Abstract
Rice products, including those given to infants, could be naturally polluted with arsenic. This issue for all age groups should be a top priority for the world food industry and the public. Food regulators assume incorrectly that infants’ food and other rice products are safe, and health, agriculture and commerce authorities follow no clear guidelines. A common measure has been to place a ML on the amount of iAs in white rice and food intended for children and pregnant women. Although oAs is less toxic than iAs, it is still toxic; consequently, the ML of arsenic for the different age groups should be also specified. However, the ML of iAs in polished white rice for infants is very low (100 μg/kg for infants and 200 μg/kg for adults) and is difficult to measure. Using neutron activation for research is very useful in improving safety standards in the food industry. The second purpose of this review study is to report on the experimental results and methods used for measurements adopted at the Delft Reactor in the Netherlands with a colleague of the quantity of arsenic in 21 samples of different rice products from a variety of brands.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The process of producing safe and clean rice for both local and international food industries is extremely demanding. Rice’s extraordinary ability to react with arsenic and absorb it from the soil and irrigation water is more pronounced compared to other related plants. Furthermore, brown rice can be polluted more heavily than white rice and should, therefore, be avoided in the manufacturing of baby food and foods targeted at pregnant women and be forbidden in the baby food industry generally.
Although arsenic is a catalytic element, it is also very hazardous. It exists everywhere and could be considered the second-most hazardous element after mercury in nature. Further, when it enters the body or industrial activities, it stays there. The main types of arsenic in the rice plant are iAs and oAs. All rice products worldwide will have some amount of arsenic. Even if no arsenic is found in rice samples, measurements should be repeated or also be taken for mercury and cadmium.
One of the causes of the differences between the level of oAs in rice between Asia and the USA is the African rice (Oryza glaberrima), which was brought to the USA several centuries ago and has been interbred with Asian species of rice. “The grain is much like common rice, although the husk around it is usually red” (Ruskin 1996)Footnote 1 or brown. The red color of the African rice (Oryza glaberrima) bran and some Asian rice bran indicates the existence of hazardous materials and chemical poisons. As a result, more studies are needed to measure the amount of arsenic, cadmium, bromine, iodine, mercury, and selenium in the rice husk separated from the seed.
According to the [FAO/WHO] (2015)Footnote 2, during CCCF8, some countries found it difficult to measure iAs in polished rice. It was, thus, agreed that the following footnote be inserted into the Standard: “Countries or importers may decide to use their own screening when applying the ML for As-in in rice by analysing total arsenic (As-tot) in rice. If the As-tot concentration is below the ML for As-in, no further testing is required and the sample is determined to be compliant with the ML. If the As-tot concentration is above the ML for As-in, follow-up testing shall be conducted to determine if the As-in concentration is above the ML”.
This footnote is quoted in a great many scientific papers and legal documents without explaining how the objective expressed therein is best achieved. The present work aims to fill this gap. I collected all possible forms of arsenic components in rice (Table 1). when you measure their (= forms of arsenic) amounts in rice you will have different values for the same samples depending on the used instrument.
Forms of arsenic
Some clarification is needed here, and the results are also highly dependent on the instruments used:
“Different arsenic-containing compounds vary substantially in their toxicity to mammals. Arsine gas (AsH3) is clearly the most toxic, followed in order of generally decreasing toxicity by inorganic trivalent compounds, organic trivalent compounds, inorganic pentavalent compounds, organic pentavalent compounds, and, finally, elemental arsenic”(Discussant et al. 1988)Footnote 3.
“Demethylation of methylarsonic acid DMA and dimethylarsinic acid MMA from rice occurs rise to the amount of ‘iAs’ species available for metabolism in cells” (González De Chávez Capilla 2018)Footnote 4. Since “DMA’s and MMA’s relatively short half-lives in soil” (Jackson and Punshon 2015b)Footnote 5 are not stable components, they are transformed into other forms of arsenic and the new forms are released in the surrounding places. Like mercury, arsenic does not disappear and keeps existing in the environment, including inside the body.
From Fig. 1, it is clear that DMA is unstable. “Arsenosugars are biotransformed by humans to at least 12 arsenic metabolites” (Francesconi et al. 2002)Footnote 6. Arsenobetaine (C5H11AsO2) is a form of arsenic, which is found in crustaceans and fish and not rice (Nordberg and Fowler 2019)Footnote 7.
Relationship between arsenic species and sum of total Arsenic species (Liang et al. 2010a)
From different sources, the concentration of Arsenic in the human body is between 80 and 250 μg/kg, where the total toxic level is 100 μg/kg; nails: 430–1080 μg/kg; urine 5–40 μg /day.
The problem with iAs and oAs is the difference between the medical analyses in vitro and vivo. When measuring the form of arsenic outside the human body, it is inorganic, but inside the body, it is mostly organic. Arsenic is also able to accumulate both in animals and humans. The best places to measure its presence are in the kidney, liver, prostate, and brain. See Bashir et al. (2006) and Ghosh et al. (2007).Footnote 8,Footnote 9
In practice, the measurements of iAs in urine are not necessarily accurate because an unknown quantity of arsenic will pass through the heart, liver, prostate, kidneys, and fat. A widespread contradictory notion implies that arsenic is not a bio-accumulator in the body like mercury and cadmium because of its ability to react. Only hair, fat, and nails can preserve it over a long period.
The analytical equivalence of the tAs and arsenic fractionation in urine was evaluated by some studies (Hackenmueller and Strathmann 2014).Footnote 10 The quantitative agreement was determined using the tAs concentration and the sum of quantified arsenic species in the fractionation assay.
When existing in food, iAs is considered the most toxic compared to the other two forms. “Arsenic is known to accumulate in rice at higher levels than in other crops, and is estimated to absorb up to 10 times the amount of other grains.” ([BNF] 2017).Footnote 11 It has higher levels of iAs because, as the rice plant grows, the plant and grain tend to absorb arsenic from the environment more than other crops. Maybe, this is because of the silica (SiO2) in the husk (Fig. 2). “Rice husk contains about 30–50% of organic carbon. In the course of a typical milling process, the husks are removed from the raw grain to reveal whole brown rice which upon further milling to remove the bran layer will yield white rice. Rice husk constitutes about 20% of the weight of rice and its composition is as follows: cellulose (50%), lignin (25–30%), silica (15–20%), and moisture (10–15%).Every 100 kg of husks burnt in a boiler for example will yield about 25 kg of RHA”. (Singh 2018).Footnote 12
a Spatial location of mercury and arsenic species in the rice grain as identified by synchrotron XRF (Jackson and Punshon 2015a); b Vacuolar sequestration of arsenic to reduce, from root-to-shoot, the amount of arsenic in the grain (Deng et al. 2018); c Vitamins and minerals are in the bran of the raw paddy rice ([RISO] 2020); d rice processing ([IRRI] 2002)
Misunderstanding the issue of arsenic in rice
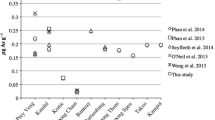
Most studies on arsenic in rice have not differentiated between black, brown and white rice, and have declared that all infants’ food containing rice and other rice products is safe, seeFootnote 13 (Karagas et al. 2016). Rice can “contain very high levels of non-threshold carcinogen iAs, at concentrations of around 1 mg/kg” (Signes-Pastor et al. 2017).Footnote 14 While rice from California, Louisiana and Texas has more oAs compared to that from the West Bengal Delta (Bangladesh and East India) and although Americans consume less cooked rice, they consume more rice ingredients. The most polluted places are Inner Mongolia, France, the USA, “Hunan, Anhui and Guangxi provinces in China” (Liang et al. 2010a, b, c),Footnote 15 and the Atacama Desert in Chile.
As shown in (Tables 2, 3), iAs/tAs is higher when rice crackers are made from the husk of brown or black rice because they are cheap and have a cacao-like color. Consequently, the color and the degree of rice peeling are important. “The daily intake of total arsenic from food and beverages is generally between 20 and 300 µg/day” (Panigrahi et al. 2016).Footnote 16
More experiments on rice recipes that include fried rice were conducted in a study by CZ Wei (2018).Footnote 17
Chung et al. (2014)Footnote 18 stated that, in Hong Kong, “the percentages iAs in rice noodles (64%) and plain steamed rice-rolls (one of the not mentioned mixed dishes) (65%) are similar to those of cooked white rice (48%) and unpolished rice (64%). Hence, the cooking process, including steaming, baking and deep frying, could only reduce a small portion of arsenic from the inorganic to the organic form. Therefore, cereal and their products become the main source of dietary intake of iAs for different countries”his means that the amount of iAs is not reduced enough to be considered safe.
However, some words have not been well defined, including “cooked,” “boiled,” “fried,” “stir-fried” and “a mix of rice products.” Accordingly, counter-interpretations exist. Another commonly-made error is taking into account only iAs rather than both iAs and oAs, that is, the tAs.
