Abstract
Semi-solid static mode was applied to brew Huyou (Citrus changshanensis) vinegar, called HV-SS, and changes of main nutrients, volatile compounds and antioxidant capacity at different brewing stages, i.e. Huyou puree (HP), Huyou wine (HW) and HV-SS, were investigated. Meanwhile, all these parameters of HV-SS and Huyou vinegar brewed by liquid static mode (HV-LS) were compared. The contents of the main nutrients except for vitamin C (Vc) and reducing sugar increased significantly after fermentation from HP to HV-SS. DPPH radical-scavenging capacity and reducing power of HW was the highest and ABTS+ scavenging capacity of HV-SS was the highest. Acids, flavonoids, phenolics and Vc jointly played important roles in the antioxidant capacity of HV-SS. Forty-six aroma compounds were detected in HP and alcohols were the principal components (15 alcohols accounted for 46.2%). During HW fermentation, most of the compounds in HP (d-limonene, hexanal, hexanol and 3-hexen-1-ol) were nearly disappeared, and some new alcohols, esters and alkanes were formed, being the abundant compounds in HW and accounting for 62.99%, 18.44% and 10.52% respectively. There were 51 aroma compounds detected in HV-SS and 10 more than those in HV-LS. The types or contents of esters (22.13%), acids (50.49%) and aldehydes (7.33%) in HV-SS were markedly different from those in HP, HW and HV-LS, giving the unique aroma and good flavor to HV-SS. Therefore, semi-solid static fermentation mode was more suitable for brewing Huyou vinegar on the aspect of balancing the nutrients characteristics and flavor compounds formation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Huyou (Citrus changshanensis), a hybrid of Citrus sinensis and Citrus grandis, is originally planted in Quzhou area of Zhejiang province and now produced and consumed in several provinces in south China (Xu et al. 2006). The fruit is rich in nutrients and bioactive compounds and it has the effect of regulating blood sugar level (Abeysinghe et al. 2007; Zhang et al. 2012). Huyou fruit is usually eaten in fresh and has a bitter flavor although the dried immature sour one has been recently accepted as the material of traditional Chinese medicines. The high yield of fresh fruits in recent year has reached to its saturation point in the local fresh edible markets. On the other hand, some Huyou fruits with bruised or inferior sensory quality are discarded and become rotten, causing serious waste of resources and environmental pollution. It is necessary to make full use of these fruits and develop products that are popular to the public while the original nutrients in Huyou fruits are retained.
Vinegar is a traditional fermented food and consumed mainly in Asian countries, but it is widespread all over the world nowadays (Ho et al. 2017). In the recent few years, new products made from various fruits have appeared on the market. Many studies have been published on developing new vinegar products with fruits, such as apple (Qi et al. 2017), orange (Cejudo-Bastante et al. 2016), strawberry (Ubeda et al. 2016) and so on. The popularity of fruit vinegar in the world nowadays is due to the dual nutritional value and healthy function of both fruit and vinegar (Ho et al. 2017). However, there is no Huyou vinegar product yet in terms of sales and research.
There are mainly two modes for making vinegar: traditional solid processes and liquid submerged method. The solid mode could bring abundant nutrients and flavor substances, and has been accepted for the traditional grain vinegar brewing. However, this mode is labor-intensive and time-consuming (Liu et al. 2004; Tesfaye et al. 2002). The liquid submerged fermentation is commonly applied for fruit vinegar brewing with fruit juice due to its higher efficiency and better control, however, the nutrients and flavor substances in the vinegar are relatively less (Tesfaye et al. 2002). Semi-solid state fermentation as a compromise once has been proposed to increase the biodiesel production from sweet sorghum (Economou, et al. 2010). Therefore, this method will be used as reference to balance the flavor, nutrients and productivity of fruit vinegar.
Recently, the studies on fruit vinegar brewing have been focusing on bacteria isolation, process conditions and aromatic profiles, etc. (Callejón et al. 2010; Song et al. 2016; Ubeda et al. 2016). The nutrients and aroma compounds are important factors reflecting the quality of vinegar and theoretical basis of process optimization. It is essential to investigate the changes of the main nutrients and aroma compounds at different stages of different fermentation modes, aiming at selecting fermentation mode, controlling the quality of vinegar and optimizing the brewing conditions.
