Abstract
Phenolic composition of red wines from Stanušina, a grape variety indigenous of the Republic of Macedonia, was compared with the regional Vranec and the international Cabernet Sauvignon. The extent of skin contact (i.e. maceration time) on levels of phenolic compounds and antioxidant activity of wines was evaluated. A total of 19 phenolic compounds were identified and quantified. Among these malvidin-3-glucoside and its derivatives were the major compounds, while caftaric acid was the predominant cinnamic acid derivative, followed by catechin, the main flavan-3-ol. The concentration of hydroxycinnamic acids, anthocyanins and (+)-catechin ranged from 224 to 511 mg/L, 22 to 360 mg/L and 26 20 to 375 mg/L, respectively and peaked at 3rd, 6th and 9th day of maceration, respectively. However, prolong maceration slightly decreased their concentration. Stanušina wines presented high levels of hydroxycinnamic acids and antioxidant activity.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The quality of red wines depends to great extent on their phenolic composition, the most important components which determine the colour, mouthfeel, astringency and bitterness of the wine (Casassa and Harbertson 2014). These compounds origin from different parts of the grape: (i) grape skins contain anthocyanins, flavan-3-ols, flavonols, dihydroflavonols, hydroxycinnamoyl tartaric acids, hydroxybenzoic acids and hydroxystilbenes, whereas (ii) flavan-3-ols and gallic acid are dominant in the seeds, and (iii) hydroxycinnamoyl tartaric acids are mainly present in the juice (Adams 2006; Doshi et al. 2015). During the last decades, the interest on bioactive phenolic compounds has significantly increased due to their benefits to human health preventing coronary heart disease or acting as antioxidants and antitumoral agents (Shahidi and Wanasundara 1992).
Phenolic composition mainly depends on the grape variety, but also on climate conditions, soil, ripening stage, vine cultivation as well as on winemaking practices that are applied for wine production (Sacchi et al. 2005; Kennedy et al. 2006). Maceration as well as pre-fermentative cold maceration (González-Neves et al. 2015), fermentation temperature, yeast strain, racking duration, type of lees (Sharma et al. 2015) influences the phenolics extraction from grapes into the must. Longer skin contact leads to higher extraction of phenolics, especially at the beginning of maceration when anthocyanins and flavonols are the first compounds that are extracted from the skins, followed by extraction of the seed flavan-3-ols in the later days of maceration. In fact, seed flavan-3-ols are the last extracted compounds since they are protected with lipidic layer which is disrupted when appropriate content of alcohol is formed, allowing their releasing from the seeds (Sun et al. 2011).
During the last decade many studies have been performed on analysis of phenolic compounds in wines. Although recent data on the phenolic composition of Macedonian red wines Vranec, Merlot and Syrah are available (Ivanova-Petropulos et al. 2015), the importance of maceration time on the individual phenolic composition was highlighted on Vranec wines only (Ivanova et al. 2011). In this view, Stanušina is a red grape variety indigenous of the Republic of Macedonia and herein only cultivated, i.e. it is found nowhere else in the world. Consequently, there are no data available in the international scientific literature about the chemical composition of this grape variety.
Therefore, the aim of this study was to evaluate the phenolic composition of Stanušina wines (exclusive of Republic of Macedonia), and then to compare them with Vranec wines (the most popular in Republic of Macedonia, as well as in the Balkan region) and Cabernet Sauvignon wines (the most known international red variety). In particular, Stanušina wine was studied for the first time, and the extraction of phenolic compounds during maceration was monitored with the time (3, 6 and 9 days).
Materials and methods
Chemicals and reagents
The following chemicals and reagents were from commercial source: methanol, acetonitrile, perchloric acid, formic acid (Merck, Darmstadt, Germany), 2,2-diphenyl-1-picrylhydrazyl radical (DPPH), gallic acid, (+)-catechin, (−)-epicatechin, caffeic acid, syringic acid (Sigma-Aldrich, Milano, Italy), protocatechuic acid, vanillic acid, p-hydroxybenzoic acid, p-coumaric acid (Extrasynthese, Genay, France) and 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid (Trolox©) used for DPPH assay calibration from Fluka Analytical (Sigma-Aldrich, Milano, Italy).
Grapes
Grapes from V. vinifera L. varieties Stanušina, Vranec and Cabernet Sauvignon cultivated in the Tikveš wine region (Republic of Macedonia) were harvested in September 2013, at optimal technological maturity: 24.3, 20.6 and 22.6°Brix, respectively (levels between 18 and 22°Brix are desirable as objective criteria for estimating optimal grape maturity). Stanušina grapes were collected from 23-year-old vineyards with area of 0.7-ha, while Vranec and Cabernet Sauvignon grapes were grown at 0.5 and 1-ha, 15- and 17-year-old vineyards, respectively. The distance between the rows was 1.5 m and the distance between the vines was 1.0 m. Grapes were manually harvested early in the morning and placed in crates.