Additional intake of arsenic in the rice husk originates from various contaminated meat and poultry products where binders are permitted (e.g., hot dogs, meatballs, and chicken patties), and rice bran is set not to exceed 3.5% of this product formulation. “The total daily intake of arsenic from food and beverages is generally within the range of 20–300 μg/day” ([IARC] 2012).Footnote 19 Physicians usually advise patients with diabetes to consume brown rice instead of white rice to decrease the level of sugar in the blood. In this case, the treatment is counterproductive for patients’ overall health.
The problem with rice husks
Polished rice contains less iAs than husked rice because polishing removes the bran layer, which contains most of the iAs. Thus, husked rice polished at a higher polishing rate results in polished rice with lower arsenic concentrations. However, the consumption of husked rice carries risks and benefits ([FOW/WHO] 2017).Footnote 20 More minerals are beneficial if they are not polluted. Special treatments can extract arsenic, silver and silica. When the rice husk is burned in a filtered oven, for instance, minerals, including arsenic, can be extracted safely without escaping into the atmosphere. Otherwise, they can escape into the environment. A study from Yao et al. (2020)Footnote 21 on a number of different elements showed that the median of arsenic in whole rice across China is 158 µg/kg, in rice bran 628 µg/kg, and in polished rice 98 µg/kg. The bran-polished rice ratio is 6.7 and the element contents loss is 44%; only approximately 10% of rice bran was milled to powder form.
Increasing cell vacuolar sequestration can prevent arsenic translocation into the rice seeds in the food chain. Deng et al. (2018)Footnote 22 revealed in their research reduced root-to-shoot and internode-to-grain translocation in transgenic rice plants, resulting in a 70% decrease in arsenic accretion in the brown rice.
However, the vague definition of “polished rice” results in inconsistent measurements of arsenic in husked and polished rice grains. Polished rice should be husk-free; that is to say, all bran has been completely removed. This is important in the food industry and for infants’ and pregnant women’s foods, where water is often added to a powdered mixture of several types of ingredients without rinsing these first.
In an Indian study, “520 samples of husked rice were analyzed over a period of three months; iAs was detected in 513 samples (98.7%)”. In another set of experiments, “the highest levels of iAs were detected in husked rice samples were from Canada (340 μg/kg), China (570 μg/kg), the EU (550 μg/kg), India (290 μg/kg), Japan (590 μg/kg), R. Korea (260 μg/kg), Thailand (390 μg/kg), and the USA (250 μg/kg).”([FAO/WHO] 2016).Footnote 23
In spite of abundant evidence, many countries, including those mentioned in the introduction, do not feel compelled to enforce strict controls. For this reason, the delegation of FAO/WHO noted that there would be a need to revise the Codes of Practice, specifically the so-called occurrence data, later in 2022 ([FAO/WHO] 2019).Footnote 24
Arsenic in rice products
People ingest arsenic from food chains, such as rice with fish, poultry products, and vegetables as demonstrated in Table 4.
One of the missing regulations in Table 4 is found in the food regulation in Hong Kong ([GLD] 2018); rice, which could be polluted with arsenic, and fish, which could be polluted with mercury are the main foods in Asia, including Hong Kong. The ML (tAs) for rice bran oil (Edible fats and oils, other than fish oil) is 100 μg/kg for adults.
Rice bran extract is used in the aqueous mixture of sunflower lecithin ([USDA-FSIS] 2012)Footnote 25. Husked rice (black or brown) has more arsenic in it, so that the ML should be less rather than more, namely 200/1.6 = 125 μg/kg. In many cases, the level of iAs could far exceed the ML. For that measuring the tAs is more practical and more economical. This can be done by control or the health authorities as an initial standard measure. If the sample exceeds the ML of 200 μg/kg, then fractional analysis can be carried out.
Based on Table 5, brown rice has almost 67% more iAs compared to white rice. In spite of using the weighted mean method for this problem, the percentage in some locations could be much higher.
Some arsenic will be absorbed into the rice seed by the reactions of starch arsenic (III) or arsenic (V) ions to form arsenic starches. There is no need for high temperatures to achieve this; arsenic can react at hot-weather conditions (30–50 °C) and even at room temperature. Therefore, the control of movement of rice starch in food industries should be a priority for health agencies. More specifically, white rice products should be more tightly controlled, whereas black and brown rice and their starches should be forbidden or restricted.
Precooking or percolating rice
The collective problems FAO/WHO seek to solve were appointed by (Rahman 2008):Footnote 26 “In Bangladesh, for example, while cooking, people using the rice stalks, contaminated more than drinking waters with arsenic, breathe the produced fumes, Agencies there monitor arsenic in drinking water without risk evaluation from topsoil contamination.”. The situation did not improve much at the end of 2021.
Cooked and parboiled rice (also called “converted rice”) is a rice type that has been partially boiled in the husk. The three basic steps of parboiling are soaking, steaming, and drying. High concentrations of total arsenic exist in rice grains, rice-based products, bran and germ. Depending on the method of food processing, temperature and time, changes in the tAs concentrations and arsenic species may occur. “The arsenic content in cooking water seems to be of special importance because it determines whether the arsenic concentrations in the prepared food may be higher or lower compared to the raw product.” ([EFSA/CONTAM] 2009).Footnote 27 “Cooking rice in excess water efficiently reduces the amount of As in the cooked grain. Excess water cooking reduces average inorganic As by 40% from long grain polished, 60% from parboiled and 50% from brown rice”.Footnote 28 (Gray and Conklin 2016). This method may also remove some key nutrients. The efficacy of removing arsenic according to different data for this method is 10–96%, but it is not accurate because a need will arise to calibrate the temperature and cooking time (in some tests, researchers spent five minutes boiling the water, after which they discarded the polluted water). At the same time, this method suggested by the FDA and other Western agencies is not suitable for the preparation of infants’ food, which is a mix of different products, including milk and oils. The method of percolating rice has been used in several studies, including in one from Signes-Pastor et al. (2017), which aimed at minimizing the iAs content in rice bran. The bran was percolated in arsenic-free water. The researchers observed up to a 96% removal of iAs for a range of rice bran products, with the quantity of iAs removed related to the volume of cooking water used. In many countries, however, water is not freely available. More concerningly, water in Nepal, Bangladesh and eastern India contains relatively high amounts of arsenic. The main source of arsenic apparently from a lot of data is the mountains of Nepal. In addition, this process reduces the content of beneficial minerals, such as copper, potassium, and phosphorus content. Therefore, the process of repeatedly washing rice or doing so with large quantities of water is either not practical or is self-defeating. What is needed is arsenic-free water, which is not available even for drinking in many countries.
It was shown in a study by Püssa (2013)Footnote 29 that by “Using an in vitro gastrointestinal simulator, it was shown that the bioavailability of arsenic from cooked rice was 37 and 58% at a total concentration of arsenic in rice of 0.389 and 0.314 mg/kg, respectively.In addition, humans assimilate daily about 10 μg of arsenic from drinking water and < 1 μg from the air”.
Rice bran oil
Rice Bran Oil, as a valuable by-product of the rice industry, is extracted from the inner husk of rice, and is popular in Asian cuisine. It was thought that PRO is one of the healthiest edible oils. However, there is little data on the amount of iAs and oAs as well as the tAs in PRO. Arsenic in oil is mostly organic, though this requires confirmation through further research.
In a study by Bakota et al (2015)Footnote 30 “Levels of (total) arsenic, lead, mercury, zinc, and cadmium were investigated in brands of rice bran oil. Arsenic and mercury concentrations differed between the methods used to collect the data. In the case of lead and cadmium, concentrations were extremely low. In the case of zinc, levels were higher but likely do not pose a toxicity threat. Arsenic and mercury were found above permissible UN FAO limits by in-house testing but not via independent analyses, indicating that further work is needed to determine the best method for analysis of these metals in edible oils. The toxicity of rice-derived food products containing arsenic should be further explored, and the possibility of establishing maximum acceptable levels of heavy metals for a variety of food products should be considered”.
Furthermore, PCBs are pollutants that were found to be the cause of Yusho disease. In one such case in western Japan, Kanemi rice oil was contaminated with PCB, (Schecter and Masuda 2012);Footnote 31 (Suzuki and Kobayashi 1968).Footnote 32 The contaminated RBO was sold as a feed supplement to consumers for use in cooking. About 14,000 people in Japan who consumed the contaminated rice oil were affected with various ailments.