In this study, the nutrients and aroma compounds at different stages of brewing Huyou vinegar were comparatively investigated. The advantages of semi-solid static mode for Huyou vinegar brewing were illustrated. The results would provide some theoretical and technological information for industrial production.
Materials and methods
Materials
Huyou fruits were collected from an orchard of standardized cultivation in Changshan County, Zhejiang, China. The fruits were ripe with yellow coloration and 10.8°Brix in soluble solid content in average.
The active dry yeast for brewing wine, Saccharomyces cerevisiae (CCTCC M 205128), was purchased from Angel Yeast Co. Ltd. in China. Acetobacter pasteurianus HN 1.01 (CICC 20001), a commonly commercialized strain used in grain vinegar brewing in China, was obtained from China Industrial Microbes Collection Center and stored at − 80 °C in our lab. Pectinase (200,000 units of enzyme activity per gram) was purchased from Imperial Jade Bio-technology Co. Ltd., Yinchuan, China).
Methods
Pretreatment of materials
Huyou fruits after peeling were crushed with a beater to obtain Huyou puree (HP). Pectinase in a concentration of 2.5 g/kg was added into HP and the mixture was incubated at 50 °C for 2 h to get Huyou puree in enzymatic hydrolysis (HP-E).
Huyou vinegar brewing-Alcoholic fermentation
The soluble solid content of HP after enzymolysis was adjusted to 18°Brix by adding sucrose. One gram per liter of S. cerevisiae (dry weight) activated previously in 5% sucrose solution at 38 °C for 20 min was inoculated in 3.5 L HP and cultured statically at 28 °C for 7 days, obtaining Huyou wine (HW).
Huyou vinegar brewing-acetic fermentation
Two different static fermentation modes were adopted at the stage of Huyou vinegar (HV) brewing, liquid static mode and semi-solid static mode. When liquid static mode was adopted, HW broth was centrifuged at 1500×g for 15 min and the supernatant was used for acetic fermentation, obtaining Huyou vinegar brewed by liquid static mode (HV-LS). When semi-solid static mode was adopted, 300 g/kg sterilized mixture of bran and rice husk (2:1, w/w) was added into the HW broth and mixed fully for fermentation, obtaining Huyou vinegar brewed by semi-solid static mode (HV-SS). The bran and rice husk were the conventional matrix in solid fermentation for grain vinegar, and they were in favor of the aeration during acetic fermentation. In both cases, 3% (v/v) A. pasteurianus HY05 was inoculated and cultured statically at 32 °C for 8 days. The residue of fermentation mash was mixed with boiled water at a ratio of 1:2 and soaked for 12 h. Then the mixture was filtered and the residues were soaked in boiled water twice under the same conditions as the first time. The supernatant after filtration of 3 times was mixed and pasteurized at 85 °C for 15 min, obtaining HV. Samples taken from HP, HW, HV-LS and HV-SS were analyzed.
Analysis
Nutritional substances
The content of reducing sugar was analyzed by the 3,5-dinitrosalicylic acid colorimetric method (DNS method). Total acidity was determined by automatic potentiometric titrator (848 Titrino plus, Metrohm Co. Ltd., Switzerland). Total flavonoid content (TFC) and total phenolics content (TPC) content were determined by colorimetric method and vitamin C (Vc) was determined by HPLC (Abeysinghe et al. 2007). Protein concentration was analyzed by Bradford assay.
Antioxidant capacity
The reducing power and the antioxidant capacity (including DPPH radical-scavenging capacity and ABTS+ scavenging capacity) were measured by the methods as reported (Ozturk et al. 2015; Tagliazucchi et al. 2008) with some modification. Briefly, vinegar samples were filtered using a filter of 0.45 μm and 0.3 mL of filtrate was mixed with 0.2 mL PBS buffer (0.2 mol/L, pH 6.6) and 0.5 mL of 1% K3Fe(CN)6 successively, and incubated for 20 min at 50 °C. The mixture was added with 1 mL of 10% trichloroacetic acid after cooled to terminate the reaction. After centrifuged for 10 min at 3000 rpm, 1.5 mL of supernatant was added with 0.2 mL of 1% FeCl3 and 3.0 mL of distilled water, mixed, stood for 5 min, and absorbance of the mixture was measured at 700 nm using a spectrophotometer.