Wine-making
Manually harvested grapes (100 kg) of each variety were transported to the Povardarie winery (Negotino, R. Macedonia) and grapes were processed separately. After processing of grapes with mechanical crusher/destemmer, the must of each grape variety was collected in a plastic tank (50 L) and then added with 80 mg/L of SO2 in a form of 5 % sulphurous acid, before the inoculation with commercial dry yeast Excellence SP Saccharomyces cerevisiae (Lamothe-Abiet, Canejan/Bordeaux, France). The yeast was previously rehydratated in water (20 g/hL, at 35 °C × 15 min). To reinforce the yeast activity, a commercial mixture of nutrients, vitamins, minerals, fatty acids and sterols (20 g/hL Oenostim activator, Lamothe-Abiet, France) was added to the musts.
Soon after the addition of SO2 and yeast, Stanušina, Vranec and Cabernet Sauvignon must was divided into three replicated sub-samples to monitor the extraction of phenolics compounds from skins with macerated for 3, 6 and 9 days, obtaining 9 sets of wines in total for each variety. In order to obtain representative samples for each wine variety and each maceration time, all samples were prepared by mixing wines from three tanks produced with the same technological treatment. During the alcoholic fermentation, the “pumping over” was applied twice a day for all sub-samples, and after the maceration period, wines were separated from the pomace by mechanically pressing and stabilized at 4 °C for a period of three weeks before bottling. High Performance Liquid Chromatography (HPLC) and spectrophotometric analyses were performed after 4 months of storage of the wines in the cellar at 12–13 °C.
Chemical composition
In order to determine the general chemical composition of wines, official methods of analysis of wines (OIV 2016) were used and following parameters were analyzed: alcohol (OIV-MA-AS312-01 A), dry extract (OIV-MA-AS2-03B), specific density (OIV-MA-AS2-01 A), total acidity (OIV-MA-AS313–01), volatile acidity (OIV-MA-AS313–02), total SO2 and free SO2 (Ivanova-Petropulos and Mitrev 2014).
Spectrophotometric analyses
Spectrophotometric analyses were performed at the following wavelength: 280 nm (total phenols), 420 nm (browning degree), 520 nm and 620 nm (anthocyanins) nm with a UV-VIS spectrophotometer (Shimadzu, UV-mini 1240, Milan, Italy) using a cuvette with 1 cm optical path against the blank, i.e. water (Harbertson and Spayd 2006). Based on the Vis measurements, the following parameters were calculated: colour intensity (CI = Σ 420 + 520 + 620 nm) and colour tonality (H = 420/520 nm) (OIV 2016).
Total phenols content, expressed as mg/L gallic acid equivalent (GAE/L), was determined by reading of the absorbance of diluted samples (1:100 dilutions in distilled water) at 280 nm, and using a calibration curve of gallic acid standard solution in the range of 1.95 to 31.25 mg/L (Ribéreau-Gayon 1970).
Antioxidant activity of wines
Antioxidant activity of wines was determined as a radical scavenging ability following the procedure described by Brand-Williams et al. (1995). Briefly, a volume of 200 μL of wine was added to 3 mL of a methanol solution of the radical DPPH with concentration of 0.025 mol/L, and measured at 515 nm after 1 h storage at dark. Antioxidant activity was calculated from a calibration curve constructed using methanol solutions of Trolox with concentrations ranged between 0.19–93 mg/L, and expressed as mg Trolox equivalent/L (TE/L).
HPLC analysis
The HPLC system was equipped with temperature control oven, photodiode array detector (DAD) and a Chromeleon chromatography manager software v. 6.60 SP2 (Dionex DX500, Milano, Italy) and used for identification and quantification of anthocyanins, phenolic acids and flavan-3-ols in wines. The samples were filtered using 0.20 μm cellulose acetate membrane (Millipore, Milano, Italy) before direct injection into the HPLC system, kept at 30 °C.
Anthocyanins and related pigments were analyzed with the Gemini RP-C18 column (250 × 4.6 mm; 5 μm particle size; 110 Å porosity; Phenomenex, Bologna, Italy) using the following mobile phases: water/methanol (70/30, v/v) containing 6 mL/L of 70 % perchloric acid (solvent A) and water/methanol (25/75, v/v) containing 6 mL/L of 70 % perchloric acid (solvent B). The flow rate was 0.9 mL/min and the gradient elution for solvent B was as follows: 0 min, 0 %; 23 min, 25 %; 51 min, 70 %; 60 min, 100 %; 65 min, 0 %. Anthocyanins were recorded at 530 nm (Ivanova-Petropulos et al. 2015).
