Abstract
This study was carried out to evaluate the effect of PEF process using static treatment chamber on fresh sugarcane juice with and without addition of lemon and ginger with respect to microbial content, chemical properties, nutrient content and shelf life extention of the product. The fresh sugar cane juice without addition of lemon and ginger treated at different field strengths (30 kV cm−1 and 50 kV cm−1) and different pulse numbers (150, 300) was initially investigated by storage at room temperature (31 °C) and refrigeration temperature (4 °C) for 30 days. The PEF effect on fresh sugar cane juice at room temperature and refrigerated temperature was compared with untreated sample (31 °C). At the end of the storage period samples treated at field strength 30 kV cm−1, 150 pulses were found to be stable compared with untreated sample. The second experimental study of PEF process was done on fresh sugarcane juice with the addition of lemon and ginger for fourteen days at different electric field intensities (10 kV cm−1, 20 kV cm−1 and 30 kV cm−1) with the same pulse number (150 pulses) and stored 4 °C. Even better reduction of microbes was achieved with PEF treatment condition of field strength 20 kV cm−1, 150 pulses in the presence of lemon and ginger. The sensory attributes of untreated fresh sugarcane juice were maintained up to only two days, but for the PEF treated sample, shelf life was extened up to seven days. Further, addition of lemon and ginger in the PEF treated sugarcane juice doubled the shelf life up to fourteen days.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The first systematic studies on the non-thermal lethal effect of homogeneous pulsed electric fields on microbes were conducted by Sale and Hamilton (1967). They observed that electric field strength and total treatment time were the most important factors involved in the inactivation of bacteria. Damage to the cell membrane, causing an irreversible loss of its function as semi permeable barrier between the cell and its environment, was proposed as the cause of the cell death. The fundamental effects and mechanisms of electric fields on the disruption of biological cells were introduced by Neumann and Rosenheck (1973) and Zimmermann (1986). The cell membrane was considered to act as a capacitor containing a perfectly elastic dielectric. The electro-mechanical instability observed due to the influence of an external electric field on biological cells is still one of the most accepted theories for microbial inactivation (Hamilton and Sale 1967; Sale and Hamilton 1967, 1968).
Pulsed electric field (PEF) is an emerging technology that has been extensively studied for non-thermal food processing, by a number of researchers across a wide range of liquid and semi liquid foods. This method of food preservation uses short burst of electricity for microbial inactivation and causes minimal or no detrimental effect on food quality attributes, retaining their fresh aroma, taste, and appearance (Castro et al. 1993). The sensory attributes of juices are reported to be well preserved, and the shelf life is extended. Application of PEF technology has been successfully demonstrated for the pasteurization of foods such as juices, milk, yogurt, soups, and liquid eggs (Qin et al. 1995). When a plant tissue is treated with high pulsed electric field, the cell membranes are ruptured leading to an increase in permeability of the cell walls and subsequent increase in juice yield (Eshtiaghi and Knorr 1999). Increased yield of sugarcane juice by PEF treatment during extraction has been observed (Kuldiloke et al. 2010; Eshtiaghi and Yoswathana 2012). Our earlier study reported the preservation of tomato juice using PEF technology (Kayalvizhi and Antony 2014). Microbial inactivation depends on properties of pulse, food, chamber, energy, repetition rate etc. The issues with PEF include problems such as the potential for electric arcs and a temperature increase inside the chamber, corrosion or a deposit of solids on a high-voltage electrode indicating some electrolysis of the product and the possibility of transferring some electrode particles to the treated food (FDA 2009). The present study documents the preservation of fresh sugarcane juice (with and without lemon and ginger) using PEF technology.
According to the worldwide top ten sugarcane production surveys of Indian Horticulture Database (2014), India was the second largest producer of sugar cane. Sugarcane is grown mainly for producing sweeteners such as sugar, jaggery/gur (unrefined sugar made from the sap of palm trees or sugarcane) and khandasari (finely granulated crystallized sugar). The preservation of raw sugarcane juice is very difficult because it turns brown soon after extraction and gets spoiled due to fermentation within hours.
Khare et al. (2012) reported that fresh sugarcane juice is popular and commonly used as a delicious drink in both urban and rural areas of India, especially in the northern states of Punjab, Haryana, Himachal Pradesh, Rajasthan and Uttar Pradesh. This drink is also preferred in various countries, especially where sugarcane is commercially grown such as South East Asia, Egypt and Latin America. In Egypt, fresh sugarcane juice is a popular drink served by almost all fruit juice vendors, who can be found abundantly in most cities. In Indonesia and Malaysia, sugarcane juice is sold nationwide especially among street vendors. It is bottled for local distribution in some regions and sold at food courts daily. In Singapore, it is sold in food courts only (Ponting 2000). India is one of the leading manufacturers and exporters of automatic sugarcane juice extractor machines (Solomon 2011).