For DPPH radical-scavenging capacity analysis, 0.1 mL of filtrate was mixed with 5 mL of DPPH solutions (0.1 mmol/L) and vigorously mixed with a vortex. Following the incubation for 30 min at 27 °C in dark conditions, absorbance of the mixture was measured at 517 nm using a spectrophotometer.
For ABTS+ scavenging capacity analysis, 0.3 mL of filtrate was mixed with 4 mL of ABTS solution and vigorously mixed with a vortex. Following the incubation for 10 min at 27 °C in dark conditions, absorbance of the mixture was measured at 734 nm using a spectrophotometer.
Elimination ratio (E, %) of radical was described as following Eq. (1):
where A0 and A represent absorbance of control (DPPH/ABTS solution) and the sample, respectively.
Two milliliters of 1, 2, 3, 4, 5% HP, HP-E, HW and HVs solution was respectively used for analyzing the correlation coefficients between antioxidant capacity and HP, HP-E, HW and HVs. Two milliliters of 4% HP, HP-E, HW and HVs solution was used for comparing the antioxidant capacity of HP, HP-E, HW and HVs.
Volatile compounds
Volatile compounds of HP, HW and HV were extracted using headspace solid phase micro-extraction (HS-SPME) (Ozturk et al. 2015). Five milliliters of sample and 2.0 g NaCl were mixed in the head space bottle and sealed with a silicone cap. Then the bottle was incubated at 50 °C for 30 min and the sample was extracted by the extraction fiber of 50/30 µm DVB/CAR/PDMS (Supelco, Sigma-Aldrich Co. Ltd., USA) at 50 °C for 40 min.
The extracted fiber was inserted into the inlet of GC and desorbed at 250 °C for 3 min. Then the aroma compounds extracted from the samples were analyzed by GC–MS (Agilent 7890A-Agilent 5975C, Agilent Technologies USA) equipped with a VF-WAXms column (30 m × 0.25 mm × 0.25 µm). The injection temperature was programmed from 35 °C (held for 3 min) at 2 °C/min to 150 °C firstly, and then it was raised to 250 °C (held for 10 min) at 5 °C/min. Helium as carrier gas was at a flow rate 1.0 mL/min. For MS detection, the electron ionization energy was 70 eV, and the temperature of ion source and quadrupole was set at 230 and 150 °C respectively. Electron ionization mass spectra data range (m/z) was 33–500 amu. All aroma compounds were identified by analogy of the retention index (RI) in the digital library of mass spectral database (NIST 08) or confirmed by retention times of standards in mass spectral. The relative contents were calculated at the ratios between the peak area of one component and the total peak area of all compounds.
Statistical analysis
All experiments reported here were performed in triplicate, and all data were the means of triplicates with standard deviation. The statistical analysis and the level of significance were conducted using Microsoft office 2010 and SPSS Statistics 22.0 software (IBM, USA). Differences between two groups were considered significant when the value of P was less than 0.05. Correlation between main nutrients and antioxidant capacity were analyzed by linear analysis in SPSS Statistics 22.0 software (IBM, USA) and the absolute value of r (|r|) was used for indicating significance level. There would be a significant correlation between two items when |r| was more than 0.95.
Results and Discussion
Main nutrients at different fermentation stages
The contents of main nutrients at different brewing stages of Huyou vinegar are shown in Table 1. The content of Vc decreased continuously during the whole process of pretreatment and fermentation (p < 0.05), because Vc was unstable and more easily oxidized (Klopotek et al. 2005). The contents of all other nutrients were increased in different extent after pectinase treatment, especially for reducing sugar and total phenolics. It was demonstrated that enzymolysis could facilitate the release of nutrients in HP as reported by Ubeda et al. (2013). During the first stage for HW fermentation, the content of reducing sugar and protein decreased significantly (p < 0.05) due to cell growth and metabolism, and the contents of total flavonoids and total phenolics were increased slightly. Some flavonoids and phenolics in the HW were from microorganism metabolism, and the others were dissolved from HP with the accumulation of ethanol because alcohol could accelerate the extraction of flavonoids and phenolics (Abeysinghe et al. 2007; Tagliazucchi et al. 2008).
At the second stage for acetic fermentation with semi-solid mode, parts of sugar, protein and other nutrients contained in the bran could be dissolved with the increase of acetic acid content or decomposed by microorganism (Li et al. 2014). Therefore, the contents of reducing sugar, total flavonoids and protein in HV were increased significantly (p < 0.05). On the other hand, some intracellular protein might also be released along with the autolysis of aging yeast at the end of fermentation.