Hydroxycinnamic acid derivatives and flavan-3-ols were analyzed with the Aquapore ODS-300 RP-C18 column (250 × 4.6 mm; 7 μm particle size; 300 Å porosity; Applied Biosystems, San Jose, CA, USA) using the following mobile phases: solvent A (water/formic acid, 98/2, v/v) and solvent B (acetonitrile/water/formic acid, 80:12:2, v/v/v), at flow rate of 0.5 mL/min. The proportion of solvent B was: 0–50 min, 9 %; 65–70 min, 10 %; 77 min, 30 %; 80–97 min, 0 %. Protocatechuic, p-hydroxybenzoic and vanillic acids were quantified at 256 nm, (+)-catechin, (−)-epicatechin, gallic and syringic acids at 280 nm, p-coumaric acid at 308 nm, whereas caftaric, caffeic, coutaric and fertaric acids at 324 nm (Ivanova-Petropulos et al. 2015).
Statistical analysis
To evaluate the effect of maceration time (3, 6 and 9 days) on the bioactive phenolic composition of analyzed red wines (three replicated sub-samples), the one-way ANOVA, regression and statistics was performed by using TANAGRA 1.4.28 software (Lyon, France). Moreover, Principal Component Analysis (PCA) was carried out to evaluate the hidden relationship among the selected variables, i.e. the wine composition in terms of selected bioactive compounds (anthocyanins, flavan-3-ols, hydroxybenzoic acids and hydroxycinnamic acids) and the grape varieties (Stanušina, Vranec and Cabernet Sauvignon).
Results and discussion
Chemical composition
Table 1 shows chemical composition of red wines Stanušina, Vranec and Cabernet Sauvignon. Alcohol content of wines ranged from 11.44 % to 13.64 % v/v, which was within the regulatory limit of 20 % v/v for wines of area C III b (Reg. CE 606/2009). Average alcohol content in Stanušina (13.57 %), Cabernet Sauvignon (12.97 %) and Vranec wines (11.52 %) was related to the sugar content of grapes at harvest (24.3, 22.6 and 20.6 °Brix, respectively). The high total acidity (range: 5.5–6.5 g/L) prevents microbial growth during storage and aging, and confers sourness to the wines, in agreement with previous findings for other wines from Macedonia (Ivanova-Petropulos et al. 2015) and the near Balkan region as well (Rajković and Sredović 2009; Košmerl et al. 2013). The volatile acidity content (0.57 ± 0.1 g/L) did not affect the quality of wines that was protected from further oxidation and microbial contamination by addition of free SO2 (30–42 mg/L), while the total SO2 ranged from 62 to 93 g/L.
Total phenols, colour intensity, hue and antioxidant activity
Total phenols (TP) ranged from 733 to 1631 mg/L GAE (mean 1128 mg/L GAE) (Table 1). On average, Cabernet Sauvignon wines showed the highest TP content (1574 mg/L), followed by Vranec (1014 mg/L) and Stanušina (795 mg/L), which were lower than those reported for the Macedonian Vranec (mean 3841 mg/L, GAE) and Merlot (mean 2838 mg/L, GAE) wines produced under different winemaking conditions (Ivanova et al. 2009; Ivanova et al. 2012). The effect of skin maceration time on the total phenolics content of wines was highest at day 9, with a lack of significant difference (p > 0.05) between wines produced with 6 and 9 days of maceration, regardless of the variety.
All wines showed high values of antioxidant activity (range: 100–117 mg/L TE) (Table 1), regardless of the variety and maceration time, with the highest average antioxidant activity observed in Cabernet Sauvignon (115 mg/L, TE), followed by Vranec (108 mg/L, TE) and Stanušina wines (102 mg/L, TE). As expected, the total phenols content was well correlated with the antioxidant activity (r 2 = 0.922) (Fig. 1), which explained the significant contribution of the total phenols in terms of antioxidant properties of red wines. In particular, according to Zúñiga et al. (2014) anthocyanins and gallic acid were the compounds with the greatest effect on the antioxidant capacity of Carménère red wines.
