatty acid (Alves et al. 2019). Effective extraction of insect oil from Acheta dometicus and T.molitor using high pressure processing assisted technique has been investigated. Study revealed that high pressure processing at 500 Mpa for 15 min as an effective extraction condition for lipids with less change in functional properties (Ugur et al. 2020). Optimization of the Cicadidae species oil extraction techniques using ethanol-assisted aqueous extraction technique in comparison with conventional hexane extraction technique has been performed. The study revealed that oil yielded by ethanol assisted aqueous extraction technique was 19.5% in comparison with the conventional method as 27%. The extracted oils from two different approaches showed very least difference in respective fatty acid profile (Mahmoudikordi et al. 2021). Effective supercritical CO2 extraction of Bombyx mori larvae oil has been studied. At optimal conditions the highest of 30.10% oil yield has been obtained such as the temperature of 45 °C, extraction time of 145 min and pressure of 203 bar (Srinivas et al. 2019). Aqueous extraction technique has been employed for oil extraction of four diverse insect species has been carried out. The oil yield of respective species was; yellow mealworm –60.29%, lesser mealworm–58.24%, house cricket–18.06% and Dubia cockroach –40.82% (Tzompa-sosa et al. 2019). Bombyx mori (silk worm) pupae oil has been extracted using petroleum-ether as a solvent in a soxhlet extractor (Patil et al. 2019). The oil yield was 34.6% with 12.75% saturated and 86.6% unsaturated fatty acid profile. In Laroche et al. (2019) effect of six lipid extraction procedures has been carried out on Acheta domesticus and T.molitor. Six techniques employed are, supercritical CO2 extractor under 325 bar pressure for 75 min; Soxhlet extractor with hexane, ethanol, ethyl actetate and petroleum ether as solvents; three phase partitioning with ammonium and t-butanol. Soxhlet extraction with ethanol yielded highest lipid content of 22.7 and 28.8% and supercritical CO2 extractor yielded the lowest rate of 11.9 and 22.1% respectively for both species.
Protein and its extraction
Due to rapid change in population dynamics and increased insist for protein, the availability of agricultural landis also being inhibited for the processing of plant-based protein and the sustainability of meat for protein also represents a serious challenge for the future. However, insects compiles abundant source of protein with high profiling essential amino acids, it also has a superior protein digestibility rate of around 98% (Kim et al. 2020). Protein extracted from various insect species can be worn as a wholesome ingredient in different food applications and found to have greater success in provisions of acceptance as food. The impact of food neophobia and disgust in consumption of the whole insect as food and convincing mentality by society could be considered by application of protein extract in different food formulations. From various researches, its revealed the maximum solubility of insect protein occurs in a range of pH 8 to 11 (Kim et al. 2019). The processing steps and extraction techniques have a direct influence on the quality, quantity and purity of obtained protein concentrate. The rate of extraction and characteristics are mostly depending on species, extraction technique and solvent used thus has a direct effect on extraction yield, protein solubility, physicochemical and functional properties. There are several extraction procedures that has been implicated for protein extraction, procedure starts with 48 h of fasting to clear the insect’s gastrointestinal tract for any residual food before killing it (Kurdi et al. 2021). Zielińska, Karaś, and Baraniak (2018) studied and characterized the various functional property (oil and water holding capacity, emulsifying property, foaming stability and protein solubility) of insect proteins potentially extracted from three different edible species (Tenebrio molitor, G. sigillatus, S.gregaria) using the alkaline method. The percentage yield of protein from three species was reported as 52.3, 70 and 76% respectively. The percentage solubility of extracted protein was found to have 90–96% at pH 11. The application of the dry fractionation extraction technique in the extraction of protein from T.molitor and the percentage yield of protein has been recorded as 72% (Purschke et al. 2017). Protein from three edible species Allomyrina dichotoma, Tenebrio molitor and Protaetia brevistarsis seulensis were isolated using acid precipitation and the yield was found to be 71.32, 89.05 and 85.38% respectively. It is also revealed the increase in essential amino acids, foaming capacity and its stability (Kim et al. 2020). used Three different extraction techniques pH shift extraction, alkaline and aqueous methods has been used for the extraction of T.molitor larvae protein and the yield has been found as 87.74, 55.18 and 42.93% respectively. FT-IR spectra show the presence of amide-I, II & III bands in the pH-shift and alkaline protein extracts and only amide- I & III are present in the aqueous protein extract. Among three extraction techniques,alkaline and aqueous protein extraction beholds less efficiency than pH-shift extraction of defatted larvae flour in terms of structural conformation and high extraction yield (Santhosh et al. 2019). The combined potential application of n-hexane and Sonication assisted technique for isolation of protein from Bombyx mori of 94%, G. bimaculatus of 37% and Tenebrio molitor of 35% (Choi, Wong, and Auh 2017).