In addition, there were cases of children with poor cognitive development. An almost identical case occurred in central Taiwan in 1979, where a mass outbreak of poisoning occurred due to the ingestion of PRO contaminated with polychlorinated biphenyls (PCBs), dibenzofurans (PCDFs), and quaterphenyls (PCQs). The incident was called PCB poisoning or “The Yu-cheng Rice Oil Poisoning Incident”. “The major PCB and PCDF congeners in the toxic oil, blood, and tissues of the poisoned patients were characterized by gas chromatography and gas chromatography-mass spectrometry using highly efficient glass capillary columns. The levels of toxic agents in the rice oil samples collected from the factory, school cafeterias, and the families of the poisoned patients are in the range of 53–99 ppm, 0.18–0.40 ppm and 25–53 ppm for PCBs, PC and PCQs, respectively”. (his-Sung Chen et al. 1984).Footnote 33
Cadmium versus arsenic in rice
Arsenic generally is more hazardous than cadmium. The Regulatory Standards of cadmium in rice Mainland China and the European Union have set the ML for cadmium in rice at 200 μg/kg; rice from Guangdong Province, China, and japan could have a high level of cadmium. While in Brazil un old codex still in use, the maximum limit for Cd is (400 μg/kg) set by national legislation, while for total As the legislation limit is (300 μg/kg). (Kato et al. 2019).Footnote 34 It is likely, therefore, that the limit set for arsenic in rice will soon be 100 μg /kg soon. In this regard, cadmium and arsenic may become competitors. According to Rong et al. (2020),Footnote 35 “Cd and As exhibit opposite geochemical behavior in paddy soil, using appropriate remediation materials to reduce their migration and inhibit their uptake by rice is a great challenge”.
Potassium cyanide (KCN) or sodium cyanide (NaCN)
The starch from the tropical root of cassava that is used as a mixture with rice starch in children’s food contains a small amount of cyanide. According to China Daily (2005),Footnote 36 “Cassava is poisonous unless it is peeled and thoroughly cooked. If it is eaten raw or prepared incorrectly, one of its chemical constituents will be attacked by digestive enzymes, resulting in the deadly poison cyanide. Even only two small cassava roots can contain a fatal dose. In 2005, 27 children perished after eating cassava at Philippine school and a further 100 became sick”.
Cassava originally comes from East Asia, mostly from Thailand, where all soil-contact products should be under control. As a result, it could be polluted by either mercury, arsenic or even cyanide. Mercury and cyanide are forbidden in infants’ food and this should be highlighted in the instructions on the food-packaging pouch.
Accumulation effect of a mixed food
The limitation of iAs in water to 10 μg/L = 10 ppb is promoted by the World Health Organizations guideline values for naturally occurring chemicals that are of health significance in drinking-water ([WHO] 2012).Footnote 37 However, in India and Bangladesh, the accepted level is < 50 μg /L in the absence of an alternative source of portable water in the affected area (Das et al. 2013).Footnote 38 In the US, the recommended ML of arsenic in juice is 23 μg/kg. Both water and juice are similar in nature and used in mixed food. It is not clear why the same limit is not applied to drinking water; many people drink juice, especially apple juice. This limit should, therefore, be reviewed.
Similar to water used for the irrigation of rice (flood irrigation), some studies, such as (Bhatti et al. 2013),Footnote 39 have found “a higher concentration of arsenic in vegetables grown by flood irrigation compared to non-flood irrigation”. For instance, “the arsenic concentration in spinach leaves exceeds the Chinese maximum permissible concentration for iAs (50 μg/kg fresh weight) by a factor of 1.6 to 6.4 times. Hence, spinach presents a direct risk to human health, where water for flood irrigation contains an arsenic concentration greater than 50 μg L−1. The primary route of arsenic exposure for the general population is via the ingestion of contaminated food or water”.
Unanswered questions
In nature, arsenic always comes with two more elements: antimony and lead. Although these two elements are both less hazardous than arsenic, they can indicate its existence. Selenium, another hazardous element, is important as a daughter isotope of arsenic.
However, an open question remains in relation to arsenic and iAs content when the food in question is being boiled in water or cooked in oil. It is necessary to know the physical conditions, including the temperature, time, the amount of water used for rinsing the food, and amount of water used for cooking in the case of boiling. While cooking, the form of arsenic changes constantly. Therefore, the key question is not the form of arsenic but the tAs as it pertains to the degree of threat in foods and drinks. Another important issue is that arsenic is used by nature as a catalyst, producing hundreds of inorganic and organic components. Arsenic is not stable in any form. Its elimination requires bonding it to a stable chemical band or chelation in the treatment of arsenic as well as mercury poisoning.
Another inorganic form of arsenic is arsine (AsH3), which has not received much attention in the literature. “It is a colorless, flammable, non-irritating toxic gas with a mild garlic odor” and “formed when arsenic is exposed to an acid” ([CDC] 2018).Footnote 40 According to Fowler et al. (2007)Footnote 41 “arsine may be also released when the hydrogen ion is formed by hydrolysis and in the reaction of moisture with arsenic-containing dross”.
The question is what if arsine could form in humans’ and animals’ stomachs, even in very low amounts. This, in turn, raises the question of methods of detection and explanations of the results. From the results of (Cullen and Reimer 2016)Footnote 42 a conflict in conclusions with other articles about the toxicity of arsenic components becomes apparent:
Based on environmental studies, humidity (water) rather than air is the most important factor in the bio-transformation and the concentration of particular arsenic species. It is important to add that the metabolized arsenic is different from rice and seafood. The role of mercury will be discussed below.
The results in Table 6 (Gilbert-Diamond et al. 2011)Footnote 43 show that iAs was 0.28 μg/kg for rice-eating females whereas tAs was 5.27 μg/kg, Urinary arsenic metabolites were measured via HPLC. These measurements of urinary arsenic metabolites were taken in the US, where rice typically contains more oAs, via HPLC. According to this method of data analysis, 4.09 μg/L of DMA is generally considered several times less toxic than iAs, but it is still unsafe. The differences in iAs is 0.07 (or + 33%) and in tAs it is 1.89 μg/L (or 1.56 times more than the non-rice eaters), so that about 56% comes from rice products. These results may differ outside of the US.
The study from Gilbert-Diamond et al. (2011) further assumed that Americans eat only American rice, which is not necessarily true. Moreover, arsenic is not stable inside the human body during the digestion process. For maximum safety, the tAs should also be measured and assigned a ML. The toxicity of iAs can be compared with that of organic mercury. It is crucial to note the difference between the amounts of arsenic detected in urine and the food itself. Again, arsenic may still be present even if the chosen method cannot detect it.
There are issues that deserve consideration. Arsenic is found, albeit in varying amounts, not only in the husk but all parts of the rice plant, including in the roots, stem, inner grain bran, and inner seed. Therefore, particularly, in the manufacture of eating and drinking utensils, such as plates and cups, it is not safe to utilize any part of the rice plant. Products made from rice are commonly considered as healthier and safer than, for instance, those made from plastic since rice is a natural material. However, such products can be even more hazardous due to arsenic contamination. Arsenic can only be found if attempts are made to search for it and not by traditional methods of analysis.
A specific example is drinking straws. After 2018, drinking straws made from rice husks with a mixture of starch, such as cassava root starch, or from the stems (culms) of the rice plant became increasingly popular in Southeast Asian countries, especially with the approaching international ban on plastic straws in 2020. What environmentalists and manufacturers alike did not appear to realize is that they were inadvertently introducing arsenic into consumers’ diets. What is more, the process of manufacturing these straws is mostly manual and, therefore, increases the exposure of workers to arsenic.
Some companies are trying to make paper from the stems of the rice plant, believing that they are protecting the environment while still giving workers a safe work environment. This is, however, not the case, as this process produces polluted water and paper. It is important, therefore, for the husk rice industries to avoid manufacturing eating and drinking utensils made from rice husks and stems. By heating these, the arsenic can be released, but this would create air pollution.
Any new technology can avoid or extract arsenic chemically only by heating the sample above 614 °C, allowing the arsenic to be sublimated. However, again, this would create air pollution.
Arsenic in the infants and pregnant women diet foods
Pregnant women should be made aware of the arsenic content in rice products, water, seaweed (Hizikia fusiforme), and fish (basin fish are considered under control). In the international regulations, there is a misunderstanding between the allowed ML of iAs between adults and infants’ rice food products as well as between iAs and oAs. Long-term exposure to arsenic originating from drinking water and food can cause cancer and skin lesions. It has also been associated with cardiovascular diseases and diabetes. In utero and early childhood, arsenic exposure has been linked to negative impacts on cognitive development and increased deaths in young adults. As a result, a need arises for the community to understand the risks of high arsenic exposure and the sources of arsenic exposure, including the intake of arsenic by crops (e.g. rice) from irrigation water and the intake of arsenic into food from cooking water. The FDA found that iAs exposure in infants and pregnant women can result in a child’s decreased performance on certain developmental tests that measure learning based on epidemiological evidence, including dietary exposure through food.