100 mL of sugarcane juice provides 40 Kcal of energy, 10 mg of iron and 6 μg of beta carotene (Parvathy 1983). It is rich in enzymes both polyphenoloxidase (PPO) and peroxidase (POD) and has many medicinal properties providing water (75–85 %), reducing sugar (0.3–3.0 %), and non-reducing sugar (10–21 %) (Marques et al. 2013). Sugarcane juice is a great preventive and healing source for sore throat, cold and flu. It has a low glycemic index of 30–40 which keeps the body healthy and can therefore be consumed by diabetics in moderation (Khare et al. 2012). It hydrates the body quickly when exposed to prolonged heat and physical activity. It is an excellent substitute for aerated beverages (containing CO2) and cola drinks; it refreshes and energizes the body instantly as it is rich in carbohydrates (Swaminathan 1995).
Based on a preliminary survey we found that fresh sugarcane juice had high microbial load and short shelf life. Eshtiaghi and Knorr (2002) observed increased yield of sugarbeet juice by PEF treatment during extraction and subsequent pasteurization change the taste and smell of the juice.
Despite its popularity and wide consumption, the preservation of juice by processing is limited. Most often the fresh juice is extracted and supplied to the consumers. Very recently fresh sugarcane juice in aseptic pack (Tetra Pak) with shelf life of six months has been commercialized in Pune, India (Sinha et al. 2014). Scientific documentation on the preservation of sugarcane juice is also very limited; probably due to its low shelf life of about 4 h. Chauhan et al. (2007) reported that the sugarcane juice could be preserved with ascorbic acid and potassium metabisulphite (150 mg L−1). Sugarcane juice preserved with potassium metabisulphite showed minimum changes in sensory qualities during storage, both at room and refrigeration temperature for 90 days.
Research attempts in this area will help to improve the shelf life, preserve its nutrition quality and make the product available to a wide group of consumers. The effect of minimal and non-thermal processing methods on sugarcane juice has not been studied till date and hence was taken up for study.
The primary objective of the study was the application of high voltage electric pulse with different field strengths and pulse numbers for a particular time interval to the fresh sugarcane juice which was obtained from the street vendors in order to extend the shelf life.
Materials and methods
Raw material
The fresh sugar cane juice samples (with and without the addition of lemon and ginger) were purchased from street vendors in air tight sterile containers near Anna University, Chennai. They were transported at ambient temperature to the laboratory and immediately subjected to processing and analysis.
Equipment
The equipment used in this study was designed and assembled in the High Voltage Engineering Division of the Faculty of Electrical Engineering, College of Engineering Guindy campus, Anna University, Chennai (Sathyanathan et al. 2012).
PEF Treatment
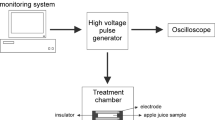
PEF treatment of sugarcane juice using static treatment chamber is shown in Fig. 1. The Marx’s generator circuit was used to produce impulse voltages of 1.3/45 μsec. Sugarcane juice with and without lemon and ginger was treated with optimized test voltages and pulse rate as shown in Table 1. For all PEF experiments 300 mL of sugar cane juice was added inside the static treatment chamber under sterile condition. After PEF treatment the sample was collected in a sterilized, wide-mouthed, screw-capped glass bottle and immediately stored at different temperatures - 31 °C (room temperature) and 4 °C (refrigeration temperature) until analysis. All experiments were repeated twice.
Analysis of shelf life stability
Chemical and microbial tests on untreated and treated samples were carried out in the Food Safety Laboratory of National Agro Foundation, Anna University campus, Taramani to analyze shelf life of the sugar cane juice with and with out addition of lemon and ginger. These tests were carried out every week for 30 days (31 °C and 4 °C) and every alternate day for 14 days (4 °C) in fresh sugar cane juice with out and with the addition of lemon and ginger respectively. The PEF treatments were carried out on two separate batches of fresh sugarcane juice (with and with out lemon and ginger) on two different days and all chemical, nutritional and microbial analyses were done in duplicates. All chemical and reagents used were of analytical grade.