Chitin
The physico-chemical composition of EI are found to have a substantial level of fiber available as chitinform in their exoskeleton. About 20–50% dry weight of chitin has been present in the three layers exocuticle, endocuticle and epiculticle of insects. Exocuticle, the most external component of insects which gets eliminated and emerges during metamorphosis has a high amount of chitin, whereas, epicuticle has a negligible amount of chitin. Chitin is considered to be an abundant polysaccharide in nature next to cellulose; on deactylation of chitin under alkaline condition forms Chitosan, a straight chain polysaccharide compound. Chitosan has profound functional properties, biocompatibility, anti-microbial, anti-inflammatory and anti oxidant properties thus impacts great potential in smart and novel packaging materials such as edible film, edible coating, etc. Commonly and majorly used chitin and chitosan extraction technique involves three major steps demineralization (acid treatment); deproteinization (alkali treatment) and deacetylation (organic acid treatment). The protein digestibility of bees is 71.5% with chitin, compared to 94.3% without it, suggesting that removing chitin from insects before to eating could enhance the quality and bioaccessibility of dietary proteins (Soares de Castro et al. 2018). da et al. (2020) has performed one step enzymatic deproteinization and deacetylation on wastes of T.molitor breeding cuticles to obtain chitin. About 85% of chitin obtained by enzymatic deproteinization and overall yield of cuticle to chitosan has been found as 31.9%. In Shin, Kim, and Shin (2018) isolation of chitin has been reported from larvae stage, adult range and super worm of Mealworm and larvae stage, pupa stage and adults of Rhinoceros beetle by both deproteinization method and demineralization techniques. Yields of chitin and chitosan were recorded as 4.68 and 80% in larvae; 8.4 and 78.33% in adult; 3.9 and 83.33% in super worm of Mealworm beetle whereas, 10.53 and 83.37% in larvae; 12.7 and 83.37% in pupa; 14.2 and 75% in adult of Rhinoceros beetles respectively. Wang et al. (2020) studied the changes occurs on chitin’s physicochemical structure of Hermetia illucens (Black soldier fly) during its developing stages. Chitin has been determined during development stages such as 3.6% in larvae; 3.1% in prepupae; 14.1% in puparium and 2.9% in adults. Kaya et al. (2015) performed isolation and comparison of chitin from seven grasshoppers species namely, Pyrgomorpha cognate, Oedipoda caerulescens, O.miniata, Aiolopus strepens, A.simulatrix, Duroniella fracta and D.laticornis found that the dry weight of chitin from these seven species varied between 5.3 to 8.9%. Chitin and its chitosan extracted from all specimens of Type 1 and 2 two spotted field cricket (Gryllus bitraculatus) has been investigatigated. The chitin and chitosan obtained from Type 1 and 2 are found to be 20.9; 21.68 and 86.30%; 87.75% respectively (Kim et al. 2017a). The study on extraction of chitin and chitosan content in larvae stage and adults of Leptinotarsa demlineata (Colorado potato beetle) revealed that dry weight of chitin yielded from adult as 20% and larvae 7% and chitosan of 72% in adult and 67% from larvae (Kaya et al. 2014). Adults and nymphs of D.maroccanus has been used for extraction of chitin and its chitosan using three step method. The dry mass of chitin obtained from adult has been established to be 14% and nymphs 12%. Chitosan yield has been recorded as 81.6% in adults and 77.38% for nymphs (Kaya et al. 2014). Extraction study of chitosan from larval exoskeleton of Hermetiaillucens has been recorded under varying treatment conditions. The dry weight of highest chitosan yield was determined as 47% under specified deacetylation condition (Hahn et al. 2020).
Product formulation
In the major parts of the world inclusion of EI in human diet because of its high quality fat, protein, minerals and vitamins with acceptable sensory attributes. The EI Protein market surge has been estimated to be esteemed at 144 million USD in 2019 and will projected to surge at 45.0% CAGR, reaching the value of 1336 million USD by 2025 (Liceaga and Andrea 2021). Apart from various drawbacks regarding food neophobia in many countries insects has been marketed and considered as delicacy. It is available in various forms such as flours, heat-dried larvae and pupae, protein isolate, canned products, extruded products, snack, candies, chocolate dipped and liquor infusion. More than 250 products are commercially made by various small and large scale industries which have been evolved over a past decade. Presence of high level of proteins and quietly well-balanced amino acid profile made its major breakthrough in quest of researchers as a potential food ingredient for nutrition rich sports supplement for instance hydrolysates, protein isolates / concentrates, energy bars and protein based drinks. Modern researches are focused on application of EI as novel food ingredients on fortification into cookies, bread, burger patties, pastas and sausages in that way increasing its nutritional rich value and its market competability in commercialization of EI based food. The developing pattern toward smart dieting and hybrid type meat items are in the midst of the critical drivers of the essentially sped up foothold inside insect protein market. Expanding innovative work in insect food innovation, and above all, the administrative headways up held by certain media inclusion and more noteworthy accessibility of insect food products have additionally set out development open doors in this market. Aspergillus oryzae and Bacillus lichenfernis powders are used as substrate in developing fermented liquid seasoning for addition to soy sauce fermentation process (Cho et al. 2017). Fortification of around 90% of mealworm larvae in the formulation of minced meat-like products and preserved under modified atmospheric packaging (Stoops et al. 2017). Silkworm pupae powders are incorporated into meat batters to prepare frankfurters thus significantly lowers cooking loss weight and improved its textural properties (Park et al. 2017). Extruded products was developed using sun-dried termites along with germinated amaranth, corn and soy oil to combat infant malnutrition (Kinyuru et al. 2015). Enriched bread formulated with Nauphoeta cinerea flour with concentration of 5 to 15% and characterized its physical and sensory properties (Oliveira, Jantzen, and Cadaval 2017). Cookies prepared using cricket flour along with amaranth, rice, oat meal, etc. and are well accepted by children (Kim et al. 2017).