Correlation between the main nutrients and anti-oxidation
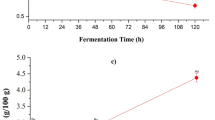
Antioxidant capacity is a healthy indicator of a product and it can prevent some diseases resulted by overloaded free radicals, such as diabetes, cardiovascular disease, cancer and so on. Generally the antioxidant capacity was increased during the brewing process from HP to HV, as shown in Fig. 1. DPPH radical-scavenging capacity and reducing power of HW was the highest and ABTS+ scavenging capacity of HV-SS was the highest. However, the total antioxidant capacity of HV-SS was higher than those of others. It was indicated that some substrates with reducing capacity were released when HP was transformed into HV, and it happened to show in Table 1 that the content of total acids, flavonoids, phenolics and protein was increased significantly during the fermentation process from HP to HV. Therefore, the correlation coefficients between the main nutrients and anti-oxidation were investigated, as shown in Table 2. There was a varied correlation between the main nutrients and the anti-oxidation, except for protein. A high negative correlation between reducing sugar and the anti-oxidation was observed, especially for the ABTS+ scavenging capacity (p < 0.05). The contents of flavonoids and phenolics were in proportion to the antioxidant capacity, and there was an extremely significant correlation between phenolics content and reducing power (p < 0.05). Vc content had a huge positive impact on the anti-oxidation capacity, especially for the DPPH radical-scavenging capacity and reducing power (p < 0.05). Therefore, the antioxidant capacity of HV was mainly dependent on acids, flavonoids, phenolics and Vc jointly.
Volatile compounds at different fermentation stages
The contents of volatile compounds mainly responsible for aroma at different fermentation stages of HV brewing are shown in Fig. 2 and Table 3. Forty-six volatile compounds were detected in HP, mainly including alcohols, esters and aldehydes. Alcohols were the most abundant compounds, their total amount accounting for 46.4%, among which, ethanol, 1-hexanol and 3-hexen-1-ol was 6.0, 11.0, 13.9% respectively. These compounds were mainly detected in HP and they were responsible for the fresh grass odor (Cejudo-Bastante et al. 2016; Chen et al. 2013). The content of hexanal, with a flavor of vanilla or apple, which played an important role in the fragrance of HP (Chen et al. 2013; Tønder et al. 1998), was the highest (24.2%). However, the characteristic aroma compounds of Huyou were d-limonene with typical citrus flavor, linalool with a strong flavor of lily of the valley and 4-terpenol with the flavor of green tea (Cejudo-Bastante et al. 2016; Pérez-López et al. 2006). These terpenoids took an important role in the characteristic flavor of HP, even though their contents were low but their odor threshold value were also low (Tønder et al. 1998).
At alcoholic fermentation stage, the typical aroma compounds of Huyou fruit decreased or disappeared and some representative compounds of wine were generated. Thirty-nine aroma compounds were detected in HW and the types of alcohols decreased significantly, but the total content of alcohols increased and compared with that (46.4%) in HP, it accounted for 63% of all compounds, among which ethanol (41.9%), 2-phenylethanol (8.3%) and 3-methyl-1-butanol (11.0%) were the dominant components. The 2-phenylethanol with an aroma of rose and jasmine was identified as the major aroma compound in HW to ease the pungent odor of some alcohols and it rendered a fresh and soft feeling. It was formed by the Strecher degradation of phenylalanine in S. cerevisiae (Câmara et al. 2006; Callejón et al. 2009). The types and relative contents of esters and alkanes responsible for wine aroma were increased obviously. Most esters have floral or fruity flavors, such as ethyl decanoate and diethyl succinate with slight grape aroma and sweet fragrance, ethyl benzoate with flora odor, ethyl palmitoleate with creamy aroma and weak scent of wax, and ethyl acetate with sweet fruity aroma (Chen et al. 2013; Su and Chien 2010; Tønder et al. 1998; Yu et al. 2012). On the other hand, the types and contents of ketones, aldehydes and heterocyclic compounds were decreased obviously during this phase. Only acetoin, furfural and benzaldehyde were detected in the HW. Acetoin had a pleasant yogurt odor and a fatty creamy butter taste, and it was a precursor of other flavor and fragrance agents (Xiao and Lu 2014). Furfural with a sweet and caramel aroma is mainly generated by the Maillard reaction and benzaldehyde was widely used for preparing food with flavor of almond and coco (Câmara et al. 2006). They were common intermediates of most aromatic substances. Therefore, some acids with sweet caramel flavor and flower fragrance were generated from corresponding aldehydes, such as octanoic acids, sorbic acid and benzoic acid (Ubeda et al. 2016). While some acids with unpleasant acidic smell, such as hexanoic acid, dodecanoic acid, and nonanoic acid (Selli et al. 2003), were disappeared or decreased greatly in HW. They might be converted into corresponding or related esters since the content of hexyl acetate, ethyl decanoate and γ-nonalactone was increased as shown in Table 3.