Chemical and nutritional analysis
The physicochemical characteristics of juices considered in quality assessment include pH, titratable acidity (TA), total soluble solids (°Brix) and vitamin C. pH value was measured using AP-1plus pH meter (Susima Chennai) and the total soluble solids present measured by a handheld refractometer (Arico India) and expressed as °Brix. The titratable acidity (% citric acid) was estimated in sugar cane juice filtrate by titration with 0.1 N sodium hydroxide to pale permanent pink end point with phenolphthalein. Further, vitamin C content of the juice was tested by the reduction of the blue dye 2, 6 dichlorophenolindophenol by ascorbic acid (AOAC 2000).
Microbial analysis
Both untreated and treated samples were analyzed for the presence of microbes like bacteria, yeast and molds by serial dilution. The total mesophilic aerobic bacterial count of samples were estimated by the pour plate method using standard plate count agar (Hi Media Mumbai). Plates were incubated at 37 °C for 48 h. The colony counts were carried out using a digital colony counter (Lapiz Mumbai). For the sugarcane juice, two different studies were done. In the first study, we analysed the bacterial count in PEF treated fresh sugarcane juice without addition of lemon and ginger which had been stored for one month and based on that result, we designed the second study in fresh sugarcane juice with addition of lemon and ginger. The yeast and mould count was determined by spread plate method using potato dextrose agar (Hi Media Mumbai) in PEF treated juice that was stored for fourteen days. Plates were incubated at 25 °C for 5 days as described by Harrigan and McCance (1998).
Results and Discussion
Chemical analysis of sugarcane juice (without addition of lemon and ginger stored at 31 °C)
Table 2 shows the physico-chemical changes in PEF treated sugar cane juice (without addition of lemon and ginger) such as pH, acidity, total soluble solids (TSS), vitamin C and color values of both untreated and treated samples stored at room temperature (31 °C) for 24 h, for the application of different field strengths (30 kV cm−1 and 50 kV cm−1) and different pulse numbers (150, 300). There was a significant (p < 0.05) decrease in pH (from 5.10 ± 0.30 to 3.98 ± 0.33), vitamin C (from 87.5 ± 0.14 to 81.54 ± 0.15 mg %) and a significant (p < 0.05) increase in acidity from 0.2 ± 0.10 to 0.5 ± 0.10 %; while, TSS showed no significant (p < 0.05) change (from 17.5 ± 0.00 to 17.0 ± 0.00 °Brix) between the untreated and treated sugar cane juice stored at room temperature (31 °C) for 24 h. After 24 h, the chemical property of PEF treated fresh sugarcane juice (without the addition of lemon and ginger) changed and spoilage was noted; suggesting that storage at room temperature was not effective in preserving the PEF treated sugarcane juice. Without PEF treatment the juice underwent spoilage after 4 h. Table 2 and Fig. 2 show the color changes of both untreated and treated fresh sugarcane juice (without the addition of lemon and ginger) before and after 24 h at room temperature (31 °C). Significant darkening of the color was evident from the decrease in the L values and the color change to yellow seen from the decrease in a* and increase in b* values.
Chemical analysis of sugarcane juice (without addition of lemon and ginger at 4 °C)
Table 3 shows that the PEF treated juice stored at 4 °C was better preserved compared to storage at 31 °C. At the end of one week, there was a little change in pH (from 5.10 ± 0.30 to 4.95 ± 0.30) and TSS (from 17 ± 0.00 to 19.0 ± 0.00 °Brix) while acidity increased from 0.2 ± 0.10 to 5.7 ± 0.10 %. The vitamin C content was retained (87.5 ± 0.14 mg %) up to 7 days when low intensity of PEF at 30 kV cm−1, 150 pulses was used. Higher field intensities and pulse numbers caused significant (p < 0.05) reduction in the ascorbic acid (from 87.5 ± 0.14 to 60.50 ± 0.10 mg %). It was also seen that longer storage up to 14 days led to loss of vitamin C in all cases. Although the color values L, a*, b* showed variations during storage (Table 3), perceptible changes were seen only at end of the second week (Fig 2 c).
Thus from the one month storage study it was observed that 30 kV cm−1,150 pulse condition was better in retaining the chemical properties and color of the juice compared to higher field strengths and pulse numbers when stored at refrigeration temperature (4 °C) for seven days. Therefore in the subsequent study on sugarcane juice with added lemon and ginger, lower field strength and pulse was used for PEF treatment (10 kV cm−1/150 pulse, 20 kV cm−1/150 pulse and 30 kV cm−1/150 pulse) and the treated juice was stored at refrigerated temperature (4 °C). The reason behind the addition of lemon and ginger was that this was the preferred form of consumption by the consumers for its enhanced flavor. Further, it is likely to prevent oxidation of the juice thereby minimizing the color change and the increased acidity may also inhibit microbial growth.