Hazards and safety regulations
Rearing insects for consumption as food for human has major issue in their potential safety because of their biological structure, usage of waste as insect feed, microbial safety, allergenic reactions, toxicological contamination, inorganic compounds and impalpability. Implication of insect for human consumption must have the account on traditional knowledge of insect consumption in different countries and implementation of food safety and regulatory issues by addressing the insect food chain right from species, origin, nature and environmental condition. The exponential amount of research on insects majorly focused on sustainability, economic and market value, nutritional value and their traditional aspects. However, very less number of studies has been published on food safety and regulatory issues of insect consumption over many years. Insects are associated with micro-organisms in two ways; external microbiome includes farming environment and nature and internal microbiota includes gut, cuticle and other anatomical compartments. There is a high possibility of prevalence of pathogenic microbes in the insects collected from wild than reared in farms/laboratories (Finke et. 2014). Though various microbes has been reported in different insect species they are very rarely found to be pathogenic and spoilage causing.
Ng et al. (2018) Studied the comparison of gut microbiota of laboratory reared and wild caught cricket and reported the presence of more diversified microbial community in laboratory reared ones. Marshall, Dickson, and Nguyen (2016) Isolated and characterized various species such as Cladosporium, Aspergillus, Pencillium, Bacillus cereus, E.coli, Klebsiella pneumonia from Gonim brasiabelina (mopane caterpillar). Belluco et al. (2013) conducted a study on isolation of gram positive population in four different commercial species such as Zoophobas morio, Galleria mellonella, T.molitor, & Acheta domesticus and reported the absence of Micrococcus Spp., Lactobacillus Spp., Salmonella Spp. and Lysteria monocytogenes. In (Megido et al. 2018) microbial safety of various commercial EI found on markets of Belgium has been investigated. Microbial isolation on fresh mealworms, house crickets, smoked termites and caterpillar has been done. The various effective processing technologies such as blanching using steam or hot water, freeze drying and sterilization on above mentioned products has been done to reduce the microbial load. Among three processing procedures freeze drying technique and sterilization treatments are found to be effective techniques for reduction in microbial flora. Garofalo et al. (2019) presented a complete overview of edible insect microbiota and their associated potential risks for human health from the published literature between 2000–2019. Poma et al. (2017) evaluated the developmental stages of edible insect intrinsic microbiota of cuticle, gut and other anatomical structures. Fasolato et al. (2018) isolated and report the occurrence of Bacillus cereus and B.cytotoxicus as an initial processing step for hazard identification in EI based products. It also proved the absence of food-borne Lysteria monocytogenes in processed insects from e-commerce. Grabowski and Klein (2016) conducted the survey on microbial analysis of 38 processed insect products such as deep-fried and ready to eat spiced up (Acheta domesticus,Omphisa fuscidentalis and Locusta migratoria), processed along with soy sauce (Vespula flaviceps, Bombyx mori, and Oxya yezoensis), powdered form (H. illucens, T. molitor) and dried form (A. domesticus, Hermetia illucens, L.migatoria, Tenebrio molitor, Bombyx mori, Alphitobius diaperinus,and Musca domestica) were examined for the presence of Enterobacteriaceae, Escherichia coli, Listeria monocytogenes, Bacilli, Staphylococci, Salmonellae, yeasts and moulds. Results obtained revealed that dried form and powdered insects contains elevated microbiota than the cooked and deep fried EI. All samples has been reported negative over L. monocytogenes, Salmonellae, E. coli and Stapyhlococcus aureus, but dried form and powdered insects, contained Bacillus cereus, Serratia liquefaciens, Coliforms, Listeria ivanovii, Penicillium spp., Mucor spp., Aspergillus spp., and Cryptococcus neoformans. (Wynants et al. 2019) in this study reported that feeding of Tenebrio molitor with wheat bran of contaminated quality resulted in the diffusion of Salmonella sp. to the insect. Contamination was found to be higher in larvae fed with contaminated bran as 7 log CFU/g, while there was no deduction of Salmonella sp. at contamination intensity of 2 log CFU/g. Like other protein-rich foods, insects also induce allergic reactions such as eczema, rhinitis, dermatitis, angioedema, conjunctivitis, bronchial asthma and congestion in sensitive human due to presence of various allergens (Ribeiro et al. 2018). Gautreau et al. (2016) reported anaphylaxis in two cases simultaneously followed up by ingestion of silkworm pupae.Verhoeckx et al. (2014) in this study, presence of cross-reactive yellow meal worm