The types and contents of aroma compounds in HV were obviously different from those in HW, especially in HV-SS. The main change was found in acids, alcohols, ketones and aldehydes. Forty-one and fifty-one aroma compounds were detected in HV-LS and HV-SS respectively. The types of alcohols were increased slightly, but the total content was decreased by 65–81%. The alcohols were basically converted into acids, especially for ethanol. Acids content in HV was about 50% of all aroma compounds. The acetic acid content was increased in dozens of folds and accounted for about 30–40% of all compounds. The content of ethyl acetate was increased about 10 times and it had pleasant fruity aroma (Su and Chien 2010). Some new esters compounds were generated by the joint action of multiple microbes and esterification reaction of acids and alcohols, including methyl acetate with a fruity flavor and slight bitter taste, hexyl acetate with apple and strawberry aroma and fennel taste, phenethyl acetate with sweet flavor of the rose and γ-nonalactone with coconut aroma and fennel taste (Chen et al. 2013; Gholami et al. 2008). The types of aldehydes was increased in vinegars, particularly in HV-SS. Some of them were deemed to be related to the addition of bran and rice husk, such as 5-methyl furfural with cereal and coffee aroma (Kong et al. 2015). Benzaldehyde and acetaldehyde with floral flavor mainly come from the degradation of amino acids and metabolism of microbials. Heterocyclic compounds, such as 2,4,5-trimethyl-1,3-oxazole and 2,3,5-trimethylpyrazine with coco aroma, might be produced by Maillard reaction (Asikin et al. 2014; Xu et al. 2013).
Difference in volatile compounds between HV-LS and HV-SS
A total of 51 aroma compounds in HV-SS and 41 aroma compounds in HV-LS were detected. The main difference between them was esters, aldehydes, alkanes and phenolics. There were 5 ester compounds more in HV-SS than those in HV-LS, and ethyl acetate content was the highest in both cases (15.2% and 12.0% respectively).
Ten alcohols were detected in both vinegars, but the total content of alcohols in HV-LS was twice of that in HV-SS. Furthermore, 2-phenylethanol content (12.9%) in HV-LS was much higher than that in HV-SS (3.3%). Both the types and content of aldehydes in HV-SS (6 types and 6.70%) were more than those in HV-LS (3 types and 2.81%), especially for benzaldehyde. The 2-phenylethanol had a flavor of rose (Andreu-Sevilla et al. 2013), while benzaldehyde had a flavor of almond and coco (Câmara et al. 2006). Therefore, the differences in the content of 2-phenylethanol and benzaldehyde led to different aroma of the vinegars brewed by different modes. In addition, some heterocyclic compounds were only found in HV-SS, such as 2,4-di-tert-butylphenol, 2-methoxy-4-methylphenol, 2,3,5-trimethylpyrazine, 2,3-butanedione, α-ethylidene-benzeneacetaldehyde, 2-hydroxy-benzaldehyde, 5-methyl furfural. They were also the important aroma compounds in vinegar (Fan et al. 2007), and mainly produced by microorganism and Maillard reaction (Chen et al. 2013; Frank et al. 2011; Gonzalez-Rios et al. 2007). These aroma compounds were also detected in the famous traditional grain vinegar in China, Zhenjiang balsamic vinegar and Shanxi aged vinegar (Bai 2013; Sun et al. 2010). Therefore, semi-solid fermentation mode could be used for brewing fruit vinegar with better flavor.