It is essential as a guidance for the food industry to analyze the level of iAs in infants’ (babies’) and pregnant women’s food. iAs is classified as more toxic, especially for infants. In April 2016, the FDA proposed an action level or a limit of 100 μg/kg for iAs in infants’ rice cereal. The FDA’s data show that 53% of more than 400 samples infants’ rice cereals sampled from retail stores in 2014 did not meet the level of 100 μg/kg of iAs and a large majority (78%) was at or below 110 μg/kg of iAs. The FDA expects that food manufacturers can produce infants’ rice cereal that meet or are below the proposed limit by utilizing good manufacturing practices, such as sourcing rice with lower iAs levels ([FDA] 2016).Footnote 44
In terms of body weight,” Dietary exposure to inorganic arsenic for children under three years of age is in general estimated to be from 2 to 3-fold that of adults.” ([EFSA] 2009).Footnote 45 “The highest mean dietary exposure estimates at the lower bound (LB) were in toddlers (0.30 μg/kg body weight (bw) per day), and in both infants and toddlers (0.61 μg/kg bw per day) at the upper bound (UB). At the 95th percentile, the highest exposure estimates (LB–UB) were 0.58 and 1.20 μg/kg bw per day in toddlers and infants, respectively. In general, UB estimates were two to three times higher (UB). The mean dietary exposure estimates (LB) were overall below the range of benchmark dose lower confidence limit (BMDL01) values of 0.3–8 μg/kg bw per day established by the EFSA Panel on Contaminants in the Food Chain in 2009. Validated analytical methods with adequate sensitivity are needed to quantify iAs, together with extraction methods that guarantee minimal (change in valency) or no changes to the original species” (Arcella et al. 2021).Footnote 46 This signifies that the risk for babies is at least three times greater compared to adults.
Some of the “arsenic-containing lipids (arsenolipids), compounds of milk from nursing mothers, are highly toxic to human cells. The concentrations of arsenolipids in tested milk were low (combined tAs content approximately 0.5 μg/kg) compared to the current recommended ML of arsenic in water (10 μg/kg). The potential concern is the possibility of the lipids crossing the blood–brain barrier at the critical stage of brain development in the newborn child” (Stiboller et al. 2017).Footnote 47 Rice-fortified foods have significantly higher tAs concentrations than non-rice-based foods.
“As(III) is the main species of arsenic with lower concentrations of DMA, and As(V) is also present in rice and in derived products like starch, flour, and syrup, which are used to fortify a number of processed baby foods, jarred purees, strained foods, and snack foods.” see (Jackson et al. 2012).Footnote 48
On the subject of arsenic in infant foods, Jackson et al. (2012) p. 221, reported the following results:”
-
If a 10 kg infant (~ 1 year old) consumed 3 full jars at the median total arsenic concentration of 1.3 μg each day, that is more than twice the 0.17 μg kg−1 d−1 safe adult arsenic exposure level.
-
The daily exposure would be 0.05 μg kg−1 d−1 solely from these jarred rice foods, before other potential arsenic sources,
-
Despite relatively low concentrations of arsenic (< 0.3–22 μg/kg) in formulas, purees and multiple ingredient infant foods, these levels are potentially still of concern because arsenic is present mainly in the more toxic inorganic form.”
According to Guillod-Magnin et al. (2018a)Footnote 49 there are particular groups of children who are at risk of arsenic exposure via rice and rice-based products. “These include those
-
with coeliac disease and who consume rice and rice-based products instead of gluten-containing cereals;
-
with a cows’ milk allergy and who consume rice drinks as a replacement for cows’ milk products;
-
with a vegan diet, who likewise consume rice drinks as a replacement for cows’ milk products;
-
and those from specific ethnic groups, for example Asians, who cover their carbohydrate needs mainly through rice.”
In a study from Gu, de Silva and Reichman (2020),Footnote 50 “nearly 75% of samples had inorganic As exceeding the EU maximum levels for infants and children (0.1 mg/kg) and the mean iAs percentage of total reached as high as 84.8%. High tAs concentration was positively correlated with rice content and also related to brown (wholegrain). Estimates of dietary exposure showed that infants consuming large amounts of rice pasta or crackers will have an increased risk of health impact associated with excess intake of As through dietary exposure”.
However, by comparing the discrepancies between the results in Table 6, it is clear that the tAs rather than only iAs should be measured. A recent study [HOUSE], [2021] 52 Toxic heavy metals endanger infant neurological development and long-term brain function. Specifically, the Subcommittee reports that: Arsenic was present in baby foods made by all responding companies. • One company sold baby foods after tests showed they contained as much as 180 parts per billion (ppb) of inorganic arsenic. Over 25% of the products tested before sale contained over 100 ppb of inorganic arsenic. Similar results are the other companies.
Errors in sample preparation
The technique employed might not consider the origin of the food or distinguish between different species of arsenic. For example, ICP-AES (or ICP-OES) can detect iAs in urine. ICP-MS uses a mixture of nitric and hydrofluoric acids with microwave digestion, where many labs (wrongly) dry their samples in an oven at 105–120 °C. This could produce different results for the same samples, in contrast to all forms of arsenic being measured via HPLC. For medical research, it is strongly recommended, therefore, that different methods of arsenic detection be combined, for example ICP-AES-HPLC. For more details about laboratory performance by technique see (Taylor et al. 2016; Cordeiro, Fernando 2016).[Footnote 51,Footnote 52]
Heating the sample in an oven or in a microwave could lower the arsenic content and change its chemical form. Therefore, a dual-seal vessel is recommended for measuring arsenic content. The dual-seal function vessels can be provided for control of the byproducts from digestions or even volatile analytes, such as arsenic, selenium and mercury.
There are possible errors in preparing the samples because of heating, frying and washing them not by the same method used internationally. This illustrates the need for a new protocol from NIST and other SRMs’ calibration institutes.
SRM 1568a, a type of rice flour, was used in many studies on arsenic in rice, and was replaced by 1568b ([NIST] 2013).Footnote 53 Consequently, all old studies that used SRM 1568a should not be trusted anymore.
“The iAs (As(III), oAs(V)) species was predominant, accounting for approximately 72% of the total As in rice” (Liang et al. 2010a, b, c). Most of the samples included in the FDA’s data ([FDA] 2016), those from Liang et al (2010a, b, c), and in Tables 5, 6, 7, 8 are above the ML for infants’ food (100 μg/kg). The bromine in rice requires separate and thorough investigation. iAs accounts for over 80% of the tAs in the samples from Hunan Province, the largest rice‐producing province in China (Ma et al. 2016).Footnote 54 The predominant arsenic species found in rice and rice-based products, except in rice drinks, was As(III) with 60–80% of the tAs content, followed by approximately 20% DMA(V) and 15% As(V), except in rice drinks, which contain 11% and 50%, respectively (Guillod-Magnin et al. 2018b).
Table 7 illustrates that the highest proportions of iAs to the tAs are in rice drinks, baby food in dry form, rice crackers, and rice grains. It also reveals that rice cereals, rice grains (brown), and rice crackers are above the ML of iAs.
Another question regards the elements contained in rice. Kongsri et al. (2016) used one of the most effective techniques to identify what other elements exist with arsenic (see Table 8). The analysis of the major elements existing in the rice seeds shows that arsenic and bromine are trace elements, both of which carry health risks, while potassium and manganese are the most prominent. These two elements are safe and are essential for regulating cell function in the human body. The carbohydrates (starch) contained in rice along with manganese and potassium, are potential reasons for the popularity of rice as a food source.
It is known neither whether the rice samples in these experiments were brown or white nor polished or husked.
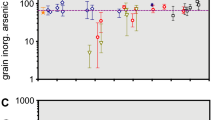
The original experimental results produced for this paper, which were collected at the Reactor Institute Delft in the Netherlands, on the tAs in rice are shown in Table 9. They show that, as expected, all 21 samples from international markets (9 from Turkey and 12 from the Netherlands) contained arsenic. The brands of rice tested are consumed by both infants and adults. However, no warning about arsenic and the effects on children were mentioned on the food packing. Moreover, 10 of the 21 samples of the products exceeded the ML (100 μg/kg) for infants and children’s foods.
Measuring the tAs is much more straightforward than measuring the iAs in most countries of the world. One sample (N12) of powdered brown rice mixed with liquorice was particularly heavily polluted, as both components (rice and liquorice) have a higher natural propensity for accumulating arsenic. The rice products are not pure rice and, consequently, no average has been considered.
Table 9 also shows the results of the experimental analyses of the tAs in rice products by k0-INAA. The sample mass is in mg (generally, approximately 260 mg). The concentrations are in μg/kg. All products are consumed by both infants and adults. Most of the samples are in a form of a mixed powder (T for samples from Turkey markets and N for Netherlands). In Turkey, all brands are consumed by children.
Conclusion
Arsenic is an active catalyst for many chemical reactions. It continually changes its chemical form and is not stable in any form. The form of arsenic depends on temperature and humidity levels. Water and rice in some places in the world, including Nepal, Bangladesh, and some regions in China and India, are naturally polluted with high levels of arsenic. This prevents the rest of the world from reaching an international agreement regarding arsenic content. Consequently, the international trademarks of rice products should be treated separately from these certain places in terms of the ML of iAs, oAs and the tAs.
Brown, red, and black rice husks, and their starch should be forbidden in infants’ food and those intended for pregnant women, as well as in pharmaceutical beauty industries. If this is not feasible, the ML of the tAs should be set at 125 μg/kg. Workers in the food industry who are in direct contact with rice husks should be protected with masks. Additional studies should be conducted on liquorice, an additive in rice products, popular herbal medicines and beverages, as a further source of arsenic pollution.