protein by Immunoglobulin-E mediated allergy risks in human. Musundire et al. (2016) research revealed the handling or storage of EI at sub-optimal conditions may be accountable in mycotoxin contamination. Reported the presence of negligible amount of aflatoxin B1 in the Encosternum delegorguei (stink bug) packed and stored using recycled containers previously used for grain storage. The chemical hazard of wild harvested EI considered for its dietary components for consuption which has been majorly concerned on variouspotentially dangerous pesticides worn against to kill them, rather than controlled farming. Poma et al. (2017) investigated and studied the presence of different organic contaminants (i.e. dioxin compounds, flame retardants, pesticides, Dichlorodiphenyltrichloroethane, Polychlorinated biphenyls) and various metals (Zn, As, Pb, Cu, Ni, Co, Cr) in multiple samples of various species of EI (migratory locust, greater waxmoth, buffalo worm, mealworm beetles) and selected four commercial insect-based food products of Belgium. The result shows the occurance of relatively low fractions of organic chemical found to be lower when compared with compounds tested in commercial animal products. The untargeted screening analysis reported the presence of vinyltoluene, ributylphosphate, pirimiphos-methyl. The levels of metals Zn and Cu present in these EI were found to be similar with those analysed in meat and sea foods. In spite of large number of available data in scientific publications on EI, there has been very limited data availabile on their respective EI toxicological characteristics used in diet from both traditional and commercial way. Gao et al. (2018) investigated and reported that only under 34 EI species, belongs to Lepidoptera, Diptera, Hemiptera, Coleoptera, Hymenoptera, Orthoptera, Blattodea, Amorphosceloidea has been assessed thoroughly for heterogenous toxicological studies which can be used in future for further assessment.
Safety regulations
EI still lack its position as food under Codex Alimentarius, an international guideline for food safety. Currently insects are referred as foreign bodies or impurities in the Codex Alimentarius. Regulations over the world strongly varies between all the countries and most of the western countries implicitly includes insects. Thus the establishment of global market for EI has been hindered and slowed down because of this differeing non-standardized legislations throughout the world. In European Union implicated the insect food under Novel food (Regulation 2283/2015) and the existing Regulations 2468/2017 and 2469/2017 explained and orchestrated standards concerning insect food, thus far, its perspective on legal aspects should be accounted as ‘gray area’. These days, EI are without a doubt considered to be 'novel food sources'. In effect, Regulation 2283/2015 indicates that classifications of food which comprise under novel food varieties encloses both entire EI and its parts. From 1st January of 2018, insects along with insect oriented foods should be approved prior to being put available and the system requires in any event 17 months. Nevertheless, the guideline offers a worked-on approval method for novel food varieties that are fresh for the business sectors of European Union, however had been customarily utilized in three nations. Despite the consumption of insects as a traditional food among the regions of South America and SouthEast Asia, there is still no regulations for the processing of EI and their products.
Conclusion
Insect consumption has always been a pre-historic food habits in world wide practice. Tremendous researchwork and progressive development in the area of EImade it to be a very promising alternative source of food and feed. Further research on its safety and regulation frameworks will further decrease the neophobia and increase the consumption of EI which is not just good for healthly diet but also for the world. EI will be an economically cheaper source of proteins, since it requires less capital investment with minimal techniqual knowledge thus farming and rearing of EI should be encouraged and promoted as a social inclusive activity. The review demonstrates the insects as a whole food category amdon the basis of these research considerations. Emerging and innovative food processing technologies must be explored toinorder to endorse entomophagy, formulation of various functional ingredients into ready to eat snackablesas a whole and identifiable forms. For such, protein concentrates extracted from EI with high foaming, increased solubility and better emulsifying properties, in addition to other techno-functional charecteristics. Insects are predominent source of polyunsaturated fatty acid content and has increased ω´-3/ω´-6 ratio. The chitin, one more fuctional compound found in chemical composition of EI found to be in similar with shrimp, making them an attainable and supportable choice for supplanting some presently accessible functional foods. These perspectives ought to be a focal point of future examination and innovative turn of events.