With regard to colour intensity (CI), Cabernet Sauvignon wine showed the highest average values (14.51 AU) followed by Vranec (5.18 AU) and Stanušina (1.90 AU) (Table 1), observed at day 3, followed – as expected – by color drop with time due to the (i) copigmentation and (ii) loss of anthocyanins by precipitation, their adsorption on the pomace and/or oxidative polymerization of monomeric anthocyanins. Hue (H) values ranged between 0.33 to 0.51 for Vranec and Cabernet Sauvignon (Table 1), respectively, which are characteristic for young red wines (Glories 1984a, 1984b) and were in agreement with values from the literature (Tsanova-Savova et al. 2002; Kontkanen et al. 2005; Ivanova-Petropulos et al. 2015). Stanušina wines showed high hue values (1.12, on average) due to the low content of red compounds that conferred a characteristic pale red colour compared to Vranec and Cabernet Sauvignon, as well as other red grape varieties (Tsanova-Savova et al. 2002; Kontkanen et al. 2005).
Anthocyanins composition
The concentration of individual anthocyanins in Stanušina, Vranec and Cabernet Sauvignon red wines sampled at different time (3, 6 and 9 days) during winemaking was determined by HPLC-DAD analysis (Table 2) (Fig. 2a).
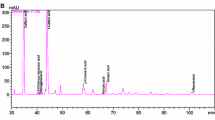
UV-Vis chromatogram of Cabernet Sauvignon wine sample recorded at 530 nm, 324 nm and 280 nm for separation and quantification of anthocyanins, flavan-3-ols/hydroxybenozic acids and hydroxycinnamic acids derivatives, respectively. Peak identification: gallic acid, (1); (+)-catechin, (2); (−)-epicatechin adducts (3); caftaric acid, (4); coutaric acid, (5); caffeic acid, (6); fertaric acid, (7); delphinidin-3-glucoside, (8); petunidin-3-glucoside, (9); peonidin-3-glucoside, (10); malvidin-3-glucoside, (11); vitisin B, (12); petunidin-(6 acetyl)-3-glucoside, (13); peonidin-(6 acetyl)-3-glucoside, (14); malvidin-(6 acetyl)-3-glucoside, (15); peonidin-coumaroyl-3-glucoside, (16); malvidin-coumaroyl-3-glucoside, (17)
In total, 9 anthocyanins were identified and quantified in wines including four monoglucosides, three acetylglucosides and two p-coumaroylglucosides. Malvidin-3-glucoside was the main anthocyanin in all wines regardless the variety and maceration time, as expected for V. vinifera varieties, followed by petunidin-3-glucoside (Table 2). Cyanidin-3-glucoside was detected in samples, but its concentration was below the limit of quantification. Similarly, peonidin-3-glucoside and delphinidin-3-glucoside were below the limit of quantification in Stanušina wines. Amongst different wines, malvidin-3-glucoside was highest in Vranec wines (577 mg/L, on average), followed by Cabernet Sauvignon and Stanušina (mean values: 384 and 101 mg/L, respectively). Similar trend was observed for petunidin-3-glucoside, presenting highest average value in Vranec (40.7 mg/L), then in Cabernet Sauvignon (15.9 mg/L) and Stanušina (4.03 mg/L), probably due to the genetic expression of each grape variety. In general, Vranec wines showed highest mean value of total anthocyanins (813 mg/L), followed by Cabernet Sauvignon (700 mg/L) and Stanušina (132 mg/L).
The group of anthocyanin monoglycosides represented the highest proportion of all anthocyanins in all wines, ranging on average from 58.1 to 80.1 % for Cabernet Sauvignon and Stanušina wines, respectively. Acetyl derivatives were highest in Cabernet Sauvignon (36.6 %, on average) and lowest in Stanušina wines (7.14 %), while the p-coumaroylglucosides ranged from 5.39 % (Cabernet Sauvignon) to 12.8 % (Stanušina). The ratio of acetylglucosides and p-coumaroylglucosides (ΣAcGlc/ΣcoumGlc) (Table 2) proposed as an authenticity index for varietal of red wines, was calculated and found values for Vranec wines (mean 1.53) in agreement with previous data for Vranec wines (range: 1.0–1.7) (Ivanova-Petropulos et al. 2015). All analyzed wines, regardless the extent of skin contact, showed similar values for anthocyanins with Cabernet Sauvignon, Cencibel and Syrah wines produced in the region of La Mancha in Spain (Hermosín Gutiérrez et al. 2005), but higher concentration of malvidin-3-glucoside and anthocyanin monoglucosides compared to Cabernet Sauvignon and Merlot wines produced in the region of Madrid in Spain (Ortega et al. 2008).