The experimental methods reported in many papers for measuring arsenic and mercury content are not sufficiently accurate. More importantly, some arsenic and mercury are lost in the process of grinding the dried samples using mortar into a homogenized powder. In addition, no heat should be applied, and the natural lab temperature should be considered. Therefore, a new protocol for volatile substances is needed. Some methods of analysis are sensitive to iAs or oAs rather than only the tAs. The INAA is one of the best control techniques to determine the tAs in rice and rice-based products, but it is not for daily practice.
Including on the packaging or the health certificate the process by which the amount of one type or the total amount of all types of arsenic, selenium, mercury, and cadmium was measured is strongly encouraged. The relationship between arsenic and mercury is inversely proportional. This measurement can be facilitated as part of the quality control or a certification within the ISO 22000 family (food safety management), or within a new ISO certification. Developing countries need hands-on international assistance with arsenic analysis equipment, data processing data, and professional exchange.
It bears reiterating as important findings of this paper that arsenic can only be found when active measures are taken to test for it, and not by traditional methods of analysis. If the material is heated, the arsenic content can be released, but this process produces air pollution. It is important for industries that use rice husks in their manufacturing processes to avoid using them for eating and drinking utensils as well as other household items. This will help minimize the amount of arsenic, particularly iAs, from entering the body.
Finally, the overarching ambition of this contribution is to inject a new, or greater, sense of urgency for the (accurate) measurement of arsenic in rice as well as other food products and to demonstrate that this can be done in a matter of hours.
Data availability
(Data transparency) All rules for scientific research were respected.
Notes
Ruskin, F. R. (Ed.). (1999). Lost Crops of Africa: Grains. DIANE Publishing, p.7.
[FAO/WHO], Codex alimentarius, (2015). ‘Joint FAO/WHO Food Standards Programme CODEX Committee on Contaminants in Foods’. http://apps.who.int/iris/bitstream/10665/98388/1/9789241209830_eng.pdf.
Discussant, Michael S, Albuquerque Gorby, Ralph C Williams, M Ichael, and S Gorby. (1988). ‘IL IWsXW Arsenic Poisoning’, p. 308., https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1026413/pdf/westjmed00133-0060.pdf.
De Chávez Capilla, T. G. (2018). The Metabolism of Arsenic in Humans: Bioaccessibility in the Gastrointestinal Tract, Diffusion across Lipid membranes and Biotransformations in Liver Cells (Doctoral dissertation, University of Canberra). https://researchsystem.canberra.edu.au/ws/portalfiles/portal/33678845/file
Jackson, Brian P., and Tracy Punshon. (2015a). ‘Recent Advances in the Measurement of Arsenic, Cadmium, and Mercury in Rice and Other Foods’. Current Environmental Health Reports. Springer International Publishing, p. 4. https://doi.org/10.1007/s40572-014-0035-7.
Francesconi, K. A., Tanggaar, R., McKenzie, C. J., & Goessler, W. (2002). Arsenic metabolites in human urine after ingestion of an arsenosugar. Clinical chemistry, 48(1), 92–101.
Nordberg, G. F., & Fowler, B. A. (2018). Risk Assessment for Human Metal Exposures: Mode of Action and Kinetic Approaches. Academic Press.
Bashir, S., Sharma, Y., Irshad, M., Gupta, S. D., & Dogra, T. D. (2006). Arsenic‐induced cell death in liver and brain of experimental rats. Basic & clinical pharmacology & toxicology, 98(1), 38–43. https://doi.org/10.1111/j.1742-7843.2006.pto_170.x.
Ghosh, D., Datta, S., Bhattacharya, S., & Mazumder, S. (2007). Long-term exposure to arsenic affects head kidney and impairs humoral immune responses of Clarias batrachus. Aquatic toxicology, 81(1), 79–89. https://doi.org/10.1016/j.aquatox.2006.11.004.
Hackenmueller, S. A., & Strathmann, F. G. (2014). Total arsenic screening prior to fractionation enhances clinical utility and test utilization in the assessment of arsenic toxicity. American journal of clinical pathology, 142(2), 184–189. https://doi.org/10.1309/AJCPHUB0YEHPRAWA.
[BNF]. 2017. ‘Arsenic in rice – is it a cause for concern?—British Nutrition Foundation. Accessed: 2021–06-23.
https://www.nutrition.org.uk/nutritioninthenews/headlines/arsenicinrice.html.
Singh Bhupinder. 5218. 13—Rice husk ash, Civil and Structural Engineering, Waste and Supplementary Cementitious Materials in Concrete, Woodhead Publishing, Pages 417–460, ISBN 9,780,081,021,569, https://doi.org/10.1016/B978-0-08-102156-9.00013-4.
Karagas, M. R., Punshon, T., Sayarath, V., Jackson, B. P., Folt, C. L., & Cottingham, K. L. (2016). Association of rice and rice-product consumption with arsenic exposure early in life. JAMA pediatrics, 170(6), 609–616. https://jamanetwork.com/journals/jamapediatrics/article-abstract/2514074.
Signes-Pastor, Antonio J., Manus Carey, and Andrew A. Meharg. (2017) ‘Inorganic Arsenic Removal in Rice Bran by Percolating Cooking Water’. Food Chemistry 234 (November): 76–80. https://doi.org/10.1016/j.foodchem.2017.04.140.
Liang, Feng, Yulan Li, Guilin Zhang, Mingguang Tan, Jun Lin, Wei Liu, Yan Li, and Wenwei Lu. (2010a). ‘Total and speciated arsenic levels in rice from China`, Food Additives & Contaminants: Part A 27 (6): 813–14. https://doi.org/10.1080/19440041003636661.
Panigrahi, A., Chattopadhyay, A. K., Paul, G., & Panigrahi, S. (2016). HIV, Cardiovascular Diseases, and Chronic Arsenic Exposure co-exist in a Positive Synergy. Retrieved from http://arxiv.org/abs/1602.05981.
Cai Zhi Wei. 2018. ‘Determination of Arsenic Levels in Daily Rice Products and Influence of Arsenic Levels upon Modifications’. Delft University of Technology.
Chung, Stephen Wai cheung, Chi ho Lam, and Benny Tsz pun Chan. (2014a). ‘Total and Inorganic Arsenic in Foods of the First Hong Kong Total Diet Study’. Food Additives and Contaminants—Part A Chemistry, Analysis, Control, Exposure and Risk Assessment 31 (4): 655. https://doi.org/10.1080/19440049.2013.877162.
International Agency for Research on Cancer. (2012). IARC Monographs: Arsenic, Metals, Fibers and Dusts. Volume 100C. A Review of Human Carcinogens.
[FOW/WHO]. (2017). ‘REPORT OF THE 11th SESSION OF THE CODEX COMMITTEE ON CONTAMINANTS IN FOODS.’ Geneva, Switzerland. http://www.fao.org/fao-who-codexalimentarius/sh-proxy/en/?lnk=1&url=https%253A%252F%252Fworkspace.fao.org%252Fsites%252Fcodex%252FMeetings%252FCX-735-12%252FWD%252FREP17_CFe.aspx.
Yao, Bao Min, Peng Chen, and Guo Xin Sun. (2020). ‘Distribution of Elements and Their Correlation in Bran, Polished Rice, and Whole Grain’. Food Science and Nutrition 8 (2): 982–92. https://doi.org/10.1002/fsn3.1379. https://onlinelibrary.wiley.com/doi/epdf/10.1002/fsn3.1379.
Deng, F., Yamaji, N., Ma, J. F., Lee, S. K., Jeon, J. S., Martinoia, E., … & Song, W. Y. (2018). Engineering rice with lower grain arsenic. Plant biotechnology journal, 16(10), 1695. https://doi.org/10.1111/pbi.12905.
[FAO and, & WHO]. (2016). JOINT FAO/WHO FOOD STANDARDS PROGRAMME CODEX COMMITTEE ON CONTAMINANTS IN FOODS Tenth Session Rotterdam, The Netherlands, 4–8 April 2016 DISCUSSION PAPER ON THE DEVELOPMENT OF MAXIMUM LEVELS FOR MYCOTOXINS IN SPICES AND POSSIBLE PRIORITIZATION OF WORK, (CX/CF 16/10/6), 3. Retrieved from http://www.fao.org/fao-who-codexalimentarius/sh-proxy/fr/?lnk=1&url=https%253A%252F%252Fworkspace.fao.org%252Fsites%252Fcodex%252FMeetings%252FCX-735-10%252FWD%252Fcf10_14e.pdf.
JOINT FAO/WHO FOOD STANDARDS PROGRAMME CODEX ALIMENTARIUS COMMISSION 42nd Session, Geneva, Switzerland 8—12 July 2019, REPORT OF THE 13th SESSION OF THE CODEX COMMITTEE ON CONTAMINANTS IN FOODS, Yogyakarta, Indonesia 29 April – 3 May 2019.