Chemical analysis of sugarcane juice (with addition of lemon and ginger stored at 4 °C)
The color changes of PEF treated fresh sugarcane juice (with lemon and ginger) on 1st day, 7th and 14th day of storage at 4 °C is shown in Fig. 3. Color was well retained in the treated juice for seven days, while color change was noticeable in the entire treated sample at the end of second week. Figure 4 shows changes in pH and acidity (% citric acid). All the treated samples maintained their pH till the 9th day and after the 9th day there were slight variations in the pH, but in the untreated sample there was decrease in the pH from the 5th day onwards. The percentage of acidity in the treated sample was retained between 0.2 % - 0.3 % for seven days, while in the untreated sample, it increased markedly after the 3rd day. As seen from Fig. 5, TSS (°Brix) value which indicates the sugar content increased (30 %) after the 7th day and peaked on the 9th day. The vitamin C content also increased (30 %) after the 5th day and peaked on the 7th day. Although similar changes were seen in the untreated, the levels of both parameters were comparatively low (Fig. 5). These increases may be attributed to the rate of microbial growth during storage. The higher growth rate in the untreated samples would account for the decrease in TSS, while cell lysis and secretion of metabolies could contribute to the increase (Fig. 7). Yeast present in the untreated and treated samples, which are known to produce ascorbic acid may have contributed (Sankhla et al. 2012).
Microbial analysis
PEF treatments in sugarcane juice without lemon and ginger with storage at refegeration temperature (4 °C) caused significant (p < 0.05) reduction (2.71 log cfu mL−1) in total aerobic mesophilic counts compared to room temperature (31 °C) as seen from Fig. 6. The field strength of 30 kV cm−1, 150 pulses/110 s pulse treatment had higher bacterial reduction, when compared to the 50 kV cm−1, 150 pulses and 50 kV cm−1, 300 pulses, indicating that lower pulse treatment resulted in effective microbial reduction (Fig. 6).
From the second set of exeperiments on fresh sugarcane juice with the addition of lemon and ginger, PEF treatment with 10 kV cm−1, 150 pulses and 20 kV cm−1,150 pulses treatment maintained low yeast counts for 7 days and after the 7th day started to increase (Fig. 7). Molds were not detected in the juice. In contrast, in the untreated sample the yeast growth was numerous from the 3rd day (about 1log increase). In both the set of exeperiments higer microbial counts with higer intensites of treatments may be explained by the elevated temperature of the sample corresponding to the higer field intensites and pulses (Ayman El-hag et al. 2008). Microbial contaminants of the fruit juice were below 106 cfu mL−1 and thus within acceptable limit for human consumption (ICMSF, 1986).
Conclusion
PEF processing significantly increased microbiological safety and stability of sugarcane juice, while reducing unfavorable changes in nutritional and sensory properties of the beverage. Encouraging results have been obtained with PEF treatment with lower intensity at the laboratory level. PEF treated juice without added lemon and ginger could be stored at room temperature (31 °C) only for 24 h. Storage of the same juice at refrigerated temperature (4 °C) extended shelf life to seven days. With the addition of lemon and ginger, sugarcane juice could be preserved with even lower PEF treatment (20 kV cm−1, 150pulses) and the product quality was maintained for seven days with lower microbial counts. Further experiments are needed to optimize the process and enhance the shelf life.
References
AOAC (2000) Official Methods of Analysis: Method No. 967. 21, 17th ed. Association of Official Agricultural Chemists. Gaithersburg MD USA.