The effect of winemaking on anthocyanins content of wine showed that, regardless the variety, after 6 days of skin maceration, anthocyanins content was the highest, including the anthocyanin monoglycosides, acetyl glucosides and p-coumaroylglucosides. By prolonging the extent of skin maceration (9 days), the abovementioned compounds showed a slight decrease for each variety. These results were consistent with previous findings, that highlighted the early extraction of anthocyanins during winemaking (Gil-Muñoz et al. 1997; Bautista-Ortín et al. 2004; Herjavec et al. 2012), followed by a decreasing trend till the end of malolactic fermentation. Furthermore, longer vinification was very often accompanied by oxidative polymerization of the monomeric anthocyanins and their complexation with other phenolics, whereupon oligomeric and polymeric pigments were formed that may got precipitated and decreased the red colour, enhancing the brown colour of wine (Somers 1971; Cheynier et al. 2006).
Non-anthocyanin composition
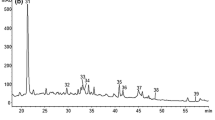
Phenolic acids analysis of wine, recorded at 280 nm and 324 nm, is presented at Fig. 2b and c, respectively.
Three hydroxybenzoic acids, namely protocatechuic acid, gallic acid and syringic acid, were identified and quantified. Gallic acid was the main hydroxybenzoic acid in all wines, with an average value of 263 mg/L in Cabernet Sauvignon wines, 139 mg/L in Vranec wines and 122 mg/L in Stanušina wines (Table 3). These results are in agreement with those reported on Italian (Tarola et al. 2007), Hungarian red wines (Pour Nikfardjam et al. 2006) and Thai local wines (Vichapong et al. 2014). Gallic acid mainly originates from the grapes whereas it could be present in a form of tartaric acid esters; also, the presence of gallic acid in wines could result from the breakdown of both hydrolyzable and condensed tannins, the latter if containing gallate esters. In terms of total hydroxybenzoic acids, Cabernet Sauvignon presented highest mean value (289 mg/L), followed by Vranec (195 mg/L) and Stanušina (148 mg/L), which was in accordance with other studies (Ertan Anli and Nilüfer 2009; Mendoza et al. 2011; Vichapong et al. 2014).
Extend in the skin contact, resulted into increase in the content of gallic acid during fermentation, with highest content in Cabernet Sauvignon and Stanušina wines at day 9, while the Vranec wine showed high content of gallic acid at day 6. In general, the content of total hydroxybenzoic acids increased during winemaking and reached to the highest concentration in the wines after 9 days of maceration, which was in agreement with previous results (Plavša et al. 2012).
Within the hydroxycinnamic acids, five compounds were detected including caftaric, coutaric, fertaric, caffeic and p-coumaric acids. Among them, caftaric acid was the most important (mean values: Stanušina 409 mg/L, Vranec 170 mg/L and Cabernet Sauvignon 120 mg/L), followed by coutaric acid (mean values: Vranec 26.1 mg/L, Stanušina 23 mg/L and Cabernet Sauvignon 15.1 mg/L) and caffeic acid (mean values: Stanušina 37.8 mg/L, Vranec 12.7 mg/L and Cabernet Sauvignon 8.78 mg/L) (Table 3). Comparing the varieties, Stanušina wines showed highest average concentration of total hydroxycinnamic acids and derivatives (492 mg/L), followed by Vranec (240 mg/L) then Cabernet Sauvignon (170 mg/L), most probably because of the varietal differences.
Influence of skin contact period was noticed on the content of hydroxycinnamic acids also. In particular, caftaric acid and hydoxycinnamates reached to maximum concentration after 3 days of maceration, regardless the variety, followed by decrease with maceration time (day 6 and 9, respectively). A similar trend was noticed for caffeic acid in Stanušina and Cabernet Sauvignon wines. The lost of these compounds during winemaking was probably due to their ability to bind anthocyanins, stabilizing the red wine colour, as well as their involvement in non-enzymatic autoxidation of vicinal dihydroxyphenols (Schwarz et al. 2003). In general, the amount of both hydroxybenzoic and hydroxycinnamic acids seemed to be grape variety dependent.
With regard to flavan-3-ols, only (+)-catechin was identified and quantified (average value) in wines as follows: Cabernet Sauvignon (252 g/L), Stanušina (205 mg/L) and Vranec wines (164 mg/L) (Table 3). In particular, the content of catechin in Vranec wines was lower compared with the commercial Macedonian Vranec wines (highest average amount: 319 mg/L) (Ivanova-Petropulos et al. 2015), while the level of catechin in Cabernet Sauvignon was higher compared to Cabernet Sauvignon wines from the same study (mean content 170 mg/L). In comparison with Serbian red wines, in which the average content of catechin was 34.2 mg/L (Radovanović et al. 2012), as well as wines from Argentina (42.6 mg/L, on average) (Fanzone et al. 2012), Macedonian red wines Stanušina, Vranec and Cabernet Sauvignon showed significantly higher concentration (207 mg/L, on average) regardless the extent of skin contact.