[USDA-FSIS]. (2012). U.S. Department of Agriculture Food Safety and Inspection Service. Safe and suitable ingredients used in the production of meat and poultry products. (Vol. 2012, p. 53). Retrieved from https://www.fsis.usda.gov/wps/wcm/connect/bab10e09-aefa-483b-8be8-809a1f051d4c/7120.1.pdf?MOD=AJPEREShttp://www.fsis.usda.gov/OPPDE/rdad/FSISDirectives/7120.1.pdfhttps://www.fsis.usda.gov/wps/wcm/connect/bab10e09-aefa-483b-8be8-809a1f051d4c/7120.1.pdf?M
Rahman, Mahmuder, (2008). An interview with Mahmuder Rahman: Bangladesh’s arsenic agony. (2008). Bulletin of the World Health Organization, 86(1), 11–12. https://doi.org/10.1590/S0042-96862008000100008.
[EFSA/ CONTAM]. (2009). ‘Scientific Opinion on Arsenic in Food`, EFSA Journal, 7, 10, p. 146., https://efsa.onlinelibrary.wiley.com/doi/epdf/10.2903/j.efsa.2009.1351?src=getftr.
Patrick J. Gray, Sean D. Conklin, Todor I. Todorov & Sasha M. Kasko (2016) Cooking rice in excess water reduces both arsenic and enriched vitamins in the cooked grain, Food Additives & Contaminants: Part A, 33:1, 78–85.
PÜSSA, TÖNU. (2013). `Principles of Food Toxicology`, 2nd ed. CRC Press, Taylor & Francis Group, p 200. https://books.google.com.tr/books?id=1DPSBQAAQBAJ&source=gbs_navlinks_s&hl=en.
Bakota, E. L., Dunn, R. O., & Liu, S. X. (2015). Heavy metals screening of rice bran oils and its relation to composition. European Journal of Lipid Science and Technology, 117(9), 1452–1462. https://doi.org/10.1002/ejlt.201400443.
Schecter, A., & Masuda, Y. (2012). The Yusho and Yucheng Rice Oil Poisoning Incidents. In Dioxins and Health: Including Other Persistent Organic Pollutants and Endocrine Disruptors: Third Edition (pp. 521–551). John Wiley and Sons. https://doi.org/10.1002/9781118184141.ch16.
SUZUKI T., KOBAYASHI, H. (1968). ‘Case Details > Contamination of Rice Bran Oil with PCB Used as the Heating Medium by Leakage through Penetration Holes at the Heating Coil Tube in Deodorization Chamber’. http://www.shippai.org/fkd/en/hfen/HB1056031.pdf.
his-Sung Chen, P., M. L. Luo, C. K. Wong, and C. J. Chen. (1984). ‘Polychlo-rinated Biphenyls, Dibenzofurans, and Quaterphenyls in the Toxic Rice-Bran Oil and PCBs in the Blood of Patients with PCB Poisoning in Taiwan’. American Journal of Industrial Medicine 5 (1–2): 133–45. https://onlinelibrary.wiley.com/doi/pdf/10.1002/ajim.4700050112.
Kato, Lilian Seiko et al. (2019). ‘Arsenic and Cadmium Contents in Brazilian Rice from Different Origins Can Vary More than Two Orders of Magnitude’. Food Chemistry 286(February): 644–50. https://doi.org/10.1016/j.foodchem.2019.02.043.
Rong, Q., Zhong, K., Li, F., Huang, H., Li, C., Nong, X., & Zhang, C. (2020). Combined Effect of Ferrous Ion and Biochar on Cadmium and Arsenic Accumulation in Rice. Applied Sciences, 10(1), 300. https://doi.org/10.3390/app10010300.
China Daily. 2005. ‘27 Children Die after Eating Cassava at Philippine School’. 2005. http://www.chinadaily.com.cn/english/doc/2005-03/10/content_423641.htm.
[WHO]. (2012). Potable Water Specification,2nd Ed, Rwanda Bureau of Standard,Kigali. https://apps.who.int/iris/rest/bitstreams/1080656/retrieve.
Das N.K., Ghosh P., Sil A. (2013) Arsenicosis. In: Kretsinger R.H., Uversky V.N., Permyakov E.A. (eds) Encyclopedia of Metalloproteins. Springer, New York, NY. https://doi.org/10.1007/978-1-4614-1533-6_488.
Bhatti, S. M., Anderson, C. W. N., Stewart, R. B., & Robinson, B. H. (2013). Risk assessment of vegetables irrigated with arsenic-contaminated water. Environmental Sciences: Processes and Impacts, 15(10), 1866–1875. https://doi.org/10.1039/c3em00218g.
[CDC]. (2018). Arsine (SA) or Stibine Facts About Arsine Case Definition: Arsine or Stibine Poisoning Toxic Syndrome Description: Arsine or Stibine Poisoning. https://emergency.cdc.gov/agent/arsine/facts.asp.
Fowler, Bruce A., Selene J. Chou, Robert L. Jones, and C. J. Chen. (2007). ‘Arsenic’. In Handbook on the Toxicology of Metals, Academic Press, 397. https://www.sciencedirect.com/science/article/pii/B9780123694133500744?via%3Dihub.
Cullen, William R, and Kenneth J Reimer. (2016). ‘CHAPTER 7. Arsenic in Food’. In Arsenic Is Everywhere: Cause for Concern?, 200. https://doi.org/10.1039/9781782626633-00190.
Gilbert-Diamond, Diane et al. (2011). ‘Rice Consumption Contributes to Arsenic Exposure in US Women’. Proceedings of the National Academy of Sciences 108(51): 20,657. https://www.pnas.org/content/pnas/108/51/20656.full.pdf.
[FDA]. (2016). ‘Arsenic in Rice and Rice Products Risk Assessment Report. https://www.fda.gov/media/96071/download.
[EFSA/ CONTAM]. 2009. ‘Scientific Opinion on Arsenic in Food’EFSA Journal 7(10): 1. https://efsa.onlinelibrary.wiley.com/doi/epdf/10.2903/j.efsa.2009.1351.
Arcella, D., Cascio, C., & Gómez Ruiz, J. Á. (2021). Chronic dietary exposure to inorganic arsenic. EFSA Journal, 19(1), pp1,43. https://doi.org/10.2903/j.efsa.2021.6380.
Stiboller, M., Raber, G., Lenters, V., Gjengedal, E. L. F., Eggesbø, M., & Francesconi, K. A. (2017). Arsenolipids detected in the milk of nursing mothers. Environmental Science and Technology Letters, 4(7), 273. https://doi.org/10.1021/acs.estlett.7b00181.
Jackson, B. P., Taylor, V. F., Punshon, T., & Cottingham, K. L. (2012). Arsenic concentration and speciation in infant formulas and first foods. Pure and Applied Chemistry, 84(2), 215–223. https://doi.org/10.1351/pac-con-11-09-17.
Guillod-Magnin, R., Brüschweiler, B. J., Aubert, R., & Haldimann, M. (2018). Arsenic species in rice and rice-based products consumed by toddlers in Switzerland. Food Additives & Contaminants: Part A, 35(6), 1176. https://doi.org/10.1080/19440049.2018.1440641.
Gu, Z., de Silva, S., & Reichman, S. M. (2020). Arsenic concentrations and dietary exposure in rice-based infant food in Australia. International Journal of Environmental Research and Public Health, 17(2). https://doi.org/10.3390/ijerph17020415.
Taylor, A., Barlow, N., Day, M. P., Hill, S., Patriarca, M., & White, M. (2017, March 1). Atomic spectrometry update: Review of advances in the analysis of clinical and biological materials, foods and beverages. Journal of Analytical Atomic Spectrometry. Royal Society of Chemistry. https://doi.org/10.1039/c7ja90005h.
Cordeiro, Fernando & de la Calle Guntiñas, Maria. (2016). Determination of total & inorganic arsenic in rice. https://doi.org/10.2787/679027.
NIST]. (2013). ‘Certificate of Analysis for Standard Reference Material® 1568b Rice Flour’, no. 1568b: 1–6. https://www-s.nist.gov/srmors/certificates/1568B.pdf.
Ma, L., Yang, Z., Tang, J., & Wang, L. (2016). Simultaneous separation and determination of six arsenic species in rice by anion-exchange chromatography with inductively coupled plasma mass spectrometry. Journal of Separation Science, 39(11), 2105–2113. https://doi.org/10.1002/jssc.201600216.