References
Aldrich JR (1988) Chemical ecology of the Heteroptera. Annu Rev Entomol 33(1):211–238. https://doi.org/10.1146/annurev.ento.33.1.211
Alegria C, Pinheiro J, Gonçalves EM, Fernandes I, Moldão M, Abreu M (2010) Evaluation of a pre-cut heat treatment as an alternative to chlorine in minimally processed shredded carrot. Innov Food Sci Emerg Technol 11(1):155–161
Alves AV, de Lima FF, da Silva TG, de Oliveira VS, Kassuya CAL, Sanjinez-Argandona EJ (2019) Safety evaluation of the oils extracted from edible insects (Tenebrio molitor and Pachymerus nucleorum) as novel food for humans. Regul Toxicol Pharmacol 102:90–94
Antonietta B (2020) Edible insects: An overview on nutritional characteristics, safety, farming, production technologies, regulatory framework, and socio-economic and ethical implications. Trends Food Sci Technol 100:35–50. https://doi.org/10.1016/j.tifs.2020.03.040
Bußler S, Rumpold BA, Jander E, Rawel HM, Schlüter OK (2016) Recovery and techno-functionality of flours and proteins from two edible insect species: meal worm (Tenebrio molitor) and black soldier fly (Hermetia illucens) larvae. Heliyon 2(12):e00218
Chakravorty J (2014) Diversity of edible insects and practices of entomophagy in India: an overview. J Biodivers Biopros Dev 1(3):124
Choi BD, Wong NA, Auh JH (2017) Defatting and sonication enhances protein extraction from edible insects. Korean J Food Sci Animal Res 37(6):955
Churchward-Venne TA, Pinckaers PJ, van Loon JJ, van Loon LJ (2017) Consideration of insects as a source of dietary protein for human consumption. Nutrit Rev 75(12):1035–1045
Collavo ALBERTO, Glew RH, Huang YS, Chuang LT, Bosse REBECCA, Paoletti MG (2005) House cricket small-scale farming. Ecological Implications of Minilivestock: Potential of Insects, Rodents, Frogs and Snails 27:515–540
da Silva L, Andressa J, Eliézer QO, Helena Leão GC, Héctor ML, Carolina Dias MS, Carlos P (2021) Extraction, physicochemical characterization, and morphological properties of chitin and chitosan from cuticles of edible insects. Food Chem 343:128550. https://doi.org/10.1016/j.foodchem.2020.128550
Deka B, Tomberlin JK, Babu A (2021) Insects as food and feed: a promising agricultural sector with special reference to India. J Insects Food Feed 7(4):471–482. https://doi.org/10.3920/JIFF2020.0131
Donovan SE, Jones DT, Sands WA, Eggleton P (2000) Morphological phylogenetics of termites (Isoptera). Biol J Lin Soc 70(3):467–513
Finke MD, Santos R, Nanna R, van Huis Arnold, Yen AL (2015) The European Food Safety Authority scientific opinion on a risk profile related to production and consumption of insects as food and feed.". J Ins Food and Feed 1(4):245–247
Frigerio J, Giulia A, Andrea G, de Mattia Fabrizio, Massimo L, Antonia B (2020) Tasting the differences: Microbiota analysis of different insect-based novel food.". Food Res Int. 137:109426. https://doi.org/10.1016/j.foodres.2020.109426
Gao Y, Wang D, Xu ML, Shi SS, Xiong JF (2018) Toxicological characteristics of edible insects in China: a historical review. Food and Chem Toxicol 119:237–251
Garofalo C, Vesna M, Federica C, Lucia A, Francesca C, Andrea O (2019) Current knowledge on the microbiota of edible insects intended for human consumption: a state-of-the-art review. Food Res Int 125:108527. https://doi.org/10.1016/j.foodres.2019.108527
Gautreau M, Marc R, Kevin S, Weisberg Stacy N (2017) Familial anaphylaxis after silkworm ingestion.". Prehospital Emergency Care. 21(1):83–85. https://doi.org/10.1080/10903127.2016.1204035
Ghosh S, Jung C, Meyer-Rochow VB (2018) What governs selection and acceptance of edible insect species? Edible insects in sustainable food systems. Springer, Cham, pp 331–351
Grabowski NT, Günter K (2017) Microbiology of processed edible insect products–results of a preliminary survey. Int J Food Microbiol 243:103–107. https://doi.org/10.1016/j.ijfoodmicro.2016.11.005
Hahn T, Roth A, Ji R, Schmitt E, Zibek S (2020) Chitosan production with larval exoskeletons derived from the insect protein production. J Biotechnol 310:62–67
Janssen RH, Vincken JP, Arts NJ, Fogliano V, Lakemond CM (2019) Effect of endogenous phenoloxidase on protein solubility and digestibility after processing of Tenebrio molitor, Alphitobius diaperinus and Hermetia illucens. Food Res Int 121:684–690
Jesse S, Vandeweyer D, Crauwels S, Verreth C, Boeckx H, Van Der Borght M, Claes J, Lievens B, Van Campenhout L (2017) Minced meat-like products from mealworm larvae (Tenebrio molitor and Alphitobius diaperinus): microbial dynamics during production and storage. Innov Food Sci Emerg Technol 41:1–9. https://doi.org/10.1016/j.ifset.2017.02.001
Jiang Y, Yifan Z, Yuanrong Z, Zhenmin L, Yu Z, Yun D, Yanyun Z (2021) Effects of salting-in/out-assisted extractions on structural, physicochemical and functional properties of Tenebrio molitor larvae protein isolates. Food Chem 338:128158. https://doi.org/10.1016/j.foodchem.2020.128158
Joo-Hyoung C, Zhao H-L, Kim J-S, Kim S-H, Chung C-H (2018) Characteristics of fermented seasoning sauces using Tenebrio molitor larvae. Innov Food Sci Emerg Technol 45:186–195. https://doi.org/10.1016/j.ifset.2017.10.010
Kamau E, Mutungi C, Kinyuru J, Imathiu S, Tanga C, Affognon H, Fiaboe KKM (2018) Moisture adsorption properties and shelf-life estimation of dried and pulverised edible house cricket Acheta domesticus (L.) and black soldier fly larvae Hermetia illucens (L.). Food Res Int 106:420–427
Kaur, N., W. P. Jessie, C. S. Sullivan, D. L. Walenta, E. C. Verhoeven, and N. P. Anderson. "Evaluation of bifenthrin resistance in field-collected clover seed weevils." NP Anderson, AG Hulting, DL Walenta, and CA Mallor-Smith (eds.) (2019).