Conclusion
Semi-solid static fermentation mode was proposed to brew HV using Huyou puree. Total acids, flavonoids, phenolics and content were increased significantly in HV compared with those in HP. The typical aroma compounds of Huyou fruit almost disappeared and some new compounds were generated after two-step fermentation from HP to HV-SS. During the fermentation, the types and the contents of esters, acids and aldehydes increased significantly, while the types and contents of alcohols and aldehydes were decreased greatly. In addition, there were also 10 aroma compounds in HV-SS more than those in HV-LS, and those compounds were basically responsible for caramel aromas and fruit sweet fragrance. Furthermore, the operation process and industrialization of semi-solid static mode was simple and economic. In conclusion, the semi-solid static mode was superior to the liquid static one for brewing Huyou vinegar on the view of the nutrients characteristics, flavor compounds formation as well as cost.
References
Abeysinghe DC, Li X, Sun C, Zhang W, Zhou C, Chen K (2007) Bioactive compounds and antioxidant capacities in different edible tissues of citrus fruit of four species. Food Chem 104:1338–1344
Andreu-Sevilla AJ, Mena P, Martí N, Viguera CG, Carbonell-Barrachina ÁA (2013) Volatile composition and descriptive sensory analysis of pomegranate juice and wine. Food Res Int 54:246–254
Asikin Y, Kamiya A, Mizu M, Takara K, Tamaki H, Wada K (2014) Changes in the physicochemical characteristics, including flavor components and Maillard reaction products of non-centrifugal cane brown sugar during storage. Food Chem 149:170–177
Bai J (2013) Metabolics analysis of volatile components in four kinds of Shanxi mature vinegar. J Shanxi Agric Univ 33:2218–2224
Callejón RM, Tesfaye W, Torija MJ, Mas A, Troncoso AM, Morales ML (2009) Volatile compounds in red wine vinegars obtained by submerged and surface acetification in different woods. Food Chem 113:1252–1259
Callejón RM, Torija MJ, Mas A, Morales ML, Troncoso AM (2010) Changes of volatile compounds in wine vinegars during their elaboration in barrels made from different woods. Food Chem 120:561–571
Câmara JS, Alves MA, Marques JC (2006) Changes in volatile composition of Madeira wines during their oxidative ageing. Anal Chim Acta 563:188–197
Cejudo-Bastante C, Castro-Mejías R, Natera-Marín R, García-Barroso C, Durán-Guerrero E (2016) Chemical and sensory characteristics of orange based vinegar. J Food Sci Technol 53:3147–3156
Chen S, Xu Y, Qian MC (2013) Aroma characterization of Chinese rice wine by gas chromatography-olfactometry, chemical quantitative analysis, and aroma reconstitution. J Agric Food Chem 61:11295–11302
Economou CN, Makri A, Aggelis G, Pavlou S, Vayenas DV (2010) Semi-solid state fermentation of sweet sorghum for the biotechnological production of single cell oil. Bioresour Technol 101:1385–1388
Fan W, Xu Y, Zhang Y (2007) Characterization of pyrazines in some Chinese liquors and their approximate concentrations. J Agric Food Chem 55:9956–9962
Frank S, Wollmann N, Schieberle P, Hofmann T (2011) Reconstitution of the flavor signature of Dornfelder red wine on the basis of the natural concentrations of its key aroma and taste compounds. J Agric Food Chem 59:8866–8874
Gholami M, Hayasaka Y, Coombe BG, Jackson JF, Robinson SP, Williams PJ (2008) Biosynthesis of flavor compounds in Muscat Gordo Blanco grape berries. Aust J Grape Wine Res 1:19–24
Gonzalez-Rios O, Suarez-Quiroz ML, Boulanger R, Barel M, Guyot B, Guiraud JP et al (2007) Impact of “ecological” post-harvest processing on coffee aroma: II. Roasted coffee. J Food Compos Anal 20:297–307
Ho CW, Lazim AM, Fazry S, Zaki UKHH, Lim SJ (2017) Varieties, production, composition and health benefits of vinegars: a review. Food Chem 221:1621–1630
Klopotek Y, Otto K, Böhm V (2005) Processing strawberries to different products alters contents of vitamin C, total phenolics, total anthocyanins, and antioxidant capacity. J Agric Food Chem 53:5640–5646