As expected, the content of (+)-catechin increased with maceration time similarly in all wines due to the enhanced extraction of flavan-3-ols from seeds during the late phases of fermentation, when appropriate level of alcohol is formed (Glories and Saucier 2000). The results were in agreement with previous findings on anthocyanins and tannins in grape and wine which emphasise that longer skin contact time can prompt higher extraction of tannins from skins and seeds (Sacchi et al. 2005).
Principal component analysis
Principal Component Analysis (PCA) was applied using the dataset of individual phenolic compounds obtained from the HPLC analysis. PCA was used to explore the effect of each variable (grape variety and skin contact of 3, 6 and 9 days) based on the phenolic profile of the analysed wines, i.e. Stanušina, Vranec and Cabernet Sauvignon. The first two principal components, PC1 and PC2, accounted for 79.54 % of the total variance (53.36 % for PC1 and 26.18 % for PC2), thus explaining a significant information in the dataset. The projection of the wine samples on the first two principal components showed a clear separation according to the variety (Fig. 3a): Vranec wines (V) were located in the down negative part of PC1 and Cabernet Sauvignon (CS) in the upper positive part of PC1, while Stanušina wines (S) were located on the left side of PC1. The plot also highlighted the effect of maceration time on the composition of red wines in the following increasing order: Stanušina, Vranec and Cabernet Sauvignon.
Principal components responsible for the differences in the phenolic composition of the wines produced from different varieties and maceration time were determined and presented in the scatter plot in Fig. 3b. The responsible components for the separation of Cabernet Sauvignon wines were acetyl-3-glucosides of malvidin, petunidin and peonidin and gallic acid which prevailed in the positive part of the first principal component, while anthocyanin monoglucosides and p-coumaroylglucosides were characteristic for Vranec wines. In addition, hydroxycinnamic acids caftaric and caffeic acids were dominant compounds in Stanušina wines. In general, separation of the wines was performed according to the varietal characteristics.
Conclusions
Present work provides new evidence on the phenolic composition of the Stanušina, Vranec and Cabernet Sauvignon red wines. Monitoring of winemaking showed a complex extraction pattern that vary with maceration time and grape variety, with hydroxycinnamic acids and anthocyanins were observed to be present in the highest content after 3 and 6 days of maceration, respectively, followed by a slight decrease with time. Hydroxybenzoic acids and (+)-catechin content was highest at 9 days of maceration. Compared to Cabernet Sauvignon and Vranec, Stanušina wines showed low level of anthocyanins, but relatively high content of hydroxycinnamic acids, such as caftaric and caffeic acids, and antioxidant activity as well. Obtained results will improve the understanding of phenolic extraction during red winemaking, especially important for the exclusive autochthonous Stanušina red grape variety.
References
Adams DO (2006) Phenolics and ripening in grape berries. Am J Enol Vitic 57:249–256
Bautista-Ortín AB, Fernádez-Fernádez JI, López-Roca JM, Gómez-Plaza E (2004) Wine-making of high coloured wines: extended pomace contact and run-off of juice prior to fermentation. Food Sci Technol Int 10:287–295. doi:10.1177/1082013204047565
Brand-Williams W, Cuvelier ME, Berset C (1995) Use of free radical method to evaluate antioxidant activity. LWT Food Sci Technol 28:25–30. doi:10.1016/S0023-6438(95)80008-5
Casassa LF, Harbertson JF (2014) Extraction, evolution, and sensory impact of phenolic compounds during red wine maceration. Annu Rev Food Sci Technol 5:83–109. doi:10.1146/annurev-food-030713-092438
Cheynier V, Dueñas-Paton M, Salas E, Maury C, Souquet J-M, Sarni-Manchado P, Fulcrand H (2006) Structure and properties of wine pigments and tannins. Am J Enol Vitic 57:298–305