Kaya M, Talat B, Sevil E, Ayfer M, Meltem AÖ, Yavuz SÇ (2014) Physicochemical comparison of chitin and chitosan obtained from larvae and adult Colorado potato beetle (Leptinotarsa decemlineata). Mater Sci Eng, C 45:72–81. https://doi.org/10.1016/j.msec.2014.09.004
Kaya M, Sevil E, Abbas M, Talat B (2015) Comparison of chitin structures isolated from seven Orthoptera species. Int J Biol Macromol 72:797–805. https://doi.org/10.1016/j.ijbiomac.2014.09.034
Khampakool Apinya, Soisungwan Salinee, You SangGuan, Park Sung Hee (2020) Infrared assisted freeze-drying (IRAFD) to produce shelf-stable insect food from Protaetia brevitarsis (white-spotted flower chafer) larva.". Food Sci Animal Res 40(5):813
Kim M-W, Song Yong-Su, Han Yeon Soo, Jo Yong Hun, Choi Myung Hyo, Park Young-Kyu, Kang Seung Ho, Kim Sun-Am, Choi Changyong, Jung Woo-Jin (2017) Production of chitin and chitosan from the exoskeleton of adult two-spotted field crickets (Gryllus bimaculatus). Entomol Res 47(5):279–285. https://doi.org/10.1111/1748-5967.12239
Kim HW, Setyabrata D, Lee Y, Jones OG, Kim YHB (2017) Effect of house cricket (Acheta domesticus) flour addition on physicochemical and textural properties of meat emulsion under various formulations. Journal of Food Sci 82(12):2787–2793
Kim T-K, Yong Hae In, Kim Young-Boong, Kim Hyun-Wook, Choi Yun-Sang (2019) Edible insects as a protein source: a review of public perception, processing technology, and research trends.". Food Sci Animal Res 39(4):521
Kim T-K, Yong Hae In, Chun Ho Hyun, Lee Mi-Ai, Kim Young-Boong, Choi Yun-Sang (2020) Changes of amino acid composition and protein technical functionality of edible insects by extracting steps. J Asia-Pacific Entomol 23(2):298–305. https://doi.org/10.1016/j.aspen.2019.12.017
Kinyuru JN, Mogendi JB, Riwa CA, Ndung’u NW (2015) Edible insects—a novel source of essential nutrients for human diet: learning from traditional knowledge. Animal Front 5(2):14–19
Kröncke N, Böschen V, Woyzichovski J, Demtröder S, Benning R (2018) Comparison of suitable drying processes for mealworms (Tenebrio molitor). Innov Food Sci Emerg Technol 50:20–25
Kurdi P, Patspon C, Jirathit W, Chanida H (2021) Studies on microbial quality, protein yield, and antioxidant properties of some frozen edible insects. Int J Food Sci. https://doi.org/10.1155/2021/5580976
Lange KW, Yukiko N (2021) Edible insects as future food: chances and challenges. J Fut Foods 1(1):38–46. https://doi.org/10.1016/j.jfutfo.2021.10.001
Laroche M, Véronique P, Alice M, Alexia G, Julien C, Alain D (2019) Comparison of conventional and sustainable lipid extraction methods for the production of oil and protein isolate from edible insect meal. Foods 8(11):572
Lawrence JF., Adam Ś, Ainsley E. Seago, Margaret K. Thayer, Alfred F. Newton, and Adriana E. Marvaldi. "Phylogeny of the Coleoptera based on morphological characters of adults and larvae." In Annales zoologici, vol. 61, no. 1, pp. 1–217. Museum and Institute of Zoology, Polish Academy of Sciences, 2011. https://doi.org/10.3161/000345411X576725.