Kong H, Zhao G, Wang W, Yan W (2015) Identification and comparison of bound aroma components in six kinds of fruits by GC–MS. Food Sci 36:140–144
Li T, Lo YM, Moon B (2014) Feasibility of using Hericium erinaceus as the substrate for vinegar fermentation. LWT Food Sci Technol 55:323–328
Liu D, Zhu Y, Beeftink R, Ooijkaas L, Rinzema A, Chen J et al (2004) Chinese vinegar and its solid-state fermentation process. Food Rev Int 4:407–424
Ozturk I, Caliskan O, Tornuk F, Ozcan N, Yalcin H, Baslar M et al (2015) Antioxidant, antimicrobial, mineral, volatile, physicochemical and microbiological characteristics of traditional home-made Turkish vinegars. LWT Food Sci Technol 63:144–151
Pérez-López AJ, Saura D, Lorente J, Carbonell-Barrachina ÁA (2006) Limonene, linalool, α-terpineol, and terpinen-4-ol as quality control parameters in mandarin juice processing. Eur Food Res Technol 222:281–285
Qi Z, Dong D, Yang H, Xia X (2017) Improving fermented quality of cider vinegar via rational nutrient feeding strategy. Food Chem 224:312–319
Selli S, Cabaroglu T, Canbas A (2003) Flavor components of orange wine made from a Turkish cv. Kozan. Int J Food Sci Technol 38:587–593
Song NE, Cho HS, Baik SH (2016) Bacteria isolated from Korean black raspberry vinegar with low biogenic amine production in wine. Braz J Microbiol 47:452–460
Su MS, Chien PJ (2010) Aroma impact components of rabbiteye blueberry (Vaccinium ashei) vinegars. Food Chem 119:923–928
Sun Z, Zhao J, Zou X, Chen Q, Li G (2010) Changes of characteristic aroma component in Zhenjiang fragrance vinegar during processing and analysis of their formation mechanism. J Chin Inst Food Sci Technol 10:120–128
Tagliazucchi D, Verzelloni E, Conte A (2008) Antioxidant properties of traditional balsamic vinegar and boiled must model systems. Eur Food Res Technol 227:835–843
Tesfaye W, Morales ML, García-Parrilla MC, Troncoso AM (2002) Wine vinegar: technology, authenticity and quality evaluation. Trends Food Sci Technol 13:12–21
Tønder D, Petersen MA, Poll L, Olsen CE (1998) Discrimination between freshly made and stored reconstituted orange juice using GC odor profiling and aroma values. Food Chem 61:223–229
Ubeda C, Callejón RM, Hidalgo C, Torija MJ, Troncoso AM, Morales ML (2013) Employment of different processes for the production of strawberry vinegars: effects on antioxidant activity, total phenols and monomeric anthocyanins. LWT Food Sci Technol 52:139–145
Ubeda C, Callejón RM, Troncoso AM, Moreno-Rojas JM, Peña F, Morales ML (2016) A comparative study on aromatic profiles of strawberry vinegars obtained using different conditions in the production process. Food Chem 192:1051–1059
Xiao Z, Lu JR (2014) Strategies for enhancing fermentative production of acetoin: a review. Biotechnol Adv 32:492–503
Xu C, Bao L, Zhang B, Bei Z, Ye X, Zhang S, Chen K (2006) Parentage analysis of huyou (Citrus changshanensis) based on internal transcribed spacer sequences. Plant Breed 125:519–522
Xu Q, Liu J, Song H, Zou T, Liu Y, Zhang S (2013) Formation mechanism of volatile and non-volatile compounds in peptide-xylose Maillard reaction. Food Res Int 54:683–690
Yu YJ, Lu ZM, Yu NH, Xu W, Li GQ, Shi JS et al (2012) HS-SPME/GC-MS and chemometrics for volatile composition of Chinese traditional aromatic vinegar in the Zhenjiang region. J Inst Brew 118:133–141
Zhang J, Sun C, Yan Y, Chen Q, Luo F, Zhu X et al (2012) Purification of naringin and neohesperidin from Huyou (Citrus changshanensis) fruit and their effects on glucose consumption in human HepG2 cells. Food Chem 135:1471–1478
Acknowledgements
This research was supported by the Special Action Program for Enriching Farmers and Strengthening Counties by Ministry of Science and Technology in China.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lu, S., Cao, Y., Yang, Y. et al. Effect of fermentation modes on nutritional and volatile compounds of Huyou vinegar. J Food Sci Technol 55, 2631–2640 (2018). https://doi.org/10.1007/s13197-018-3184-0
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-018-3184-0