Doshi P, Adsule P, Banerjee K, Oulkar D (2015) Phenolic compounds, antioxidant activity and insulinotropic effect of extracts prepared from grape (Vitis vinifera L) byproducts. J Food Sci Tech Mys 52:181–190. doi:10.1007/s13197-013-0991-1
Ertan Anli R, Nilüfer V (2009) Antioxidant phenolic substances of Turkish red wines from different wine regions. Molecules 14:289–297. doi:10.3390/molecules14010289
Fanzone M, Zamora F, Jofré V, Assof M, Gómez-Cordovés C, Peña-Neira Á (2012) Phenolic characterisation of red wines from different grape varieties cultivated in Mendoza province (Argentina). J Sci Food Agric 92:704–718. doi:10.1002/jsfa.4638
Gil-Muñoz R, Gómez-Plaza E, Martínez A, López-Roca JM (1997) Evolution of the CIELAB and other spectrophotometric parametars during wine fermentation. Influence of some pre and post-fermentative factors. Food Res Int 30:699–705. doi:10.1016/S0963-9969(98)00029-5
Glories Y (1984a) La couleur des vins rouges. Mesure, origine et interprétation. Partie I. Connaiss Vigne Vin 18:195–217
Glories Y (1984b) La couleur des vins rouges. Mesure, origine et interprétation. Partie II. Connaiss Vigne Vin 18:253–271
Glories Y, Saucier C (2000) Tannin evolution from grape to wine. Effects on wine taste. In: Ratz J (ed) The ASEV 50th Anniversary Annual Meeting. ASEV, Davis, pp. 353–355
González-Neves G, Favre G, Gil G, Ferrer M, Charamelo D (2015) Effect of cold pre-fermentative maceration on the color and composition of young red wines cv. Tannat J Food Sci Tech Mys 52:3449–3457. doi:10.1007/s13197-014-1410-y
Harbertson JF, Spayd S (2006) Measuring phenolics in the winery. Am J Enol Vitic 57:280–288
Herjavec S, Jeromel A, Maslov L, Jagatić Korenika AM, Mihaljević M, Prusina T (2012) Influence of different maceration times on the anthocyanin composition and sensory properties of Blatina wines. Agric Conspec Sci 77:41–44
Hermosín Gutiérrez I, Sánchez-Palomo Lorenz E, Vicario Espinosa A (2005) Phenolic composition and magnitude of copigmentation in young and shortly aged red wines made from the cultivars, cabernet sauvignon, Cencibel, and Syrah. Food Chem 92:269–283. doi:10.1016/j.foodchem.2004.07.023
Ivanova V, Stefova M, Vojnoski B (2009) Assay of the phenolic profile of merlot wines from Macedonia: effect of maceration time, storage, SO2 and temperature of storage. Maced J Chem Chem En 28:141–149
Ivanova V, Dörnyei Á, Márk L, Vojnoski B, Stafilov T, Stefova M, Kilár F (2011) Polyphenolic content of Vranec wines produced by different vinification conditions. Food Chem 124:316–325. doi:10.1016/j.foodchem.2010.06.039
Ivanova V, Vojnoski B, Stefova M (2012) Effect of winemaking treatment and wine aging on phenolic content in Vranec wines. J Food Sci Tech Mys 49:161–172. doi:10.1007/s13197-011-0279-2
Ivanova-Petropulos V, Mitrev S (2014) Determination of SO2 and reducing sugars in Macedonian wines. Yearbook Fac Agric 12:7–18 ISSN 1409-987X
Ivanova-Petropulos V, Ricci A, Nedelkovski D, Dimovska V, Parpinnelo GP, Versari A (2015) Targeted analysis of bioactive phenolic compounds and antioxidant activity of Macedonian red wines. Food Chem 171:414–420. doi:10.1016/j.foodchem.2014.09.014
Kennedy JA, Saucier C, Glories Y (2006) Grape and wine phenolics: history and perspective. Am J Enol Vitic 57:239–248
Kontkanen D, Reynolds AG, Cliff MA, King M (2005) Canadian terroir: sensory characterization of Bordeaux-style red wine varieties in the Niagara peninsula. Food Res Int 38:417–425. doi:10.1016/j.foodres.2004.10.010
Košmerl T, Bertalanič L, Maraš V, Kodžulović V, Šućur S, Abramovič H (2013) Impact of yield on total polyphenols, anthocyanins, reducing sugars and antioxidant potential in white and red wines produced from Montenegrin autochthonous grape varieties. Food Sci Technol 1:7–15. doi:10.13189/fst.2013.010102
Mendoza L, Matsuhiro B, Aguirre MJ, Isaacs M, Sotés G, Cotoras M, Melo R (2011) Characterization of phenolics acids profile from Chilean red wines by high performance liquid chromatography. J Chil Chem Soc 56:688–691
OIV (International organization of vine and wine) (2016) Compendium of international methods of analysis of wines and musts. Paris, France, Vol. 1. Available from: http://www.oiv.int/public/medias/4231/compendium-2016-en-vol1.pdf