Leni GL, Soetemans J, Jacobs S, Depraetere N, Gianotten L, Bastiaens A. Caligiani, Sforza S (2020) Protein hydrolysates from Alphitobius diaperinus and Hermetia illucens larvae treated with commercial proteases. J Ins Food and Feed 6(4):393–404
Liceaga AM (2021) Processing insects for use in the food and feed industry. Current Opinion in Insect Science 48:32–36
Liya Yi, Van Boekel Martinus AJS, Boeren Sjef, Lakemond Catriona MM (2016) Protein identification and in vitro digestion of fractions from Tenebrio molitor.". European Food Res Technol 242(8):1285–1297. https://doi.org/10.1007/s00217-015-2632-6
Luca F, Cardazzo B, Carraro L, Fontana F, Novelli E, Balzan S (2018) Edible processed insects from e-commerce: Food safety with a focus on the Bacillus cereus group. Food Microbiol 76:296–303. https://doi.org/10.1016/j.fm.2018.06.008
Mahmoudikordi F, Mohammad B, and Hamid-Reza A (2021) Optimization of ethanol assisted aqueous oil extraction from a cicadidae Sp. bioRxiv. https://doi.org/10.1101/2021.01.24.427958
Megido RC, Christine P, Marjorie E, Marisa L, Christophe B, Sabine D, Eva T et al (2018) Effect of household cooking techniques on the microbiological load and the nutritional quality of mealworms (Tenebrio molitor L. 1758). Food Res Int 106:503–508. https://doi.org/10.1016/j.foodres.2018.01.002
Mintah BK, Ronghai H, Agyekum Akwasi A, Mokhtar D, Golly Moses K, Haile M (2020) Edible insect protein for food applications: Extraction, composition, and functional properties. J Food Process Eng 43(4):e13362. https://doi.org/10.1111/jfpe.13362
Mitsuhashi J (2016) Edible insects of the world. CRC Press. https://doi.org/10.1201/9781315367927
Musundire R, Osuga Isaac M, Xavier C, Janet I, Baldwyn T (2016) Aflatoxin contamination detected in nutrient and anti-oxidant rich edible stink bug stored in recycled grain containers. PloS one. 11(1):e0145914. https://doi.org/10.1371/journal.pone.0145914
Nadeau L, Isaac N, Frank F, Florence D (2015) The potential for entomophagy to address undernutrition. Ecol Food Nutr 54(3):200–208. https://doi.org/10.1080/03670244.2014.930032
Ng SH, Stat M, Bunce M, Simmons LW (2018) The influence of diet and environment on the gut microbial community of field crickets. Ecology and Evolution 8(9):4704–4720
Nongonierma AB, FitzGerald RJ (2017) Unlocking the biological potential of proteins from edible insects through enzymatic hydrolysis: a review. Innov Food Sci Emerg Technol 43:239–252
Ophelia D, Reade B, Spence C (2015) The insectivore’s dilemma, and how to take the West out of it. Food Qual Prefer 44:44–55. https://doi.org/10.1016/j.foodqual.2015.02.007
Otero P, Alba G-D, Joaquin N, Guillermo R, Diana M (2020) Extracts from the edible insects acheta domesticus and tenebrio molitor with improved fatty acid pro fi le due to ultrasound assisted or pressurized liquid extraction”. Food Chemistry 314:126200. https://doi.org/10.1016/j.foodchem.2020.126200
Park YS, Choi YS, Hwang KE, Kim TK, Lee CW, Shin DM, Han SG (2017) Physicochemical properties of meat batter added with edible silkworm pupae (Bombyx mori) and transglutaminase. Korean J Food Sci Animal Res 37(3):351
Patil AR, Gagan D, Meenatchi R, Moses JA, Bhuvana S (2019) Extraction and characterization of silkworm pupae (Bombyx mori) oil by LC-MS/MS method. Int. J. Pure App. Biosci. 7(3):503–509
Poma G, Matthias C, Elvio A, Chiara C, Jean FF, Adrian C (2017) Evaluation of hazardous chemicals in edible insects and insect-based food intended for human consumption. Food Chem Toxicol 100(70–79):70–79. https://doi.org/10.1016/j.fct.2016.12.006
Purschke B, Henrik B, Rafaela S, Henry J (2018) Effect of pre-treatment and drying method on physico-chemical properties and dry fractionation behaviour of mealworm larvae (Tenebrio molitor L.). Eur Food Res Technol 244(2):269–280. https://doi.org/10.1007/s00217-017-2953-8
Raheem D, Conrado C, Oluwatoyin BO, Nieuwland Maaike, Saraiva Ariana, Millán Rafael, Raposo António (2019) Traditional consumption of and rearing edible insects in Africa, Asia and Europe.". Critical Rev Food Sci Nutrition 59(14):2169–2188. https://doi.org/10.1080/10408398.2018.1440191
Ramos-Elorduy J (2005) Insects: a hopeful food source. Ecological implications of minilivestock. 263–291
Ribeiro JC, Luís MC, Bernardo S-P, João F (2018) Allergic risks of consuming edible insects: a systematic review. Mol Nutr Food Res 62(1):1700030
Rumpold BA, Schlüter Oliver K (2013) Nutritional composition and safety aspects of edible insects. Molec Nutrit Food Res. 57(5):802–823. https://doi.org/10.1002/mnfr.201200735
Santhosh R, Yadav BK, Rawson A (2019) Optimization of protein extraction from yellow mealworm larvae. Int J Chem Stud 7(3):4577–4582