Ayman H, El-Hag, Otunola A, Jayaram SH, Anderson WA (2008) Reduction of microbial growth in milk by pulsed electric fields. IEEE International Conference on Dielectric Liquids 1–4
Chauhan OP, Dheer Singh SM, Tyagi, Balyan DK (2007) Studies on preservation of sugarcane juice. J Food Properties 5:217–229
Castro AJ, Canovas GVB, Swanson BG (1993) Microbial inactivation of foods by pulsed electric fields. J Food Processing and Preservation 17:47–73
Eshtiaghi MN, Knorr D (1999) Process for treatment of sugar beet, European Patent Patent-Nr.: EP 99923708
Eshtiaghi MN, Knorr D (2002) High electric field pulse treatment: potential for sugar beet processing. J Food Engg 52:265–272
Eshtiaghi MN, Yoswathana N (2012) Laboratory scale extraction of sugarcane using high electric field pulses. World academy of science. Eng Technol 6:1215–1220
FDA (US Food and Drug Administration) (2009) Kinetics of microbial inactivation for alternative food processing technologies - Pulsed Electric Fields. Center for Food Safety and Applied Nutrition: 2–37, http://www.steribeam.com/technology/FDA-on-PEF-Rev 2009.pdf
Hamilton WA, Sale AJH (1967) Effects of high electric fields on microorganisms-II. Mechanism of Action of the Lethal Effect Biochem Biophy Acta 148:789–800
Harrigan WF, McCance ME (1998) Laboratory method in food and dairy microbiology. Academic Press, London. ISBN 0123260434.
ICMSF (International Commission on Microbiology Specification for Food) (1986) Sampling for microbiological Analysis. Principles and Specific Application, University of Toronto press, Toronto: 203–205.
Indian Horticulture Database (2014) Available from: <agricoop.nic.in/imagedefault/whatsnew/handbook2014.pdf>. [7 October 2014]
Kayalvizhi V, Antony U (2014) Microbial and Physico-chemical changes in tomato juice subjected to pulsed electric field treatment. Afr J Microbial Res 8(37):3382–3391
Khare A, Behari Lal A, Singh A, Pratap (2012) Shelflife enhancement of sugarcane juice. Croat J Food Tech, Biotech and Nutr 7:179–183
Kuldiloke J, Eshtiaghi MN, Neatpisarnvanit C (2010) Application of high electric field pulses for sugarcane processing. KMITL Science and Technology J 8:75–83
Marques TA, Rampazo EM, Marques PA (2013) Oxidative enzymes activity in sugarcane juice as a function of the planting system. Food Sci and Technol, Campinas 33:146–150
Neumann E, Rosenheck K (1973) Potential difference across vesicular membrane. J Membrane Bio 14:194–196
Parvathy K (1983) Bottling of sugarcane juice proceedings of scheme for studies on postharvest technology. ICAR Coimbatore Centre India. Annual report: 13–16
Ponting C (2000) World History: A New Perspective, Chatto & Windus (eds), London
Qin BL, Zhang Q, Barbosa-Canovas GV, Swanson BG, Pedrow PD (1995) Pulsed electricfield treatment chamber design for liquid food pasteurization using the finite element method. Trans ASAE 38(2):557–565
Sale AJH, Hamilton WA (1967) Effects of high electric fields on microorganisms I. killing of bacteria and yeast. Biochimt Biophys Act 148:781–788
Sale AJH, Hamilton WA (1968) Effects of high electric fields on microorganisms-lysis of erythrocytes and protoplasts. Biochimica and Biophysica Acta 163:37–43
Sankhla S, Chaturvedi A, Kuna A, Dhanalakshmi K (2012) Preservation of sugarcane juice using hurdle technology. Sugar Tech 14(1):26–39
Sathyanathan T, Kayalvizhi V, Sree VG, Sundararajan R (2012) Microbial growth reduction in tomato juice by pulsed electric field with co-axial treatment chamber. IEEE 10th International Conference on the Properties and Applications of Dielectric Materials
Sinha R, Mishra AK, Singh HK (2014) The branding of sugarcane juice in India. J Economics and Business Research 20:193–202
Solomon S (2011) The Indian sugar industry: an overview. Sugar Tech 13:255–265
Swaminathan V (1995) Food science chemistry and experimental foods. Bangalore printing and publishing co. ltd first edition, Bangalore
Zimmermann U (1986) Electric breakdown, electropermeabilization and electrofusion. Rev Physiol Biochem Pharmacol 105:176–256
Acknowledgments
We gratefully acknowledge Division of High Voltage Engnieering, Anna University, Chennai http://www.annauniv.edu/HighVoltage/index.php and Food Safety and Standards Division, “National Agro Foundation”, Centre for Food Technology, Department of Biotechnology, Taramani, Chennai for the provision of laboratory facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kayalvizhi, V., Pushpa, A.J.S., Sangeetha, G. et al. Effect of pulsed electric field (PEF) treatment on sugarcane juice. J Food Sci Technol 53, 1371–1379 (2016). https://doi.org/10.1007/s13197-016-2172-5
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-016-2172-5