Abbreviations
- As(III):
-
Arsenite (oxidation state + 3), trioxidoarsenate
- As(V):
-
Arsenate (oxidation state + 5), pentavalent arsenic
- BfR:
-
The German Institute for Risk Assessment (German)
- CCCF8:
-
Codex Committee on Contaminants in Foods 8th Session
- CDC:
-
Centers for Disease Control
- CFS:
-
Centre for Food Safety in Hong Kong
- CONTAM:
-
The EFSA panel on contaminants in the food chain (CONTAM Panel)
- DMA(III):
-
Dimethylarsinic acid III
- DMA(V):
-
Dimethylarsinic acid V
- DW:
-
Dry weight
- EFSA:
-
European Food Safety Authority
- EU:
-
European Union
- FAO:
-
The Food and Agriculture Organization
- FDA:
-
US Food and Drug Administration
- GLD:
-
The Government Logistics Department
- HPLC:
-
High-performance liquid chromatography
- IARC:
-
International Agency for Research on Cancer
- iAs:
-
Inorganic arsenic
- ICP-MS:
-
Inductively coupled plasma mass spectrometry
- ICP-OES (= ICP-AES):
-
Inductively coupled plasma-optical emission spectrometry, or inductively coupled plasma-atomic emission spectrometry
- INAA, NAA:
-
Instrumental neutron activation analysis; K0-method-NAA
- IRRI:
-
International Rice Research Institute
- KCN:
-
Potassium cyanide
- ML:
-
Maximum limit
- ML:
-
Maximum limit
- MMA(III):
-
Monomethylarsonic acid III
- MMA(V):
-
Monomethylarsonic acid V
- NaCN:
-
Sodium cyanide
- NIST:
-
National Institute of Standards and Technology
- oAs:
-
Organic arsenic
- PCBs:
-
Polychlorinated biphenyls
- PCDFs:
-
Polychlorinated dibenzofurans
- PCQs:
-
Polychlorinated quaterphenyls
- PRO:
-
Rice bran oil
- RHA:
-
Rice husk ash
- RISO:
-
The Association for the Promotion of Rice Consumption
- SRM:
-
Standard reference materials
- tAs:
-
Total arsenic (As-tot)
- USDA-FSIS:
-
United States Department of Agriculture, Food Safety and Inspection Service
- WHO:
-
World Health Organisation
- WW:
-
Wet weight
- XRF:
-
X-ray fluorescence spectrometry
References
Arcella D, Cascio C, Gómez Ruiz JA (2021) Chronic dietary exposure to inorganic arsenic. EFSA J 19(1):1–43. https://doi.org/10.2903/j.efsa.2021.6380
Bakota EL, Dunn RO, Liu SX (2015) Heavy metals screening of rice bran oils and its relation to composition. Eur J Lipid Sci Technol 117(9):1452–1462. https://doi.org/10.1002/ejlt.201400443
Bashir S, Sharma Y, Irshad M, Gupta SD, Dogra TD (2006) Arsenic-induced cell death in liver and brain of experimental rats. Basic Clin Pharmacol Toxicol 98(1):38–43. https://doi.org/10.1111/j.1742-7843.2006.pto_170.x
Bhatti SM, Anderson CWN, Stewart RB, Robinson BH (2013) Risk assessment of vegetables irrigated with arsenic-contaminated water. Environ Sci Process Impacts 15(10):1873. https://doi.org/10.1039/c3em00218g
BNF (2017) Arsenic in rice-is it a cause for concern? British Nutrition Foundation. https://www.nutrition.org.uk/nutritioninthenews/headlines/arsenicinrice.html
CDC (2018) Arsine (SA) or stibine facts about arsine case definition: arsine or stibine poisoning toxic syndrome description: arsine or stibine poisoning. https://emergency.cdc.gov/agent/arsine/facts.asp
Chen PHS, Luo ML, Wong CK, Chen CJ (1984) Polychlorinated biphenyls, dibenzofurans, and quaterphenyls in the toxic rice-bran oil and PCBs in the blood of patients with PCB poisoning in Taiwan. Am J Ind Med 5(1–2):133–145. https://doi.org/10.1002/ajim.4700050112
Chung SWC, Lam CH, Chan BTP (2014) Total and inorganic arsenic in foods of the first Hong Kong total diet study. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 31(4):650–657. https://doi.org/10.1080/19440049.2013.877162
Codex alimentarius FAO-WHO (2015) Joint FAO/WHO food standards programme CODEX committee on contaminants in foods. http://apps.who.int/iris/bitstream/10665/98388/1/9789241209830_eng.pdf
Cordeiro F, Cizek-Stroh A, de la Calle B (2016) Determination of total and inorganic arsenic in rice. IRMM-PT-43 Profic Test Rep. https://doi.org/10.2787/679027
Cullen WR, ReimerKJ (2016) Chapter 7. Arsenic in food. In: Arsenic is everywhere: cause for concern?, p 200. https://doi.org/10.1039/9781782626633-00190
China Daily (2005) 27 Children die after eating cassava at Philippine school. http://www.chinadaily.com.cn/english/doc/2005-03/10/content_423641.htm
Das NK, Pramit G, Amrita S (2013) Arsenicosis. In: Kretsinger RH, Uversky VN, Permyakov EA (eds) Encyclopedia of metalloproteins. Springer, New York, pp 173–188. https://doi.org/10.1007/978-1-4614-1533-6_488
De Chávez Capilla TG (2018) the metabolism of arsenic in humans: bioaccessibility in the gastrointestinal tract, diffusion across lipid membranes and biotransformations in liver cells. http://www.canberra.edu.au/researchrepository/file/b3826d19-a801-4451-8605-bda09c8b8d50/1/full_text.pdf
Deng F, Yamaji N, Ma JF, Lee SK, Jeon JS, Martinoia E, Lee Y, Song WY (2018) Engineering rice with lower grain arsenic. Plant Biotechnol J 16(10):1695. https://doi.org/10.1111/pbi.12905
Discussant MS, Gorby A, Williams RC, Ichael M, Gorby S (1988) Arsenic poisoning [Clinical Conference]. West J Med 149:308–315. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1026413/
EFSA/CONTAM (2009) Scientific opinion on arsenic in food. EFSA J 7(10):146. https://doi.org/10.2903/j.efsa.2009.1351
EU (2015) Commission regulation (EU) 2015/1006 officialournal of the European Union L 161/14 June: 14–16. https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32015R1006&from=EN
FAO and WHO (2016) Joint FAO/WHO Food standards programme codex committee on contaminants in foods tenth session Rotterdam, The Netherlands, 4–8 April 2016 Discussion paper on the development of maximum levels for mycotoxins in spices and possible prioritization of work. No. CX/CF 16/10/6: 2–3. http://www.fao.org/fao-who-codexalimentarius/sh-proxy/fr/?lnk=1&url=https%253A%252F%252Fworkspace.fao.org%252Fsites%252Fcodex%252FMeetings%252FCX-735-10%252FWD%252Fcf10_14e.pdf
FAO/WHO (2019) Rep19/Nfsdu Joint FAO/WHO food standards programme codex alimentarius commission. No. Nov 2018: 64–65
FDA (2016) Arsenic in rice and rice products risk assesment report arsenic. No. March: 24. http://www.fda.gov/Food/FoodScienceResearch/RiskSafetyAssessment/default.htm
Fontcuberta M, Calderon J, Villalbí JR, Centrich F, Portaña S, Espelt A, Duran J, Nebot M (2011) Total and inorganic arsenic in marketed food and associated health risks for the catalan (Spain) population. J Agric Food Chem 59(18):10013–10022. https://doi.org/10.1021/jf2013502
FOW/WHO (2017) Report of the 11th session of the codex committee on contaminants in foods. Geneva, http://www.fao.org/fao-who-codexalimentarius/sh-proxy/en/?lnk=1&url=https%253A%252F%252Fworkspace.fao.org%252Fsites%252Fcodex%252FMeetings%252FCX-735-12%252FWD%252FREP17_CFe.aspx
Fowler BA, Chou SJ, Jones RL, Chen CJ (2007) Arsenic in handbook on the toxicology of metals. Academic Press, p 397. https://doi.org/10.1016/B978-012369413-3/50074-4
Francesconi KA, Tanggaard R, McKenzie CJ, Goessler W (2002) Arsenic metabolites in human urine after ingestion of an arsenosugar. Clin Chem 48(1):92–101. https://doi.org/10.1093/clinchem/48.1.92
Ghosh D, Datta S, Bhattacharya S, Mazumder S (2007) Long-term exposure to arsenic affects head kidney and impairs humoral immune responses of clarias batrachus. Aquat Toxicol 81(1):79–89. https://doi.org/10.1016/j.aquatox.2006.11.004
Gilbert-Diamond D, Cottingham KL, Gruber JF, Punshon T, Vicki Sayarath A, Gandolfi J, Baker ER, Jackson BP, Folt CL, Karagas MR (2011) Rice consumption contributes to arsenic exposure in US women. Proc Natl Acad Sci 108(51):20656–20660. https://doi.org/10.1073/pnas.1109127108
Gray PJ, Conklin Sean D, TodorovTodor I, Kasko Sasha M (2016) Cooking rice in excess water reduces both arsenic and enriched vitamins in the cooked grain. Food Addit Contam Part A 33(1):78
Gu Z, de Silva S, Reichman SM (2020) Arsenic concentrations and dietary exposure in rice-based infant food in Australia. Int J Environ Res Public Health 17(2):1. https://doi.org/10.3390/ijerph17020415
Guillod-Magnin R, Brüschweiler BJ, Aubert R, Haldimann M (2018a) Arsenic species in rice and rice-based products consumed by toddlers in Switzerland. Food Addit Contam-Part A Chem Anal Control Expo Risk Assess 35(6):1176. https://doi.org/10.1080/19440049.2018.1440641