Selaledi L, Monnye M (2021) The influence of drying methods on the chemical composition and body color of yellow mealworm (Tenebrio molitor L.). Insects. 12(4):333. https://doi.org/10.3390/insects12040333
Seo M, Hwa JL, Joon HL, Minhee B, In-Woo K, Sun YK, Jae-Sam H, Mi K (2019) A study of the anti-inflammatory effect of protein derived from tenebrio molitor larvae. J Life Sci 29(8):854–860
Shin C-S, Do-Yeong K, Weon-Sun S (2019) Characterization of chitosan extracted from Mealworm Beetle (Tenebrio molitor, Zophobas morio) and Rhinoceros Beetle (Allomyrina dichotoma) and their antibacterial activities. Int J Biol Macromol 125:72–77. https://doi.org/10.1016/j.ijbiomac.2018.11.242
Singer MC, Parmesan C (2010) Phenological asynchrony between herbivorous insects and their hosts: signal of climate change or pre-existing adaptive strategy? Ann Rev Entomol 365(1555):3161–3176
Smets R, Peter G, Claes Johan, Van Der Borght Mik (2021) Optimisation of the lipid extraction of fresh black soldier fly larvae (Hermetia illucens) with 2-methyltetrahydrofuran by response surface methodology.". Separ Purific Technol. 258:118040. https://doi.org/10.1016/j.seppur.2020.118040
Srinivas G, Nidoni U, Ramachandra CT, Ramappa KT, Ashoka J (2019) Supercritical fluid extraction of pupae oil from mulberry silkworm (Bombyx mori L.). J Pharmac Phytochem 8(3):4507–4513
Tzompa-Sosa Daylan Amelia, Yi L, Van Valenberg HJF, Lakemond CMM (2019) Four insect oils as food ingredient: physical and chemical characterisation of insect oils obtained by an aqueous oil extraction. J Ins Food and Feed 5(4):279–292. https://doi.org/10.3920/JIFF2018.0020
Ugur AE, Bolat B, Oztop MH, Alpas H (2021) Effects of high hydrostatic pressure (HHP) processing and temperature on physicochemical characterization of insect oils extracted from Acheta domesticus (house cricket) and Tenebrio molitor (yellow mealworm). Waste and Biomass Valoriz 12(8):4277–4286
Vandeweyer D, Lenaerts S, Callens A, Van Campenhout L (2017) Effect of blanching followed by refrigerated storage or industrial microwave drying on the microbial load of yellow mealworm larvae (Tenebrio molitor). Food Control 71:311–314
Verhoeckx KC, van Broekhoven S, den Hartog-Jager CF, Gaspari M, de Jong GA, Wichers HJ, Knulst AC (2014) House dust mite (Der p 10) and crustacean allergic patients may react to food containing Yellow mealworm proteins. Food and Chem Toxicol 65:364–373
von Dohlen CD, Moran NA (1995) Molecular phylogeny of the Homoptera: a paraphyletic taxon. J Molec Evol 41(2):211–223
Wang H, Kashif Ur R, Weijian F, Dan Y, Rashid R, Minmin C, Jibin Z, Ziniu Y, Longyu Z (2020) Physicochemical structure of chitin in the developing stages of black soldier fly.". Int J Biolog Macromolec 149:901–907
Williams JP, JR Williams, Kirabo A, Chester D, Peterson M. Nutrient content and health benefits of insects. In: Insects as sustainable food ingredients, pp. 61–84. Academic Press, 2016. https://doi.org/10.1016/B978-0-12-802856-8.00003-X
Wynants E, Lotte F, Sabine van Miert A, Geeraerd J. Claes, Van Campenhout L (2019) Risks related to the presence of Salmonella sp. during rearing of mealworms (Tenebrio molitor) for food or feed: Survival in the substrate and transmission to the larvae. Food Control 100:227–234. https://doi.org/10.1016/j.foodcont.2019.01.026
Funding
This study has been supported by Indian Institute of Technology Kharagpur, West Bengal.
Author information
Authors and Affiliations
Contributions
Gnana Moorthy Eswaran U implemented the idea, collected the literature, analyzed the content, and wrote the initial manuscript draft; Sangeetha Karunanithi, Rakesh Kumar Gupta, Srutee Rout contributed to improving the initial draft and reviewed the manuscript, and Prem Prakash Srivastav conceived the idea, supervised the work and formatted the manuscript appropriately for submission.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The work described has not been published before and it is not under consideration for publication elsewhere. The submission to JFST publication has been approved by all authors as well as the responsible authorities at the institute where the work has been carried out. If accepted, it will not be published elsewhere in the same form, in English or in any other language, including electronically without the written consent of the copyright holder. JFST will not be held legally responsible should there be any claims for compensation or dispute on authorship.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gnana Moorthy Eswaran, U., Karunanithi, S., Gupta, R.K. et al. Edible insects as emerging food products–processing and product development perspective. J Food Sci Technol 60, 2105–2120 (2023). https://doi.org/10.1007/s13197-022-05489-y
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-022-05489-y