Ortega T, De La Hera E, Carretero ME, Gómez-Serranillos P, Naval MV, Villar AM, Prodanov M, Vacas V, Arroyo T, Hernández T, Estrella I (2008) Influence of grape variety and their phenolic composition on vasorelaxing activity of young red wines. Eur Food Res Technol 227:1641–1650. doi:10.1007/s00217-008-0888-9
Plavša T, Jurinjak N, Antunović D, Peršurić Đ, Kovačević Ganić K (2012) The influence of skin maceration time on the phenolic composition and antioxidant activity of red wine Teran (Vitis vinifera L.). Food Technol Biotechnol 50:152–158
Pour Nikfardjam MS, Mark L, Avar P, Figler M, Ohmacht R (2006) Polyphenols, anthocyanins, and trans-resveratrol in red wines from the Hungarian Villany region. Food Chem 98:453–462. doi:10.1016/j.foodchem.2005.06.014
Radovanović AN, Jovančićević BS, Radovanović BC, Mihajilov-Krstev T, Zvezdanović JB (2012) Antioxidant and antimicrobial potentials of Serbian red wines produced from international Vitis vinifera grape varieties. J Sci Food Agric 92:2154–2161. doi:10.1002/jsfa.5601
Rajković MB, Sredović ID (2009) The determination of titratable acidity and total tannins in red wine. J Agric Sci 54:223–346
Ribéreau-Gayon P (1970) Le dosage des composés phénoliques totaux dans les vins rouges. Bull Soc Chim Fr 52:627–631
Sacchi KL, Bisson LF, Adams DO (2005) A review of the effect of winemaking techniques on phenolic extraction in red wines. Am J Enol Vitic 56:197–206
Schwarz M, Wabnitz TC, Winterhalter P (2003) Pathway leading to the formation of anthocyanin-vinylphenol adducts and related pigments in red wines. J Agric Food Chem 51:3682–3687. doi:10.1021/jf0340963
Shahidi F, Wanasundara PK (1992) Phenolic antioxidants. Crit Rev Food Sci Nutr 32:67–103. doi:10.1080/10408399209527581
Sharma AK, Kumar R, Ahmad Azad ZRA, Adsule PG (2015) Use of fine wine lees for value addition in ice cream. J Food Sci Tech Mys 52:592–596. doi:10.1007/s13197-013-1007-x
Somers TC (1971) The polymeric nature of wine pigments. Phytochemistry 10:2175–2186. doi:10.1016/S0031-9422(00)97215-7
Sun B, Neves AC, Fernandes TA, Fernandes AL, Mateus N, De Freitas V, Leandro C, Spranger MI (2011) Evolution of phenolic composition of red wine during vinification and storage and its contribution to wine sensory properties and antioxidant activity. J Agric Food Chem 59:6550–6557. doi:10.1021/jf201383e
Tarola AM, Milano F, Giannetti V (2007) Simultaneous determination of phenolic compounds in red wines by HPLC–UV. Anal Lett 40:2433–2445. doi:10.1080/00032710701577666
Tsanova-Savova S, Dimov S, Ribarova F (2002) Anthocyanins and color variables of Bulgarian aged red wines. J Food Compos Anal 15:647–654. doi:10.1006/jfca.2002.1100
Vichapong J, Santaladchaiyakit Y, Burakham R, Srijaranai S (2014) Cloud-point extraction and reversed-phase high performance liquid chromatography for analysis of phenolic compounds and their antioxidant activity in Thai local wines. J Food Sci Tech Mys 51:664–672. doi:10.1007/s13197-011-0556-0
Zúñiga MC, Pérez-Roa RE, Olea-Azar C, Laurie VF, Agosin E (2014) Contribution of metals, sulfur-dioxide and phenolic compounds to the antioxidant capacity of Carménère wines. J Food Compos Anal 35:37–43. doi:10.1016/j.jfca.2014.04.003
Acknowledgments
This work was financially supported by JoinEU-SEE IV, Erasmus Mundus Action 2 Partherships, which is gratefully acknowledged, covering the study stay of Violeta Ivanova-Petropulos at the University of Bologna, whereas the HPLC analyses of wines were performed.
Author information
Authors and Affiliations
Corresponding author
Additional information
Research Highlights
• HPLC-DAD analysis of phenols in Stanušina, Vranec and Cabernet Sauvignon wines was performed.
• Individual phenols were determined for the first time in Stanušina wines.
• Maceration time influenced the phenolics content and composition of wines.
Rights and permissions
About this article
Cite this article
Ivanova-Petropulos, V., Durakova, S., Ricci, A. et al. Extraction and evaluation of natural occurring bioactive compounds and change in antioxidant activity during red winemaking. J Food Sci Technol 53, 2634–2643 (2016). https://doi.org/10.1007/s13197-016-2235-7
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-016-2235-7