Guillod-Magnin R, Brüschweiler BJ, Aubert R, Haldimann M (2018b) Arsenic species in rice and rice-based products consumed by toddlers in Switzerland. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 35(6):1164–1178. https://doi.org/10.1080/19440049.2018.1440641
Hackenmueller SA, Strathmann FG (2014) Total arsenic screening prior to fractionation enhances clinical utility and test utilization in the assessment of arsenic toxicity. Am J Clin Pathol 142(2):184–189. https://doi.org/10.1309/AJCPHUB0YEHPRAWA
IARC (2012) IARC Monographs: arsenic, metals, fibres, and dusts. IARC monographs on the evaluation of carcinogenic risks to humans, vol 100C
IRRI (2002) What is rice milling? http://www.rice-milling-machine.com/solution/rice-milling-process.html
Jackson BP, Taylor VF, Punshon T, Cottingham KL (2012) Arsenic concentration and speciation in infant formulas and first foods. Pure Appl Chem 84(2):215–223. https://doi.org/10.1351/pac-con-11-09-17
Jackson BP, Punshon T (2015a) Recent advances in the measurement of arsenic, cadmium, and mercury in rice and other foods. Curr Environ Health Rep. https://doi.org/10.1007/s40572-014-0035-7
Jackson BP, Punshon T (2015b) Recent advances in the measurement of arsenic, cadmium, and mercury in rice and other foods. Curr Environ Health Rep. https://doi.org/10.1007/s40572-014-0035-7
Karagas MR, Punshon T, Sayarath V, Jackson BP, Folt CL, Cottingham KL (2016) Association of rice and rice-product consumption with arsenic exposure early in life. JAMA Pediatr 170(6):609–616. https://doi.org/10.1001/jamapediatrics.2016.0120
Kato LS, De Nadai EA, Fernandes AR, Bacchi MA, Feldmann J (2019) Arsenic and cadmium contents in Brazilian rice from different origins can vary more than two orders of magnitude. Food Chem 286:644–650. https://doi.org/10.1016/j.foodchem.2019.02.043
Kongsri S, Srinuttrakul W, Sola P, Busamongkol A (2016) Instrumental neutron activation analysis of selected elements in thai jasmine rice. Energy Proc 89:361–365. https://doi.org/10.1016/j.egypro.2016.05.047
Liang F, Li Y, Zhang G, Tan M, Lin J, Liu W, Li Y, Wenwei Lu (2010a) Total and speciated arsenic levels in rice from China. Food Addit Contam Part A 27(6):814. https://doi.org/10.1080/19440041003636661
Liang F, Li Y, Zhang G, Tan M, Lin J, Liu W, Li Y, Wenwei Lu (2010b) Total and speciated arsenic levels in rice from China. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 27(6):810–816. https://doi.org/10.1080/19440041003636661
Liang F, Li Y, Zhang G, Tan M, Lin J, Liu W, Li Y, Wenwei Lu (2010c) Total and speciated arsenic levels in rice from China. Food Addit Contam Part A 27(6):810–816. https://doi.org/10.1080/19440041003636661
Ma Li, Yang Z, Tang J, Wang L (2016) Simultaneous separation and determination of six arsenic species in rice by anion-exchange chromatography with inductively coupled plasma mass spectrometry. J Sep Sci 39(11):2105–2113. https://doi.org/10.1002/jssc.201600216
NIST (2013) Standard Reference Material 1568b Rice Flour. National Institute of Standards and Technology. https://www-s.nist.gov/srmors/certificates/1568B.pdf
Nordberg GF, Fowler BA (2019) Examples of risk assessments of human metal exposures and the need for mode of action (MOA), toxicokinetic-toxicodynamic (TKTD) modeling, and adverse outcome pathways (AOPs). Risk Assess Human Metal Expo. 8.2. Arsenic (As), p 233. https://doi.org/10.1016/b978-0-12-804227-4.00008-x
Panigrahi A, Chattopadhyay AK, Paul G, Panigrahi S (2016) HIV, cardiovascular diseases, and chronic arsenic exposure co-exist in a positive synergy, 3. http://www.imdb.com/title/tt0036613/
Püssa T (2013) Principles of food toxicology principles of food toxicology, 2nd edn. CRC Press
Rahman M (2008) News. Bulletin of the World Health Organization. http://www.who.int/mediacentre
RISO (2020) RISO: Das Paraboiling-Reis-Verfahren Mit Bild! https://www.riso.ch/de/reis/verarbeitung/parboiled-reis/?oid=1874&lang=de
Rong Q, Zhong K, Li F, Huang He, Li C, Nong X, Zhang C (2020) Combined effect of ferrous ion and biochar on cadmium and arsenic accumulation in rice. Appl Sci 10(1):1. https://doi.org/10.3390/app10010300
Ruskin FR (1996) Lost crops of Africa, vol I. https://doi.org/10.17226/2305
Schecter A, Masuda Y (2012) The yusho and yucheng rice oil poisoning incidents. Dioxins and health including other persistent organic pollutants and endocrine disruptors, 3rd edn. Wiley, Hoboken, pp 521–551. https://doi.org/10.1002/9781118184141.ch16
Signes-Pastor AJ, Carey M, Meharg AA (2017) Inorganic arsenic removal in rice bran by percolating cooking water. Food Chem 234:76–80. https://doi.org/10.1016/j.foodchem.2017.04.140
Singh B (2018) Rice husk ash. Waste and supplementary cementitious materials in concrete: characterisation, properties and applications. Elsevier Ltd. https://doi.org/10.1016/B978-0-08-102156-9.00013-4
Stiboller M, Raber G, Lenters V, Gjengedal ELF, Eggesbø M, Francesconi KA (2017) Arsenolipids detected in the milk of nursing mothers. Environ Sci Technol Lett 4:273. https://doi.org/10.1021/acs.estlett.7b00181
Suzuki T, Kobayashi H (1968) Case details and gt; contamination of rice bran oil with PCB used as the heating medium by leakage through penetration holes at the heating coil tube in deodorization chamber. http://www.shippai.org/fkd/en/cfen/CB1056031.html
Taylor A, Barlow N, Day MP, Hill S, Patriarca M, White M (2016) Atomic spectrometry update: review of advances in the analysis of clinical and biological materials, foods and beverages. J Anal Atomic Spectrom. https://doi.org/10.1039/c6ja90005d
USDA-FSIS (2012) US Department of agriculture food safety and inspection service safe and suitable ingredients used in the production of meat and poultry products, vol 2012. https://www.fsis.usda.gov/wps/wcm/connect/bab10e09-aefa-483b-8be8-809a1f051d4c/7120.1.pdf?MOD=AJPERES
WHO (2012) Potable water specification, 2nd edn. Rwanda Bureau of Standard, Kigali
Yao BM, Chen P, Sun GX (2020) Distribution of elements and their correlation in bran, polished rice, and whole grain. Food Sci Nutr 8(2):982–992. https://doi.org/10.1002/fsn3.1379
ZhiWei C (2018) Determination of arsenic levels in daily rice products and influence of arsenic levels upon modifications. Delft University of Technology
BfR (2014) EU-Höchstgehalte für anorganisches Arsen in Reis und Reisprodukten durch Ver-zehrsempfehlungen zum Schutz von Säuglingen, Kleinkindern und Kindern ergänzen—Aktualisierte Stellungnahme Nr. 017/2015 des BfR vom 06. https://www.bfr.bund.de/cm/343/eu-hoechstgehalte-fuer-anorganisches-arsen-in-reis-und-reisprodukten-durch-verzehrsempfehlungen-zum-schutz-von-saeuglingen-kleinkindern-und-kindern-ergaenzen.pdf
GLD (2018) Health and food adulteration (metallic contamination) regulation, Food adulteration (metallic contamination) regulation. Hong Kong. Retrieved from https://www.gld.gov.hk/egazette/pdf/20182223/es220182223113.pdf
Acknowledgements
The author would like to thank Menno Blaauw, Albert van de Wiel, Mehmet Sarilar, TU Delft/Reactor Institute Delft, Nederland for their support in the experimental analyses, and Sevilay Hacıyakupoğlu, Sema Akyıl Erentürk, and İskender A. Reyhancan, Head of the Nuclear Researches Division, Energy Institute, Istanbul Technical University, Turkey, for their assistance in conducting this study. The author also thanks Chris J. Cookson, Centre for Open Learning, University of Edinburgh, for his efforts in editing this paper.
Funding
No funding was provided. Only free help because of the importance of the research for the food safety. There is an attached file from Istanbul technical University.
Author information
Authors and Affiliations
Contributions
One author.
Corresponding author
Ethics declarations
Conflict of interest
The authors have not disclosed any competing interest.
Consent to participate
One author.
Consent for publication
(Appropriate statements regarding publishing an individual’s data or image) All rules for scientific research were respected.
Ethics approval
All ethical rules for scientific research were respected.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sabbagh, S. Arsenic contamination in rice, radiation and chemical methods of measurement, and implications for food safety. J Food Sci Technol 60, 1870–1887 (2023). https://doi.org/10.1007/s13197-022-05469-2
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-022-05469-2